Abstract
Background and objective:
Breast cancer liver and lung metastases are common and associated with poor prognosis. Personalized medical treatment of advanced breast cancer, based on established predictive factors, has improved survival during the last decades. In contrast, there is no consensus regarding indications for surgery. The aim of this narrative review is to summarize the current knowledge on the outcome of surgical treatments for breast cancer liver and lung metastases.
Method:
A narrative review of existing evidence for diagnosis and treatment of breast cancer liver and lung metastases.
Results:
There are no randomized trials to evaluate surgery as an adjunct to medical treatment. In this review, data are reported from case–control studies and meta-analyses on surgery for liver and lung metastases. Selected patients have an improved survival after surgery compared to those who only received medical treatment. The survival benefit is, however, uncertain when adjusting for prognostic factors, and prospective trials are warranted. Anecdotal cases have long-term survival and surgery is safe.
Conclusion:
Surgery for breast cancer liver and lung metastases may be considered in selected cases within prospective studies.
Context and relevance
Breast cancer is a common disease, but the benefit of local treatment of distant metastases remains unclear. There are several case series published suggesting improved survival after surgical treatment of breast cancer metastases, but there is still a lack of large, randomized trials to rule out selection bias. Lung and liver are the second and third most common sites for breast cancer metastases. This narrative review summarizes the current knowledge on the diagnosis and surgical treatment of disseminated breast cancer and describes the modalities for local treatment in the lung and the liver. Recent studies highlight both the importance of patient selection prior to treatment and the need for prospective trials.
Introduction
Breast cancer is the leading cause of cancer-related death among women in the world. 1 Screening programs combined with modern surgical and oncological treatment strategies have improved survival among breast cancer patients in developed countries. 2 Still, the breast cancer mortality is constant due to an increased incidence. 3 Systemic disease may ultimately result in visceral crisis and death. 4
Locoregional breast cancer is associated with excellent survival rates while the prognosis remains dismal in metastatic disease. 3 The St. Gallen classification of molecular breast cancer subtypes has prognostic value and predicts response to oncological and endocrine treatments. The classification is based on the Nottingham histologic grade (NHG), the proliferation marker Antigen Kiel 67 (Ki-67) and expression of estrogen receptors (ERs), progesterone receptors (PgRs), and amplification of human epidermal growth factor receptor 2 (HER2) gene. 5 Expression of ER and PgR is associated with improved prognosis, 6 while the oncogene HER2 has a treatment predictive value owing to targeted anti-HER2 therapy. 7 Still, metachronous metastases may arise long after primary diagnosis, even in patients with a favorable breast cancer subtype. This results in long-term anxiety for relapse among breast cancer patients. Bone is the most common site of distant spread, particularly in estrogen receptor-positive cancer. These patients respond well to endocrine treatment, and progression can often be delayed for years. 8
Breast cancer lung and liver metastases are the second and third most common metastases, respectively. 8 This visceral spread is associated with poor prognosis. 9 The median survival is estimated to be approximately 30 months. Breast cancer cells with HER2 amplification or loss of hormonal receptor expression are more common in these sites, compared to bone.8,9 The hormonal receptor status may also convert between the primary tumor and the metastases, leading to another, often more aggressive, molecular subtype in the metastases.10–14
Surgical treatment of breast cancer metastases is debated. Advanced breast cancer, with spread beyond locoregional lymph nodes, is normally considered a systemic disease, and there are no general recommendations for surgery. 4 Numerous case series and retrospective studies have, however, suggested that surgical treatment may improve survival in selected patients, especially in an oligometastatic setting.15–26 In an era of improved medical treatment for advanced breast cancer, it is important to ask whether surgery improves survival? This review summarizes the current knowledge regarding the outcome of local treatment of breast cancer liver and lung metastases.
Diagnosis of breast cancer metastases
Synchronous distant metastases are diagnosed during initial staging examinations and primarily result in recommendation on systemic treatment. If the primary breast cancer is asymptomatic, it is often left without surgical treatment. There is no general surveillance guideline for early diagnosis of metachronous distant metastases in early locoregional cancer. Instead, these may be diagnosed as incidental findings, due to symptoms or clinical suspicion based on risk assessment. 4 The tumor marker Ca15-3 may also indicate disease progression. 27
Surgery of metastases is normally restricted to a limited spread, termed oligometastases, and often defined as 1–5 lesions. De novo oligometastases are separated from induced oligometastases when multiple lesions have vanished after medical treatment and ≤5 remain. 28 Prior to surgical resection, a detailed map of the disease is of outmost importance, and a positron emission tomography (PET) can be considered for optimal staging. 4 Magnetic resonance imaging (MRI) with liver-specific contrast and diffusion-weighted images has the highest sensitivity for liver metastases, 29 while computed tomography (CT) is enough for pulmonary metastases. 30 Surgery is mostly considered after response to systemic treatment, and the RECIST-criteria can be applied to determine treatment response. 31 In contrast to surgical treatment of colorectal liver and lung metastases, a biopsy of breast cancer metastases is always motivated. This is due to a frequent shift in the molecular subtype between the primary tumor and metastases. In 21% of patients, liver metastases have a different subtype than the primary tumor and most commonly a loss of hormonal receptor expression or gain of HER2 amplification (Fig. 1). 10 Regarding lung metastases, a biopsy is also warranted to discriminate a metastatic lesion from a primary lung cancer. 32 Breast cancer patients have an increased risk of lung cancer (standard incidence rate 1.4). 33 Furthermore, it is reported that adjuvant radiotherapy after breast surgery increases the risk of lung cancer, compared to breast surgery alone (relative risk 1.5).34,35
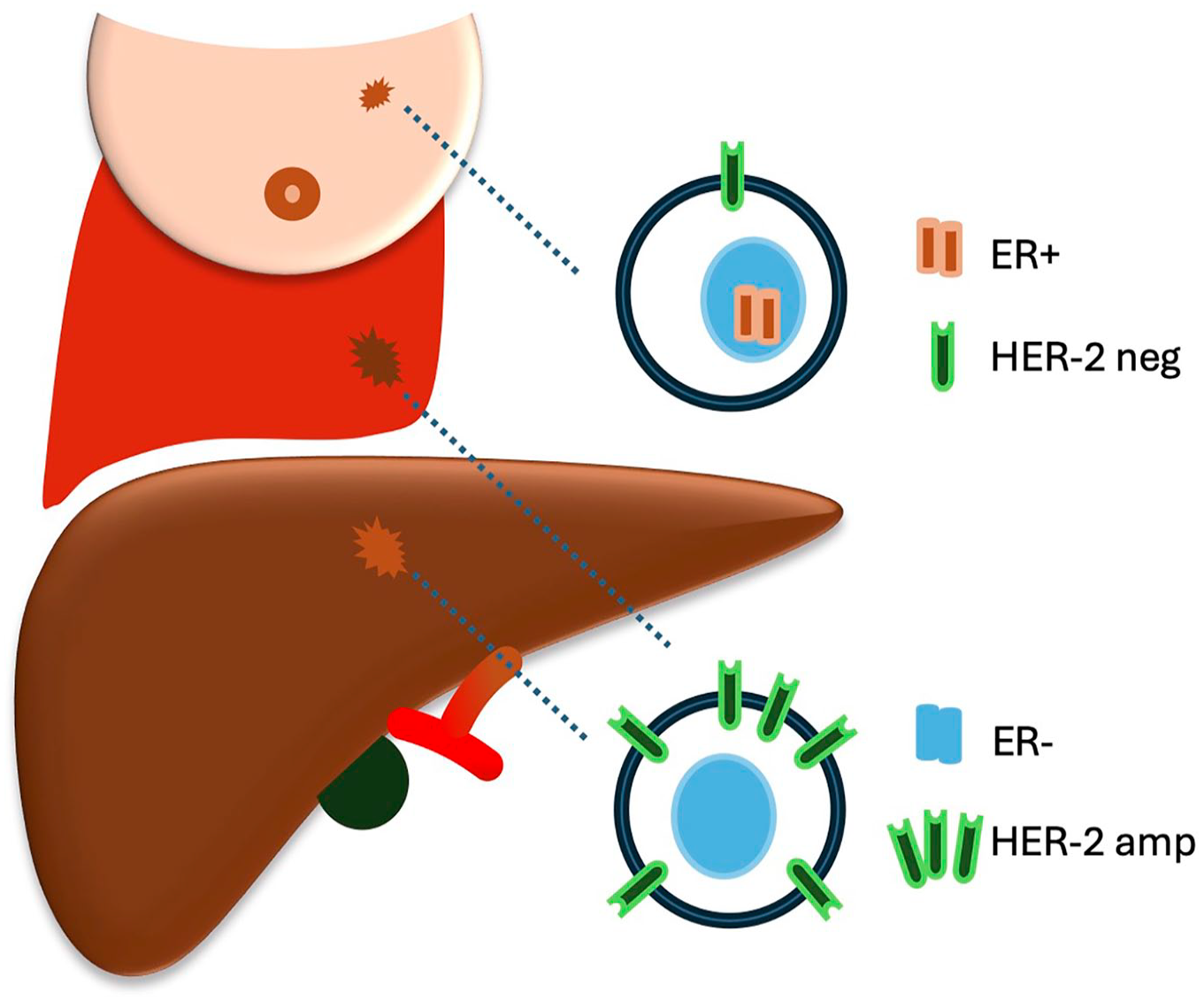
Conversion of subtype in breast cancer liver and lung metastases. Estrogen receptor (ER) and human epidermal growth factor receptor-2 (HER2) status may convert from primary tumor to metastases. For example, a HER2 negative (neg) primary tumor may establish metastases with HER2 amplified (amp) cells.
Outcome of surgery for liver metastases
There are no randomized trials evaluating surgical treatment in addition to standard oncological treatment regimens for liver metastases specifically. One trial was recently closed due to a low inclusion rate. 36 The population of surgical candidates with isolated oligometastases is probably limited. We previously analyzed data from the Swedish National Register for Cancer in the Liver and Bile Duct and identified only 29 patients operated in Sweden during 2009–2016. Surgery was safe with no reported 90-day mortality. Median overall survival was 77 months from diagnosis of breast cancer liver metastases (BCLM). A control group with isolated oligometastases without surgical treatment had significantly shorter survival (28 months, p = 0.004). However, when adjusting for hormonal receptor status and HER-2 amplification, the hazard ratio in favor of surgery failed to reach statistical significance (hazard ratio (HR) = 0.48 95%confidence interval (CI) = 0.19–1.18, p = 0.11). 21 There are several case reports, case series, and retrospective case–control series published, and most of them report improved outcomes from surgical treatment in selected patients. Table 1 summarizes the results of studies with a control group.
Retrospective studies of surgical treatment of breast cancer liver metastases.
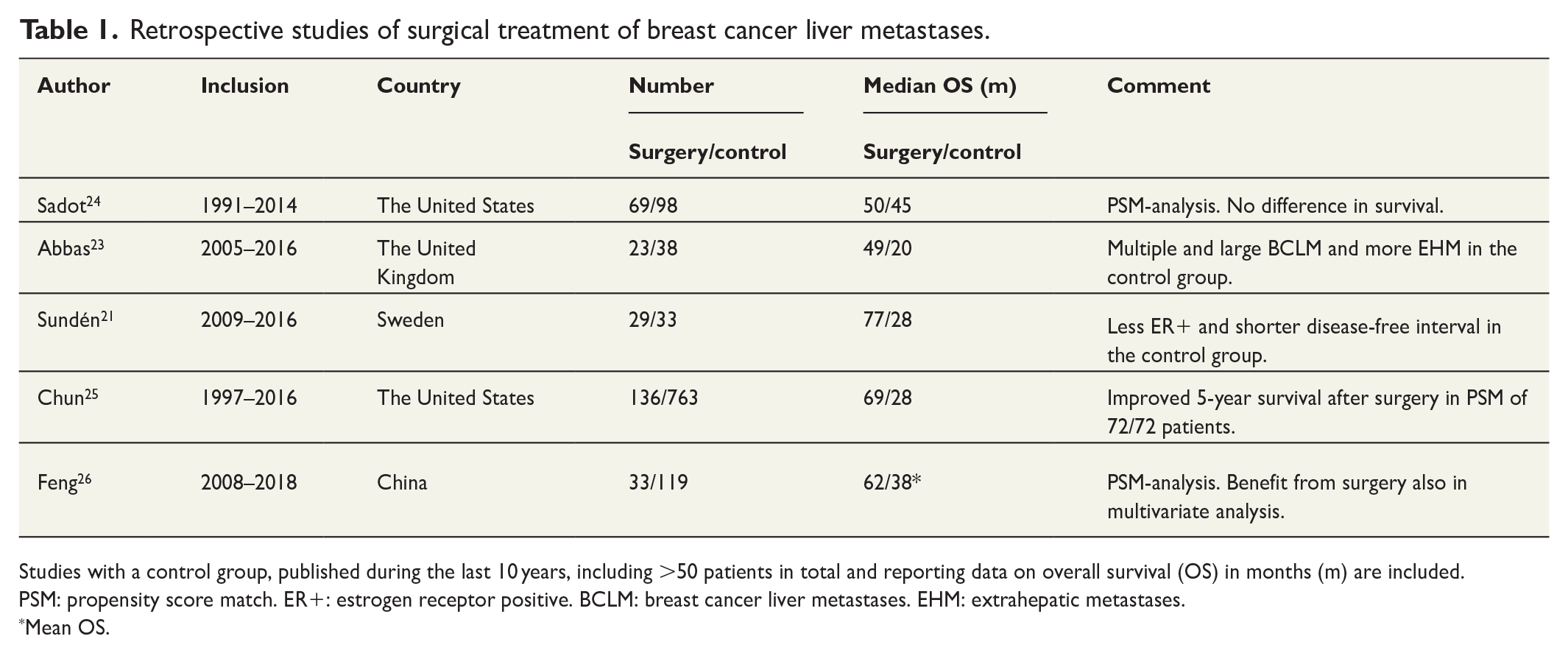
Studies with a control group, published during the last 10 years, including >50 patients in total and reporting data on overall survival (OS) in months (m) are included.
PSM: propensity score match. ER+: estrogen receptor positive. BCLM: breast cancer liver metastases. EHM: extrahepatic metastases.
Mean OS.
A meta-analysis of case–control studies reported 46.0% 5-year survival after surgery, compared to 24.7% after systemic treatment only (p < 0.001). 37 Selection bias in the original studies cannot, however, be ruled out. The ESO-ESMO International Consensus Guideline for Advanced Breast Cancer recommends surgical resection or ablation only in highly selected patients with oligometastases isolated to the liver and after response to systemic treatment. 4
Ablation, transarterial treatment, and radiotherapy of liver metastases
In addition to liver resection, there are several options for local treatment of liver metastases, and these are illustrated in Fig. 2. Radio frequency or microwave ablations are thermal techniques to destroy tumor tissue. These are established in adjunction to liver resection for the treatment of hepatocellular carcinoma (HCC) and colorectal liver metastases. 38 Percutaneous needle placement is guided by ultrasound or CT and navigation systems. Ablation is also an option during laparotomy or laparoscopy, either as a single intervention or in combination with liver resection. Superficial lesions can only be targeted with caution of surrounding tissues, and destruction of hilar metastases may result in bile duct injuries or cancer relapse due to survival of cancer cells close to a cooling blood stream in larger vessels. Yet, ablative treatment is associated with minor morbidity, compared to liver resection, and is often a treatment of choice for lesions up to 3 cm.39,40 Zou et al. 41 recently reported 132 patients with ablative treatment and did a propensity scored match in a control cohort of 836 patients. Among patients with up to three liver metastases <3 cm, median overall survival was 65.5 months in the ablation cohort, compared to 40.4 months in the matched control group (p = 0.003).
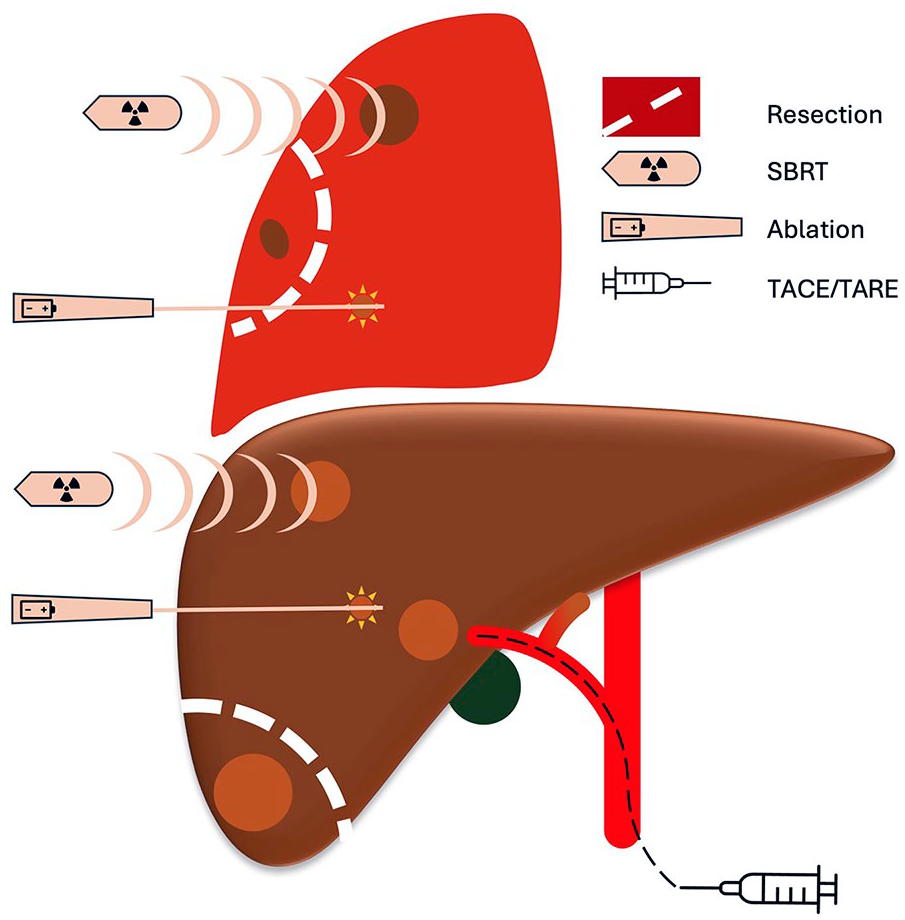
Local treatment modalities of liver and lung metastases.
Trans-arterial chemo-embolization (TACE) is an established treatment for local control or down-sizing of HCC, but there is no evidence to support treatment of breast cancer liver metastases outside clinical trials. A single-center study reported the median survival of 0.8 years in 549 patients, although the survival was better in patients with multimodal interventions including ablations. 42 Trans-arterial radio-embolization (TARE) with yttrium-90 microspheres is another therapy for HCC, and it has also been tested for breast cancer liver metastases. Experimental TACE and TARE treatments have mostly been applied after the development of resistance to standard medical treatment. The benefit of these treatments is uncertain in patients with late stages of breast cancer. 43
Stereotactic body radiation therapy (SBRT) may be an option to treat liver metastases when other local therapies are unavailable. Most studies on SBRT, however, are case series with diverse origin of the primary tumors. While these studies show that local control can be obtained with acceptable toxicity, there is no evidence for improved survival rates. 44 Franceschini et al. 45 recently shared results from a prospective phase II trial on SBRT for up to four liver or lung metastases and report 51.9% overall survival and 16% distant metastases-free survival after 3 years.
Outcome of surgery for lung metastases
Pulmonary metastasectomy is rare, but when performed, it is usually restricted to minor sublobar resections. The indication for mediastinal lymph node dissection is controversial due to the lack of evidence for a survival benefit. 46
A German study of 81 patients operated for lung metastases between 1982 and 2007 showed improved survival compared to control groups matched for age and TNM-stage or age and hormone receptor status. Importantly, a small proportion of patients survived more than 10 years after metastasectomy. 47 A similar small Korean study by Yhim et al. 48 reported 82% 4-year survival after metastasectomy, compared to 31.6% in a control group (p = 0.001). A systematic review summarized nine studies of lung surgery for breast cancer metastases and the 5-year survival ranged from 35% to 72%. 49 They also identified prognostic factors frequently reported in the studies, including the disease-free interval, the number of metastases, complete R0 resection, and hormone receptor status. Of note is that most patients were operated more than 20 years ago.
Radiotherapy and ablation for lung metastases
SBRT has emerged as an option to treat oligometastases and is most frequently used against bone-, lung-, and lymph node metastases. 50 SBRT is generally safe and among 1597 patients (whereof 227 with breast cancer), SBRT was associated with 0.5% acute grade ≥3 adverse events. 51 SBRT can result in local control of the disease, but the effect on survival remains to be determined. Local control is, however, associated with improved survival. 45 CT-guided percutaneous ablation of lung metastases has been evaluated in colorectal cancer and may be part of a multimodal treatment approach. 52 Regarding breast cancer lung metastases, there are only minor case series published on ablation and the treatment can be associated with adverse events such as pneumothorax, pleural effusion, and infection. 53
Discussion
Metastatic breast cancer is a common cause of cancer-related death in women. 3 While diagnostics and medical treatments improve, there is still a general lack of knowledge regarding the value of local treatment for oligometastases in the lung and liver. International guidelines state that prospective trials are urgently needed. 4 However, randomization may be challenging due to a low incidence of patients fulfilling suggested inclusion criteria such as limited disease burden, metastases amenable to local treatment, preserved performance status, and response to treatment. In addition, randomization demands acceptance and compliance from study participants. Chmura et al. 54 have presented an abstract from a randomized trial on local treatment for 1–4 extracranial metastases. In the experimental arm, most had SBRT and only 1/60 had a surgical resection. The addition of local treatment to standard systemic therapies had no impact on progression-free or overall survival. International guidelines do not recommend routine ablation of asymptomatic oligometastases outside of clinical trials. 55 Current studies mainly focus on SBRT, 56 and the Taormina-trial 57 is still recruiting participants in Sweden. This study includes patients with up to five metastases in one or two organs and randomizes between SBRT toward all metastases or systemic treatment alone in a 2:1 ratio. A study design of multimodal treatments of oligometastatic breast cancer may be feasible to answer the question of if local treatments improve outcomes. Glemarec et al. 58 recently presented this in a single institution study and reported a hazard ratio (HR) 0.31 in favor of local ablative techniques compared to systemic treatment only.
Surgical treatment of breast cancer metastases may be valid in highly selected patients. It should only be performed after thorough discussions at multidisciplinary team (MDT) conferences and preferably within prospective studies. The MDT should consider performance status, disease-free interval, response to systemic treatment, and prognostic factors. Estrogen-positive breast cancer is a positive prognostic factor and predictive for response to endocrine treatments. HER-2 amplified breast cancer cells have a more aggressive phenotype and may thrive in viscera, but that are also sensitive to targeted anti-HER-2 treatment. Accordingly, we recently reported that HER-2 amplification was associated with improved survival among patients with breast cancer liver metastases. 10
Future advancements in diagnostics and improved knowledge about recurrence patterns may enable early detection of metastases. For example, circulating tumor DNA (ctDNA) can be detected in blood due to the release of fragmented DNA from necrotic and apoptotic tumor cells. The level of ctDNA is reflected by tumor size and can monitor treatment response. 59 It holds prognostic value at an early stage of the disease and at the time of relapse. ctDNA has also been assessed for surveillance of relapses, yet with conflicting results.60–62 Artificial intelligence (AI) models are also under development to predict distant metastases in breast cancer. 63
Due to the uncertain survival benefit, metastasectomy must be safe and associated with few complications. Minimally invasive strategies like video-assisted thoracoscopic surgery (VATS) and laparoscopic or robotic liver resection should be considered to minimize the surgical trauma. Ablative techniques may also enable local control of the disease and are associated with lower risk than operative resections. If local control can be obtained, it might provide improved quality of life and temporary cessation of heavy oncological treatment even if a survival benefit is uncertain.
In conclusion, surgical treatment of visceral breast cancer metastases should only be performed in highly selected patients. All operative and ablative local treatments of breast cancer metastases should be included in prospective trials.
Footnotes
Author contributions
O.H. drafted the article, and E.H. and C.W. revised it critically. All made a substantial contribution and approved the manuscript.
Authors’ note
Social media summary: This narrative review on local treatment of breast cancer liver and lung metastases shows that selected patients may be operated but preferably within prospective trials.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by research funds from Region Västerbotten (ALF-means).
