Abstract
Background and aims:
Neoadjuvant short-course radiotherapy (SCRT) prior to surgery has been used to reduce local recurrences in moderately advanced rectal cancer (RC). Total mesorectal excision (TME) surgery by itself can reduce the local recurrence rate in these patients. The objective was to assess whether discontinuing neoadjuvant SCRT had a negative impact on survival and local recurrence rates of moderately advanced RC.
Methods:
This retrospective study examined 137 consecutive patients (67 neoadjuvant SCRT, 70 TME without neoadjuvant therapy) treated for rT1-3N1M0 RC without extramural venous invasion (EMVI) or a threatened surgical margin between January 2016 and March 2020 in a tertiary rectal surgery referral center. The primary objective was the impact of neoadjuvant therapy on overall, cancer-specific, and disease-free survival, and local recurrence rate. Secondary outcomes were risk factors affecting survival. The analyses were conducted on the total study cohort as well as a propensity score matched cohort of the same patient group, with the matching predicated upon age, gender, and histological T score.
Results:
The oncological outcomes were similar between the two different treatment groups. The 5-year cumulative overall survival rate, calculated using the Kaplan–Meier method, was 89.6% in the neoadjuvant SCRT group and 83.5% in the surgery-only group (p = 0.570), with the corresponding cancer-specific survival rates at 97.0% and 98.4% (p = 0.219) and disease-free survival rates at 97.0% and 91.9% (p = 0.438). No local recurrencies were detected in either group during the 5-year follow-up period.
Conclusion:
The omission of neoadjuvant SCRT in rT1-3N1M0 RC with no EMVI and no threatened resection margin caused no adverse effects on the survival of the patients. Based on this series, it appears that avoiding SCRT does not compromise the oncological outcome in these patients.
Context and Relevance
Neoadjuvant therapy regimens for moderately advanced rectal cancer vary across countries. In Finland, neoadjuvant radiotherapy was previously given to patients with N1 disease without a threatened margin or extramural venous invasion (EMVI), but this practice was discontinued in 2018. However, in some other countries, these patients still receive neoadjuvant radiotherapy. We conclude that neoadjuvant radiotherapy may not affect survival and local recurrence in these cases.
Introduction
The introduction of the total mesorectal excision (TME) technique stands out as the most important advancement in the surgical management of rectal cancer in recent decades. TME has led to a notable reduction in local recurrence (LR) rates and a significant improvement in survival outcomes, even in cases where curative treatment is not pursued.1–3 Preoperative staging of rectal cancer is typically accomplished radiologically. Rectal magnetic resonance imaging (MRI) is recommended as the preferred method for assessing local staging, 4 while computed tomography of the thorax, abdomen, and pelvis is used to detect metastatic disease and critical comorbidities. 5
Early-stage rectal cancer can be effectively treated with surgery alone. 6 However, locally advanced disease harbors challenges, requiring a multimodal approach due to the complexities posed by surgical planning, high rates of locoregional recurrences, and metastatic disease development. 7 MRI serves as a reliable tool for assessing the extent of the primary tumor 8 and the presence of extramural venous invasion (EMVI). 9 However, MRI’s ability to detect local metastatic lymph nodes is limited.8,10 Coupled with mounting evidence indicating the greater prognostic significance of other mesorectal disease over lymph nodes, it has been concluded that the suspicion of few metastatic lymph nodes (rN1) alone does not reliably predict adverse prognosis. 11
Previously, in Sweden, patients with rT1-3abN1M0 rectal cancer without EMVI were typically treated with neoadjuvant short-course radiotherapy (SCRT), whereas in Norway, these patients underwent surgery without any neoadjuvant therapy. However, no significant disparities in oncological outcomes were observed between the two countries.12,13 Currently, these patients do not receive neoadjuvant therapy in the Scandinavian countries. By contrast, long-course chemoradiotherapy is recommended for patients with rT1-3N1M0 RC in the United States and Spain.14,15 In 2018, the Finnish national guidelines were revised to exclude SCRT from the treatment regimens for these patients.
The aim of this study was to determine whether there have been any changes in the oncological outcomes of overall survival (OS), cancer-specific survival (CSS), disease-free survival (DFS), and LR among patients with rT1-3N1M0 rectal cancer following the cessation of neoadjuvant SCRT in Finland.
Methods
Patient characteristics
A retrospective analysis of 137 consecutive patients who underwent TME for rT1-3N1M0 rectal cancer without mesorectal fascia involvement or radiological EMVI, at Helsinki University Hospital between January 2016 and March 2020 was performed. Data were retrieved from medical records, encompassing diverse information such as gender, age at diagnosis, histology of the biopsy, radiological assessment of tumor location, invasion depth, and metastatic lymph nodes, details of neoadjuvant therapy, type of surgery performed, locations of cancer recurrence, OS, and causes of death.
All patients underwent computed tomography (CT) scans of the chest, abdomen, and pelvis, as well as rectal MRI. Tumor histology was confirmed through preoperative endoscopic biopsies. All patients’ treatment plans were determined through multidisciplinary team meetings, which comprised colorectal surgeons, oncologists, an abdominal radiologist, and a pathologist. Adjuvant chemotherapy was considered if metastatic lymph nodes, lymphovascular or perineural invasion (pLVI/PNI), histological grade 3, tumor budding grade 3, or mucinous histology were detected in the surgical specimen.
All patients adhered to the follow-up protocol, which included clinical examinations, endoscopies, and serum carcinoembryonic antigen-level measurements in a surgical unit for 3 years. In addition, whole body CT scans were conducted at 1 and 2–3 years postoperatively for patients subjected to adjuvant chemotherapy. After 3 years, patients were yearly followed up with serum carcinoembryonic antigen measurements and clinical examinations by their primary care physicians for up to 5 years. No patients were lost to follow-up.
Data retrieval from patient records occurred at least 3 years post-surgery. The follow-up period was defined as the time, in months, from the date of surgery to the last healthcare interaction or time of death.
The study design and reporting adhere to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines, as recommended by the EQUATOR Network.
This study was approved by the Research Administration of Helsinki University Hospital (HUS 4/2024). As this was a retrospective register study with no patient intervention, ethics committee approval and informed patient consent were not required by Finnish national legislation.
Radiological technique
All patients underwent contrast-enhanced CT imaging in the portal venous phase using a multi-slice helical CT scanner. Rectal MRI was conducted using 3.0T or 1.5T scanners, incorporating T2-weighted imaging in three planes, along with diffusion-weighted imaging in the oblique plane perpendicular to the lumen of the bowel wall encompassing the tumor. Rectal filling was omitted; however, patients received a small enema before the scan to mitigate susceptibility artifacts. Interpretation of the images was performed by board-certified radiologists and reviewed by experienced abdominal radiologists for the multidisciplinary team meetings, with findings documented using a structured report template.
Statistical analysis
Propensity score matching was conducted using a 1:1 nearest neighbor matching algorithm without replacement with distances determined by logistic regression. Propensity score matching was predicated upon a set of covariates including age, gender, and T score (T1 or T2 vs T3).
Absolute standardized mean difference in distances between patients receiving neoadjuvant RT and surgery-only was found to be balanced after conditioning on the propensity score, where a difference of less than 0.1 in absolute standard mean difference after matching was considered to indicate a good balance.
Categorical analyses of patient and disease characteristics between the different neoadjuvant therapy groups were performed using the chi-squared test or Fisher’s exact test. Continuous variables were assessed for normality using the Kolmogorov–Smirnov test, and as they were normally distributed, they are expressed as the mean ± standard deviation (SD). Cumulative survival and recurrence distributions were analyzed by the Kaplan–Meier method and were compared using the log-rank test. In cases of missing data, these were excluded from the analysis to avoid bias. The statistical analyses were performed using IBM SPSS-Statistics software version 28 (IBM Corporation, Armonk, NY, USA). A p-value of <0.05 was used as a cutoff for statistical significance.
Results
Patient characteristics before and after propensity score matching
During this period, a total of 1,171 patients underwent rectal cancer surgery at the unit, of which 137 consecutive rT1-3N1M0, EMVI negative patients were included in the final analysis; 67 (48.9%) received neoadjuvant SCRT (5 × 5 Gray during the week preceding their surgery), and 70 patients were operated on without any neoadjuvant therapy. A total of 60 patients underwent surgery before the official therapy regimen change in April 2018, and 77 patients after the change. The two therapy groups were treated in parallel throughout the study period, with 15 (25.0%) patients undergoing surgery without neoadjuvant SCRT before April 2018, and 22 (28.6%) patients receiving SCRT after April 2018.
Before propensity score matching, the surgery-only group included statistically significantly more patients with adjuvant chemotherapy (71.4% vs 49.3%, p = 0.007), but this difference disappeared after matching. After propensity score matching, the material consisted of 55 matched pairs. These results are depicted in Table 1.
Baseline characteristics.
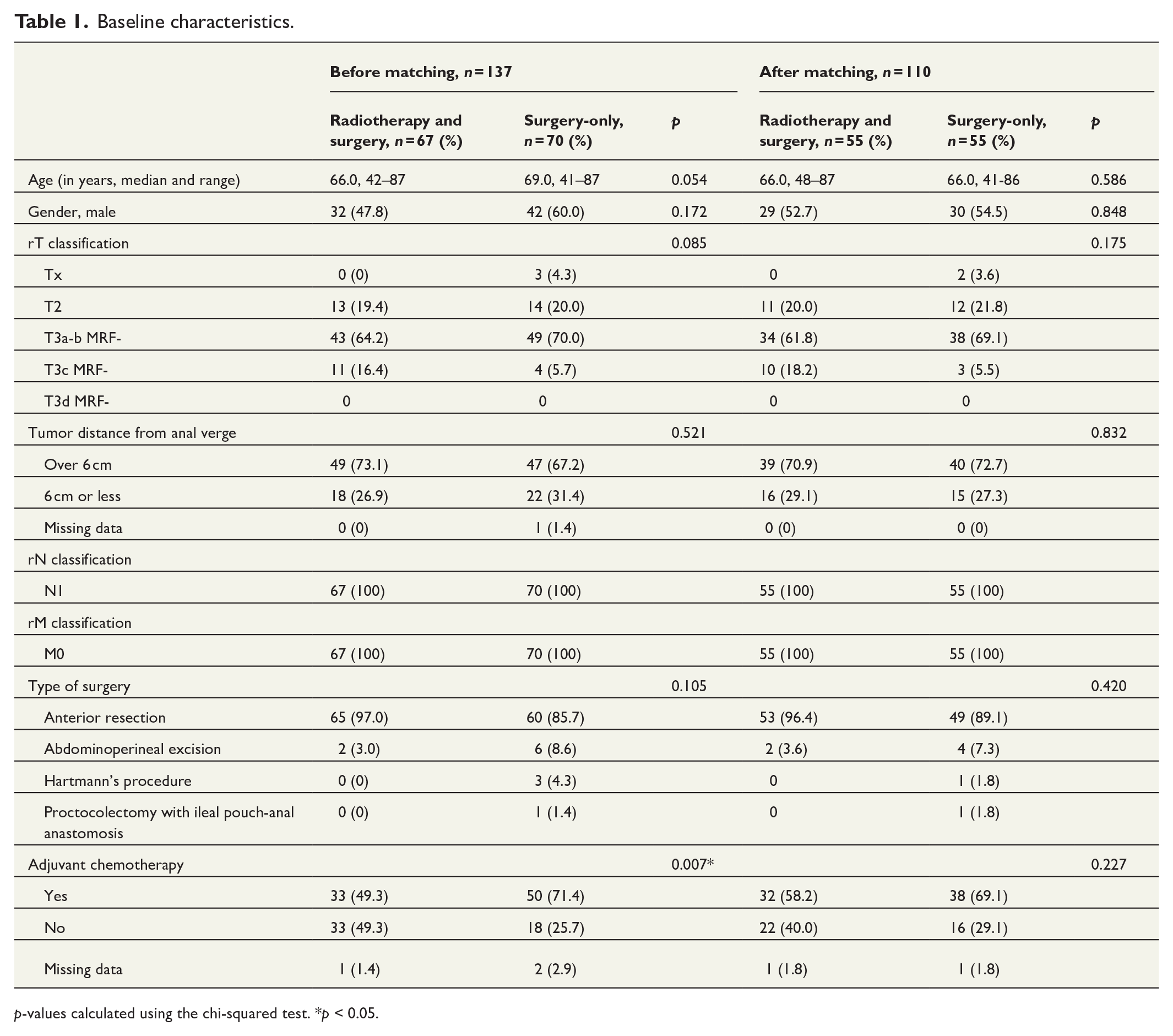
p-values calculated using the chi-squared test. *p < 0.05.
Disease characteristics and follow-up before and after propensity score matching
The differences were non-significant in terms of histopathological diagnosis, T stage in the surgical specimen, circumferential resection margin, and pLVI/PNI in the surgical specimen before and after propensity score matching. The surgery-only group exhibited a higher rate of N0 disease in the surgical specimen in comparison to the neoadjuvant SCRT before surgery group (p = 0.028). The difference disappeared after the propensity score matching. The results are shown in Table 2.
Histopathological characteristics of the tumors.
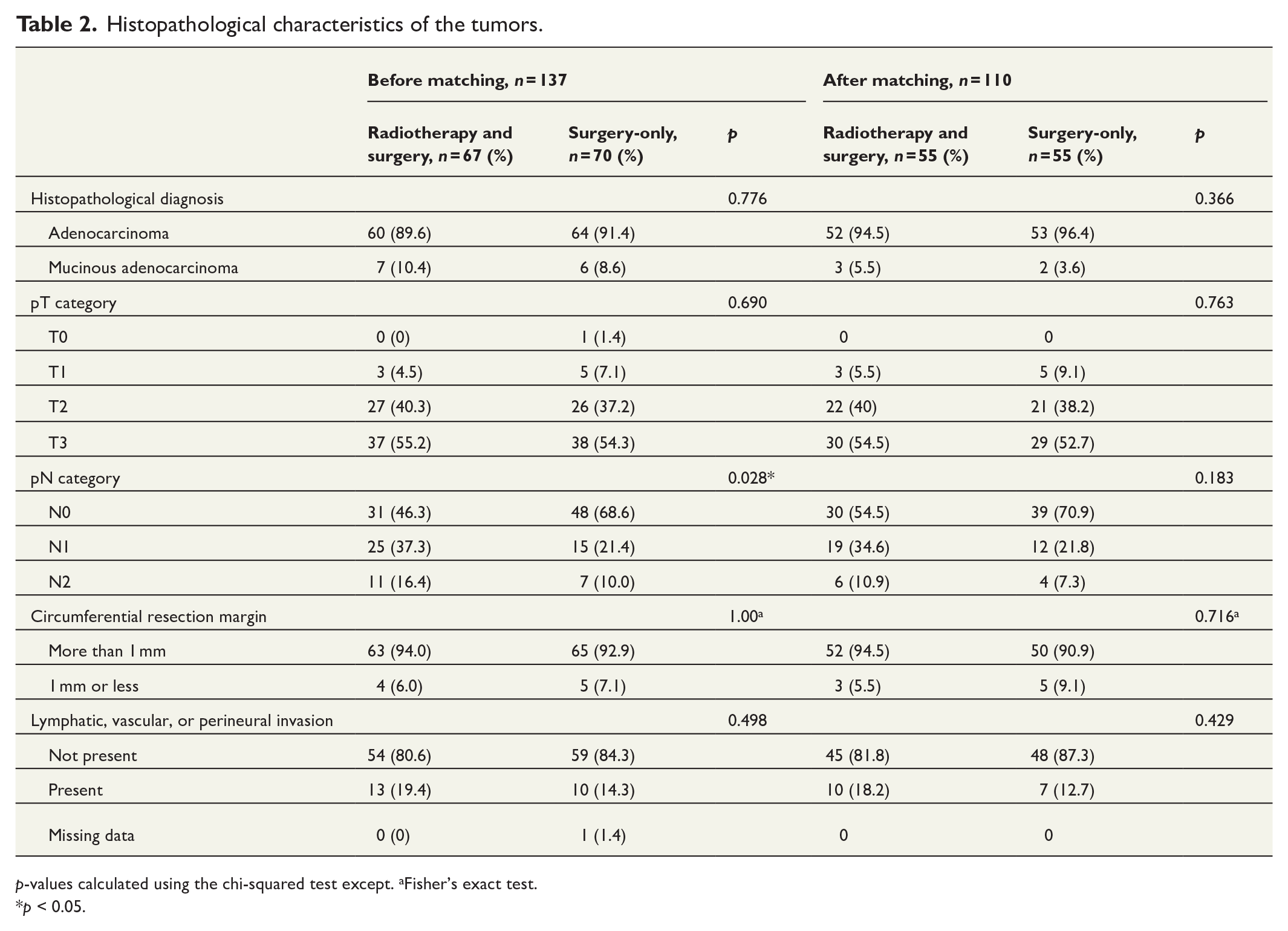
p-values calculated using the chi-squared test except. aFisher’s exact test.
*p < 0.05.
The overall mean follow-up duration for the entire study cohort was 63.6 ± 17.9 months: 71.9 ± 17.5 months for the neoadjuvant SCRT and surgery group, and 55.6 ± 14.4 months for the surgery-only group (p = 0.003). In the propensity score matched (PSM) population, the mean follow-up was 64.35 ± 17.20 months: 70.73 ± 18.32 months in the neoadjuvant SCRT and surgery group and 57.98 ± 13.38 months in the surgery-only group (p = 0.011).
Oncological outcomes
In the entire study cohort, the cumulative 3- and 5-year OS rates were 95.5% and 89.6% in the neoadjuvant SCRT and surgery group and 92.9% and 83.5% in the surgery-only group, respectively (p = 0.570), as depicted in Fig. 1. The cumulative 3- and 5-year CSS rates in the neoadjuvant SCRT and surgery group were 97.0% and 97.0%, and in the surgery-only group 100% and 98.4%, respectively (p = 0.219), as demonstrated in Fig. 2. The cumulative 3- and 5-year DFS rates were 97.0% and 97.0% in the SCRT and surgery group, and 100% and 91.9% in the surgery-only group, respectively (p = 0.438), as shown in Fig. 3. No LR were detected in either group during the 5-year follow-up period.

Cumulative OS rates in the different neoadjuvant therapy groups in the whole study cohort.

Cumulative CSS rates in the different neoadjuvant therapy groups in the whole study cohort.

Cumulative DFS rates in the different neoadjuvant therapy groups in the whole study cohort.
In the PSM cohort, the cumulative 3- and 5-year OS rates were 94.5% and 87.3% in the neoadjuvant SCRT and surgery, and 96.4% and 90.1% in the surgery-only group, respectively (p = 0.359). The cumulative 3- and 5-year CSS rates were 98.2% and 96.3% in the SCRT and surgery group, and 100% and 100%, respectively (p = 0.070). The cumulative 3- and 5-year DFS rates were 98.2% and 96.3% in the SCRT and surgery group and 100% and 92.8% in the surgery-only group, respectively (p = 0.955). No LR was detected during 5 years of follow-up.
Discussion
Previously, neoadjuvant SCRT was administered to patients with the main objective of preventing LR in moderately advanced rectal cancer, with factors such as rN1 with no additional mesorectal disease and an unthreatened circumferential resection margin. Earlier studies have demonstrated the efficacy of neoadjuvant SCRT in improving local disease control.16,17 However, these studies were conducted approximately 20 years ago, at the beginning of the TME era. Notably, in the Swedish study, the surgical techniques were not standardized. Meanwhile, in the Dutch trial, although TME was employed, the technique was still relatively novel to the surgeons. Recent studies have suggested that the universal adoption of the TME technique may diminish the potential benefits of neoadjuvant SCRT. 18 Our findings concerning moderately advanced rectal cancer did not reveal an increased rate of LR in the group that did not receive neoadjuvant SCRT prior to surgery, nor did we observe inferior outcomes in other survival parameters in the surgery-only group, aligning with previous research. 18 Across the entire study cohort and within the PSM subset, there were no statistically significant disparities in oncological outcomes between patients who underwent neoadjuvant SCRT and those who did not. Furthermore, omitting SCRT from the therapy regimen spares the patients from the side effects of the radiotherapy,19,20 while simultaneously reducing costs.
This study highlights established challenges associated with radiological diagnostics of metastatic lymph nodes.8,21 A modern rectal MRI protocol was uniformly employed for all patients, with interpretations by experienced abdominal radiologists indicating N1 status in all cases. However, surgical specimens from over two-thirds of the cases in the surgery-only group revealed no metastatic lymph nodes. In the group receiving neoadjuvant SCRT prior to the surgery, however, 46.3% of patients were reported to have a histological N0 status. This finding can suggest an enhancement in the technical quality of the images, resulting in the detection of more lymph nodes. The higher percentage of suspected lymph nodes in the surgery-only group could be attributed to the radiologists taking more time to adjust to the improved images.
We acknowledge several potential limitations of our study, including its retrospective design and a relatively small study cohort with no diagnosed LRs. To mitigate bias, we predefined our objectives and hypotheses before data collection, analyzed all consecutive patients treated during the study period, and controlled for confounding variables using propensity score matching. Despite our institution being a major rectal surgery center with over 300 annual operations, our efforts to ensure a uniformly adopted TME technique and a follow-up period of at least 3.5 years resulted in a total study cohort of 137 patients, with the PSM cohort comprising a relatively modest 110 patients. However, the study cohort is large enough to indicate that neoadjuvant SCRT may not offer benefit concerning oncological outcome in patients with rT1-3N1M0 RC without additional mesorectal disease or a threatened surgical margin.
In conclusion, cessation of neoadjuvant SCRT in rT1-3N1M0 rectal cancer with no EMVI and no threatened margin was not associated with adverse effects on the survival parameters of the patients in a retrospective analysis.
Footnotes
Author contributions
S.M. had full access to all data and takes responsibility for data integrity and accuracy of data analysis. Study concept and design: A.L. and S.M. Acquisition of data: S.M. Analysis and interpretation of data: S.M., L.K., and A.L. Statistical analysis: S.M. and L.K. Drafting of the manuscript: S.M. Critical revision of the manuscript for important intellectual content: L.K. and A.L.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partial financial support was received from the Helsinki University Hospital Research Fund. Open access funded by Helsinki University Library (open access licence CC BY-NC 4.0).
Clinical trial registration
N/a.
Trial registration and patient consent
This study was approved by the Research Administration of Helsinki University Hospital (HUS 4/2024). As this was a retrospective register study with no patient intervention, ethics committee approval and informed patient consent were not required by Finnish national legislation.
