Abstract
Background and aims:
The Fecal Incontinence Quality of Life Scale (FIQL) is a health-related questionnaire that measures the quality of life among patients with fecal incontinence. This questionnaire comprises 29 items divided into four subscales: Lifestyle, Coping/Behavior, Depression/Self-Perception, and Embarrassment. The aim of the study was to validate the FIQL questionnaire in the Finnish language.
Methods:
A multiphase validation method was used consisting of a two-panel translation process followed by a psychometric evaluation process. The validation process was used to ensure the consistency and quality of the questionnaire through the test–retest process. The internal consistency between the four subscales was measured with Cronbach’s alpha, and the stability over time was assessed by intra-class correlation coefficient (ICC) analysis. Spearman’s correlations were used to examine the construct validity.
Results:
A total of 82 patients with fecal incontinence and 10 reference participants were included in this study. The results for Finnish FIQL indicated that all four subscales had good internal consistency, except for the embarrassment subscale, which had a slightly lower Cronbach’s alpha value for the test (0.68). Between subscales, ICC ranged from 0.83 to 0.90, indicating almost perfect stability over time. Correlations were moderate between the FIQL and the severity of fecal incontinence.
Conclusions:
The Finnish version of the FIQL has been successfully validated and shown to be feasible. The questionnaire can be used for both research and clinical purposes in Finnish patients with fecal incontinence.
Context and Relevance
The Fecal Incontinence Quality of Life Scale (FIQL) is a validated quality outcome measure for patients with fecal incontinence. The International Consultation on Incontinence Committee has given a grade A recommendation (highly recommended) for the use of the FIQL instrument for the evaluation of the health-related quality of life impact. The Finnish version of the FIQL questionnaire is reliable, valid, and feasible for evaluating the quality of life among Finnish patients with fecal incontinence. It can be used in both research and clinical practice.
Introduction
Fecal incontinence (FI) is defined as the involuntary loss of solid or liquid feces. It is a physically and psychosocially debilitating condition known to have a major negative impact on quality of life (QOL). 1 Diagnosis and management are often delayed due to several reasons, such as patients’ reluctance to report symptoms and seek care. 2 Therefore, physicians must proactively evaluate their patients for symptoms specific to incontinence. One of the goals in the evaluation of patients with FI is to ascertain symptom severity and its consequences for patient QOL. An accurate and objective symptom and QOL assessment is important for diagnosing FI, clinical decision-making, and evaluating the efficacy of treatment. The patients and physicians may rank the severity of FI differently and emphasize different aspects of the symptom bother. Patient-reported outcomes (PROs) measured by symptom severity and QOL questionnaires represent the patients’ perspectives. Furthermore, a structured questionnaire provides a communication tool that helps patients bring forth their highly personal problems and allows physicians to capture relevant information that may not otherwise be shared in a traditional doctor’s visit. The current literature recommends the use of condition-specific health-related QOL questionnaires, rather than general ones, to quantify the impact of FI. 3
There is much emphasis on using standardized and validated instruments both in clinical settings and in research. The use of a validated instrument increases the certainty with which the instrument accurately reflects its intended measures. Appropriate translations and cultural adaptations are needed for an instrument to be valid in new temporal, cultural, and linguistic contexts. 4
The FIQL by Rockwood et al. 5 is a validated quality outcome measure in patients with FI. The International Consultation on Incontinence Committee has given a grade A recommendation (highly recommended) for the use of the FIQL instrument for the evaluation of health-related QOL impact. 6 The FIQL has already been successfully translated and adapted to several languages, including French, 7 Spanish, 8 Portuguese, 9 Norwegian, 10 Chinese, 11 and Japanese. 12 The psychometric analyses in these studies have indicated generally high internal consistency in three out of four subscales (lifestyle, coping/behavior, and depression/self-perception) and slightly lower for the fourth subscale (embarrassment). The FIQL proved to be highly stable over time and was associated with good responsiveness in these studies.10,13 Convergent validity has been evaluated earlier concerning symptom severity measures by fecal incontinence severity index (FISI) and Wexner scores. 14 In our institution, the Cleveland Clinic Fecal Incontinence Severity Scoring System (CCIS), often termed the Wexner score, has been used as a symptom severity measure in patients with FI. 14
This study aimed to translate and examine the psychometric properties and thereby validate the Finnish version of the FIQL.
Methods
Patients
The target population for the questionnaire is patients with FI. The participants in this study were recruited from four proctology outpatient clinics in Finland, two University Hospitals (Oulu and Helsinki) and two Central Hospitals (Jyväskylä and Seinäjoki). We followed the quality criteria recommending at least 50 participants. 15 A total of 82 patients over 18 years of age were included in this study. Ten individuals presenting upper gastrointestinal symptoms were recruited for control subjects.
The patients were asked to fill out the questionnaires twice, the first time at the outpatient clinic and the second time at home 2 weeks after the first; the controls filled the questionnaire once. Pre-stamped envelopes and the questionnaires were given to patients at the clinic to be returned by mail. When filling out the FIQL, the patients were asked to simultaneously fill out the symptom-specific questionnaire, Wexner score, and the obstructed defecation syndrome score (ODS).
Questionnaires
FIQL is a health-related questionnaire that measures the QOL among patients with FI. This questionnaire comprises 29 items divided into four subscales: Lifestyle (10 items), Coping/Behavior (9 items), Depression/Self-Perception (7 items), and Embarrassment (3 items). All items are rated on a scale from 1 to 4 points, except two items on the depression/self-perception subscale rated with 1 to 5 and 1 to 6 points. Scale scores are the mean response to all items on the scale with a 1 indicating a lower functional status of QOL, that is, higher scores indicate a better QOL.
Questions Q2 (13 items) and Q3 (14 items) included the response “Not Applicable” which we coded as a missing value in the analysis for all questions as indicated in the original paper by Rockwood. In validation of the Finnish FIQL, the equation used to calculate the score for the coping/behavior subscale was corrected, using Q3c instead of Q3d, which probably was a typographical error in the original paper. 16
The Wexner score is a severity rating score for FI. 14 It combines the five items regarding frequency and type (solid and liquid stool, gas) of stool loss, use of pads, and alteration in lifestyle. The frequency options are never, less than once a month (rarely), monthly but less than once a week (sometimes), weekly but less than once a day (usually), and daily (always) (range = 0–4). Higher scores indicate more severe FI. The ODS score is a validated score ranging from 0 to 40 and assesses symptoms of obstructed defecation. 17
Translation
The translation work is traditionally carried out using the forward translation and the back-translation methods. There is also good evidence of translating the questionnaire into the target language using the two-panel method, where attention is paid to the high-quality language and cultural adaptation process of the questionnaire instead of exact translation. The questionnaire translation process includes cultural adaptation to the target language. 18 In addition, items in the questionnaire should reflect the areas that are important to the target population using the questionnaire. 15 These perspectives were taken into consideration in the translation process.
In our validation, the translation process was carried out using a two-panel approach. The first panel translated the questionnaire from English to Finnish. This panel consisted of four Finnish- and English-speaking specialist doctors (three gastrointestinal surgeons and one anesthetist) familiar with the research topic. One had native English skills and good Finnish skills, and three others had native Finnish skills and good English skills. The second panel was the lay panel, consisting of five persons (three women, two men, aged 34–91 years) with various educational backgrounds. The lay panel assessed only the translated version of the questionnaire as recommended 18 by writing down their observations on the comprehensibility of the questionnaire and forming a group discussion about the translation. Finally, together with the first panel, one other gastrointestinal surgeon observed the translation accounting for the lay panel’s comments and formed the final version of the translation.
In the translation process, minor cultural adaptations to the original Rockwood questionnaire were authored. For example, in the original paper, proposition Q2d was written as “It is difficult for me to get out and do things like going to a movie or to church.” However, in Finland, it is less common to go to church, so we decided to translate the proposition as “It is difficult for me to get out, like going shopping, to a movie or theatre.”
Measurement properties
The translated symptom questionnaire was evaluated psychometrically by ensuring the consistency and quality of the form through the test–retest process. It is recommended to carry out the process with the target population, which consists of the people for whom the questionnaire was developed. 15 The subscales of FIQL were tested for internal consistency, test–retest reliability, construct validity, and floor and ceiling effects.
Internal consistency measures the homogeneity of the items. 15 The internal consistency between the four subscales was measured with Cronbach’s alpha. Alpha values below 0.7 indicate too high heterogeneity and lack of correlation between the items in the subscales, while values above 0.90 indicate too high similarity between items. 19 Test–retest reliability analysis was measured with the intra-class correlation coefficient (ICC), where the values between 0.81 and 1.0 indicate almost perfect agreement and the values 0.61–0.80 indicate substantial agreement. 20
The construct validity of the questionnaire was assessed on its convergent and discriminant validity. The convergent validity was analyzed by evaluating the correlation between the four subscales and Wexner scores. Discriminant validity was examined by correlating the four subscales to the ODS score. Correlations between 0.36 and 0.67 are considered moderate, from 0.68 to 1.0 strong, with 0.9 or above very high correlations. 21 Comparison to the control group was used to assess the disease specificity of the questionnaire. In addition, ceiling and floor effects were calculated and considered to be present if 15% of recruited patients achieved the lowest or highest possible score. 15
Statistical analyses
Internal consistency analysis was performed by Cronbach’s alpha coefficients. The test–retest analysis for reliability and reproducibility was assessed by using an ICC. The analysis of the convergent and discriminant validity of the FIQL concerning the Wexner score and ODS score was completed using Spearman’s correlations. Between-group comparisons were performed by using the Student’s t-test. In addition, ceiling and floor effects were calculated. A two-sided p-value of <0.05 was considered significant. SPSS 28 (Chicago, IL, USA) was used for analyses.
Ethical approval
This study was approved by the Ethics Committee of the Northern Finland Hospital District (10/2020), and all participants provided written informed consent prior to participation.
Results
Altogether 82 patients were recruited for this study and filled out the first questionnaire (test) and 67 patients filled out and posted the second questionnaire (retest). A total of 10 individuals formed a control group.
Reliability
The internal consistency was measured with Cronbach’s alpha for the four subscales. The results are presented in Table 1. The results for Finnish FIQL indicated that all four subscales had good consistency, except for the embarrassment subscale, which had a slightly lower Cronbach’s alpha value for the test (0.68). Between subscales, the ICC varied from 0.83 to 0.90 which indicates almost perfect agreement and high homogeneity (Table 1). For the internal consistency and ICC analysis, we only accepted questionnaires with fully filled subscales.
Reliability analyses of the translated FIQL. The internal consistency is presented with the Cronbach’s alpha and subscale scores.
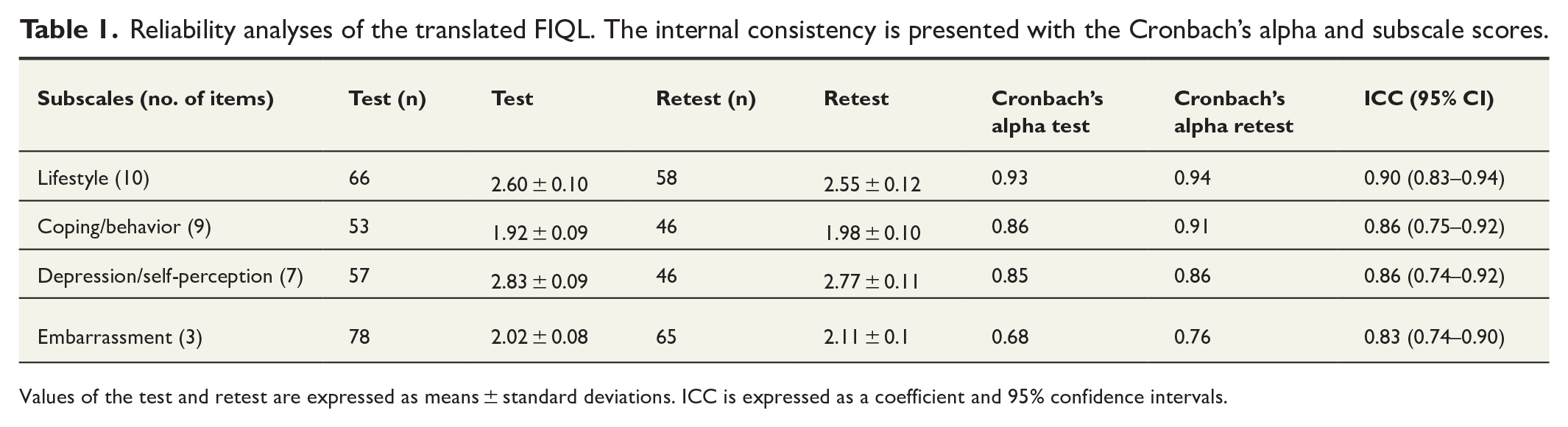
Values of the test and retest are expressed as means ± standard deviations. ICC is expressed as a coefficient and 95% confidence intervals.
We also calculated ICC for subscales with missing values, which are found in the Supplementary Table 1. For the accepted subscales, we considered subscales with at least 67% answer rate to be acceptable. For the acceptable answers, we took into account 7 responses out of 10 for the first subscale, 6 responses out of 9 for the second subscale, 5 responses out of 7 for the third subscale, and 2 responses out of 3 for the fourth subscale. ICC for the missing values ranged from 0.82 to 0.90, indicating good reliability and reproducibility over time.
Construct validity and floor and ceiling effect
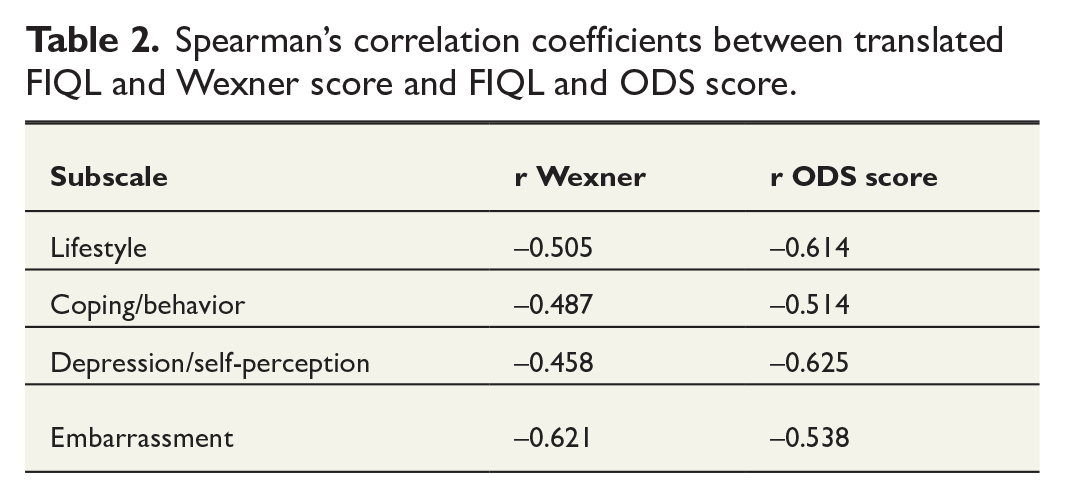
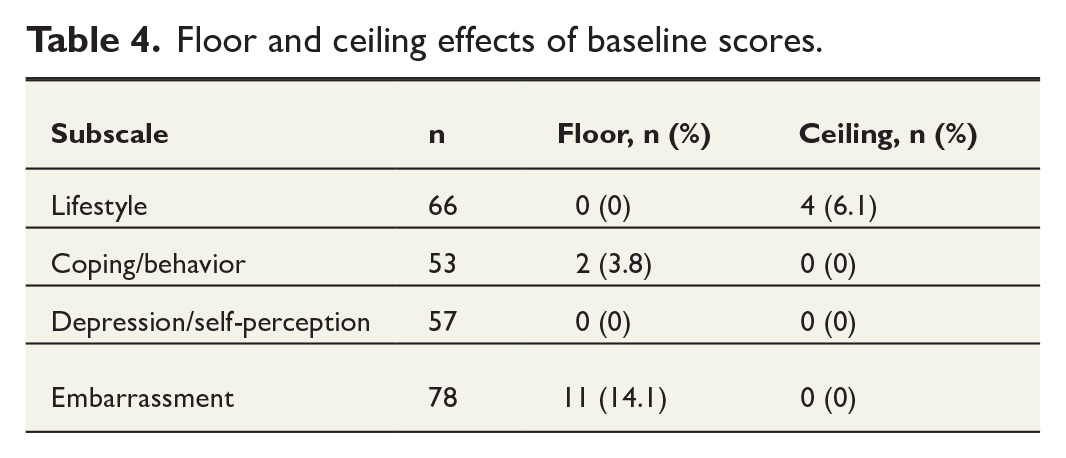
The convergent and discriminant validity of the subscale was examined by correlating the FIQL subscales and total score with the Wexner score and ODS score. The results are presented in Table 2. The correlations varied from −0.458 to −0.621 between FIQL subscales and Wexner scores and from −0.514 to −0.625 between FIQL subscales and ODS scores. These correlation coefficients indicate moderate correlation between FIQL, Wexner, and ODS. In FIQL, lower points indicate more severe symptoms, whereas in Wexner and ODS, higher points indicate more severe symptoms. Correlations for missing values are presented in the Supplementary Table 2. Spearman’s correlation coefficient ranged from −0.497 to −0.613, showing moderate correlations between questionnaires. There were significant differences in FIQL scores between FI patients and controls, indicating that it is disease-specific, as presented in Table 3. The distribution of floor and ceiling effects is shown in Table 4. None of the subscales had floor or ceiling effect.
Spearman’s correlation coefficients between translated FIQL and Wexner score and FIQL and ODS score.
Comparison of Wexner and FIQL subscale scores between FI patients and controls.
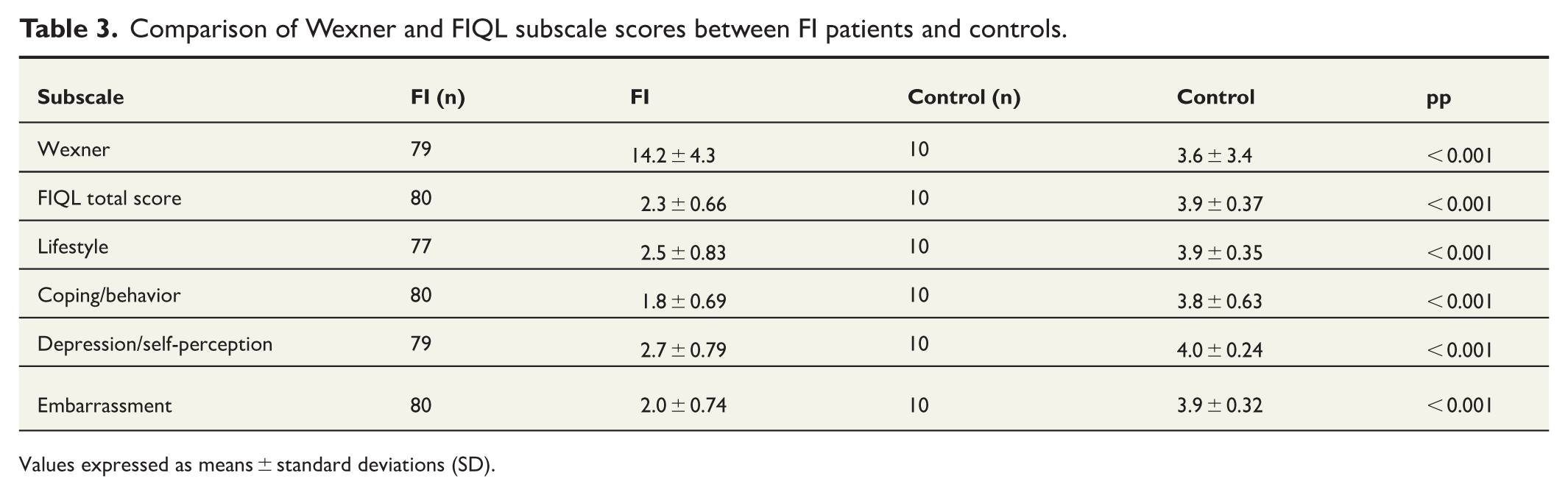
Values expressed as means ± standard deviations (SD).
Floor and ceiling effects of baseline scores.
Discussion
This article provides a successfully validated Finnish version of the FIQL (Supplementary File 1). Our results for the subscale’s internal consistency and stability over time are in line with previous validations.
The psychometrical analysis showed high internal consistency for three out of four subscales and slightly lower Cronbach’s alpha for the fourth subscale (embarrassment). Cronbach’s alpha for lifestyle subscale was above 0.9 both in test and retest as well as in retest of coping/behavior subscale, which may indicate that the items are too similar. Cronbach’s alpha is very sensitive to the number of items which may account for the lower Cronbach’s alpha seen in the embarrassment subscale, both in our and in other comparable re-validation studies, as the embarrassment subscale has a lower number of items (3) than other subscales.7,10 However, despite the lower internal consistency of this subscale, it provides results for descriptive purposes. No floor or ceiling effects were found, indicating that the questions are of adequate difficulty and variation appropriate for the target population.
The size of the study population was sufficient, taking into account some lost patients in the retest round, when the response rate for the second questionnaire was 81%. The number of participants was comparable to other re-validation studies, where study populations have varied: 50 patients (Portuguese) 9 ; 73 and 8 controls (Italian) 22 ; 76 (Norwegian) 10 ; 55 and 93 controls (Chinese) 11 ; 55 and 277 controls (Dutch) 23 ; 100 (French) 7 ; and 119 patients (Japanese). 12
A strength of our study was the versatility of the people both in the translation and in the validation processes. Our first panel consisted of both English and Finnish natives from northern and southern Finland, and the lay panel was diverse in terms of age, gender, and educational background. Recruited patients for the validation process represented different areas of Finland: northern, western, southern, and middle parts of the country. The patients were predominantly female as in previous studies, but the FIQL has also been validated for male patients. 24 Another strength of this study was the use of tested, applicable quality criteria for the evaluation of measurement properties, as proposed by Terwee et al. 15 However, one limitation of our study is that we did not include intervention in the validation process which prevented us from detecting clinically important changes over time.
As subscales with missing values were not taken into account in the original Rockwood et al.’s 5 paper, we believed it was important to assess the significance of incomplete responses for the usability of the form. Based on our experience and previous reports, it is likely that the patients will not answer all the questions, especially if the questionnaire is long. 25 Similar to the Chinese and Norwegian studies, we found that Finnish patients tended to not answer questions related to highly personal aspects of their life. 11 Therefore, we wanted to evaluate results where a certain amount of missing values are acceptable. When assessing the data with missing values, the ICC ranged from 0.82 to 0.90. This still indicated good stability over time and the correlation remained the same, which supports the usability of the Finnish questionnaire.
For convergent validity, we found moderate correlations between FIQL and Wexner scores. For the discriminant validity, we chose ODS score as it measures the “opposite” defecation symptom. We found that the correlation coefficient was marked negative for both Wexner and ODS scores. However, correlation coefficients in 3 out of 4 subscales were higher for Wexner scores than for ODS scores, as expected. This shows that our chosen score to examine discriminant validity was not quite successful, as the ODS score also includes questions relevant to patients suffering from FI (e.g. using an enema). This indicates that impaired QOL related to FI correlates not only with increased FI but also with symptoms of obstructed defecation. This also depicts that pelvic floor symptoms create complex problems, and an individual may experience both FI and obstructed defecation symptoms. In our study, the FIQL total score was significantly lower in FI patients than in controls which shows FIQL can discriminate between patients with and without FI.
We chose the FIQL instrument for Finnish translation, as it has already been successfully translated and adapted to several other languages and is widely used. The questionnaire’s differential functioning between genders has been reported to be minimal. 24 Some questions about the instrument have been raised, such as overlapping of some items in terms of their content and the lack of weighting the items to grade their significance.5,12 Suggestions for specific revisions to improve the instrument have been warranted based on evaluation using item response theory (IRT). 24 Based on high intercorrelations between the four subscales and the high Cronbach’s alpha of the global score, it has been suggested that the mean score of all items could be employed for quicker overall evaluation and easier comparison of patients and patient groups.10,12 In our study, the FIQL total score was significantly lower in FI patients than in controls. The responsiveness of the FIQL total score has also been reported to be good. 13 Despite its limitations, the FIQL instrument serves as a symptom-specific QOL measure physicians can reliably use to measure individual differences and treatment effects of patients suffering from FI and allows for research comparisons between populations from different countries.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969251337855 – Supplemental material for Translation and validation of the Finnish version of the Fecal Incontinence Quality of Life Scale
Supplemental material, sj-docx-1-sjs-10.1177_14574969251337855 for Translation and validation of the Finnish version of the Fecal Incontinence Quality of Life Scale by Helena Komokallio, Tero Rautio, Pasi Ohtonen, Tarja Pinta, Anne Mattila, Antti Turunen and Johanna Mäkelä-Kaikkonen in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-2-sjs-10.1177_14574969251337855 – Supplemental material for Translation and validation of the Finnish version of the Fecal Incontinence Quality of Life Scale
Supplemental material, sj-docx-2-sjs-10.1177_14574969251337855 for Translation and validation of the Finnish version of the Fecal Incontinence Quality of Life Scale by Helena Komokallio, Tero Rautio, Pasi Ohtonen, Tarja Pinta, Anne Mattila, Antti Turunen and Johanna Mäkelä-Kaikkonen in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
The authors thank Dr Michael Spalding for providing forward translation.
Author contributions
H.K. contributed to the concept and design of the work, acquisition, analysis and interpretation of data, and writing the article. T.R. contributed to the concept and design of the work, analysis and interpretation of data, and writing the article. P.O. contributed to the concept and design of the work, analysis and interpretation of data, and writing the article. T.P. contributed to the acquisition of data and writing the article. A.M. contributed to the acquisition of data and writing the article. A.T. contributed to the acquisition of data and writing the article. J.M.-K. contributed to the concept and design of the work, acquisition, analysis and interpretation of data, and writing the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Finnish State Research Fund, the Northern Finland Healthcare Support Foundation (Terttu Foundation), and the Oulu Medical Research Foundation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
