Abstract
Surgery has historically played a pivotal role in the management of metastatic melanoma, evolving significantly with the advances of systemic therapies. The advent of immune checkpoint inhibitors initially diminished the role of surgery in treatment paradigms; however, there has been a resurgence of interest in its application within this setting. Several retrospective studies show a survival benefit for patients treated with immune checkpoint inhibitors who are resected to no evidence of disease, especially in case of an objective response to modern therapies. This narrative review explores the role of surgery as a treatment modality in metastatic melanoma before and in the era of immune checkpoint inhibitors, highlighting indications, outcomes, and integration with systemic treatment approaches.
Introduction
The incidence of cutaneous melanoma has increased in the last decades, and the main factors underlying this are an aging population and UV-light exposure, but also increased detection and awareness. 1 Malignant melanoma can metastasize to any organ, and its clinical behavior is difficult to predict, ranging from indolent oligometastatic lesions in some patients to multifocal disseminated disease in others. 2 Patients diagnosed with American Joint Committee on Cancer (AJCC) 8th edition stage IV disease, can be classified in four distinct subgroups based on the pattern of distant metastatic involvement. 3 Patients with M1a disease (distant metastasis to skin, soft tissue and non-regional lymph nodes), M1b (distant metastasis to lung with or without M1a sites), M1c (distant metastasis to non-CNS visceral sites with or without M1a or M1b sites of disease), and M1d (distant metastasis to CNS sites with or without M1a, M1b or M1c sites of disease). 3 Overall, patients with distant metastasis have a poor prognosis; however, those with M1a as opposed to M1d disease exhibit significantly better overall survival (OS), reflecting the prognostic impact of metastatic site.3,4 The biological route of metastasis is driven by a complex process involving tumor cell detachment, local tissue invasion with entry in lymphatics, and the vascular system and colonization of distant organs. 5
Before the introduction of immune checkpoint inhibitors (ICIs) and targeted therapies (TTs), patients with stage IV disease were treated with combinations of systemic therapies and local therapies. Systemically, chemotherapeutics such as dacarbazine (DTIC), taxanes, temozolomide, or cisplatinum were administered, of which DTIC had the highest response rates of approximately 10%–15%.6,7 Between 1970 and 2000, early immunotherapeutic agents became available, such as Bacillus Calmette-Guérin (BCG), high dose interferon-alpha (HD IFNα) and interleukin-2 (IL-2).8 –11 Despite high toxicity rates and marginal efficacy, the introduction of these agents was the first step toward immunotherapy as we know it. Combinations of chemotherapy and immunotherapy only slightly increased response rates and the need for additional local treatment persisted. 6 Surgical resection resulted in 5-year survival rates of up to 25%; 7 however, these results were usually achieved in highly selected patients with isolated metastases or stage M1a disease.
In the early 2010’s, effective systemic therapies became available in clinical practice in Europe. Ipilimumab, an anti-CTLA4 antibody, first received approval by the European Medicines Agency (EMA) in 2011, quickly followed by BRAF inhibitors vemurafenib and dabrafenib in 2012 and 2013, respectively.12 –14 Subsequently, MEK inhibitor trametinib (2014), the anti-PD1 antibodies pembrolizumab and nivolumab (2015) and combination regimens were approved by the EMA and became available in clinical practice (Table 1).15 –17
Effective systemic therapies and year of EMA approval.
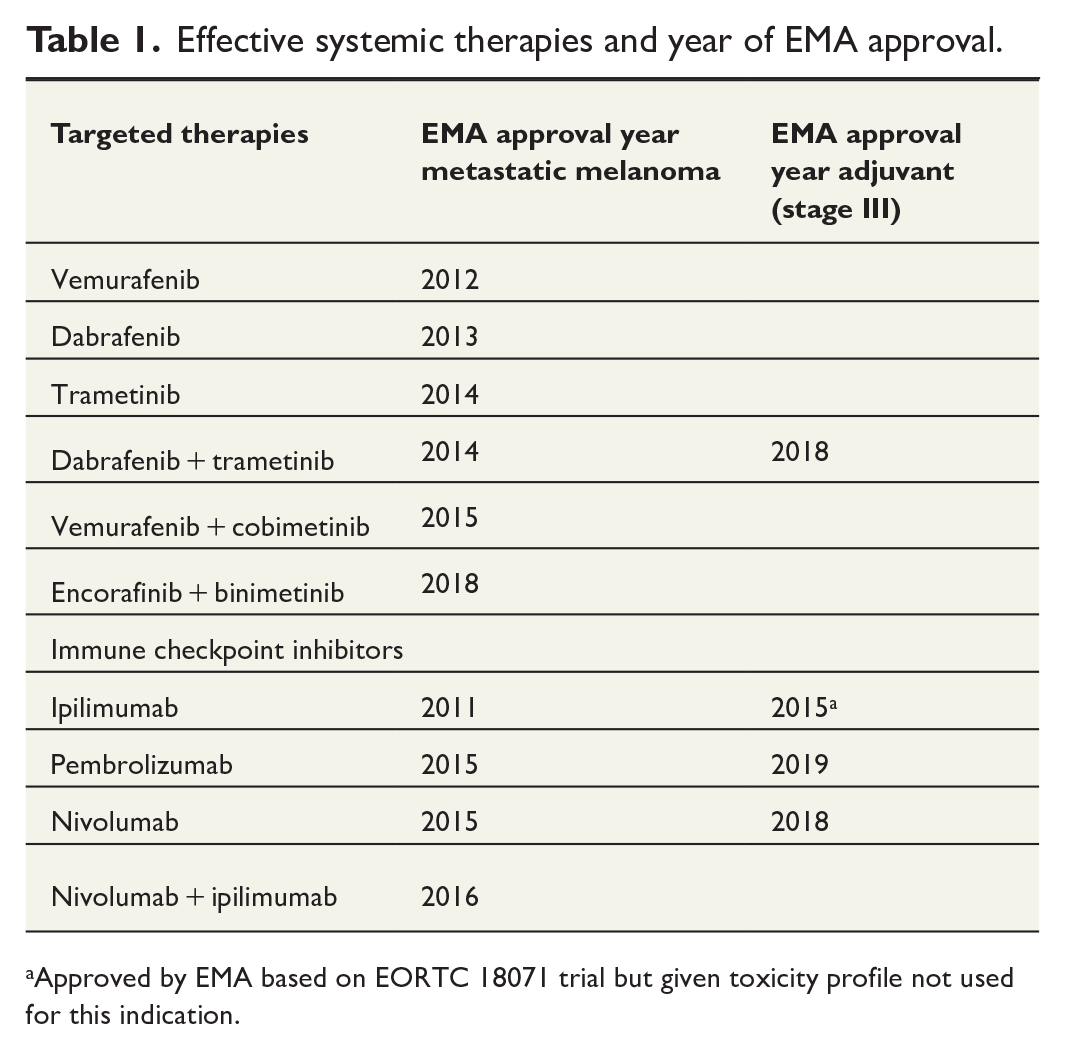
Approved by EMA based on EORTC 18071 trial but given toxicity profile not used for this indication.
With the introduction of these effective systemic therapies, the role of surgery in patients with distant metastases almost disappeared. This is now however being reconsidered and the interest in surgical intervention is increasing. 18 The most recent European Society for Medical Oncology (ESMO) guidelines state that surgery remains an option in selected patients, preferentially combined with adjuvant or neoadjuvant systemic therapies in a clinical trial setting. 19 Currently, there is no established gold standard for surgical management in stage IV disease. The aim of this narrative review is to discuss the role of surgery in the current era of effective systemic therapies, highlighting indications, outcomes, and integration with systemic treatment approaches.
Historical role of surgery in metastatic melanoma
In the era before effective systemic treatment options, surgery played a central role in the treatment of metastatic melanoma. Surgery in metastatic disease can be described as having three main purposes: symptom relief, debulking, or curation. Palliative surgery in case of symptomatic lesions has been an important treatment modality both before and in the era of effective systemic therapy. As effective systemic treatment prolongs survival in a patient population with advanced disease, the role of (local) palliative therapy potentially increases. 20 When considering palliative surgery, it is crucial to balance the potential risks of surgical complications against the burden caused by the symptomatic lesions. For example, symptoms that may warrant resection are bleeding gastro-intestinal (GI) tract lesions, obstructing GI tract lesions or nodal lesions causing pain, issues in wound management and/or mobility problems. The risk of surgical complications depend on the localization of the lesion, the size of the lesion, the general condition of the patient and the surgical approach—whether the procedure is open or laparoscopic, and whether it is performed in an emergency setting or electively. 21 In the last 40 years, surgical techniques have become increasingly safe with the introduction of less invasive techniques like laparoscopy in the 1990s and robot-assisted laparoscopy from 2010 onward. These advancements have contributed to a reduction in infectious complications, a decrease in intraoperative blood loss and shorter duration of hospital stay. 22 In addition to this, interventional radiological techniques, such as image-guided ablation and embolization have emerged as potential minimally invasive locoregional treatment options for metastatic disease.23,24
In a time without effective systemic therapies, complete surgical resection of all metastases offered the best chance for prolonged survival. 25 Only highly selected patients were amenable for this approach. Factors associated with an improved prognosis after surgery were a longer disease-free interval after resection of the primary melanoma and lower stage at initial presentation, suggesting a relatively mild tumor biology. Hence, if the surgery had any real effect on survival, or just mirrored a selection bias, is still unclear.
Integration of surgery and effective systemic therapy
Since the introduction of effective systemic therapies, surgery temporarily diminished from the treatment of metastatic melanoma and was less frequently applied as a treatment modality. Surgery would still be the cornerstone of treatment of resectable stage III melanoma. However, it did not take long for effective systemic therapies to integrate in the treatment of earlier stages of melanoma (Table 1). In 2018, anti-PD1 therapy was approved as an adjuvant treatment modality after the Checkmate 238 trial and the Keynote 054 trial demonstrated an improved recurrence-free survival compared to ipilimumab and placebo, respectively.26,27
More recently (2024), neoadjuvant therapy with anti-PD1 monotherapy and anti-PD1+ anti-CTLA4 combination therapy have become available after two positive trials, the SWOG 1801 and the NADINA trial, in patients with macroscopic stage III disease.28,29 In both the adjuvant and neoadjuvant setting, the integration of surgery and systemic therapy leads to an improved prognosis for patients with stage III melanoma.
While this synergistic approach between surgery and systemic therapies is well-established in earlier stages of melanoma, there is no gold standard regarding the use of surgery in metastatic melanoma in the context of effective systemic therapies.19,30 Nevertheless, surgery continues to have a valuable role in specific scenarios. An example of successful integration of surgery and systemic therapy is the management of melanoma brain metastasis, which historically respond relatively poorly to systemic therapies. 31 Surgery or stereotactic radiosurgery combined with combination therapy of ipilimumab and nivolumab are associated with an improved OS compared to ICIs alone.32 –34 The liver is another metastatic site that typically shows poorer response to ICIs compared to other visceral organ sites and is associated with a higher hazard of death. 35 A systematic review by Yeo et al. 36 suggests that surgical intervention for liver metastases may improve survival. However, the role of local treatments, such as surgery or ablative therapies, in the management of liver metastases in the era of ICIs remains unclear. Prospective clinical trials are needed to better evaluate the efficacy of integrating local treatment with modern systemic therapies for metastatic melanoma involving the liver.
Current role of surgery in metastatic melanoma
Despite the fact that the debate on the issue of surgery in metastatic melanoma is ongoing, there have been efforts to retrospectively study the use of surgery in the current era. 25 Generally, resection of melanoma metastasis is performed with either a therapeutic intention, aimed at curative outcomes, or with palliative intention, focused on symptom management and improving quality of life. Palliative metastasectomy, resection of symptomatic lesions, is done to relieve patients of their symptoms, and is generally not associated with an improved survival, 37 as there are often other metastases which determine the prognosis. However, in case of resection of a solitary lesion, which happens to be symptomatic, survival can be improved. Several retrospective cohort studies have identified resection to no evidence of disease (NED) as a factor associated with improved OS.38 –43 Resection to NED is often performed in a therapeutic setting and requires a highly selected patient population. To be eligible for resection to NED, a patient can only have one or a few metastatic lesions. This is the case in patients with solitary progression after a complete response (CR) to ICIs, oligoprogression after a CR to ICIs or oligometastatic disease. Although oligometastatic disease is defined as metastatic disease with no more than five lesions, 44 more lesions could be resected at the treating surgeon’s discretion. In addition to the number of lesions to be resected, it is crucial that lesions are amenable to complete resection and that the patient is sufficiently fit to tolerate the surgical procedure. Another factor associated with an improved survival in patients undergoing surgery for metastatic disease is treatment response prior to surgery. Blankenstein et al. and Bello et al. both show that response to ICI is significantly associated with survival outcome. Specifically, patients with progression of completely resectable disease perform worse in terms of outcome than patients with response of completely resectable disease, even though both have been resected to NED.38,39
Surgical outcomes other that NED are non-progressive residual disease (NPRD) and progressive residual disease (PRD). Patients with PRD understandably have worse survival outcomes and often undergo surgery in palliative setting. In case of a partial response (PR) or a stable disease (SD) to ICIs with solitary progression, patients can be resected to NPRD. Several retrospective studies show a progression-free survival of >12 months; however, an OS benefit cannot be proved with these data.41,45,46 Figure 1 depicts an overview of surgical indications and outcomes in patients with metastatic melanoma.
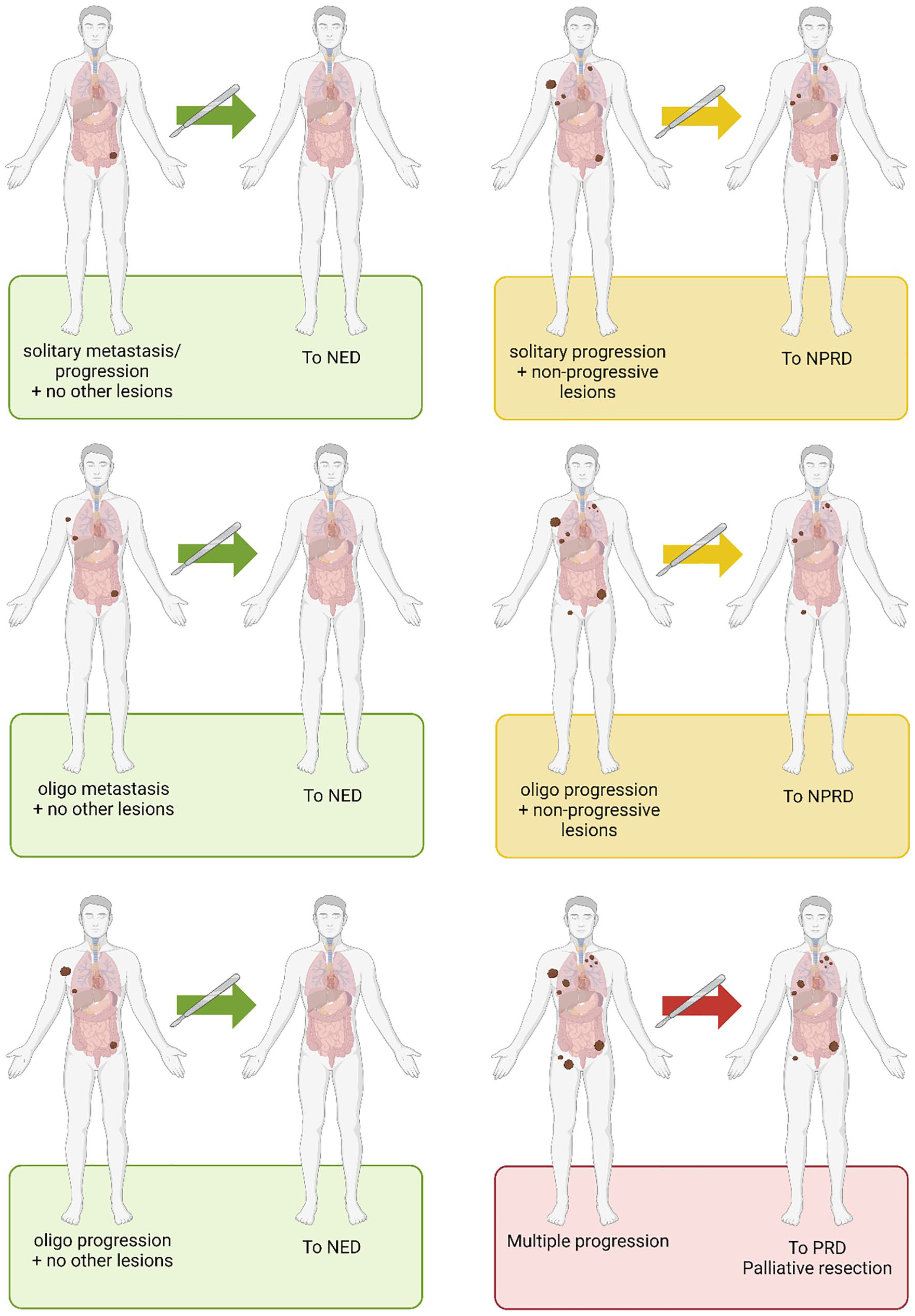
Schematic overview of surgical indications and outcomes in metastatic melanoma.
Challenges and future directions
Several challenges in defining the role of surgery in metastatic melanoma persist, primarily revolving around patient selection and the optimal timing of intervention. To identify appropriate candidates for surgery requires careful evaluation of the patient’s performance status to ensure that they are fit for the procedure. Equally important is determining the purpose of the surgery—whether it is palliative or therapeutic. For palliative resections, balancing the risk of surgical complications against the severity of the patient’s symptoms is critical for improving quality of life without undue harm. In cases of M1a disease, involving distant metastases to the skin, subcutaneous tissue, or lymph nodes, surgical risks are generally low, dependent on the size of the lesions. However, in M1b-d disease, where metastases involve visceral organs or the central nervous system, surgical planning requires meticulous consideration of both anatomical and systemic factors. When surgery is intended to be therapeutic, the goal shifts toward achieving long-term disease control, necessitating a multidisciplinary approach to integrate surgical intervention with systemic therapies.
The optimal timing and sequence of surgery and systemic therapy is another point of debate. Evidence from trials on neoadjuvant immunotherapy show that ICIs may demonstrate enhanced efficacy when the tumor remains
Currently, no randomized data exist, and the available evidence on the role of surgery in metastatic melanoma is scarce and of retrospective nature. Randomized trials are needed to draw robust conclusions on the added value of metastasectomy in stage IV melanoma. Designing clinical trials to evaluate the role of surgery in metastatic melanoma presents several challenges, beginning with the need to precisely define the study population. In the era of long-term “survivors” following treatment with ICIs, careful patient selection is vital. Notably, for patients with durable control through ICIs alone, surgery may not improve survival outcomes and could pose unnecessary surgical risks. Therefore, distinguishing between patients who might benefit from surgical intervention and those for whom systemic therapy alone suffices is essential to optimize outcomes and minimize harm. In practice, this may mean that a potential subject should achieve at least an SD or a PR to therapy or achieve a CR with subsequent solitary or oligo progression. When a potential subject is identified, resectability of lesions should be discussed in a multidisciplinary team meeting, featuring at least a surgical oncologist, medical oncologist, radiologist, and a radiotherapist. To rigorously compare surgery to the current gold standard, a randomized trial should include two arms. One with ICIs alone and another with ICIs combined with surgery. Progression-free survival and OS would serve as primary endpoints, but secondary outcomes like surgical complications, post-treatment recovery and health-related quality of life should not be overlooked. An example of such a trial is the SUMMIST trial, 47 unfortunately the trial closed prematurely due to low patient recruitment, showing the true necessity of multidisciplinary and multicenter collaboration to perform such trials in a quickly moving therapeutic landscape. Since most eligible patients are identified by medical oncologists, their active involvement and strong commitment are essential for the success of such trials.
Conclusion
The role of surgery in metastatic melanoma remains subjective to debate, but evidence suggests its potential benefits when integrated with systemic therapies in well-selected patients. Retrospective studies consistently identify resection to NED and response to ICIs prior to surgery as a significant factor associated with an improved OS. For patients with PR or SD, resection to NPRD may provide progression-free survival benefits, but data supporting OS improvement are limited. Prospective trials are required to define the precise role and timing of surgery in metastatic melanoma, paving the way for more personalized and effective treatment approaches.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
