Abstract
Background and objective:
The optimal type of anastomosis for preventing anastomotic leakage (AL) and bowel dysfunction after colorectal surgery remains uncertain. The aim of the study was to evaluate anastomotic type after anterior resection (AR) in relation to AL and functional outcome in long-term follow-up.
Methods:
This was a population-based study using data from the Swedish Colorectal Cancer Registry (SCRCR). The patients were categorized into two groups, based on anastomotic design (“J-pouch/side-to-end (STE) anastomosis” or “end-to-end (ETE) anastomosis”). AL was established using SCRCR and supplemented with review of medical records. The low anterior resection syndrome (LARS) score questionnaire and Cleveland Clinic Florida Fecal Incontinence score (CCFFIS) were used for the assessment of bowel function. The associations and the predefined confounders were adjusted for using logistic regression/linear mixed-effects models.
Results:
A total of 710 patients who underwent curative rectal cancer surgery with AR between 2007 and 2013 were included. AL occurred in 87 (15.7%) patients in the STE group and 10 (10.2%) in the ETE group. After adjustment, the type of anastomosis (STE versus ETE) did not affect the odds of AL (odds ratio (OR) 0.80 (95% CI: 0.37–1.76)). There was no association between the anastomotic technique and bowel dysfunction (LARS score: OR 1.14 (95% CI: 0.58–2.27) and CCFFIS: OR −0.08 (95% CI: −1.63 to −1.46)).
Conclusions:
This is the first study examining anastomotic type and the risk of AL and bowel dysfunction in a long-term perspective beyond 3 years among patients who underwent AR. The anastomosis type did not show any association for AL or bowel dysfunction.
Keywords
Context and Relevance
The optimal anastomosis type for preventing anastomotic leakage (AL) and bowel dysfunction after colorectal surgery remains uncertain. This long-term follow-up study found no significant association between side-to-end or end-to-end anastomosis and the risk of AL or bowel dysfunction in rectal cancer surgery patients.
Introduction
Improved surgical techniques and the use of neoadjuvant therapy, including radiotherapy and chemotherapy, have resulted in a higher number of rectal cancer patients undergoing sphincter-preserving surgery with anastomosis. The optimal type of anastomosis for preventing leaks and to reduce bowel dysfunction remains uncertain. Literature reports Anastomotic leakage (AL) rates ranging from 3% to 23%.1,2 It is known that low anastomoses have a higher risk of AL and cause reduced bowel function, these symptoms are more pronounced after neoadjuvant radiotherapy. 3
To improve the capacity of the neorectum several different surgical techniques can be used. A large meta-analysis that included thirty-two trials comparing type of anastomosis, the study concluded that transverse coloplasty (TCP) anastomosis was associated with a higher risk of AL and stricture, while colonic J-pouch (CJP), side-to-end (STE), and end-to-end (ETE) anastomosis had equivalent rates for these complications. Moreover, CJP anastomosis led to better bowel functional outcome over the other types in the short term after surgery. 4
Furthermore, AL has been suggested to increase the risk of local recurrence and decreases overall survival by delaying the induction of adjuvant treatments.5,6
Besides to AL, most patients (70%–90%) suffer from varying degrees of disturbed bowel function (low anterior resection syndrome, LARS) that resulting in sustained negative impact on quality of life in long term after rectal cancer surgery.7–11
Hain et al. 12 report symptomatic AL as predictor for major LARS in multivariable analysis (OR 3.51; 95% CI: 1.19–10.73) using no or asymptomatic leakage as the reference. Other studies were not able to demonstrate an association.13–16
The aim of the study was to evaluate anastomotic type after anterior resection (AR) in relation to AL and bowel functional outcome in long-term follow-up.
Material and methods
Study population and design
This was a population-based cohort study of all patients who had undergone curative rectal cancer surgery with total (TME) or partial (PME) mesorectal excision between January 2007 and December 2013 in Stockholm County, Sweden. Patients aged 18 years or more were identified through the Swedish Colorectal Cancer Registry (SCRCR), a registry with high completeness and accuracy. 17
The clinical data were retrieved from the SCRCR and a review of the patients’ medical records.
The SCRCR lacks a definition of AL, which was retrieved from the medical records after review by a surgeon. AL was defined as clinically as diagnosed AL (via clinical examination and blood tests), confirmed by radiology, endoscopy or both.
Data concerning surgery, neoadjuvant treatment and information on recurrent and/or disseminated disease were included. Patients without metastasis and/or recurrence at follow-up (irrespective of primary stage and neoadjuvant/adjuvant treatment) and were alive on 4 April 2017, were included. Patients with a stoma, diagnosis of dementia or recurrent disease, and those who passed away, were excluded.
In some regions in Sweden the anastomotic configuration is reported differently to SCRCR with either two alternatives (“reservoir with either CJP or STE anastomosis” or “ETE anastomosis”) or in other region with three alternatives, one for each type of anastomosis (“CJP,” “STE anastomosis,” or “ETE anastomosis”). To accommodate this inconsistency, the patients in the present study were categorized into two groups, reconstructed with either STE (CJP/STE) or ETE anastomosis.
All patients who met the inclusion criteria were invited to participate in the study. A letter was sent to each patient with questionnaires for the LARS score and Cleveland Clinic Florida Fecal Incontinence Score (CCFFIS).
Questionnaires
The LARS score consists of five questions: The range of the score is 0–42, weighted in relation to the impact on quality of life and classified as no LARS (0–20 points), minor LARS (21–29 points) and major LARS (30–42 points).18,19
The CCFFIS consists of five separate questions. Each question has five different response alternatives (range 0–4 points), resulting in a maximum of 20 points. Three questions concern incontinence for flatus and/or feces, one concerns the use of pads, and one concerns lifestyle alteration owing to bowel dysfunction. 20
A study-specific questionnaire concerning the presence of a stoma was developed for this study, which contains two questions: “Do you have a stoma?” (yes or no); and “If yes, what is the reason for you having a stoma?” (“No stoma reversal was attempted after my primary rectal cancer operation” or “I got a stoma later because of severe bowel dysfunction” or “None of the above response alternatives”).
Ethical approval
The study was approved by the local ethics committee of Stockholm (2016/1604-31/2 and 2017/605-32). Informed consent was obtained from the patients.
Statistical analysis
Descriptive statistics were used to summarize baseline characteristics and follow-up outcomes. Categorical variables were reported as frequencies and percentages, while continuous variables were summarized medians and interquartile ranges. Chi-square test or Fisher exact test, and Kruskal–Wallis test were used to calculate the p-values for baseline patient characteristics. To address missing data, multiple imputations were performed, assuming missing at random, and using the Fully Conditional Specification (FCS) method with 20 imputations. The procedure included categorical variables (type of anastomosis, gender, and smoking status) modeled using logistic regression, and continuous variables (body mass index (BMI)) modeled using predictive mean matching. The imputation accounted for patient characteristics and surgical factors. This approach ensured robust and unbiased estimates by incorporating variability in the missing data. Analyses were conducted on the imputed datasets to account for the uncertainty of missing values.
Logistic regression models were employed to evaluate associations between type of anastomosis and outcomes AL major LARS where the LARS score was categorized into binary groups (no/minor LARS and major LARS) for comparative analysis. Linear mixed-effects models were used to analyze the association between type of anastomosis and CCFI. Both models included covariates to adjust for potential confounders, and model fit was assessed using standard diagnostic techniques.
Confounder selection was based on a detailed study protocol, incorporating prior research and established knowledge. For the logistic regression model evaluating AL (primary outcome), adjustments were made for gender, age at diagnosis (continuous), neoadjuvant chemoradiotherapy, tumor level (0–5 cm, 6–10 cm, and 11–15 cm), PME/TME, body mass index (BMI) (>30 or ⩽30), preoperative smoking status (yes/no), diabetes, and cardiovascular disease.
For the models examining secondary outcomes LARS groups (no/minor versus major LARS) and CCFI score, adjustments included all the above variables plus surgical approach (open versus minimally invasive), ligature level (unknown, inferior mesentery artery (IMA), or rectal superior artery).
All statistical analyses were performed using SAS 9.4 software, with a significance level set at p < 0.05.
Outcomes
The primary outcomes was the prevalence of AL in across different anastomosis types in the total cohort. The secondary outcomes were the association between anastomosis type and major LARS, and CCFI score.
Results
In total there were 710 patients in the cohort with an anastomosis and included in the AL analysis. Out of these, 481 patients were included in the analysis with data on both AL and bowel function (questionnaire response rate 82.6 per cent, 481 of 582), Fig. 1. Of these, 478 patients completed both the LARS score and CCFFI questionnaire. Mean follow-up time was 6.7 (range 3.4–11.0) years after surgery.
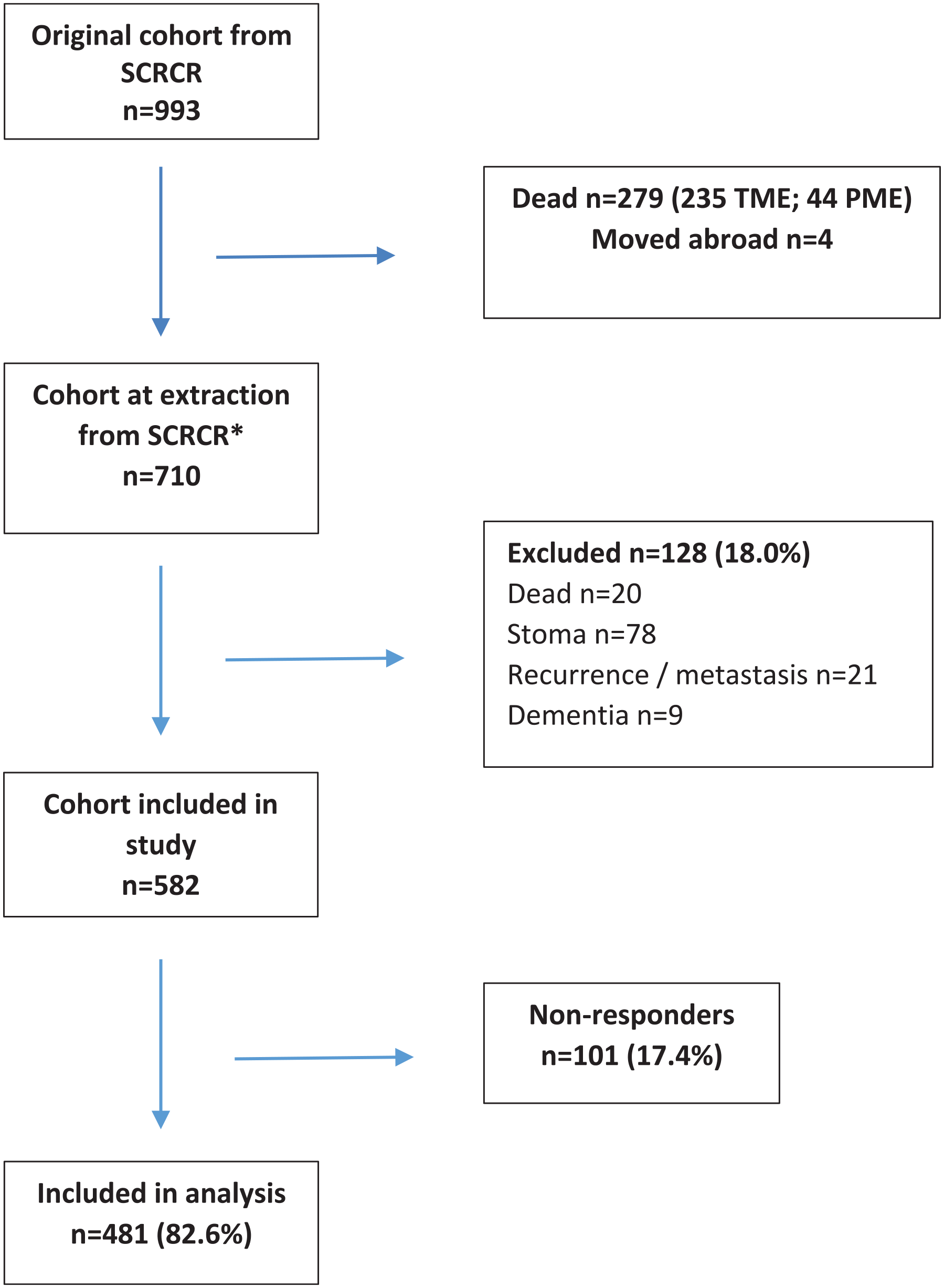
Flow diagram for the study.
In the total cohort (710 patients), 555 (78%) patients had a STE anastomosis, 98 (14%) patients with ETE anastomosis and 57 (8%) patients with unknown anastomosis (Table 1). There were only six patients with CJP in the present cohort. In the unknown group the type of anastomosis the patients’ received, were missing in both the registry and in the medical records.
Baseline patient characteristics and surgical variables after curative rectal cancer surgery.
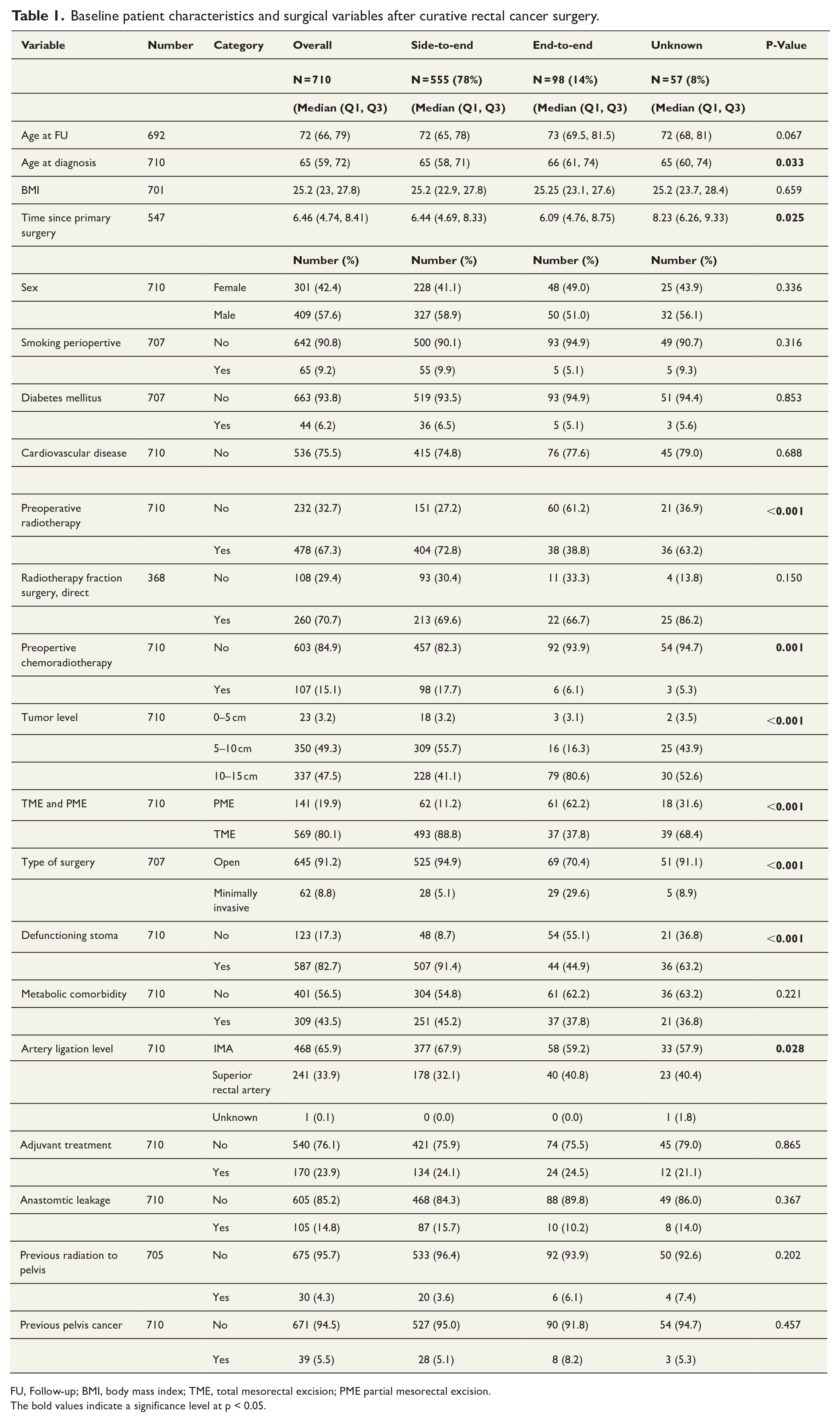
FU, Follow-up; BMI, body mass index; TME, total mesorectal excision; PME partial mesorectal excision.
The bold values indicate a significance level at p < 0.05.
The groups differed regarding age at diagnosis, with the ETE group being older (median 66 years; p = 0.033). There were a higher proportion of the patients in the STE group and unknown group that received preoperative radiotherapy (STE; 404 patients (72.8 %), ETE; 38 patients (38.8%), and unknown; 36 patients (63.2 %); p < 0.001). Furthermore, the STE group received more preoperative chemoradiotherapy (98 patients (17.7%) in comparison with the other two groups (6.1% ETE and 5.3% unknown group); p = 0.001).
The tumor location relative to the anal verge differed across groups. In the ETE group, more tumors were located at 10–15 cm compared to other anastomosis types(ETE 79 (80.6%), STE 228 (41.1%), unknown30 (52.6%); p < 0.001). At 5–10 cm, STE and unknown anastomoses were more common than ETE (STE 309 (55.7%), unknown 25 (43.9%), ETE 16 (16.3%); p < 0.001). This pattern was also reflected in the PME versus TME surgery, with PME being more frequent in the ETE group(ETE 61 (62.2%), STE 62 (11.2%) and unknown 18 (31.6%); p = 0.001). Open surgery was more frequent in the STE group than in the ETE group (STE: 525 (94.9%), ETE: 69 (70.4%), unknown: 51 (91.1%); p < 0.001). Moreover, defunctioning stomas were more frequently used in the STE group (STE: 507 (91.4%), unknown: 36 (63.2%), ETE: 44 (44.9%); p < 0.001).
Anastomotic leakage
AL occurred in 15.7% (87 out of 555 patients) in the STE group, 10.2% (10 out of 98 patients) in the ETE group, and 14.0% (8 out of 57 patients) in the unknown group, p = 0.367 (Table 1).
In the logistic regression after adjustment, the type of anastomosis (STE versus ETE) did not affect the odds of AL (OR 0.80; 95% CI: 0.37–1.76) (Table 2).
Type of anastomosis and association to anastomotic leakage.
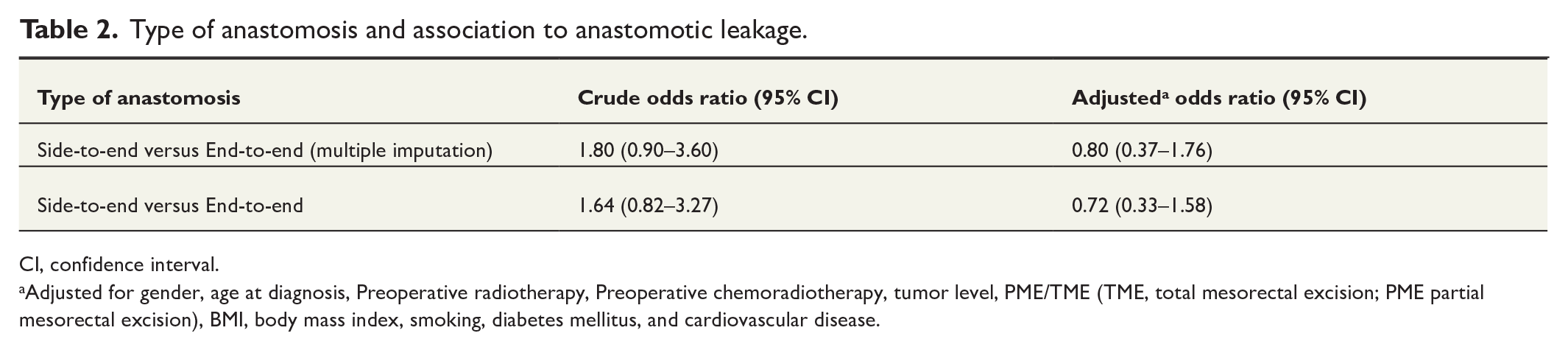
CI, confidence interval.
Adjusted for gender, age at diagnosis, Preoperative radiotherapy, Preoperative chemoradiotherapy, tumor level, PME/TME (TME, total mesorectal excision; PME partial mesorectal excision), BMI, body mass index, smoking, diabetes mellitus, and cardiovascular disease.
Functional outcomes
Non-responders and excluded patients
There were no differences in patient characteristics between included and excluded patients (data not shown).
Low anterior resection syndrome
The adjusted STE point estimate showed a trend toward association with major LARS, but this became non-significant after confounder adjustment(OR 1.14; 95% CI: 0.58–2.27) (Table 3).
Type of anastomosis and association to major LARS when compared to no/minor LARS.

LARS, low anterior resection syndrome. CI, confidence interval. Three of the 481 patients in the study did not complete the LARS score questionnaire and so are not included here.
Adjusted for gender, age at diagnosis, perioperative chemoradiotherapy, tumor level, PME/TME (TME, total mesorectal excision; PME partial mesorectal excision), BMI, body mass index, smoking, diabetes mellitus, and cardiovascular disease, type of surgery, and ligature level.
Cleveland Clinic Florida Fecal Incontinence Score
Patients with STE anastomosis had a higher crude CCFFIS scores, but the difference did not remain after adjustments (OR −0.08; 95% CI: −1.63 to −1.46), (Tables 4 and 5).
Type of anastomosis and association to Cleveland Clinic Florida Fecal Incontinence.
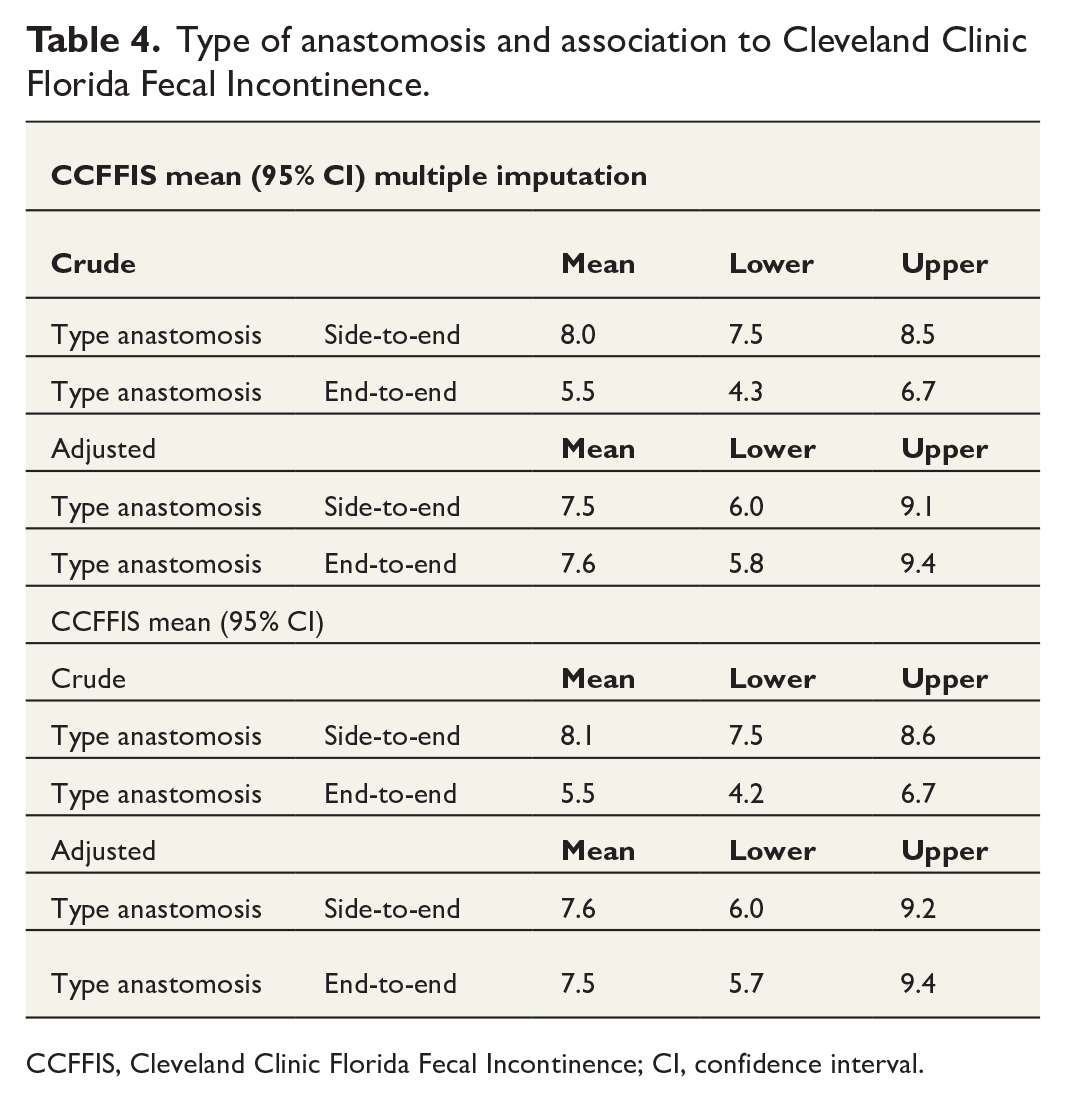
CCFFIS, Cleveland Clinic Florida Fecal Incontinence; CI, confidence interval.
Type of anastomosis and association to Cleveland Clinic Florida Fecal Incontinence Score.
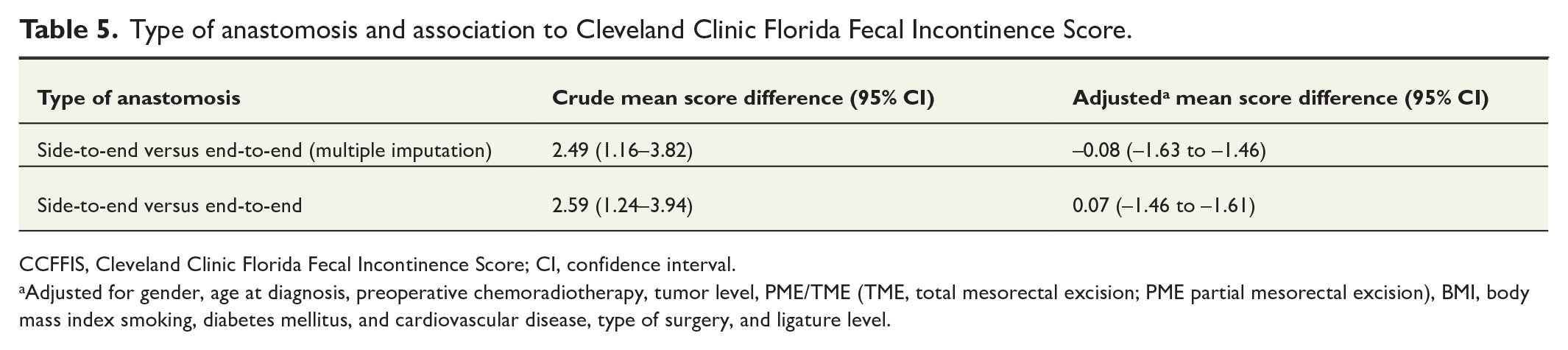
CCFFIS, Cleveland Clinic Florida Fecal Incontinence Score; CI, confidence interval.
Adjusted for gender, age at diagnosis, preoperative chemoradiotherapy, tumor level, PME/TME (TME, total mesorectal excision; PME partial mesorectal excision), BMI, body mass index smoking, diabetes mellitus, and cardiovascular disease, type of surgery, and ligature level.
Discussion
In this study, the type of anastomosis (STE versus ETE) did not show any an association with the risk of AL or bowel dysfunction (major LARS, or high CCFFIS score) after adjustment for confounders. These results are in concordant with a previous Swedish study Sandberg et al., 21 based on 494 patients. Their study found no long-term benefitof CJP/STE anastomosis on LARS (CJP/STE OR 1.05; 95% CI: 0.82–1.34). The present study, with a larger cohort and longer follow-up time (6.7 versus 3 years), confirmed these findings. The unequal group size may have influenced results, and if a ture difference exista, detecting it would require a much larger study.
More tumors were located in the higher rectum (10–15 cm) in the ETE group, while mid rectal level tumors (5–10 cm), there were more common with STE anastomosis. This aligns with the higher frequency of PME in the ETE group. The STE group had more open procedures and a higher use of defunctioning stomas, likely due to the increased surgical complexity of lower tumors. Descriptive analysis also showed a higher AL rate in the STE versus ETE group (15.7% versus 10.2%), suggesting that more challenging cases may contribute to elevated AL. However, adjusted analysis demonstrated no difference in AL between the anastomotic types. Tumor location and its distance from the anal verge are known risk factors for AL, and the AL frequency in this study remains within the reported range in the literature.22,23
In the descriptive analysis, more patients in the STE group received preoperative radiotherapy (72.8 %) compared to the ETE group (38.8%). Similar differences were observed for preoperative chemoradiotherapy, which is more common in low rectal cancers due to the higher risk of threatened circumferential resection margins in narrow pelvis and the limitations of radiotherapy in upper rectal cancers. Low tumors were likely classified as rectal cancer rather than sigmoid cancer, leading to more frequent preoperative chemoradiotherapy. Furthermore, early in the study period, ETE anastomosis was more common, and preoperative radiotherapy was used less frequently.
Compare AL rates with other studies is challenging due to inconsistent definitions. A recent systemic review by Heuvelings et al. 24 analyzing 95 studies found that only 18% reported AL severity grading, while most relied on clinical signs, biochemical markers, radiology, or reintervention findings. In this study, AL was identified through the SCRCR registry and validated via medical records, capturing some AL cases not initially registered. A standardized definition for diagnosing and reporting AL is crucial for future research. 25
After adjusting for confounders, the association between STE anastomosis and major LARS was non-significant, contrasting with earlier studies showing better short-term outcomes with CJP and STE anastomoses compared to ETE anastomosis the first year after surgery. A 2008 Cochrane review 26 reported similar findings, but long-term data remain scarce, and evidence for functional differences between the different anastomotic types over time is weak. 27
This study was limited by its retrospective design and the inclusion of only 481 of 993 eligible patients for bowel function analysis. However, strengths included a large cohort (710 patients) with a high questionnaire response rate (82.6%), validated questionnaires ensuring comparability, and a long follow-up (mean 6.7 years), providing insight into quality of life and bowel function post-AL. In addition, all medical records were reviewed to validate registry data and identifying missing AL cases. Studies on anastomotic types in rectal surgery show mixed results, with some reporting reduced AL and LARS with STE, while other find no differences.27–33 A large multi-center prospective study with standardized AL definition and both clinical and patient-reported outcome is needed to clarify the impact of anastomotic type on AL and bowel dysfunction.
Conclusion
This long-term follow-up study showed no association between anastomotic technique (STE versus ETE) and the risk for AL or bowel dysfunction in rectal cancer surgery.
Footnotes
Author contributions
EP, contributed to design of the work, Data curation, Supervision, Validation, and Writing, review and editing PL, design of the work, Investigation, Methodology, Validation, and Writing, review and editing, AJ, design of the work, Formal analysis, Investigation, Methodology, Validation, and Writing—review and editing M A-N, design of the work, Project administration, contributed to Data curation, Formal analysis. Investigation, Methodology, Supervision, Validation, and original draft preparation, review and editing.
Data availability
All data collected for the study can be obtained from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
