Abstract

Introduction
Postoperative pancreatic fistula (POPF) is the main driver of postoperative complications after pancreatectomy.1,2 Both pasireotide—a somatostatin analogue—and hydrocortisone—a corticosteroid—reduce the incidence of POPF compared to placebo.1,2 Pasireotide is thought to function via reduction of the exocrine secretion of the pancreas, 1 while hydrocortisone has anti-inflammatory features, decreasing the risk for post-pancreatectomy acute pancreatitis—a risk factor for POPF.2,3 The HYPAR trial (HYdrocortisone vs PAsireotide in Reducing pancreatic surgery complications) was a randomized clinical trial (RCT) comparing pasireotide versus hydrocortisone in patients undergoing pancreatectomy with high risk for POPF. The primary RCT outcome was Comprehensive Complication Index (CCI) at 30 days, and these short-term results suggested the superiority of pasireotide in preventing postoperative complications, especially in distal pancreatectomies (DPs). 4 It is unclear whether pasireotide or hydrocortisone affects long-term outcomes, and anti-inflammatory properties of hydrocortisone might affect oncological outcomes. Here, we report the predefined five-year outcomes of the HYPAR trial.
Methods
The HYPAR trial was conducted at Helsinki University Hospital between 19 May 2016 and 17 December 2018. The study design, study protocol, and short-term results are previously published. 4 The original trial has been registered to both ClinicalTrials.gov (NCT02775277) and EudraCT (2016-000212-16).
Briefly, the trial included patients who underwent a partial pancreatectomy with a high risk for POPF. Patients were randomized to receive perioperatively either pasireotide (900 µg, twice a day subcutaneously, until the evening of postoperative day 6) or hydrocortisone (100 mg, intravenously, three times per day until the evening of postoperative day 2). The first dose of the study drug was administered in the morning of the operation. The trial was approved by the Finnish National Committee on Medical Research Ethics, Finnish Medicine Agency, Helsinki University Hospital’s ethical committee, and Helsinki University Review Board. All patients gave written consent. The predefined secondary outcomes included disease-specific and disease-free survival. The follow-up data were collected from the electronic medical records, and the data included the date and site of possible recurrence and the date and cause of death. If medical records were incomplete, the patient was contacted by a study nurse.
Groups were compared using t-test, Mann–Whitney U, or Chi-square tests as appropriate. Survival was analyzed using Kaplan–Meier and compared using the log rank test.
Results
One hundred twenty-six patients were randomized to receive either pasireotide or hydrocortisone and were included in the analyses. Baseline and operative characteristics have been published earlier. 4 Complete 5-year follow-up (or until death) was obtained from 115 patients (91.2%, 54 in Pasireotide group, 61 in Hydrocortisone group), and the median follow-up was 64 months (range = 0–88). Both groups had statistically similar overall and disease-free survival in 1- and 5-year follow-up in the overall cohort as well as in a cohort only including patients with a malignancy (Fig. 1). In patients with pancreatic ductal adenocarcinoma (PDAC), 1-year and 5-year overall survival were 92.3% and 30.8% in Pasireotide group versus 53.8% and 15.4% in Hydrocortisone group, respectively (p = 0.222) (Fig. 1).
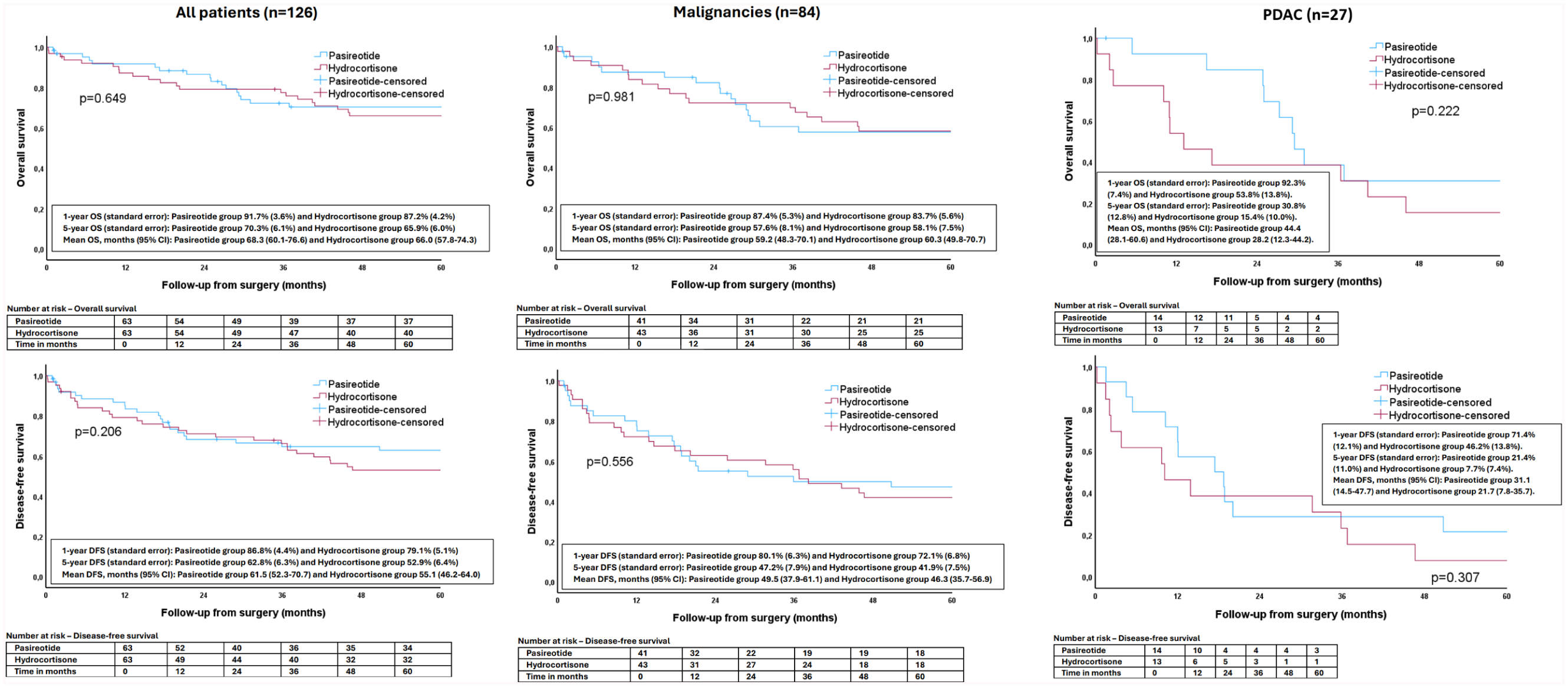
The overall and disease-free survival of the patients in the HYPAR trial.
Discussion
In this secondary analysis of the HYPAR trial, there were no statistically significant differences in long-term outcomes between patients randomized to receive perioperatively pasireotide versus hydrocortisone in the whole cohort or in the subgroups for malignant disease. However, some uncertainty remains as 1- and 5-year overall survivals were approximately double (92% versus 54 % and 31% versus 15%) in Pasireotide group versus Hydrocortisone group in patients with PDAC. Although this difference did not reach statistical significance, it is prone to type 2 error due to a small number of patients. The effect of pasireotide and other somatostatin analogues in pancreatic surgery are vastly studied, but these studies focus only on short-term outcomes.1,4 However, a study suggested that corticosteroids could reduce the risk of recurrence after pancreatic cancer resection due to their anti-inflammatory and immunomodulatory pathways, and an animal study showed that corticosteroids suppress the dissemination of cancer cells.5,6 These studies do not specifically focus on hydrocortisone, and such studies concerning pasireotide are lacking. The limitations of this study are the single-center setting without a placebo arm and the low number of patients with cancer, which may lead into false negative findings. Future studies with a larger sample size, a placebo arm, and potentially a combined pasireotide-hydrocortisone arm are warranted.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
