Abstract
Background and objective:
Bariatric surgery with Roux-en-Y gastric bypass (RYGB) is widely used to treat morbid obesity and present diagnostic and therapeutic challenges in patients with pancreatic and periampullary tumors. The aim of this study was to describe diagnostic tools and challenges in performing pancreatoduodenectomy (PD) on patients with altered anatomy after RYGB.
Methods:
Patients undergoing PD after RYGB from April 2015 to June 2022 at a tertiary referral center were identified. Preoperative workup, operative techniques, and outcomes were reviewed. A literature search was performed to identify articles reporting PD in post-RYGB patients.
Results:
Of a total of 788 PDs, six patients had previous RYGB. The majority were women (n = 5), and median age was 59 years. The patients most commonly presented with pain (50%) and jaundice (50%) with a median of 5.5 years after RYGB. The gastric remnant was resected in all cases, and reconstruction of the pancreatobiliary drainage was achieved using the distal part of the pre-existing pancreatobiliary limb in all patients. Median follow-up was 60 months. The Clavien–Dindo grade ⩾3 complications occurred in two patients (33.3%), and 90 days mortality occurred in one patient (16.6%). The literature search revealed 9 articles reporting a total of 122 cases, specifically addressing PD after RYGB.
Conclusions:
Reconstruction after PD in post-RYGB patients may be challenging. Resection of the gastric remnant and use of the pre-existing biliopancreatic limb may be a safe strategy, but surgeons should be prepared for other reconstruction options for creation of a new pancreatobiliary limb.
Introduction
Worldwide, the prevalence of obesity has nearly tripled during the last four decades. In 1997, the World Health Organization declared obesity as a major public health problem and a global epidemic. 1 Obesity has been shown to be associated with an increased risk of several cancers, including pancreatic cancer.2,3 In-line with the rising obesity epidemic, the application of bariatric procedures has increased and is a durable solution to both obesity and obesity-associated morbidities. 4 This leads to increasing odds of encountering such patients during a pancreatoduodenectomy (PD), which makes it imperative to understand the Roux-en-Y gastric bypass (RYGB) anatomy and anticipate challenging surgical considerations regarding resection and reconstruction. Some case reports on reconstruction options for PD after RYGB has been published, but there are no established guidelines.5,6 This study sought to analyze the optimal preoperative management and operative approaches for reconstruction after PD in postbariatric surgical patients based on a single-center experience and a narrative literature review.
Methods
The study was approved by the institutional review board at Oslo University Hospital (19/04710) according to the general guidelines provided by the regional ethics committee. Written consent for use of picture of the resected specimen in Fig. 1 was retrieved from the patient. During the time period April 2015 to June 2022, patients with PD after previous RYGB were identified from the institutional pancreatic database. Data regarding patient characteristics, prior surgery, presentation and diagnostic workup, operative management, postoperative complications, pathology, recurrence, and survival were reviewed.
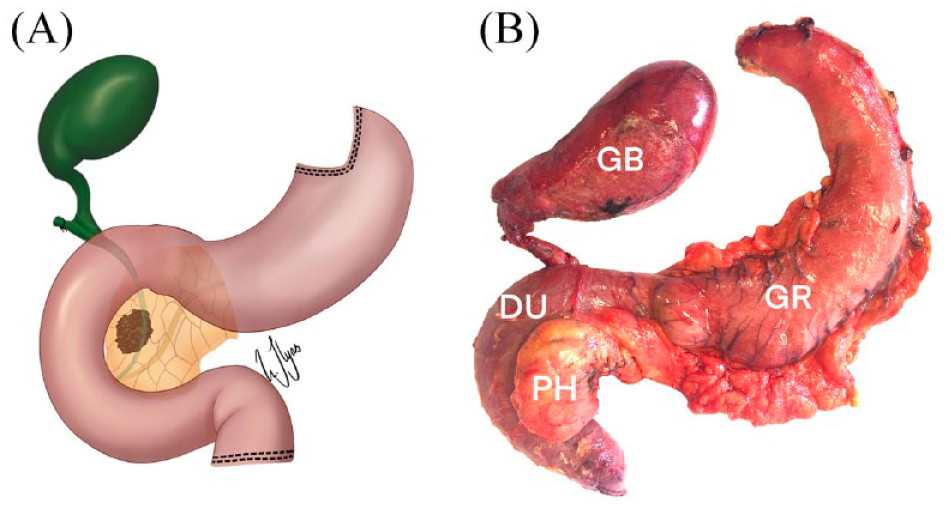
Resected specimen after pancreatoduodenectomy in a patient with prior gastric bypass. Schematic drawing of resected specimen (A) and picture of a surgical specimen (B) showing resected gastric remnant (GR), bile duct with the gall bladder (GB), duodenum (DU) and the pancreatic head (PH).
All patients with pancreatic and periampullary tumors were examined with a contrast-enhanced computed tomography (CT) of the pancreas. Due to altered anatomy in RYGB patients, endoscopic retrograde cholangiopancreatography (ERCP) was performed by balloon enteroscopy or hybrid laparoscopic transgastric ERCP if indicated in cases of obstructive jaundice. 7 If ERCP was not possible, the bile duct was drained by percutaneous transhepatic biliary drainage (PTBD) if needed.
All patients were reviewed at a multidisciplinary tumor board meeting. PD was performed with standard lymphadenectomy as previously described. 8 There were no dietary restrictions after surgery, but patients were encouraged to begin carefully and increase intake according to tolerance over postoperative day (POD) 1–4. Postoperative pancreatic fistulas (POPFs), postpancreatectomy hemorrhage, bile leaks, and delayed gastric emptying were defined and graded in accordance with the International Study Group of Pancreatic Surgery (ISGPS) definitions.9–11
Patient characteristics are presented as medians (range) (age, body mass index, time after RYGB) or counts (sex, diabetes, presenting symptoms). No statistical comparisons were performed due to the small sample size. Overall survival (OS) was calculated from the date of diagnosis to death.
A search of the English literature was conducted in PubMed/MEDLINE using the MeSH terms alone and in combination of “pancreatoduodenectomy” OR “pancreaticoduodenectomy” AND “bariatric surgery” OR “Roux-en-Y gastric bypass” OR “RYGB” OR “gastric bypass.” The search period ended on June 2022. The titles and abstracts were manually screened, and the full text was obtained for all relevant articles. The final inclusion of papers to cite and reference was made at the discretion of the authors. A formal systematic literature search according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines was not undertaken.
Results
Six patients of a total of 788 PD patients (0.76%) were operated after previous RYGB for obesity. Demographic data are presented in Table 1. Median interval between RYGB and PD was 5.5 years (range 4–10), and patients most commonly presented with pain (50%) and jaundice (50%). The operative and postoperative outcomes are summarized in Table 2. For further diagnosis, endoscopic ultrasound (EUS) was performed in one patient to obtain a fine-needle aspiration of the pancreatic mass. One patient had a significant stenosis of the celiac trunk and was treated with endovascular stent prior to PD. In all patients, the gastric remnant was resected (Fig. 1), and in five patients, the pre-existing pancreatobiliary limb was used to reconstruct the pancreatic and biliary anastomosis (Fig. 2). In one patient, the pancreatic duct was occluded with neoprene-based glue due to a high risk of pancreatic fistula, 12 and only a hepaticojejunostomy was performed using the pre-existing pancreatobiliary limb.
Patient demographics.
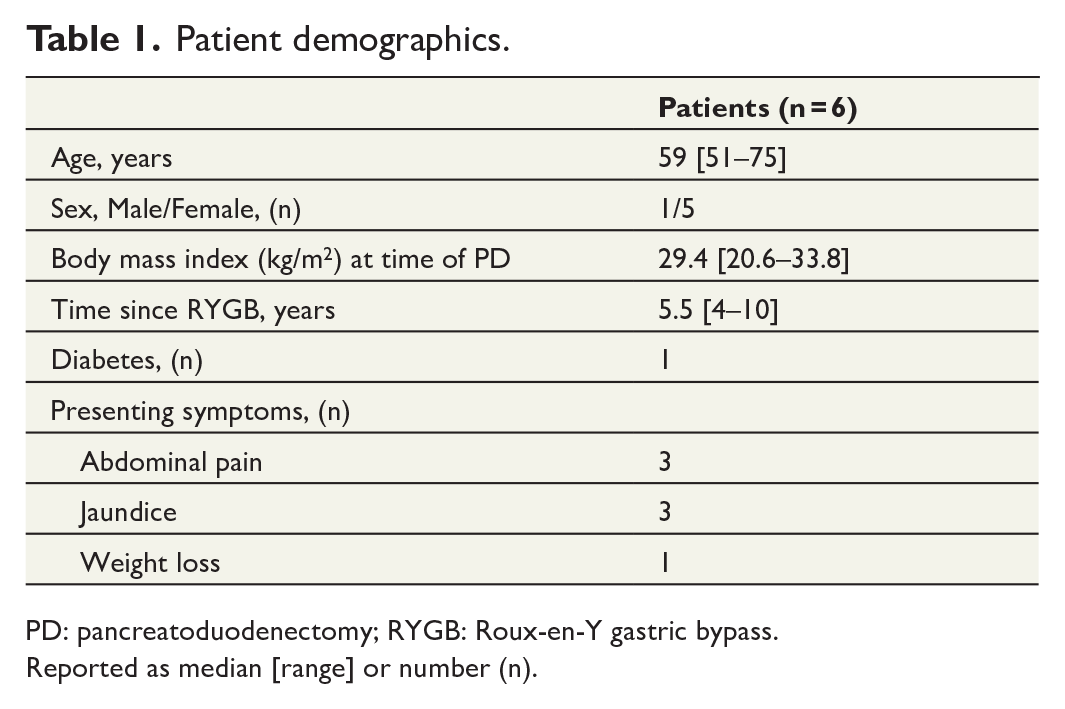
PD: pancreatoduodenectomy; RYGB: Roux-en-Y gastric bypass.
Reported as median [range] or number (n).
Perioperative details and outcome.
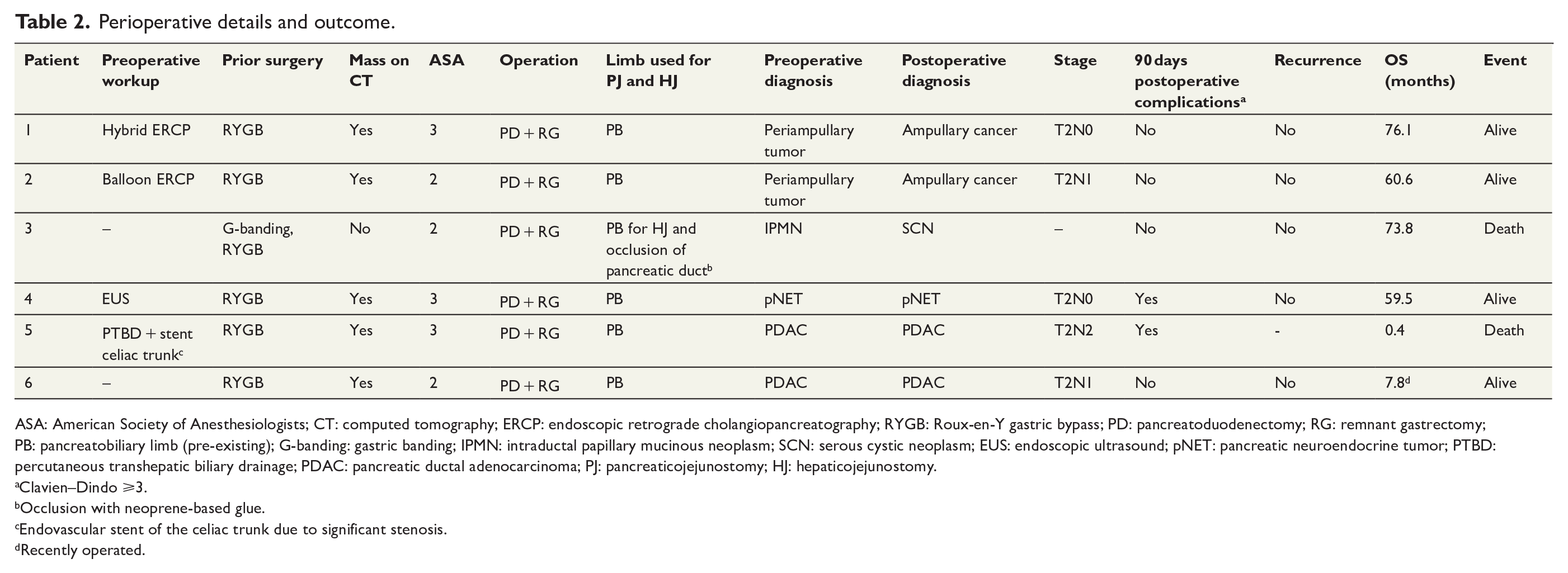
ASA: American Society of Anesthesiologists; CT: computed tomography; ERCP: endoscopic retrograde cholangiopancreatography; RYGB: Roux-en-Y gastric bypass; PD: pancreatoduodenectomy; RG: remnant gastrectomy; PB: pancreatobiliary limb (pre-existing); G-banding: gastric banding; IPMN: intraductal papillary mucinous neoplasm; SCN: serous cystic neoplasm; EUS: endoscopic ultrasound; pNET: pancreatic neuroendocrine tumor; PTBD: percutaneous transhepatic biliary drainage; PDAC: pancreatic ductal adenocarcinoma; PJ: pancreaticojejunostomy; HJ: hepaticojejunostomy.
Clavien–Dindo ⩾3.
Occlusion with neoprene-based glue.
Endovascular stent of the celiac trunk due to significant stenosis.
Recently operated.
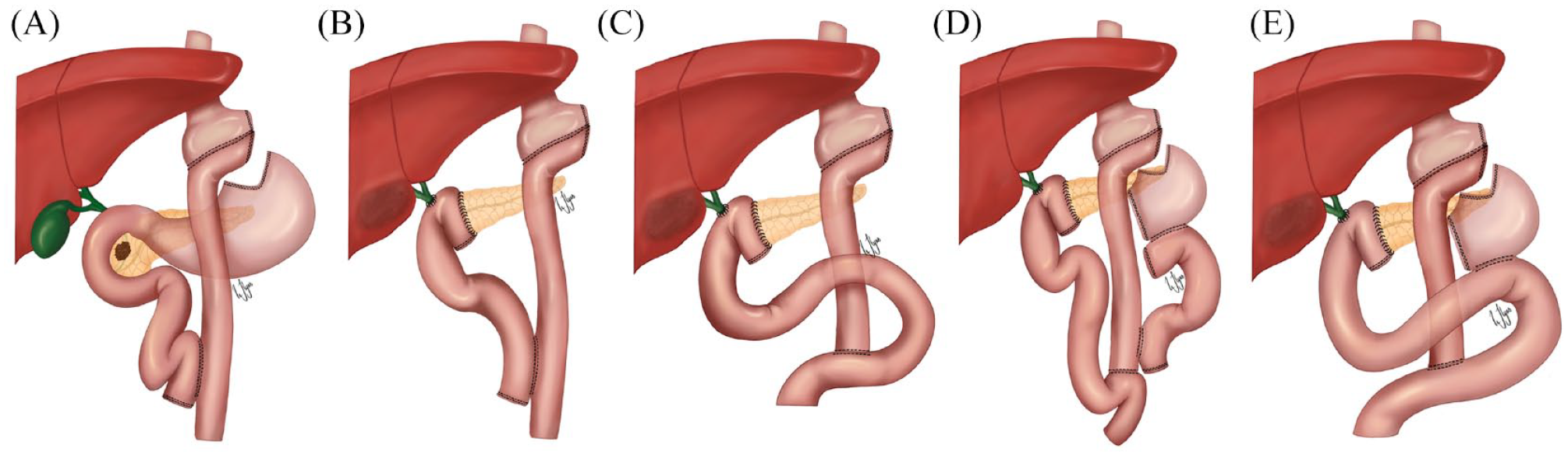
Depiction of reconstruction options after pancreatoduodenectomy (PD) in patients with prior gastric bypass. Anatomy after Roux-en-Y gastric bypass (A), and reconstruction after PD and remnant gastrectomy using the pre-existing pancreatobiliary limb for pancreatojejunostomy and hepaticojejunostomy (B). The pancreatobiliary limb can be placed retro-colic or retro-mesenteric. If the pre-existing pancreatobiliary limb is too short and has to be removed, a new Roux limb is created for pancreatojejunostomy and hepaticojejunostomy (C). If the gastric remnant cannot be removed, and part of the pre-existing pancreatobiliary limb can be saved, it may be used for drainage of the gastric remnant and a new Roux limb is created for pancreatojejunostomy and hepaticojejunostomy (D). In case where the gastric remnant cannot be removed, and the pre-existing pancreatobiliary limb is also excised, a new Roux limb is created for both pancreatojejunostomy, hepaticojejunostomy, and drainage of the gastric remnant (E).
The pre-existing pancreatobiliary limb was divided close to the ligament of Treitz and brought up into the supracolic compartment in a retro-colic (n = 5) or retro-mesenteric manner (n = 1), and a pancreaticojejunostomy and hepaticojejunostomy were performed. The previous enteroenterostomy and gastrojejunal anastomoses were left intact.
One patient developed a POPF grade C and two patients developed a POPF grade B. One patient died in hospital 12 days postoperatively from multiorgan dysfunction after developing hepatic artery thrombosis and necrosis of the liver and the stomach pouch due to preoperative high grade of atherosclerosis of the aorta and visceral arteries. This patient had a preoperative stent in the celiac trunk, but there were no evidence of stent failure (thrombosis) on CT scans. The Clavien–Dindo grade ⩾3 complications occurred in two patients (33.3%). Length of stay was 11.5 days (range = 7–24 days). Ninety-day readmission rate was none, and 90 days mortality was 16.6% (n = 1).
The literature search revealed 9 English language original articles, reporting a total of 122 cases, specifically addressing PD after RYGB (Table 3).6,13–20 The largest case series is from an international collaborative from 28 centers reporting 96 patients with previous RYGB undergoing PD. Briefly, the patients were predominantly female, in the sixth decade of life, and presented with abdominal pain, jaundice, and weight loss. The gastric remnant was resected in the majority of patients. Reconstruction of biliopancreatic drainage was achieved in the majority using the pre-existing pancreatobiliary limb. In the reporeted litterature (Table 3), twenty (16%) patients had postoperative complications are reported; eight clinically relevant POPF, three clinically relevant delayed gastric emptying, one leak from the duodenojejunostomy, and one enterocutaneous fistula. The reported 90 days mortality was 2.5%.
Published cases of pancreatoduodenectomi (PD) after Roun-en-Y gastric bypass (RYGB).
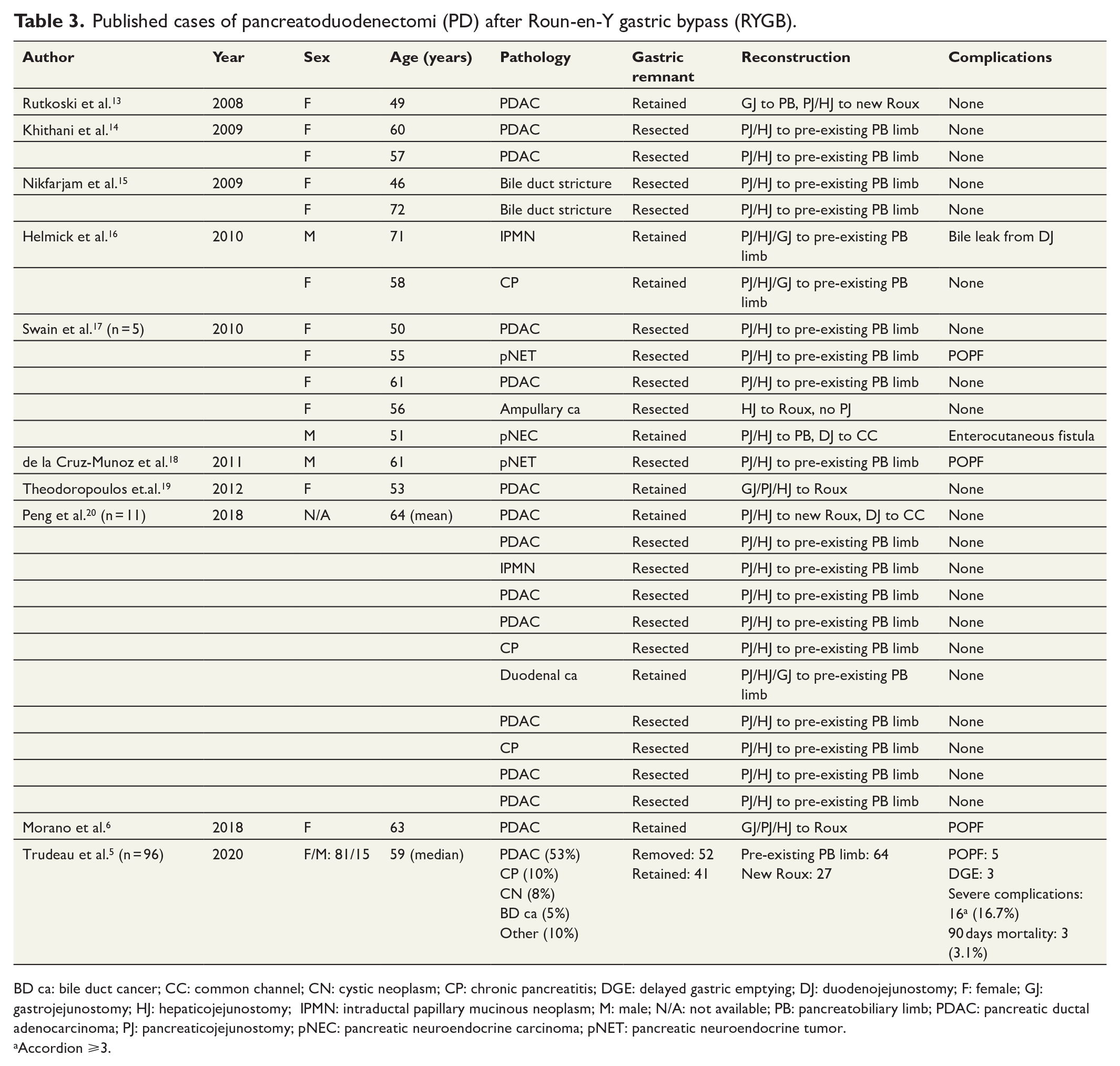
BD ca: bile duct cancer; CC: common channel; CN: cystic neoplasm; CP: chronic pancreatitis; DGE: delayed gastric emptying; DJ: duodenojejunostomy; F: female; GJ: gastrojejunostomy; HJ: hepaticojejunostomy; IPMN: intraductal papillary mucinous neoplasm; M: male; N/A: not available; PB: pancreatobiliary limb; PDAC: pancreatic ductal adenocarcinoma; PJ: pancreaticojejunostomy; pNEC: pancreatic neuroendocrine carcinoma; pNET: pancreatic neuroendocrine tumor.
Accordion ⩾3.
Discussion
Obesity is a known risk factor for pancreatic cancer.21,22 With the rising obesity epidemic and increasing number of bariatric surgeries performed, pancreatic surgeons will be encountering patients with altered anatomy. The current case series and literature review of PD after previous RYGB surgery show that resection of the gastric remnant and pancreatobiliary drainage using the pre-existing pancreatobiliary limb is feasible and can be done with an acceptable complication rate (Figs 1 and 2). Furthermore, the surgeons must have knowledge about operative reconstructions that have been used and their associated outcomes if there is insufficient length of the pre-existing pancreatobiliary limb. 6 An interesting observation is that there is a female dominance in the RYGB patients operated with PD, and the whole cohort is about 10 years younger than non-obese population described in the literature (Table 3).
The altered anatomy in RYGB patients also imposes diagnostic challenges. Although contrast-enhanced CT scan may show a pancreatic or periampullary tumor, further diagnostics is often not straight forward. Standard ERCP is not possible due to the long anatomical route, but balloon enteroscopy, as done in one of our patients, has been described to be successful. 23 An alternative method to reach the periampullary area in RYGB patients is using a hybrid approach with laparoscopically assisted transgastric ERCP. One patient in our cohort had a successful diagnosis of a periampullary tumor by this route. Both approaches are described to be safe with high rate of success in patients with altered anatomy.7,24 Another approach to the common bile duct and the ampulla after RYGB is by PTBD. 25 In the diagnostic workup of both solid and cystic pancreatic lesions, fine-needle aspiration cytology (FNAC) and/or core biopsy by EUS remain essential. After RYGB retrograde access of the afferent limb may be challenging by the use of double-balloon enteroscopy, and the EUS is often limited by length and may not be able to successfully image portions of the pancreas. 26 In these patients, percutaneous biopsy may be the only option for a diagnosis. 27 . However, one patient in our cohort had a successful EUS-guided FNAC from a pancreatic tumor. The EUS-scope was placed in the alimentary limb giving an acceptable image of the pancreas, and an FNAC was performed.
Several surgical procedures for PD in RYGB patients have previously been reported according to the literature review. A frequent discussed topic is whether the gastric remnant should be resected and how to reconstruct the biliary and pancreatic drainage.5,6 It is important that the surgeon has knowledge about the exact anatomical arrangement of the anastomoses prior to performing PD. There is a potential risk of complications with ischemia of the gastric remnant (if retained), injury to the gastric pouch or the enteric limb. Removal of the gastric remnant is favored in the literature and was also performed in all cases at our institution. Removal of the defunctionalized remnant will reduce the number of anastomoses by one, eradicate the risk of enteric leak or bleeding from this anastomosis, and remove a possible site for future malignancies. A feared complication when removing the gastric remnant is ischemia of the gastric pouch. Gastric infarction is extremely rare owing to the stomach’s extensive vascular supply. 28 The gastric pouch has the main arterial circulation from esophageal arterial branches as well as branches from the left gastric artery. When removing the gastric remnant during PD, the right gastric artery and gastroepiploic arteries are divided. However, it is important to be careful not to divide or harm left gastric artery in order to maintain sufficient blood supply to the gastric pouch. Ischemia of the gastric pouch and gastroenteric anastomosis in RYGB patient after PD has previously not been described. One patient in our cohort had severe atherosclerosis and postoperatively developed thrombosis of the hepatic artery with liver ischemia and necrosis of the gastric pouch with perforation, multiorgan dysfunction, and died 12 days postoperatively. Although the majority of surgeons prefer to remove the gastric remnant (Table 3), others have argued to retain the gastric remnant for maintaining physiologic function and make future diagnostic interventions easier.6,29 This may not be a problem if there is available service from experienced interventional radiologists and gastroenterologists performing PTBD or balloon ERCP, respectively. Removal of the gastric remnant also leads to the need of lifelong substitution of vitamin B12. 30 However, the majority of RYGB patients already take vitamin B12. Removing the gastric remnant or retaining it has not shown any differences with respect to complications, operative time, or blood loss. 5
Several methods for reconstruction after PD in RYGB patients have been described.6,31 The main four options are shown in Fig. 2 and are as follows: (1) keep the pre-existing pancreatobiliary limb right after the ligament of Treitz and creating a retro-colic or retro-mesenteric pancreatojejunostomy and hepaticojejunostomy; (2) removing the original pancreatobiliary limb and creating a new Roux limb for pancreatojejunostomy and hepaticojejunostomy; (3) retaining the gastric remnant (e.g. due to massive adhesions and risk of harming the gastric pouch) and using the original pancreatobiliary limb for drainage of the remnant while creating a new Roux limb for pancreaticojejunostomy and hepaticojejunostomy; (4) retaining the gastric remnant for reasons stated above, but in need of excising the pre-existing pancreatobiliary limb (e.g. due to very short length, tumor affection, or not possible to mobilize the limb), a new Roux limb is created for pancreaticojejunostomy, hepaticojejunostomy, and drainage of the gastric remnant. In all our patients, the gastric remnant was removed and the pre-existing pancreatobiliary limb was sufficient to make both the pancreatic and biliary anastomosis.
PD in RYGB patients is still a rare entity, and understandingly, there are no international guidelines or consensus. Based on our experience and the current literature, all patients have a diagnostic scan with CT or magnetic resonance imaging (MRI). If unresectable or borderline cancer is suspected, tissue sample is obtained by balloon ERCP, EUS or percutaneous biopsy before the initiation of chemotherapy. Upon surgical treatment with PD, we resect the gastric remnant whenever possible, and use the pre-existing pancreatobiliary limb to reconstruct the pancreatic and biliary drainage if possible.
Conclusion
With the increase in the number of bariatric procedures performed worldwide, pancreatic surgeons should be aware of the varied surgical reconstruction options for PD following RYGB.
Footnotes
Acknowledgements
The authors thank Dr Uzma Ilyas, MD, for kindly making the illustrations for this publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: S.Y. and K.J.L. received research grants from South-Eastern Norway Regional Health Authority (2020067 and 2019029).
