Abstract
Background and objectives:
Long-term studies exploring factors associated with glycemic control of type 2 diabetes mellitus (T2DM) after bariatric surgery are being requested. This prospective cohort study aimed to evaluate potential predictors of long-term glycemic control 5 years after surgery.
Methods:
Patients were operated between 2002 and 2014. Data were collected prospectively in a database after obtaining written informed consent from the patients. Surgical methods were sleeve gastrectomy (SG) or biliopancreatic diversion with duodenal switch (BPDDS). Possible predictors of postoperative long-term glycemic control (HbA1c) were investigated using multiple path regression analysis, which handles missing data.
Results:
A total of 181 patients were included consecutively, 87 after SG and 94 after BPDDS. The follow-up rate was 124/181 (69%), 57 (66%) after SG and 67 (71%) after BPDDS. We found that 39/57 (68%) of the patients who underwent SG and 54/67 (81%) of the patients who underwent BPDDS had remission of T2DM at 5 years. Lower preoperative HbA1c (P < 0.010), higher preoperative C-peptide (P = 0.004), greater percent total weight loss (P < 0.005), and the BPDDS procedure (P < 0.001) were associated with better postoperative long-term glycemic control (explained variance = 39.4%).
Conclusions:
Both procedures, especially BPDDS, were effective in achieving long-term glycemic control. Lower preoperative HbA1c, higher C-peptide levels, greater weight loss, and the BPDDS procedure were key predictors of better long-term glycemic control.
Keywords
Twitter/X summary
This study links preoperative HbA1c, C-peptide level, type of surgery, and weight loss to metabolic control (HbA1c) in patients with type 2 diabetes after bariatric surgery, supporting individualized assessments in treatment planning.
Context and relevance
A greater understanding of the factors influencing long-term glycemic control in patients with type 2 diabetes mellitus (T2DM) after bariatric surgery has been requested. In this observational study with 5-year follow-ups, 81% of the patients who underwent biliopancreatic diversion with duodenal switch and 68% of those who underwent sleeve gastrectomy obtained remission of T2DM. Long-term glycemic control (HbA1c) was associated with preoperative HbA1c levels, C-peptide levels, weight loss, and type of surgery, collectively explaining 39.4% of the variation. This study underscores the potential for individualized assessment of prognostic factors in patients with T2DM before selecting a bariatric metabolic procedure.
Introduction
Severe obesity, defined as body mass index (BMI) ≥40 kg/m² or ≥35 kg/m² with at least one obesity-related disease, can significantly impact a person’s life physically, mentally, socially, and economically.1–3 Bariatric surgery has been shown to improve health and increase life expectancy, particularly in patients with type 2 diabetes mellitus (T2DM).4,5 Therefore, surgical treatment of T2DM has become an option for patients with a BMI ≥35 kg/m² and may also be recommended for patients with a BMI of 30–35 kg/m².1,6
Bariatric surgery can reduce body weight through various mechanisms, including reduced food intake (restriction), decreased calorie absorption (malabsorption), and potentially through other mechanisms related to changes in gut peptides and body metabolism, which may differ between surgical methods. 7 Although improvement in glycemic control after bariatric surgery is closely related to weight loss, there are speculations about whether weight-independent mechanisms exist.7–9 Therefore, different types of bariatric surgery may improve glycemic control to varying extents due to differences in weight loss and possibly through other mechanisms.
Several models for predicting glycemic control after bariatric surgery have been published.8–11 Since most of these are based on 1-year follow-up data, more knowledge about factors associated with long-term glycemic control is requested. Such studies should be conducted in different populations and cultures to ensure validity. 12
The primary aim of this study was to explore factors associated with long-term glycemic control (HbA1c) in patients with T2DM, 5 years after sleeve gastrectomy (SG) and biliopancreatic diversion with duodenal switch (BPDDS) in a Norwegian cohort. Comparing SG to BPDDS also allowed the evaluation of a potential additional metabolic effect of the duodeno-jejunal exclusion in BPDDS. We also assessed the proportion of patients who achieved resolution of T2DM after 5 years.
Methods
Design, population, and ethics
This is a prospective cohort study with a 5-year follow-up of patients with T2DM who underwent bariatric surgery. The material is derived from a consent-based local quality registry at Førde Hospital, approved by the Norwegian Data Protection Authority in 2002 (ref. no. 282738). It is part of a larger research project approved by the Regional Committee for Medical and Health Research Ethics in Western Norway (ID:18371). The first patient was included in 2002, and the last in 2014. In the early years, all patients underwent BPDDS, but from 2006, some patients with less pronounced T2DM (preferably not using insulin and having a better long-term glycemic control) were offered SG.
In the initial years, all procedures were performed with open surgery using an upper transverse incision, and all the BPDDS procedures in this series were performed with open surgery. The gastric resection was performed loosely along a 40-Fr tube starting 3–4 cm from the pylorus. Total small bowel length was measured, and 60% of the upper small bowel (duodenum, jejunum) were excluded, leaving the lower 40% (i.e. 200–300 cm of ileum) in function. The common limb was 10% of the small bowel length (i.e. 50–100 cm of ileum). 13 The introduction of laparoscopic SG started in 2007. When SG was performed as a stand-alone procedure, a 32-Fr bougie was used for calibration, and the resection started 1–2 cm from the pylorus. 14
Variables
T2DM was defined as HbA1c ≥6.5% (≥48 mmol/mol) or the use of glucose-lowering treatment. 15 Diabetes status was categorized into non-diabetic, dietary, medication except insulin, or insulin treatment. Diabetes remission was defined as HbA1c <6.5% (<48 mmol/mol) without the use of glucose-lowering treatment. 11 The study evaluated predictors of metabolic control using HbA1c levels, expressed in their natural units, without applying a dichotomous cut-off. This approach was chosen because the sample size was modest, and continuous variables are more appropriate in such cases. 16 Larger sample sizes are needed to adequately explore dichotomous outcomes, such as the resolution of T2DM. Variables possibly related to metabolic control were chosen partly from published prediction models and partly based on our clinical observations (Table 1).9,11
Path regression model with HbA1c 5 years after surgery as the dependent variable (n = 181).
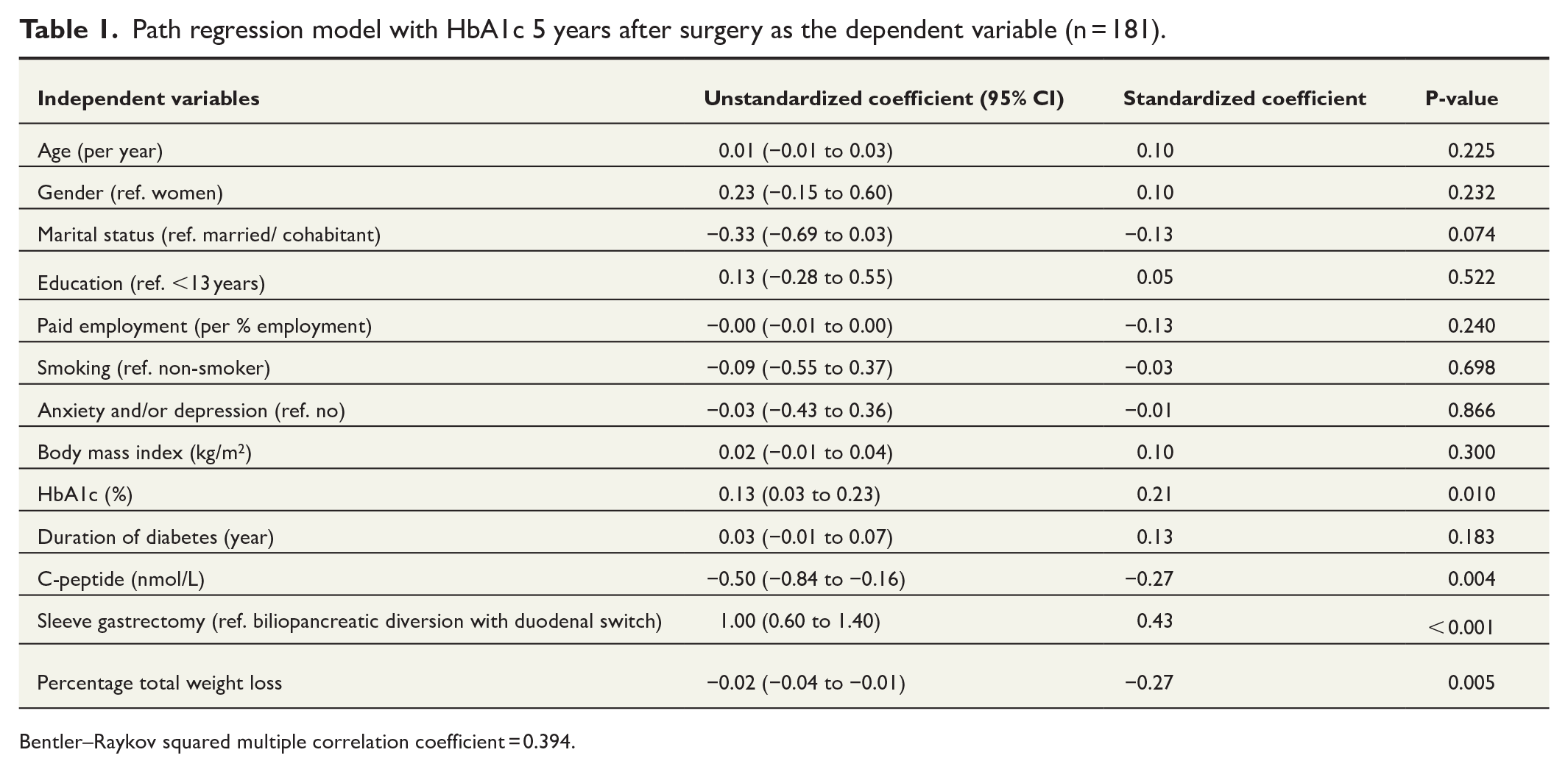
Bentler–Raykov squared multiple correlation coefficient = 0.394.
Analysis
Descriptive statistics were used to describe the sample stratified by surgical method. Drop-out analysis compared patients with and without data at 5 years based on the preoperative values in Table 2. An independent t-test was used for continuous variables, while the chi-square test was applied to categorical variables. Changes in clinical variables over time were investigated using longitudinal mixed-effect models that handle missing data. Changes in T2DM status before and after surgery were examined using McNemar’s test. Potential factors associated with postoperative metabolic control were examined using multiple path regression analysis because it handles missing data using full-information maximum likelihood. 17 Preoperative variables and postoperative percent total weight loss (%TWL) were included in the model. Two-sided P-values were considered continuous indicators of uncertainty, with values ≤0.05 indicating potentially robust estimates. 18 No adjustments were made for multiple comparisons. 19 Stata 16 was used for the multiple regression path analysis, while IBM SPSS Statistics 26 was used for the other statistical analyses.
Description of the sample (n = 181).
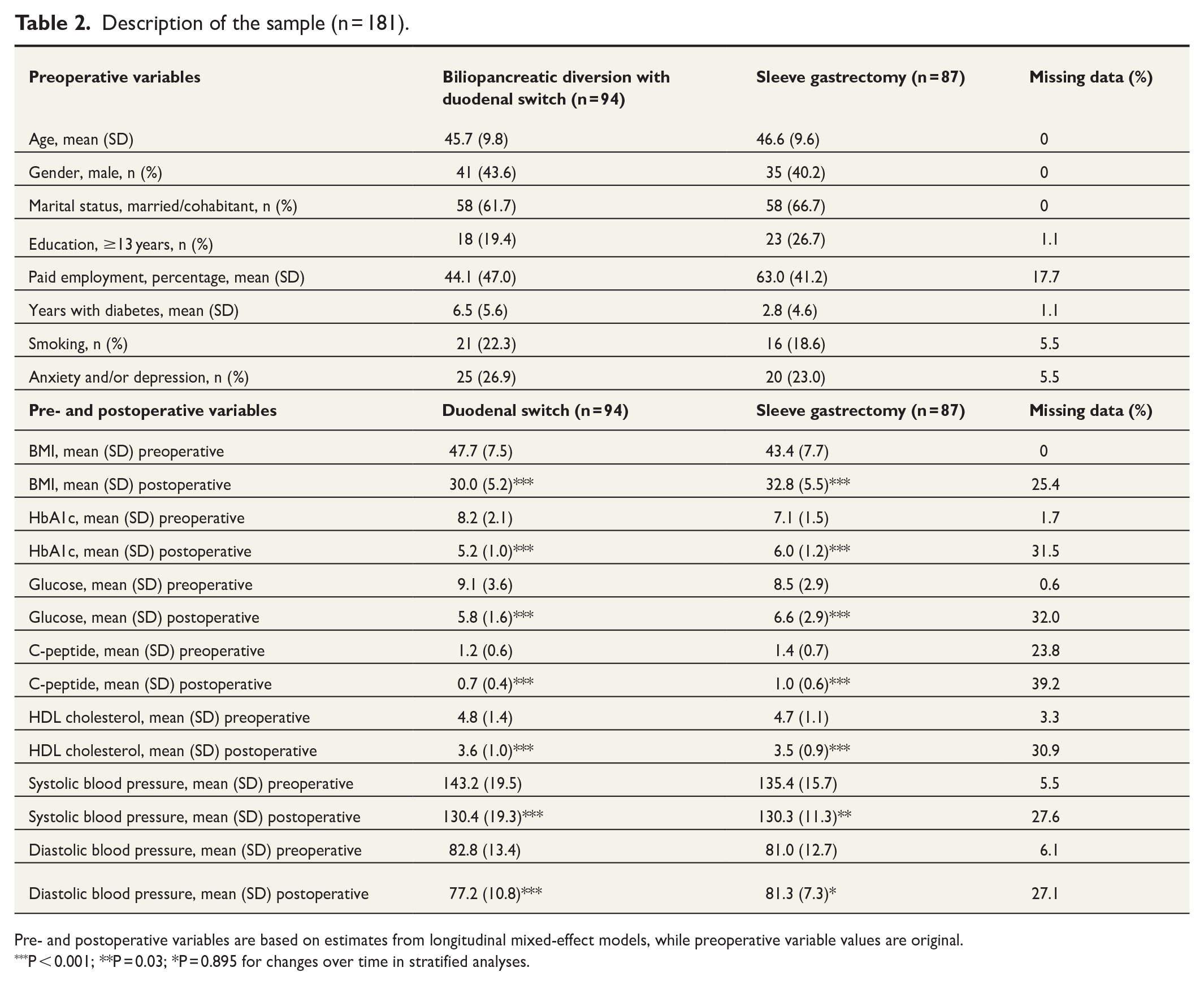
Pre- and postoperative variables are based on estimates from longitudinal mixed-effect models, while preoperative variable values are original.
P < 0.001; **P = 0.03; *P = 0.895 for changes over time in stratified analyses.
Results
The register had an inclusion rate of 95% (n = 181), and 124 of the 181 patients (69%; 67 BPDDS and 57 SG) had data at the 5-year follow-up (Table 2). Drop-out analysis revealed that patients with missing data at 5 years had a lower preoperative HbA1c (Cohen’s d = –0.36, P = 0.041). No significant differences were observed for other variables (P-values ranged from 0.095 to 0.810).
Table 2 presents demographic and clinical data. The preoperative prevalence of T2DM among all bariatric surgery patients at the hospital from 2002 to 2014 was 26%. Long-term glycemic control and other key clinical variables showed clear improvement (Table 2). Patients who had BPDDS lost more weight than those who had SG (P < 0.001, Table 3). We found that 81% of patients who underwent BPDDS and 68% who underwent SG had remission of diabetes 5 years postoperatively (Table 4).
Distribution of percent total weight loss (%TWL) in patients undergoing biliopancreatic diversion with duodenal switch and sleeve gastrectomy 5 years after surgery.

%TWL was calculated with complete data for 75 patients with biliopancreatic diversion with duodenal switch and 60 patients with sleeve gastrectomy. Biliopancreatic diversion with duodenal switch: mean %TWL = 36.2 (SD, 1.3). Sleeve gastrectomy: mean %TWL = 23.7 (SD, 1.6).
Diabetes status preoperative and postoperative after 5 years.

P < 0.001 for changes in diabetes status over time in both groups, McNemar’s test.
Lower preoperative HbA1c (P < 0.010), higher preoperative C-peptide levels (P = 0.004), greater weight loss (P = 0.005), and undergoing BPDDS (P < 0.001) were associated with better postoperative long-term glycemic control (explained variance = 39.4%) (Table 1). The number of years with diabetes and preoperative C-peptide levels were negatively correlated (r = –0.50, P < 0.001). Without C-peptide in the model, the number of years with diabetes was significantly associated with worse postoperative long-term glycemic control, but not when C-peptide was included in the analysis (data not shown).
Discussion
We found that 81% of patients who underwent BPDDS and 68% who underwent SG experienced remission of T2DM 5 years postoperatively. Preoperative HbA1c and C-peptide levels, weight loss, and the type of surgery were associated with long-term glycemic control. The treatment outcomes in this study are favorable compared to the literature, and the factors associated with remission are consistent with previous findings.4,5,7–9,12,20–25
Lower preoperative HbA1c and higher C-peptide levels were significant predictors of better long-term glycemic control, which aligns with previous findings.20,24,25 This suggests that patients with better preoperative glycemic control and insulin production are more likely to achieve remission of T2DM after bariatric surgery. The number of years with T2DM was associated with lower C-peptide levels, indicating reduced insulin production over time. These findings highlight the importance of early surgical intervention in achieving metabolic control, as demonstrated by the Scandinavian Obesity Surgery Registry. 26
Greater weight loss was associated with better postoperative glycemic control. This is consistent with existing knowledge about how weight loss impacts insulin resistance and long-term glycemic control. 22 After SG, a large variation in weight loss between institutions has been demonstrated. 27 The remission rate for T2DM after BPDDS in this study is consistent with others, while the remission rate after SG is higher than in most studies.28,29 This could be due to the significant weight loss observed after SG in this study, which may be linked to a more radical gastric resection. Differences in preoperative diabetic status and how T2DM remission is defined are other possible explanations for differences in remission rates between studies.28,29 A significant role of weight loss in glycemic control underscores the need to support patients in achieving and maintaining weight loss after surgery.
The BPDDS was associated with better long-term glycemic control than SG, even after adjusting for weight loss. This suggests that excluding 60% of the upper small bowel and stimulating the terminal ileum create a weight-independent mechanism for remission of T2DM. There is a paucity of long-term studies exploring potential mechanisms for T2DM remission after bariatric surgery, particularly after BPDDS. 30 A study by Elias et al. 31 found improved glucose homeostasis and reduced insulin resistance in association with lower levels of leptin and glucose-dependent insulinotropic polypeptide (GIP), and higher levels of glucagon-like peptide 1 (GLP-1) and peptide YY (PYY) eight years after BPDDS. Although the metabolic improvement is believed to be mainly due to the incretin effect of GLP-1, other mechanisms like a lower glucose absorption capacity of the ileum as compared to the duodenum/jejunum and bile acid stimulation of the terminal ileum causing reduced hepatic glucose production and increased tissue uptake of glucose could play a role. 32 Interestingly, a weight loss-independent effect on T2DM was not found for the Roux-en-y gastric bypass compared to the restrictive methods of gastric banding and vertical banded gastroplasty in the Swedish Obese Subjects study 2 and 10 years after surgery. 33 This could have practical implications when choosing a surgical method for patients with severe T2DM.
The strengths of the study include an extended follow-up period and considerable variation in the values of the included variables. A limitation is the follow-up rate of 68.5%. This is not uncommon in such studies, but there is uncertainty about the prognosis for those for whom we lack 5-year data. In addition, unknown factors could still influence the results while the regression model handled missing data. The finding that lower preoperative HbA1c was associated with a higher drop-out risk may be reassuring for the validity of this study, as the opposite pattern could have raised more concerns. However, the underlying reasons for this association remain unclear. It is also important to note that this study is based on a population from one hospital in Norway, and the findings may not be generalizable to other populations. We did not test the sample for interaction effects, and stratified additional analyses based on surgical methods should be conducted in future studies with larger samples. Traditional BPDDS using two anastomoses, as performed in this study, is less used today. Instead, the procedure has evolved into the single-anastomosis duodenal switch (SADI-S), which reduces operating time and short- and long-term risks while still achieving similar weight loss and effectiveness in treating obesity-related disorders as the classic BPDDS.34,35
Conclusion
This study highlights the potential for individualized assessment of prognostic factors for metabolic control in patients with T2DM before choosing a bariatric metabolic procedure. A longer diabetes duration, lower C-peptide levels, and a higher HbA1c could be arguments for using a duodenal switch configuration, which would be the SADI-S today. Future research should investigate preoperative predictors in larger and more diverse populations and the mechanisms underlying the observed associations.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Research ethics and patient consent
The data for this study were derived from a consent-based local quality registry at Førde Hospital, approved by the Norwegian Data Protection Authority in 2002 (reference no. 282738). All patients provided their informed written consent. This registry is part of a larger research project approved by the Regional Committee for Medical and Health Research Ethics in Western Norway (ID: 18371).
Data availability statement
Due to legal restrictions, the data utilized in this article cannot be publicly disclosed. However, the corresponding author will make the data available upon reasonable request.
