Abstract
Background:
The prognosis of differentiated thyroid cancer (DTC) is good, and radical surgery has been questioned, especially for small tumors. Size alone cannot, however, predict prognosis. Recurrences occur after several decades, and other risk factors should be included when deciding on an optimal treatment strategy.
Materials and Methods:
A consecutive series of 231 patients with DTC were treated in 2004–2016 at Borås Hospital in Sweden. Follow-up was performed in 2023, after a mean of 10.7 years (range = 6.5–19.1). Data on type of surgery, adjuvant radioiodine treatment, thyroglobulin levels, deaths, and recurrences were collected. Patients with negative thyroglobulin levels after treatment were compared with those who had measurable thyroglobulin or high levels of anti-thyroglobulin.
Results:
In 63/231 patients (27.3%), there was no preoperative suspicion of malignancy, 214 (92.6%) patients underwent total thyroidectomy, and the complication rate was low. There were 181 patients (84.6%) with negative thyroglobulin after treatment, and 33 patients (14.3%) with measurable thyroglobulin and/or anti-thyroglobulin levels. Fifteen patients died from thyroid cancer, and all were in the latter group. Risk factors for recurrence were tumor size, vascular invasion, extra-thyroidal growth, lymph node metastases, and male sex. Multifocality was frequent (32.5%) and patients with bi/multifocal cancers had significantly more lymph node metastases. Ten patients with recurrences had at least two of the identified risk factors.
Conclusion:
Total thyroidectomy or hemithyroidectomy in DTC is recommended, preferably including the central lymph node clearance. Total thyroidectomy has the advantage of allowing follow-up with thyroglobulin measurements to detect recurrences in time for treatment.
Introduction
The prognosis of differentiated thyroid cancer (DTC) is good. Treatment has gradually changed, from routinely performed total thyroidectomy with central lymph node clearance, to hemithyroidectomy in selected cases, or simply active surveillance, in cases with tumor diameters smaller than 10 mm.1–6 Despite accurate surveillance with ultrasonography (US), small tumors and metastases might, however, escape detection.7,8 Furthermore, patient quality of life is lowered due to fear of recurrence even years after treatment.9–11
Follow-up using the specific tumor marker thyroglobulin is useful only if total thyroidectomy is performed, 12 but only a minority of patients are followed long-term 13 even though recurrences may occur after several decades. 14 Increased thyroglobulin levels may have a benign cause but do often indicate recurrences at an early stage, and therefore it is possible to identify recurrences in time to minimize their consequence.
The aim of this study was to perform a long-term follow-up of a consecutive series of DTC patients in relation to thyroglobulin levels and risk of recurrences.
Materials and methods
Study cohort
In 2004–2016, there were 252 patients diagnosed with a thyroid malignancy at the public Borås hospital, with a catchment population of 290,000 inhabitants in the southern part of Västra Götaland County in Sweden. Out of the 252 patients, 250 underwent surgery. Patients with anaplastic cancer (n = 11), medullary cancer (n = 6), non-Hodgkin lymphoma (n = 1), and plasmacytoma in the thyroid (n = 1) were excluded, and the final DTC study cohort consisted of 231 patients with DTC.
Fine needle aspiration cytology (FNAC) of thyroid nodules was generally guided by palpation, but in four cases only seen by US and two of these were further diagnosed by cytology. The classification derived from the British Thyroid association 15 and the Bethesda system 16 was introduced in 2011. Some DTC cases were diagnosed at surgery due to various symptoms including unpleasant pressure affecting the ability to breathe or swallow, toxic goiter, or diagnosed because of signs of recurrent laryngeal nerve dysfunction. In eight cases, the tumor was discovered at surgery because of hyperparathyroidism.
To the follow-up, the patient and their families present addresses were received from the Swedish authority for National registration. If the patient was deceased, the date of death was provided. Clinical data were retrieved from medical records. If information was incomplete, the patient was contacted and asked to clarify. A second opinion of all histopathological specimens was clinical routine using the TNM classification of Malignant Tumors, the 7th edition. 17
Statistical analysis
Apart from descriptive statistics, the following potential risk factors for having measurable thyroglobulin or high levels of anti-thyroglobulin after treatment were estimated: tumor size, vascular invasion, extra-thyroidal growth, lymph node metastases, sex, and age < 45. This was performed using t-test for mean tumor size and Fisher’s exact test for categorical variables when comparing patients with negative thyroglobulin after treatment and patients with measurable thyroglobulin or high levels of anti-thyroglobulin after treatment. In addition, all these potential risk factors were included in a multivariable logistic regression model. Fisher’s exact test was also used with 2 × 2 contingency tables for investigation of consequences related to preoperative computed tomography with iodine and to bi-/multifocality.
Ethics
This study was approved by the Regional Ethical Review Board in Gothenburg in October 2018 (D759-18). Every case history in the report is described with full integrity, and 100% anonymity is preserved; identification of any of the patients is impossible.
Results
Patient and tumor characteristics
There were 175 women and 56 men with DTC (Figure 1), usually diagnosed because of symptoms such as a local swelling, lump, or discomfort. These included goiter in 87 cases, thyroid nodule in 96 cases, and in six cases a nodule that was subsequently diagnosed as a metastatic lymphatic gland. In five patients, distant metastases were the first sign. Mean age at surgery for DTC was 56.3 years (SD = 18.9; median = 58.8; range 16.7-91.8).
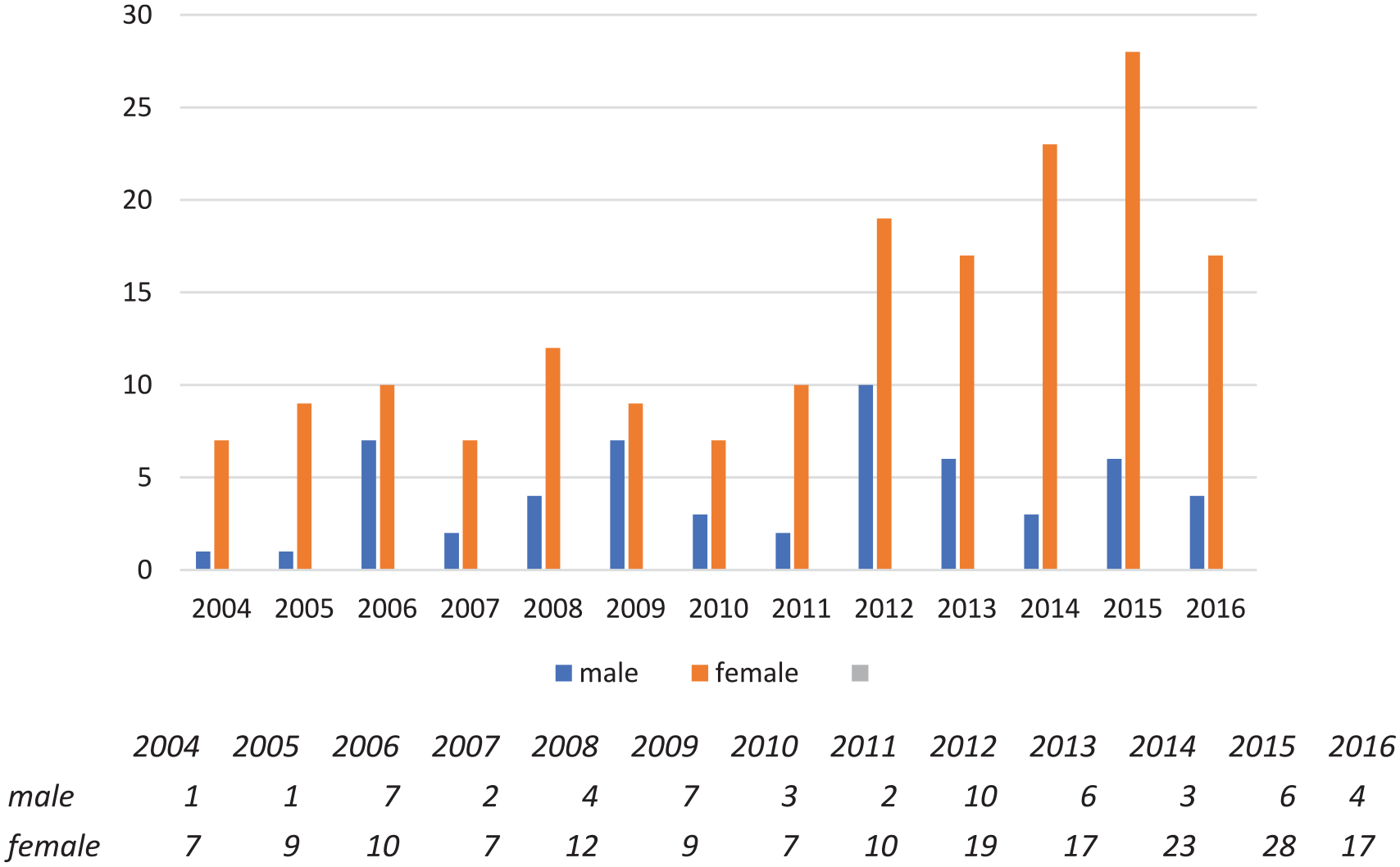
A consecutive series of 231 patients, 175 women and 56 men, treated from 2004 to August 2016 for differentiated thyroid cancer (DTC). Overall, 186 patients with papillary cancer, 98 with the follicular variant of papillary cancer, 25 with follicular cancer, and 20 with oncocytic carcinomas.
The Bethesda system 16 was applied to those with FNAC available by retrospective review, and the division was BII (n = 43), BIII (n = 17), BIV (n = 67), BV (n = 21), and BVI (n = 48), respectively. The indication for surgery was motivated in 153 cases, that is, BIII-BVI (66.2%). In 63/231 cases (27.3%), there was no preoperative suspicion of malignancy and included intrathoracic goiters unsuitable for FNAC, large thyroid glands (n = 10, mean = 200 g; range = 124–259). In some, the enlarged thyroid gland was found at computer tomography (CT), with three cases where the trachea was compressed to 4-8 mm. In four cases the tumor penetrated larynx or the tracheal wall. There were 42 patients with small tumors, out of whom 17 had toxic goiters with tumor diameters 2-8 mm (mean = 5.2). Bi- or multifocality occurred in 75/231 cases (32.5%).
Thyroid surgery
Altogether 139 patients (60.2%) with DTC were primarily operated with removal of most thyroid tissue. Total thyroidectomy was performed in 130 patients (56.3%), one patient with toxic goiter had a subtotal thyroidectomy plus radioiodine (RI), and eight patients had previously undergone a hemithyroidectomy. In 92 patients, a hemithyroidectomy was primarily performed. In 75 of 92, the remaining thyroid lobe was removed when the cancer diagnosis was confirmed. Thus, at the end, 214 patients had all thyroid tissue removed and 17 were operated by hemithyroidectomy only (7.4%). Fifteen of these patients had papillary T1-tumors, 14 with diameters of 0.5–6 mm, and one with 10 mm. There were two T3 tumors—one papillary 6 mm tumor and one oncocytic 52 mm tumor. The 91-year-old lady in the latter case wanted as little treatment as possible. She lived 10 years after that. All 17 had benign US performed 1 year after surgery.
Lymph node staging
A modified central neck dissection was performed when there were signs of malignancy, that is, the tissue adjacent to the thyroid was removed en bloc with the thyroid gland. Sometimes, lateral nodes on the tumor side were included, but in 11 cases at a later operation.
In 43 patients, ⩾15 glands were investigated (mean = 23.3; SD = 7.9), and 29/43 had metastases (67.4%). For reliable TNM classification, at least six lymph nodes should be examined, 17 which was achieved in 163/231 patients (70.6%). Of the remaining 68 patients this was not achieved/performed, 35 had microcarcinomas, 21 had Bethesda II lesions, eight had toxic goiter, and eight had follicular carcinomas.
The number of lymph nodes examined in the 214 patients with all thyroid tissue removed was 0–54. No glands in 14 patients, out of whom four with toxic goiter, six with BII lesions, and in four cases, the trachea was compromised by tumor growth. The mean number of lymph nodes examined in the remaining 200 patients was 10.9 (SD = 8.32), and out of these, 63/200 (31.5%) had metastases. In 159/200 cases with ⩾6 glands examined, 13 a total of 59 had metastases (37.1%). Among the 17 patients only undergoing a hemithyroidectomy, in 13 out of 17 cases, 1–16 lymph nodes were investigated (mean = 5.7; SD = 4.8), all without metastases.
Postoperative complications
There was one reoperation due to bleeding and one due to leakage from the thoracic duct. Two patients had temporary recurrent laryngeal nerve dysfunction after surgery. Seven patients had recurrent laryngeal nerve paresis before surgery, six of them because of tumor growth, and one recovered within one year after surgery, as the tumor was small and within the large thyroid gland weighing 165 g. Seven patients had permanent postoperative hypoparathyroidism with adjusted medication with calcium and vitamin D (7/214; 3.27%). 18
Microcarcinomas
There were 79/231 patients (34.2%) with tumor size ⩽10 mm. One was follicular cancer; the others were papillary cancers, with 25 being of the follicular variant of papillary cancer. Lymph nodes were examined in 70/79, and 18 patients had metastases (25.7%). For comparison with patients with tumor diameter > 10 mm (n = 150), there were 143 with examined lymph nodes, out of whom 46 had metastases (32.2%). The difference was not statistically significant (p = 0.43).
Among the 79 patients with microcarcinomas, five had vascular invasion (6.3%), six had extra-thyroidal growth (7.6%), and four had ⩾5 metastatic nodes (5.6%). Multifocality occurred in 26/79 cases (36.7%), and 24/26 had investigated lymphatic glands, out of whom 11 had metastases (45.8%).
Bi- or multifocal cancer
Patients with bi- or multifocal DTC (n = 65) were compared with patients with unifocal cancer tumor in the whole cohort. The shares of patients with lymph node metastases out of the numbers who had any nodes examined (n = 60) were calculated. Overall, 26/60 (43.3%) with bi- or multifocal DTC had metastases compared to those with unifocal cancers (38/153; 24.8%) (p = 0.012). The difference was also significant when looking only at microcarcinomas (p = 0.006).
Adjuvant treatment
In 2004–2010, adjuvant radioactive iodine treatment (RI) with 4000 MBq was given, while 2000 MBq was used to eliminate minimal remnants of thyroid tissue in low-risk patients. From 2011, lower doses were used, 3700 MBq and 1100 MBq, respectively. Patients with distant metastases got 8000 or 7400 MBq. Adjuvant radioactive treatment after surgery19,20 was given in 160/214 cases (74.8%). In 46 cases, treatment was delayed by a few months due to previous X-ray with iodine contrast administration. There were 54 patients without adjuvant radioactive iodine treatment, all without signs of metastases: 44 with stage T1 tumors, six with stage T2, and four with T3 tumors.
Thyroxine supplementation
Thyroxine supplementation is a consequence after surgery, and the dose is determined in relation to the TSH level. In this series, TSH was kept as low as possible, but within the normal range to avoid negative cardiovascular effects 21 with only occasional exceptions.
Thyreoglobulin levels after surgery
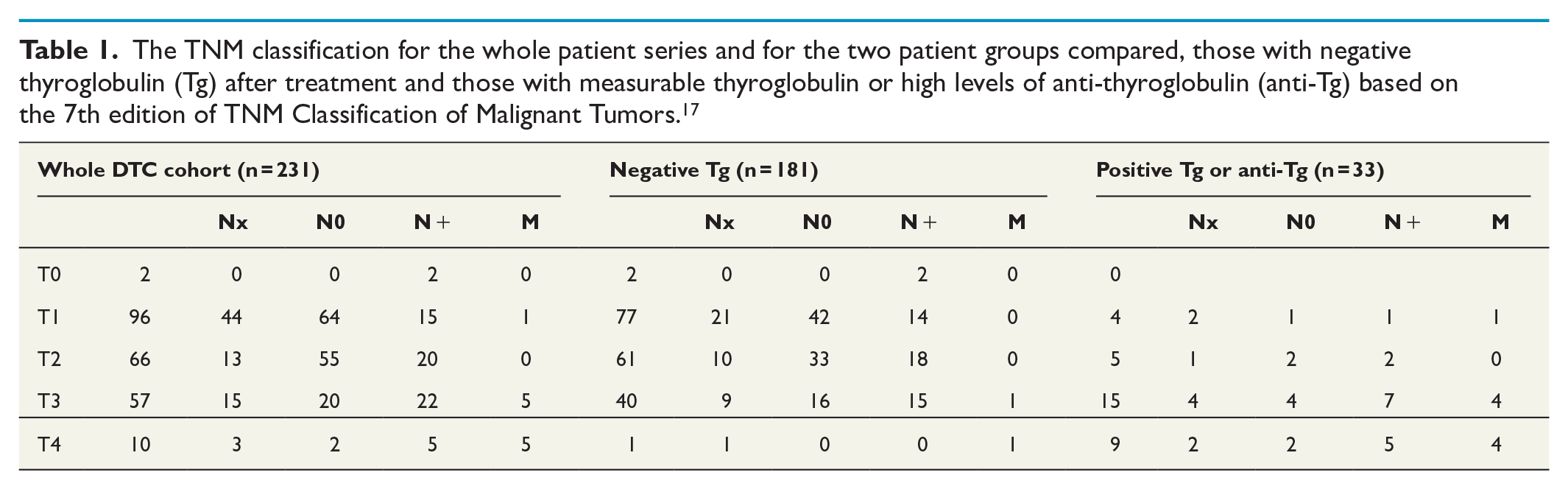
After treatment, surgery with or without adjuvant RI treatment, thyroglobulin was negative in 181/214 cases (84.6%), and 33 patients had measurable thyroglobulin levels, or high anti-thyroglobulin levels (n = 4). The characteristics of these groups are shown in Tables 1 and 2. All fifteen patients that died from DTC were in the group with measurable thyroglobulin levels or high anti-thyroglobulin levels.
The TNM classification for the whole patient series and for the two patient groups compared, those with negative thyroglobulin (Tg) after treatment and those with measurable thyroglobulin or high levels of anti-thyroglobulin (anti-Tg) based on the 7th edition of TNM Classification of Malignant Tumors. 17
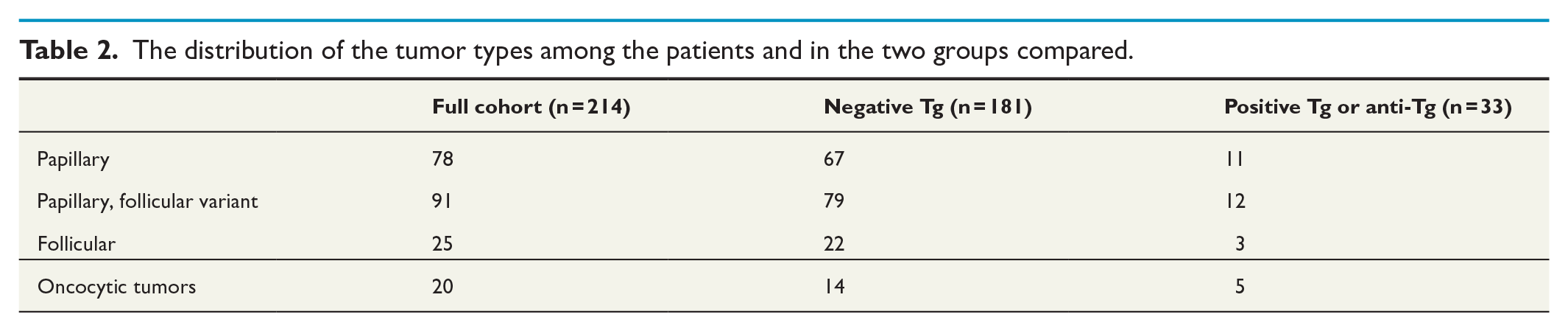
The distribution of the tumor types among the patients and in the two groups compared.
Pulmonary metastases occurred in 12 patients with thyroglobulin levels of 70–2200 µg/L and skeletal metastases in 11 patients with levels of 1200–55,000 µg/L. Some of the 33 patients had only slightly elevated thyroglobulin levels without a serious cause. One patient got her adjuvant RI treatment 6 years after surgery, as her thyroglobulin was continuously measurable at a low level, after which it has remained negative for 14 years. In one case, thyroglobulin declined slowly during 6 years from 2.2 to <0.1 µg/L after surgery + RI given at 1100 MBq, upon removal of a goiter weighing 252 g with a bifocal tumor without metastases in 15 lymphatic glands. Another patient had thyroglobulin levels of 28 µg/L after surgery and RI in 2015, that turned negative eight months later, after removal of her right ovary containing a teratoma with thyroid tissue. Two patients without RI had low thyroglobulin levels of 0.3 and 1.5 µg/L at 7 and 9 years, respectively, due to apparent aberrant benign thyroid tissue. One was located at the root of the tongue according to scintigraphy, and the other was a suspicious lymph node with the finding of an island of benign thyroid tissue instead.
In many cases measurable thyroglobulin or high levels of anti-thyroglobulin after treatment was a sign of an advanced stage. Mean tumor diameter in this group of 33 patients was 41.6 mm, and vascular invasion, extra-thyroidal growth, and metastatic lymphatic glands were frequent. The distribution of these potential risk factors with significant p-values is shown in Table 3, and also included male sex. There was a borderline significance for just one metastatic lymph node, with statistical testing performed excluding the 14 patients without investigated lymph nodes.
Risk factors among patients with negative thyroglobulin after treatment (n = 181) and among patients with measurable thyroglobulin or high levels of anti-thyroglobulin after treatment (n = 33).
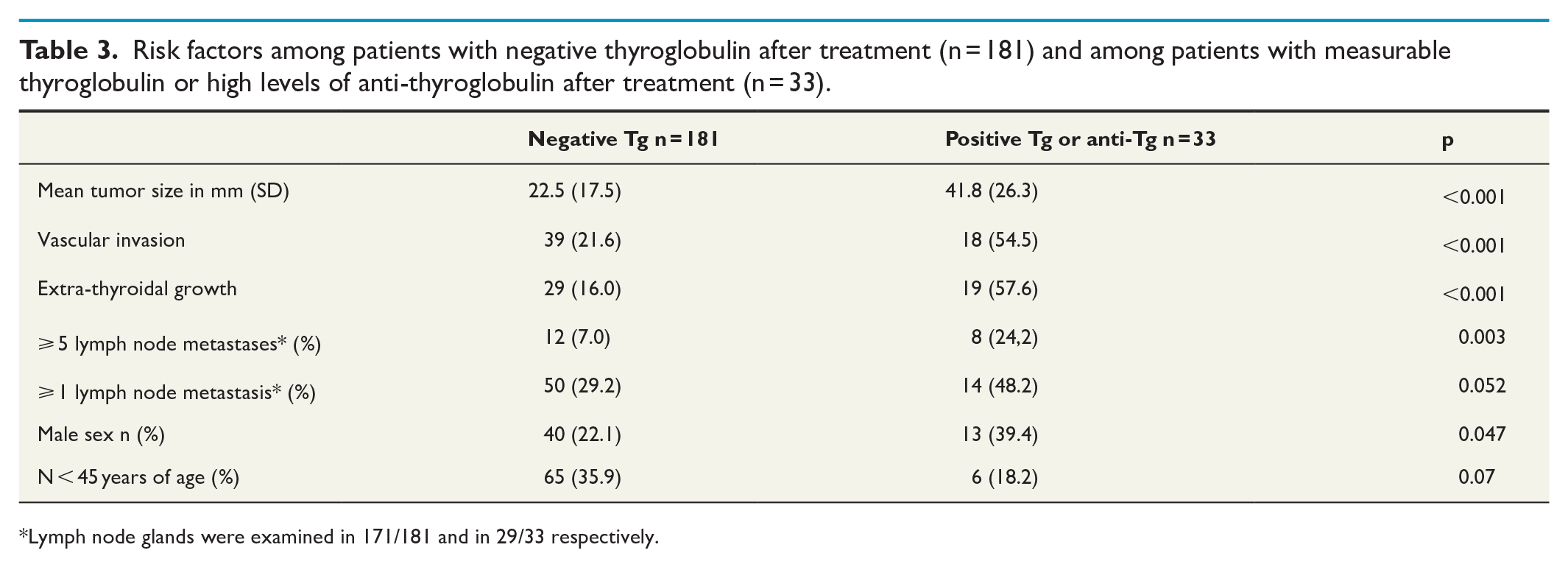
Lymph node glands were examined in 171/181 and in 29/33 respectively.
The multivariable logistic regression model showed only tumor size and extra-thyroidal growth as statistically significant independent risk factors for having measurable thyroglobulin or high levels of anti-thyroglobulin after treatment.
Effect of preoperative CT with iodine contrast
There were 29 patients with increasing thyroglobulin or anti-thyroglobulin levels at follow-up. Some of them had primarily had negative thyroglobulin that became measurable. To find out if the previous iodine administration contributed to this development, 15 patients who had had a CT with iodine contrast before surgery were compared with the 14 patients without previous iodine administration. Only patients still alive were compared, leading to exclusion of five patients with delayed RI and eight who received RI soon after surgery. Thus, 15/41 (46 patients with exclusion of 5 =41) (36.6%) were compared with 14/106 (114 patients with exclusion of 8 = 106) (13.2%). The difference is significant (p = 0.002) indicating a clearly worse situation after previous iodine contrast administration.
Survival and thyroglobulin levels
Altogether 62/231 had died at follow-up in 2023. Mean age at death was 82.0 years (SD = 9.8; range = 60.7–101.7) for women and 80.1 years (SD = 11.2; range = 56.8–99.4) for men. The average length of life in Sweden in 2021 was 84.8 years for women and 81.2 years for men. Mean follow-up time of the deceased patients was 6.4 years (SD = 4.2; range = 0.3–15.9), and of the patients still alive at follow-up of 10.7 years (SD = 3.7; range = 6.5–19.1).
Fifteen patients died because of their DTC, eight women and seven men (15/231; 6.5%). Mean age at death was 81.0 years (SD = 10.0; range = 74.2100) for women and 74.3 years (SD = 12.2; range = 56.8–89.0) for men. At the time of diagnosis, five patients had locally advanced disease, four had pulmonary metastases, six had skeletal metastases, and one patient had symptoms from a cerebral metastasis diagnosed during follow-up after her thyroidectomy. Mean follow-up time of these patients was 4.6 years (SD = 2.8; range = 0.3–11.0), but two of them did not even manage through the radioactive treatment.
Eight patients died from other causes but with measurable thyroglobulin levels: 1.5–2.2 µg/L (n = 3), 7.1–22 µg/L (n = 3), and 200 µg/L (n = 1), and one patient had anti-thyroglobulin of 378 kU/L. Five of the 17 patients who had a hemithyroidectomy had died at mean 77.8 years of age (range = 61–101) due to other causes than thyroid cancer.
Recurrences and thyroglobulin levels
In 12/54 patients (22.2%) without adjuvant RI treatment, thyroglobulin became measurable at low levels, a mean of 0.42 µg/L (range = 0.2–0.7), after a mean of 68.8 months (range = 3–133). In 23/160 patients (14.4%) who received adjuvant RI treatment, thyroglobulin also became measurable, at a mean of 0.60 µg/L (range = 0.2–2.6), after a mean of 60.7 months (range = 11–175). Four cases with high anti-thyroglobulin had increasing levels, but a few patients with primarily elevated levels of anti-thyroglobulin had declining levels during the first years, and follow-up with thyroglobulin was used.
Recurrences occurred in 10 patients (10/214; 4.7%) after a mean of 5.7 years (SD = 4.2; range = 1.1–13.6). Details on these 10 patients (one T1, four T2 and five T3 tumors), are shown in Supplemental Tables 1 and 2. In eight patients, the primary sign of recurrence was a metastatic lymph node in the lateral compartment. In one case a jugular node was not found until after pulmonary metastases occurred, and another patient died before the source of thyroglobulin increase was found.
The thyroglobulin level at which recurrence occurred was a mean of 2.7 µg/L (range = 0.3–7.1). In three cases, a second recurrence occurred later. All had adjuvant RI treatment after surgery, and all had primarily minimum two of the five risk factors. Another two patients with thyroglobulin of 0.3 and 0.7 µg/L were diagnosed after more than 8 years with small but suspicious lymph nodes at US. They have, however, declined biopsy. It is also possible that there might have been a few more unlocalized recurrences of asymptomatic nature among the patients described above with measurable thyroglobulin who died from other causes.
Only selected cases were followed for 10 years or more. After the patients became out-patients without hospital connection, thyroglobulin determinations were not performed at all. In the most recent follow-up after a mean of 10 years, 7/12 patients without adjuvant RI treatment had mean thyroglobulin of 0.3 µg/L (range = 0.1–4.4), and after a mean of 9 years, 10/23 with RI treatment had mean thyroglobulin of 2.06 µg/L (range = 0.2–10.0), one had 111 µg/L, and 2 had anti-thyroglobulin of 4000 kU/L.
Discussion
This consecutive series of patients treated for DTC draws an opposite picture to several presentations on over-diagnosis and overtreatment.1–6,22–24 DTC is not an aggressive cancer in comparison with many other cancer forms, but clearly, early treatment is preferable to late treatment. The impression is that a single complete and precise operation is better for the patient, than if further operations are needed, which is the purpose of a central compartment lymph node clearance at the first operation. If not, the patients will need ultrasound evaluations at different intervals, leading to varying degrees of anxiety and insecurity. Metastases <3–4 mm or intra-thyroidal tumor foci with diameters of 1–4 mm, that were found in more than 30% of the cases, can furthermore not be identified with certainty by ultrasound.7,8 After a total thyroidectomy with a negative thyroglobulin that would not be a problem, but the frequent occurrence of metastatic lymph nodes found in this series suggests that including these nodes is justified.
The second possibility, a hemithyroidectomy including removal of the central lymph nodes unilaterally, gives reliable data on the tumor characteristics and whether there is spread to the closest lymphatic compartment. Risk factors will be identified, and in the future, further prognostication will be possible with gene mutation mapping. 24 A hemithyroidectomy has the disadvantage of necessitating follow-up without thyroglobulin determinations. These measurements give valuable information that is important for both patients and surgeons.12,25 Moreover, during the follow-up performed for this cohort, five patients expressed uneasiness about the lack of specialized medical check-ups. Complications after surgery would be half as frequent, but the complication rate after bilateral surgery in this series can be regarded as low, 21 considering the advantage of as many as 85% becoming thyroglobulin negative.
The third possibility, active surveillance, concerns micro-carcinomas. It is obvious that important risk factors cannot be revealed with ultrasound alone nor with FNAC. For this reason, together with the small, but not negligible, numbers of findings of vascular invasion, extra-thyroidal growth, metastatic lymphatic glands, and multifocality, the strategy of active surveillance appears uncertain. The present observations contradict some descriptions in the literature of microcarcinomas as generally harmless.22,23
Follow-up using thyroglobulin appears to be a reliable marker despite its variability. As observed here, thyroglobulin levels may become measurable due to different reasons, and some are not malignant. There appears not to be a clear limit for the thyroglobulin level at which extended investigations are called for, maybe in part a consequence of the level being related to the TSH level. All thyroglobulin measurements given here are basic levels. Thyrogen-stimulated thyroglobulin determinations 2 were also performed, but not during the whole study period. A negative basic thyroglobulin level means safety all the same. A low level that is stable may be safe as well but is no guarantee that tumor growth will not take place. There is always a risk of recurrence.26–30 In this series, only two independent risk factors, tumor size and extra-thyroidal growth, were found, and obviously other factors correlate with them.
It is worth noticing that some patients had stationary skeletal metastases and/or pulmonary metastases for many years or with only slow long-term progress. The slow DTC development may be the reason for late recurrences occurring after two or three decades.1,14 Currently, thyroglobulin tests are in many cases not performed for more than a few years after treatment. The patients’ fear of recurrence does not disappear at this stage. In clinical practice, it would be reasonable to follow more patients long-term with basic thyroglobulin determinations every 1–2 years. It could easily be coordinated with the yearly blood sampling to adjust thyroxine medication.
Sweden has national guidelines for the treatment of thyroid cancer where the treatments mentioned above have been accepted since 2017, 13 although active surveillance was not included until 2025. Still, individual decisions concerning both surgery and follow-up must be made in each case. The recommendations for follow-up depend upon TNM-status and vary between less than 1 year, 2 years, 5 years, and 10 years. This suggested improvement with longer follow-up with thyroglobulin tests for out-patients has three purposes: to find recurrences in time for treatment with satisfactory results, reduce costs of extra-thyroglobulin testing by coordinating with the check-ups for thyroxine medication, and reduce patient anxiety, as thyroglobulin in most cases is a reliable and easy to interpret tool.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969251356064 – Supplemental material for Recurrence after treatment for differentiated thyroid cancer: Observations on risk factors, long-term follow-up and treatments
Supplemental material, sj-docx-1-sjs-10.1177_14574969251356064 for Recurrence after treatment for differentiated thyroid cancer: Observations on risk factors, long-term follow-up and treatments by Gunilla M Hedbäck in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
Lars Barregard, MD, PhD, Professor Emeritus, who assisted with the statistics. Roman Krenz, MD, PhD, Department of Pathology, Sahlgrenska University Hospital. Andreas Hallqvist, MD, PhD, Department of Oncology, Sahlgrenska University Hospital. Ewa Lundgren, Associate Professor, Department of Surgical Sciences, Uppsala University, Uppsala, Sweden, Lars Rostgård-Christensen, Senior Radiologist, Lidköping Hospital, Sweden for supporting this study and advice.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
