Abstract
Background:
Roux-en-Y gastric bypass (RYGB) is a standard bariatric surgical technique, associated with suboptimal response and recurrent weight gain in 25%–50% of cases. Single-anastomosis duodeno-ileal bypass with sleeve gastrectomy (SADI-S) is a simplification of duodenal switch with promising results. However, a comparison of the two techniques in a randomized clinical trial (RCT) has not yet been reported. This RCT aims to compare SADI-S and RYGB.
Methods:
BYPSADIS is a randomized, open-label, multicentre clinical trial comparing SADI-S with RYGB. Patients aged 18–60 years with body mass index (BMI) = 40–50 kg/m2 will be recruited in six hospitals. Primary endpoints are as follows: (1) efficacy, measured as percentage of patients with total weight loss (TWL) > 30% at 5 years; and (2) safety, defined as percentage of patients experiencing at least one severe complication either at 90 days postoperatively (Clavien-Dindo >2) or up to 5 years, including severe malnutrition, diarrhea, dumping, hypoglycemia, or need for surgical reintervention. Sample size was calculated for both primary outcomes, needing 432 patients to prove superiority of SADI-S in efficacy and 423 to prove non-inferiority in safety, with statistical power of 0.8, alpha risk of 0.025, assuming 10% loss throughout follow-up. To ensure power for both primary endpoints, 450 subjects will be included, 225 SADI-S and 225 RYGB. Secondary outcomes include metabolic comorbidities, quality of life, digestive symptoms (dumping, bowel habits, and GERD), need of extra supplementations, and alimentary habits. Outcomes will be compared using one-sided 95% confidence interval of proportion difference between groups.
Discussion:
To our knowledge, there is only one ongoing RCT comparing SADI-S with RYGB (SADISLEEVE trial, ClinicalTrials.gov identifier NCT03610256), which includes primary and revisional surgeries, without BMI restrictions and with varying limb lengths. The BYPSADIS trial will complement its findings and help to bridge the knowledge gap on whether SADI-S is more effective than RYGB, while offering a similar safety profile.
Trial registration:
The study protocol has been approved by the Ethics Committee of the six participating hospitals and has been registered in ClinicalTrials.gov, NCT06789965.
Keywords
Summary
A study protocol of an open-label, multicentre RCT comparing SADI-S with RY-GBP—Is SADI-S more effective than RYGB in patients with grade III obesity, with similar safety?
Context and Relevance
The most effective treatment in the long term for people living with severe obesity (body mass index (BMI) > 35 kg/m2) is metabolic and bariatric surgery. The Roux-en-Y gastric bypass (RYGBP) is currently considered the gold standard bariatric technique. It involves creating a small gastric pouch and a Roux-en-Y gastrojejunostomy. It is a well-standardized technique with large cohorts of patients followed over the long term, with a good balance between safety and efficacy. However, up to 25%–50% of patients may present suboptimal initial response or recurrent weight gain in the long term.
Single-anastomosis duodeno-ileal bypass with sleeve gastrectomy (SADI-S) is a simplification of duodenal switch (DS) which could potentially improve these results. It was described in 2007 with the aim of maintaining the efficacy of DS, minimizing its potential complications, especially the risk of malnutrition. It involves a sleeve gastrectomy and a duodeno-ileal anastomosis with preservation of the pylorus, jejunal exclusion, and a common channel of 250 or 300 cm of ileum. Even though SADI-S is a promising bariatric technique, there is lack of solid evidence on its efficacy and safety compared with RYGB.
The BYPSADIS trial is an open-label multicentre randomized study aiming to determine whether SADI-S more effective than RYGB, while offering a similar safety profile.
Background
Obesity is a major public health challenge. It directly or indirectly contributes as the leading cause of non-traumatic death in the Western population and is progressively reaching developing countries as well.1,2 In Spain, recent epidemiological studies estimate that over 20% of the population is at least overweight, and nearly 5% has severe obesity. 3 The treatment of obesity is multidisciplinary, ranging from lifestyle and dietary changes to surgery. Among all available treatments, surgery remains the most cost-effective option for patients with severe obesity.4,5 The beneficial effects go far beyond weight loss, as it has been shown to reduce and improve comorbidities, enhance quality of life, lower cardiovascular and cancer risk, reducing mortality at long term in individuals affected by obesity.6–8
DS has proven to be the most effective surgical procedure for treating severe obesity and its comorbidities.9,10 However, it currently represents a small percentage of bariatric surgeries performed worldwide, likely due to its technical complexity and the risk of long-term complications.11,12 To simplify the DS technique, the SADI-S was described in Spain in 2007. 13 The omega reconstruction, which avoids the distal ileo-ileal anastomosis, aims to decrease surgical time and reduce postoperative risks.13,14 The SADI-S involves a sleeve gastrectomy and a duodeno-ileal anastomosis with preservation of the pylorus, jejunal exclusion, and an original total common channel of 200 cm, standardized later to 250 or 300 cm to minimize the risk of nutritional deficiencies. The SADI-S can be performed as a primary direct surgery, planned in two stages, or as a revisional surgery in case of late postoperative deterioration in case of weight gain after sleeve gastrectomy. Observational studies suggest SADI-S may be associated with similar weight control and fewer long-term complications and nutritional deficiencies compared with DS, but robust comparative data are lacking.15–17
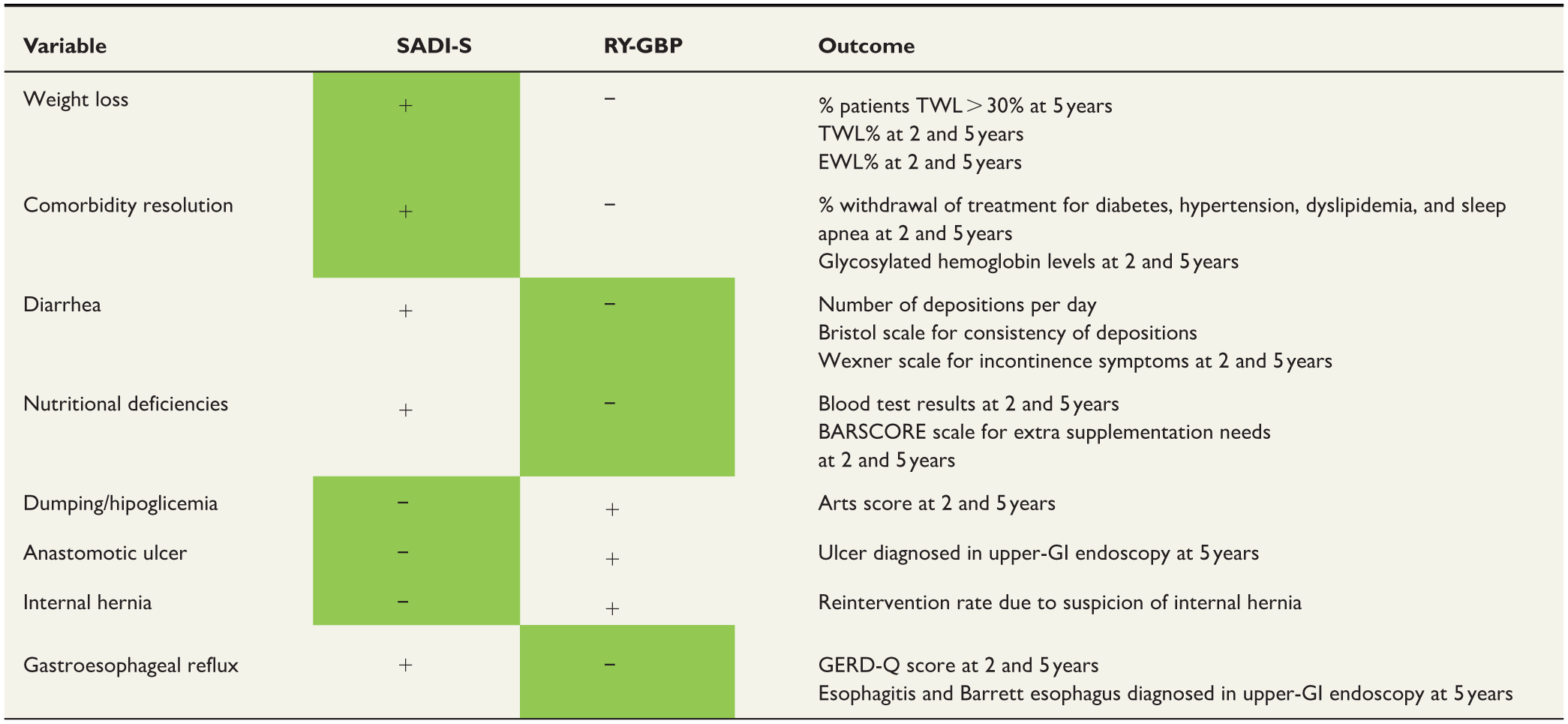
The Roux-en-Y gastric bypass (RYGB) is currently considered the gold standard of bariatric surgery. 11 It involves creating a small gastric reservoir and a Roux-en-Y gastrojejunostomy. It is a well-standardized technique associated with good initial results.18–20 Furthermore, it is the best technique for preventing or treating gastroesophageal reflux (GERD), due to its small gastric reservoir and rapid emptying. 21 However, it is associated with suboptimal weight control in the long term in up to 25%–50% of patients.22,23 Besides, it has potential specific complications resulting from the non-preservation of the pylorus and mesenteric partitioning: anastomotic ulcer, early or late dumping syndrome, hypoglycemias, and abdominal pain associated with internal hernias.24–26 All these complications are uncommon, but data from retrospective studies suggest that they are more frequent after an RYGB than after a SADI-S14–17 (Table 1).
Expected differences between surgical techniques in variables measured in the BYPSADIS trial.
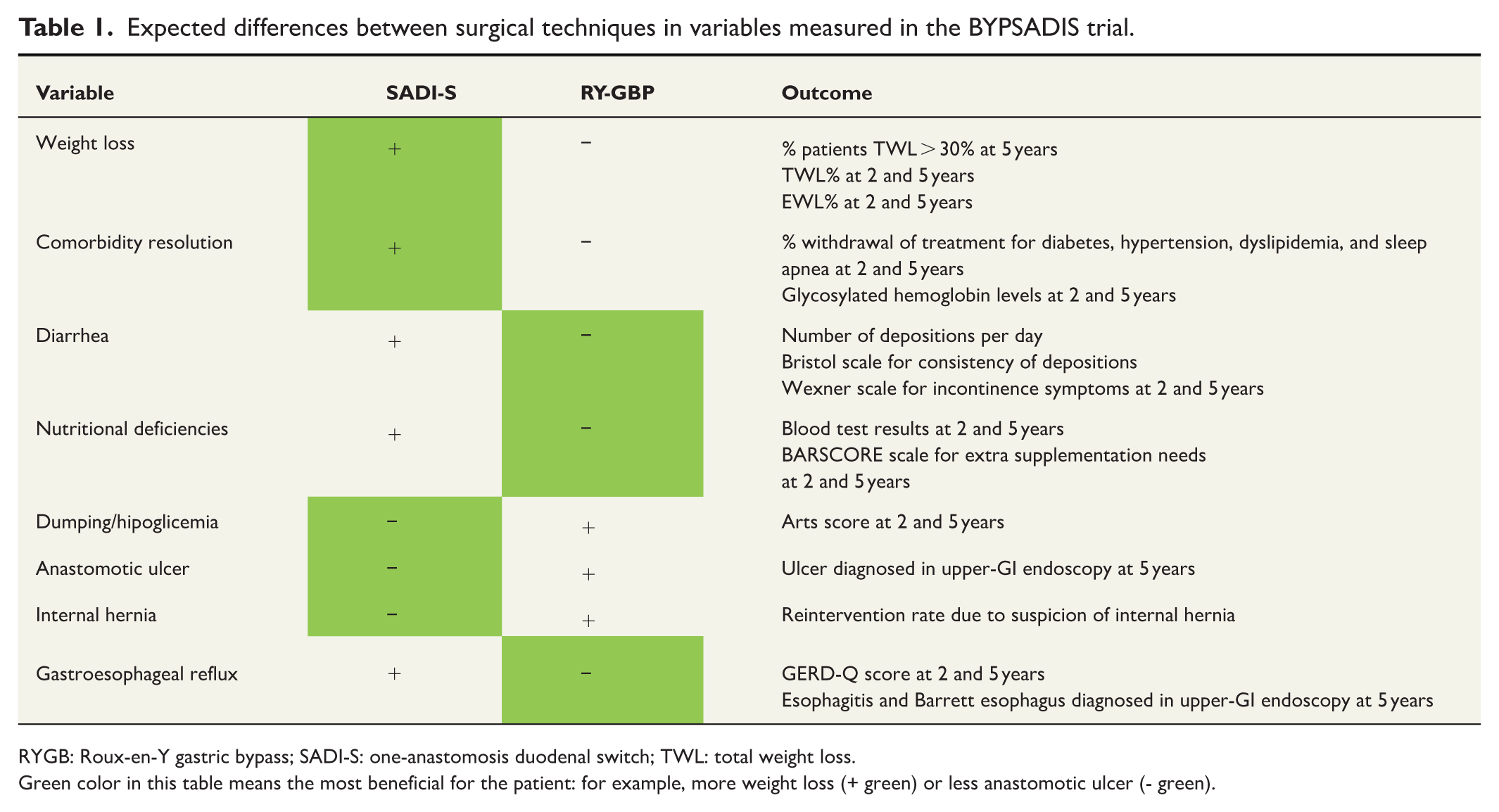
RYGB: Roux-en-Y gastric bypass; SADI-S: one-anastomosis duodenal switch; TWL: total weight loss.
Green color in this table means the most beneficial for the patient: for example, more weight loss (+ green) or less anastomotic ulcer (- green).
To our knowledge, there is currently only one ongoing prospective study with the primary aim of comparing weight loss at 2 years ensuing SADI-S or RY-GBP (SADISLEEVE, ClinicalTrials.gov ID NCT03610256). This is a French multicentre open-label RCT, the results of which will be published in the near future. It includes both patients undergoing primary surgery and those undergoing revisional procedures, without BMI restrictions and with varying lengths of limbs according to the pre-surgical BMI. At variance with that study, the present RCT aims to compare SADI-S with RYGB as primary bariatric procedures in patients with a BMI between 40 and 50 kg/m2, with a standardized homogeneous surgical technique in all participating centers. By using both weight loss and safety up to 5 years of follow-up as co-primary endpoints, we believe this study will help bridge the knowledge gap on whether SADI-S is more effective than RYGB, while offering a similar safety profile.
Methods
Study design
The BYPSADIS trial is a multicentre, open-label RCT comparing SADI-S with RYGB for the treatment of adult patients with obesity, with BMI ranging from 40 to 50 kg/m2 to ensure homogeneity. Trial enrolls patients from six high-volume hospitals, four Spanish University public centers (Hospital Clinc de Barcelona, Hospital de Bellvitge, Hospital Clínco San Carlos de Madrid, and Hospital Universitario de Navarra), and two private centers, of which one in Spain (Hospital de Sagunto) and one in Portugal (Hospital Entre Douro e Vouga). The protocol adheres to the SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) statement. 27 A flowchart of the trial is shown in Fig. 1.
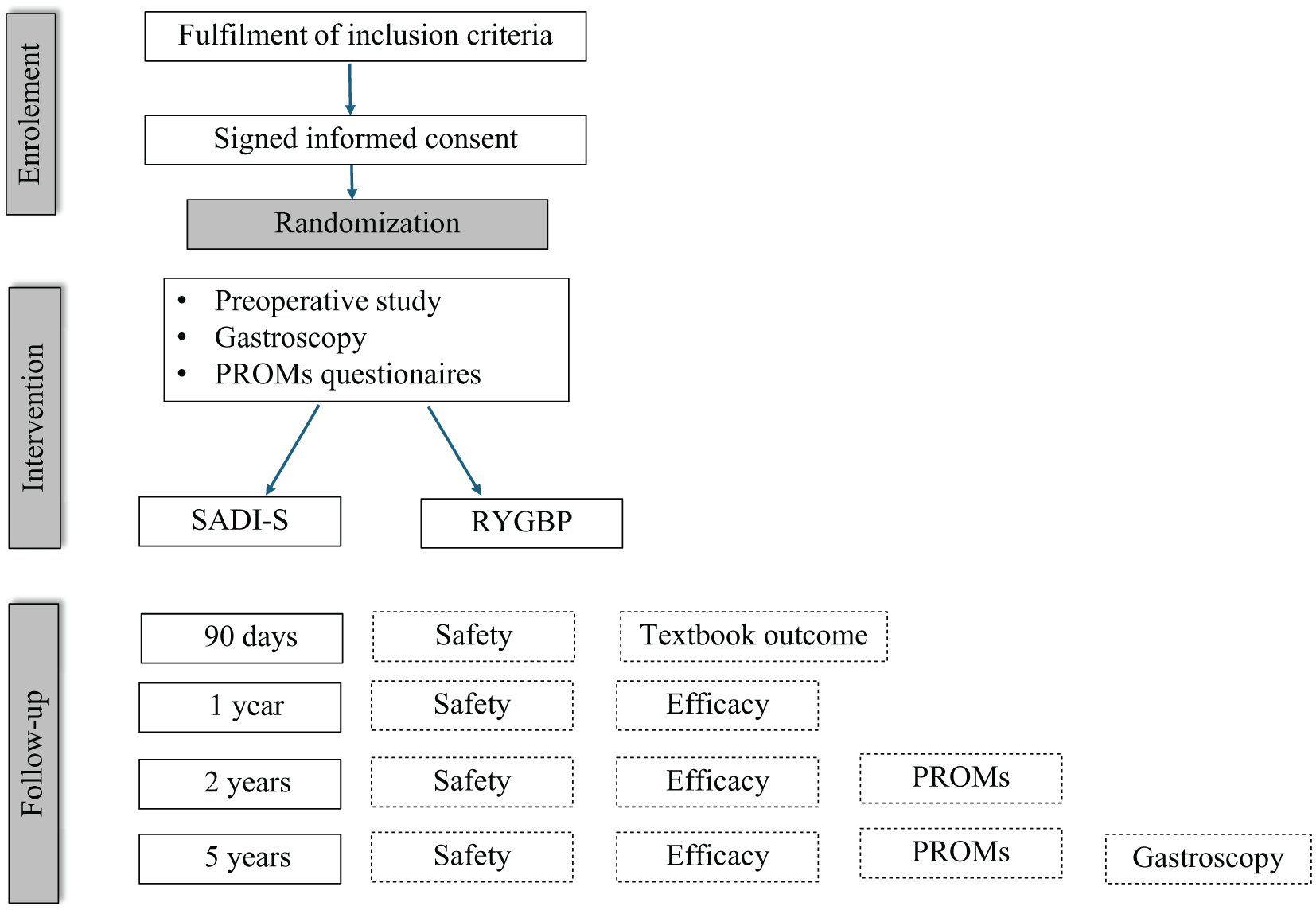
BYPSADIS flow diagram.
Patient selection
All adult patients (aged 18–60 years) with initial BMI between 40 and 50 kg/m2 for whom primary bariatric surgery is indicated at the participating centers will be screened for eligibility. Patients with previous bariatric surgery, as well as those with contraindications for hypoabsorptive or mixed bariatric surgery (such as severe liver disease, inflammatory bowel disease, refractory anemia, or bridge to solid organ transplantation), will be excluded. Patients who are unable to co-operate due to cognitive, psychiatric, or language barriers will also be excluded, as well as those refusing to give informed consent. A summary of inclusion and exclusion criteria is given in Table 2.
Detailed inclusion and exclusion criteria of the BYPSADIS trial.
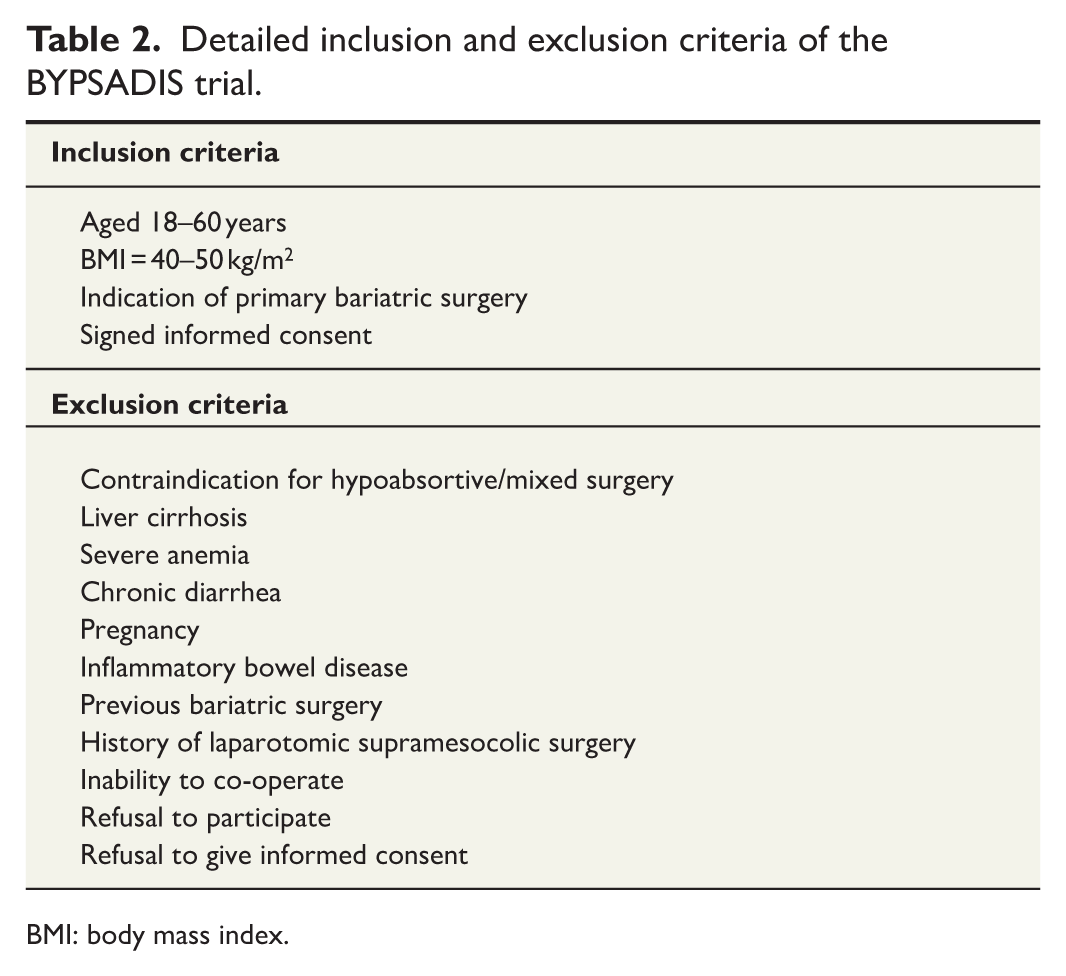
BMI: body mass index.
Randomization
After written informed consent for the BYPSADIS trial, enrolled patients will be randomized using the Research Electronic Data Capture (REDCap) online database by the attending surgeon in each study hospital, with a 1:1 equal allocation ratio to receive either SADI-S or RYGB, with no stratification. The assigned technique will be explained to the patient, and a second specific informed consent will be signed for the surgical procedure, in accordance with clinical practice and regulatory requirements.
Sample-size calculation
Sample size was calculated for both primary endpoints separately:
Efficacy (proportion of patients maintaining a TWL ⩾ 30% at 5 years): Based on prior published studies,14,15,22 it is assumed that 38% of patients submitted to RYGB and 52% of SADI-S patients will maintain TWL ⩾ 30% at 5 years. In total, 432 patients are needed to prove superiority of SADI-S, with statistical power 0.8, alpha risk of 0.025, and assuming 10% of patients will be lost during follow-up.
Safety (proportion of patients experiencing at least one severe complication up to 5 years postoperatively): It is assumed that 20.6% of patients in the RYGB group and 7.4% in the SADI-S group will experience some severe complication up to 5 years after surgery, based on prior studies. In total, 423 patients will be needed to prove non-inferiority of SADI-S, with statistical power of 0.8, alpha risk of 0.025, and assuming 10% of patients will be lost during follow-up.
To ensure enough power for both primary endpoints, 450 subjects will be included, 225 assigned to SADI-S and 225 to RYGB. First patient was included in February 2025, and recruitment primary completion is estimated in August 2026.
Interventions
All patients will be submitted to the standard preoperative circuit in each center, including esophago-gastric endoscopy and anesthetic evaluation. Endoscopy will detail the presence or absence of esophagitis (graded according to Los Angeles score), gastric or duodenal ulcers, gastritis, or bile lakes.
Besides, patients will be requested to answer the following questionnaires:
SF-12 test for quality of life
Arts questionnaire
Number of bowel movements per day
Bristol visual scale for consistency of depositions
Wexner test for fecal incontinence
GERDQ test for gastro-oesophageal reflux symptoms
BITE test for alimentary habits
All six participating surgical teams have previous experience in both techniques and have been tutored and accredited as such by the Sociedad Española de Cirugía de la Obesidad (SECO), Spanish branch of the IFSO. Surgical technique is standardized in all participating hospitals:
For SADI-S, the complete dissection of the antrum and the first portion of the duodenum are performed up to the limit of the gastroduodenal artery. A sleeve gastrectomy is created using a 38 F Foucher tube with endo-staplers. Next, the duodenum is sectioned with an endo-stapler. Then, 250 cm is measured from the ileocecal valve, where a duodeno-ileal hand-sewn end-to-side anastomosis is performed (Fig. 2A). Finally, the duodeno-ileal anastomosis is tested using intraoperative endoscopy or a methylene blue and air test. The gastrectomy specimen is removed through a trocar incision. No drain will be systematically placed, being considered in cases of special technical difficulty or intraoperative complications.
For RYGB, the lesser gastric curvature is first dissected between the cardia and the angular incisura, accessing the retrogastric space. A gastric pouch is then created with endo-staplers with a 38 F orogastric tube guide. Next, 100 cm of the biliopancreatic limb is measured, where a mechanical side-to-side anastomosis is performed, using an endo-stapler and manual closure of the enterotomy. Then, 150 cm of the alimentary limb is measured, and a side-to-side jejunojejunal anastomosis is created (Fig. 2B). A section is then made between the two anastomoses. The mesenteric and the Petersen spaces are closed with a continuous non-absorbable running suture. The anastomosis is verified using intraoperative endoscopy or a methylene blue and air test. No drain will be systematically placed, except in case of special technical difficulty or intraoperative complications.
Patients will start prompt oral liquid intake and mobilization. Hospital discharge will be considered on postoperative day 1 or 2 in the absence of complications.
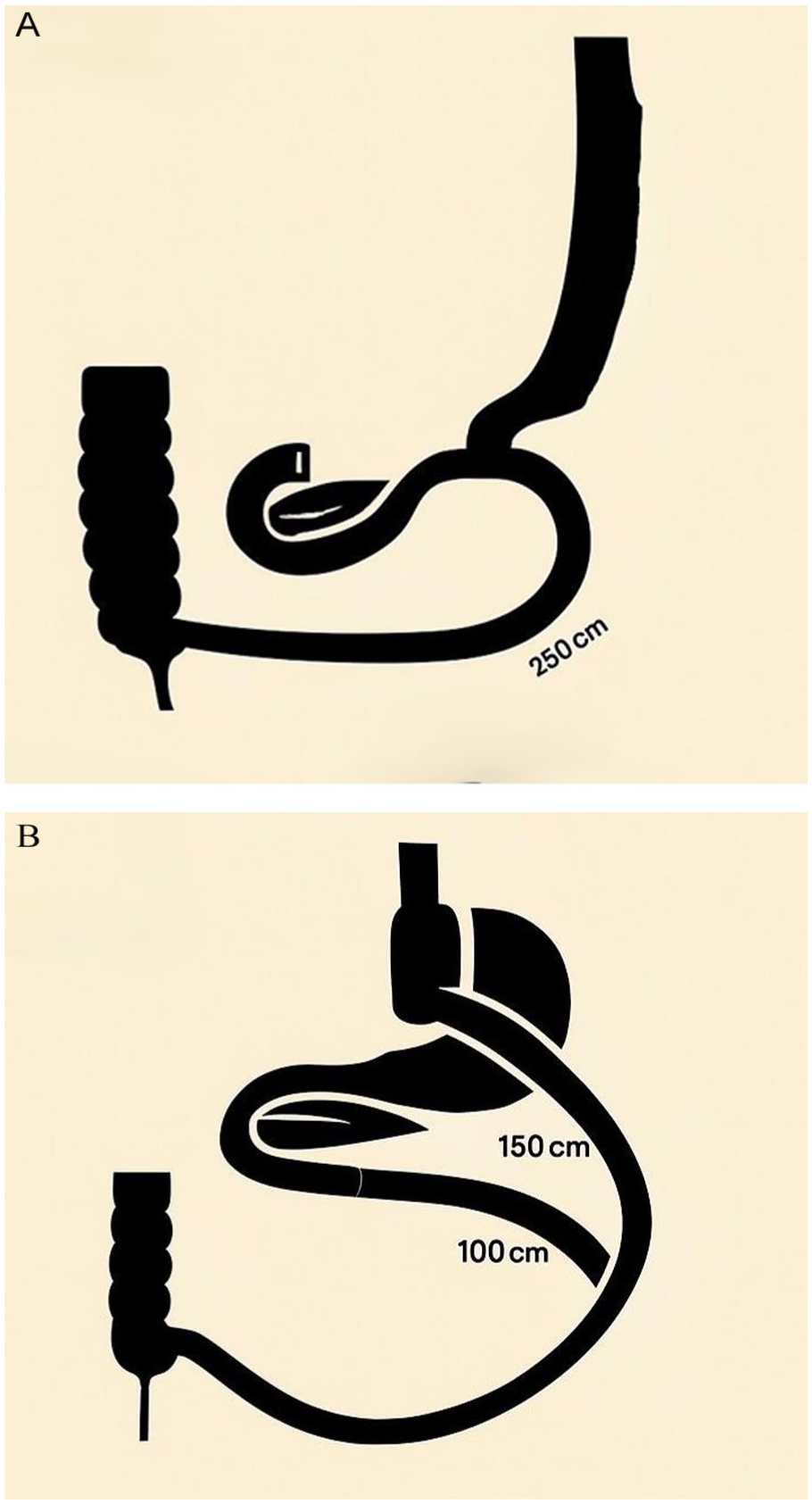
BYPSADIS surgical techniques: (A) SADI-S and (B) RYGB.
Follow-up
Face-to-face visits in outpatient clinic with the treating surgeon will be planned at least once within 90 days after surgery. Since then, follow-up with surgeons, endocrinologists, and nutritionists will be arranged up to 5 years postoperatively, with at least two visits and one complete nutritional blood test per year.
Two years after surgery, an esophago-gastric endoscopy will be performed, with detailing the presence of esophagitis (graded according to Los Angeles score), gastric or anastomotic ulcers, gastritis, or bile lakes.
At 2 and 5 years after surgery, patients will be requested to answer the following tests:
SF-12 test for quality of life
Arts questionnaire
Number of bowel movements per day
Bristol visual scale for consistency of depositions
Wexner test for fecal incontinence
GERDQ test for gastro-oesophageal reflux symptoms
BITE test for alimentary habits
Patients lost to follow-up will be contacted by phone or email by the local surgical team.
Outcome parameters
A summary of the outcomes of the BYPSADIS trial is given in Table 3.
Detailed primary and secondary outcomes of the study.
TWL: total weight loss; ICU: intensive care unit; GERD: gastroesophageal reflux disease.
Primary outcomes
There are two primary outcomes
Efficacy, measured as the percentage of patients with total TWL% > 30% at 5 years.
Safety, defined as the percentage of patients experiencing at least one severe complication: In the short term (within 90 days postoperatively), complications classified as Clavien-Dindo III or IV (those requiring reoperation, endoscopic or radio-guided interventional treatment, and/or ICU admission). From 90 days to 5 years postoperatively, any complications requiring reoperation, endoscopic or radio-guided interventional treatment, ICU admission, sustained parenteral or enteral nutrition, or specific parenteral therapy. Reasons attributable to bariatric surgery are included, such as intestinal obstruction, internal hernia, diarrhea, hypoglycemia, dumping, malnutrition, ulcer, or GERD. Reoperations for incisional hernia, cholecystectomy, or causes unrelated to bariatric surgery are excluded.
Secondary outcomes
Mortality at 5 years.
Ordered textbook outcome for bariatric surgery, at 90 days postoperatively: (0) No complications, no hospital readmissions, and hospital stay of 2 days or less from surgery. (1) Prolonged hospital stays (>2 days). (2) Mild complications (Clavien-Dindo I–II). (3) Hospital readmission within 90 days of surgical intervention. (4) Severe complications (Clavien-Dindo >IIIA). (5) Mortality.
Dumping syndrome at 2 and 5 years after surgery, according to the Arts questionnaire.
Gastro-oesophageal reflux symptoms at 2 and 5 years after surgery, according to the GERD Q test.
Number of bowel movements per day at 2 and 5 years after surgery, together with tool consistency (Bristol scale), and fecal incontinence (Wexner score).
Quality of life, determined with the SF-12 questionnaire.
Endoscopic findings at 5 years from surgery: esophagitis graded with the Los Angeles scale, gastric or anastomotic ulcer, gastritis, and bile lakes.
Resolution of metabolic comorbidities at 2 and 5 years, defined as complete withdrawal of all specific treatment: type 2 diabetes, arterial hypertension, dyslipidemia, and sleep apnea.
Metabolic and nutritional deficiencies at 2 and 5 years of surgery, measured using the BARSCORE score.
Eating habits measured with the BITE questionnaire, at 2 and 5 years from surgery.
Data collection
Study data will be collected and managed using REDCap tools hosted at University of Barcelona (Clinic Hospital). REDCap is a secure, web-based software platform designed to support data capture for research studies, providing an interface for validated data capture, audit trails for tracking data manipulation and export procedures, and interoperability with external sources.
Data collection before surgery includes age, gender, initial weight and BMI, height, weight and BMI before surgery, metabolic comorbidities (diabetes, arterial hypertension, dyslipidemia, and sleep apnea), Charlson index, preoperative patient-reported data (SF-12 test for quality of life, Arts test for dumping, number of bowel movements per day, Bristol visual scale for tool consistency, Wexner test for fecal incontinence, GERDQ test for gastro-oesophageal reflux symptoms, BITE test for alimentary habits), and endoscopic findings (esophagitis graded according to Los Angeles score, gastric or anastomotic ulcers, gastritis, or bile lakes).
Data recorded for surgery and 90 days postoperatively include intraoperative complications, surgical time, intraoperative bleeding, postoperative complications (graded with the Clavien-Dindo score), ordered textbook outcome, and weight and BMI at 90 days.
During the follow-up, up to 5 years postoperatively, the following data will be collected: weight and BMI at 6 months of surgery and then yearly, metabolic comorbidities resolution at 6 months and then yearly (diabetes, arterial hypertension, dyslipidemia, and sleep apnea), patient-reported data at 2 and 5 years of surgery (SF-12, Arts, number of bowel movements per day, Bristol visual scale, Wexner test, GERDQ test, and BITE test), and endoscopic findings at 2 years of surgery (esophagitis graded according to Los Angeles score, gastric or anastomotic ulcers, gastritis, or bile lakes).
Safety monitoring and interim analysis
To ensure patient safety in, an interim analysis will be conducted after enrollment of 200 patients. If the difference in proportion of the safety primary outcome is statistically significant and clinically relevant between groups, the study will be terminated. In addition to the interim analysis, all participating researchers will be requested to meet ad hoc, if requested by any of them.
Hypothesis
The primary objectives of the trial are to demonstrate:
The superiority of SADI-S over RYGB in maintaining a higher proportion of patients with TWL > 30% at 5 years postoperatively (efficacy endpoint).
The non-inferiority of SADI-S versus RYGB in associating a comparable proportion of patients experiencing any severe complication up to 5 years after surgery (safety endpoint).
Secondary hypotheses are as follows:
SADI-S will be associated with lower incidence of dumping syndrome.
SADI-S will be associated with lower incidence of anastomotic ulcer.
SADI-S will be associated with better quality of life than RYGB.
RYGB will be associated with a lower incidence of gastro-oesophageal reflux.
The complication rates of both techniques in the immediate postoperative period (up to 90 days post-intervention) will be similar.
SADI-S will achieve higher rates of resolution of metabolic syndrome comorbidities: type 2 diabetes mellitus, hypertension, dyslipidemia, and sleep apnea.
Patients undergoing SADI-S and RYGB will require similar supplementation to compensate for nutritional deficits in the short, medium, and long term.
Baseline eating habits are related to weight loss at 2 and 5 years within each of the two patient groups.
Statistical analysis
Quantitative variables will be compared using Student’s t-test for dependent or independent variables, as appropriate, and chi-square with n degrees of freedom for qualitative variables.
Logistic, Cox, and competing event regression will be performed to assess the influence of surgical technique, changes in weight and comorbidities, and the occurrence of metabolic complications on the various patient-reported outcomes throughout follow-up.
If major differences are detected, more complicated statistical models will be used in the analyses of primary and secondary outcomes. If needed, mixed models will be used to be able to incorporate study site as a random factor in the model. The p-values less than 0.05 will be considered statistically significant.
The analyses will be based on the intention-to-treat principle (all randomized patients, except those excluded before surgery for any cause). For the primary outcome, in cases of patients lost to follow-up, missing data will be collected from hospital registries, if possible, and therefore, no imputation of missing values will be needed. For secondary outcomes, the subjects with missing data will automatically be excluded from the analyses of the variables in concern. Statistical analyses will be performed using SAS System for Windows, Version 9.4 or later (SAS Institute, Inc., Cary, NC, USA).
Ethics consideration and confidentiality
Each patient will be informed about the nature of the study and the two surgical techniques and will be given an information sheet. They must sign the informed consent form for the Hospital’s diagnostic and therapeutic procedures, as well as the specific consent form to accept inclusion in the study.
All information (personal and clinical) collected from patients undergoing bariatric surgery will be processed in accordance with regulation (EU) 2016/679 of the European Parliament and the Council of April 27, 2016, on the protection of individuals with regard to the processing of personal data and on the free movement of such data, as well as all other current and applicable laws and regulations, such as Organic Law 3/2018, of December 5, on the Protection of Personal Data and the Guarantee of Digital Rights.
Discussion
The BYPSADIS trial aims to determine whether SADI-S offers better weight control than RYGB, without increasing complications. Both efficacy and safety are co-primary endpoints in this trial, as the balance between potential benefits and risks of each surgical technique is the milestone of decision-making in clinical practice. Most of professionals treating obesity believe hypoabsortive surgical procedures such as SADI-S are more effective than RYGB; however, general reluctance in adopting them is mainly due to fear of long-term complications.12,28,29 As sample-size calculation of most surgical and medical trials is based on efficacy as the main outcome, comparison of the observed complication taxes cannot be conclusive. In the present trial, sample size was calculated separately for both co-primary endpoints, resulting in 432 patients for efficacy and 423 for safety; definitive sample-size election was 450 patients, therefore allowing the trial to be powered for both outcomes. Even though this design is not standard, it is methodologically correct, and it may help to progress in avoiding weight-centrism in scientific debate. 30 Both weight control and safety must be considered with the same importance when defining the role of a therapeutic option.
The most feared risk associated with any hypoabsortive surgery is malabsorption. Retrospective published experience suggests SADI-S allows weight and comorbidity control like hypoabsortive techniques, such as DS, with less nutritional deficiencies.15–17 Therefore, it could be reasonable to think that SADI-S behaves as an hypoabsortive technique in weight control and as a mixt technique in supplementation needs. BYPSADIS’ results will help to confirm this possibility.
BYPSADIS trial also aims to permit a better comprehension of the potential physiological benefits of pylorus preservation. RYGB is associated with some complications derived from fast gastric emptying, such as dumping, hypoglycemia, and anastomotic ulcers, which are much less frequent after SADI-S. Besides, the Roux-en-Y reconstruction is associated with a higher risk of internal hernia, a complication very rarely described after SADI-S.24–26 On the contrary, SADI-S is more likely to be associated with a higher incidence of gastro-oesophageal reflux, both acid—due to the high-pressure system of sleeve gastrectomy—and biliary–due to the omega reconstruction and the possibility of pyloric incontinence after duodenal dissection. The real incidences of all these complications are not well known, as most SADI-S are retrospective and do not include systematic objective patient-reported outcomes nor endoscopic findings. 31 BYPSADIS trial includes prospective standardized questionnaires for dumping, depositional habit, and gastroesophageal, as well as upper gastro-intestinal endoscopies. This will allow to compare the incidence of digestive symptoms and complications after SADI-S and RYGB and to determine their influence on quality of life.
There are some limitations in this study that have already been acknowledged. The main weakness is its open-label design. Being conscious of the surgical technique may influence some decisions for treatment clinicians, such as length of hospital stay or prescription of nutritional supplementations, even though a perioperative and follow-up protocol has been agreed among the six participating hospitals. Another major challenge is the difference in some surgical practices among surgeons, such as the ligation of the right gastric artery, which could potentially influence the outcomes. However, the most important technical aspects, such as limb lengths and type of anastomosis, will be homogeneous. Besides, there is potential bias arising from subjective patient-reported outcomes; in some cases, objective determination of upper-GI findings will allow us to partially mitigate this limitation.
In conclusion, the BYPSADIS trial is an open-label multicentre randomized study aiming to determine whether SADI-S is more effective than RYGB, while offering a similar safety profile, in patients living with obesity (BMI = 40–50 kg/m2). To our knowledge, it is the first trial comparing both techniques as primary surgery in a homogeneous population. Both efficacy and safety are co-primary endpoints in this trial, as the balance between potential benefits and risks of each surgical technique is the milestone of decision-making in clinical practice.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability
Individual de-identified participant data (including data dictionary), statistical code, and any other materials of this study can be obtained by request to corresponding author.