Abstract
Background and aims:
The therapeutic management of fistulas presents significant challenges, often involving both conservative and surgical approaches. Despite these interventions, recurrence and postoperative mortality rates remain high. Endoscopic stent insertion into the fistula, along with the creation of a stent stoma, may offer a promising alternative for patients who fail surgical or conservative therapies. This study aimed to evaluate the feasibility, effectiveness, and safety of endoscopic stent insertion in the treatment of refractory small intestinal fistulas.
Methods:
Patients with refractory small intestine fistulas who underwent endoscopic stent insertion were included. The primary endpoint was defined as successful fistula treatment, which included an improvement in clinical symptoms related to the fistula, successful bridging to subsequent surgical revision, and the restoration of enteral nutrition. Secondary endpoints comprised the feasibility of the endoscopic procedure, complications, procedure-related complications, and in-hospital mortality.
Results:
Eight patients were included, with a median follow-up period of 2.7 months. The implantation of a self-expanding metal stent was successfully performed in all patients (technical success rate, 100%; n = 8/8). The clinical success rate was 87.5% (n = 7/8), indicating clinical improvement in fistula-related symptoms, wound care, and enteral nutrition. Procedure-related complications occurred in one patient (12.5%; n = 1/8), involving stent dislocation leading to small intestine perforation, which was managed endoscopically. No procedure-related mortality was observed.
Conclusions:
Endoscopic stent insertion is a feasible, effective, and safe option for the management of therapy-refractory small intestinal fistulas. The creation of a stent stoma improves patient quality of life.
Context and relevance
The management of small intestinal fistulas remains challenging and is associated with multiple complications. Endoscopic stent insertion into the fistula and the creation of a stent stoma may be a promising option for patients who fail surgical and conservative therapy. In this retrospective study, we evaluated the feasibility, effectiveness, and safety of endoscopic stent insertion in refractory small intestinal fistula treatment. Our findings highlighted the feasibility, effectiveness, and safety of endoscopic stent insertion. The quality of life of patients may be improved.
Introduction
Small intestinal fistulas are associated with high morbidity, with reported mortality rates ranging from 5% to 43%.1 –4 Primary complications include chronic and painful skin macerations from autolytic small intestine secretions, alongside the challenges posed by inadequate enteral nutrition. The management of this severe condition remains a difficult and interdisciplinary challenge.
Available management options for fistulas encompass conservative measures such as nutritional support, fistula output control, and meticulous skin care. 5 Vacuum therapy has emerged as an option in cases where conservative treatment fails. 6 Persistent intestinal fistulas often require surgical reconstruction, which may involve small intestine overstitching or resection. 5 However, surgical repair is challenging, as evidenced by a recurrence rate of 31%2,4,7 –11 and postoperative mortality rates ranging from 3.5% to 14%.12 –14 Factors such as extensive intestinal adhesions, peritoneal carcinosis, poor general health due to advanced malignancy or cachexia, and severe inflammatory changes related to small intestinal secretions can complicate surgical interventions, rendering them unfeasible in certain patients. In such cases, treatment options are largely limited to managing the defect through stoma care and costly dressing changes.
Given these challenges, endoscopic stent insertion into the fistula, along with the creation of a stent stoma, presents a promising treatment alternative for patients who do not benefit from surgical or conservative therapies.
The use of self-expanding metal stents (SEMS) has become a routine endoscopic procedure in the upper and lower gastrointestinal tracts for the recanalization of both malignant and benign stenoses, as well as the management of postoperative anastomotic insufficiencies or leaks. However, few studies have investigated the use of metal stents in the small intestine.
The primary objective of stent insertion into the fistula is to facilitate the controlled discharge small intestinal contents through a partially coated metal stent into a stoma, allowing for effective nursing care. This intervention aims to alleviate pain and improve the quality of life for affected patients.
This study aimed to determine the feasibility, effectiveness, and safety of endoscopic stent insertion for therapy-refractory small intestinal fistulas by analyzing the associated success rates, as well as morbidity and mortality outcomes.
Methods
Study design and population
All stent implantations were performed by an endoscopic surgeon across three different tertiary centers. The study included patients who met the following criteria: (1) therapy-refractory fistulas of the small intestine; (2) absence of further surgical treatment options; (3) no improvement in wound healing after weeks to months of conservative care; and (4) who underwent endoscopic stent insertion into the fistula or small intestine. Patients with other possible indications for stent implantation into a stoma, such as a parastomal abscess, ostomy retraction, stenosis, or tumor compression, were excluded.
The causes of the intestinal fistulas included anastomotic leakage, intestinal erosion due to tumor ingrowth, intestinal damage following adhesiolysis, and injury from midline abdominal wall closure. Fistulas were classified into three distinct types based on their characteristics:
Type I:
The intestinal fistula was located near the ostomy, allowing direct access via endoscopy. Indications for stent implantation included anastomotic leakage, peristomal abscess, or ostomy stenosis. A guide wire was placed orally over the fistula, followed by stent implantation under endoscopic guidance. The stent was positioned a few centimeters outside the ostomy to facilitate optimal wound dressing (Fig. 1).
Type II:
The intestinal leakage occurred in the midgut. Endoscopic access to the fistula via the ostomy was obstructed due to multiple angulations or strictures. A fine caliber endoscope was introduced into the defect or enteral cutaneous fistula under X-ray control to reach the origin of the defect. A guide wire was then placed into the oral part of the intestine under both endoscopic and X-ray guidance. Subsequently, the stent was implanted and expanded within the small intestine, leading to the body surface (Fig. 2). Intestinal contents could then be evacuated through the ostomy aboral to the stent or via regular defecation.
Type III:
The intestinal leakage causing peritonitis with abscess formation, which was inaccessible from the ostomy or anally, as described in Type II. Due to tumor stenosis or severe strictures, emptying of intestinal contents was not feasible. In these cases, a second stent (a covered bile duct stent with a 10-mm diameter) was placed in the aboral intestine (Fig. 3).
Diagnosis of leakage in the small intestine was confirmed using endoscopy and X-ray imaging. Data on patient demographics and clinical characteristics were obtained, along with information regarding leakage characteristics and procedural details from both surgical and endoscopic interventions. Follow-up was conducted until April 2024, tracking outcomes from the time of the initial therapeutic intervention to the last contact or patient death.
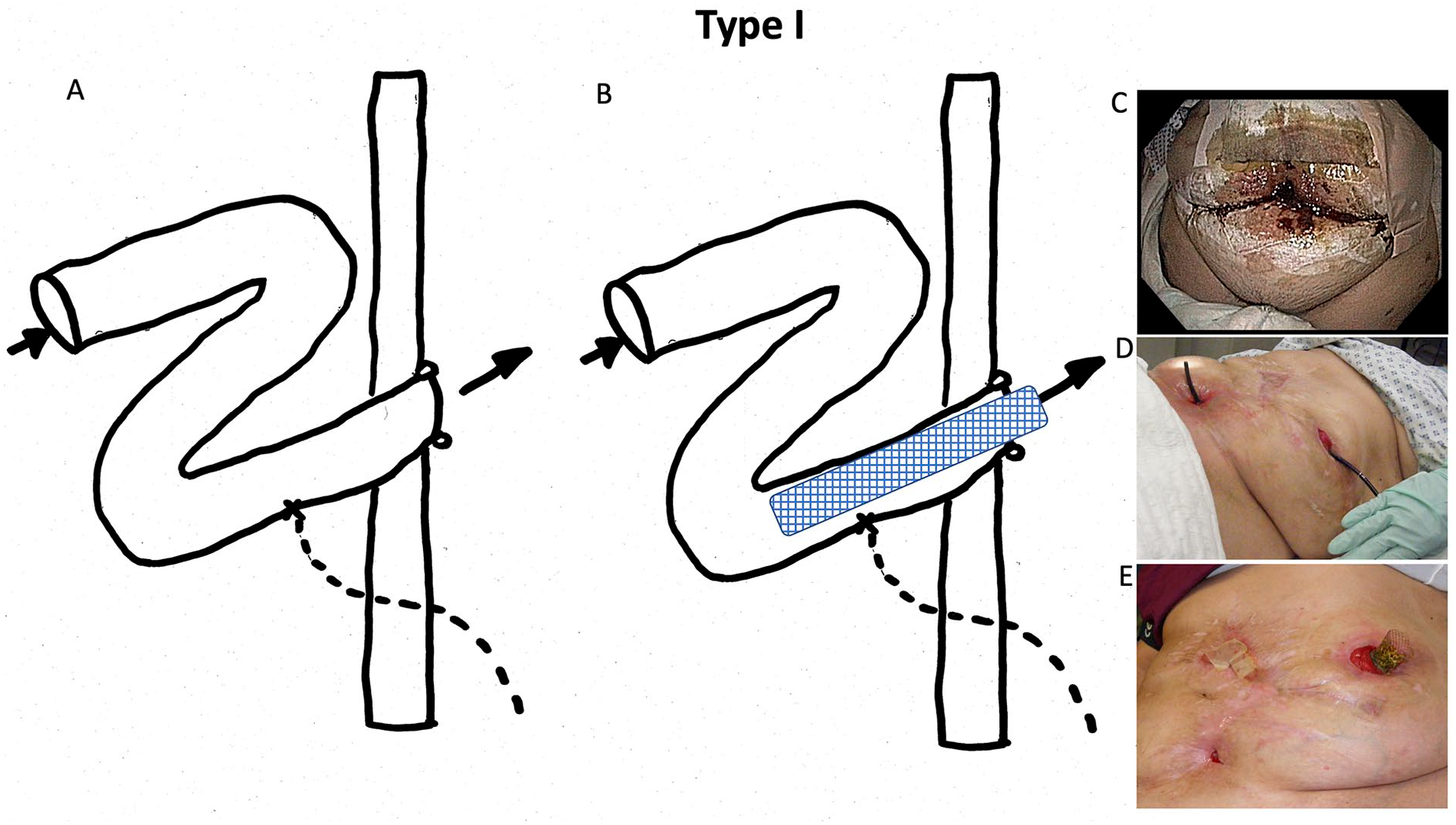
Fistula classification: Fistula Type I.
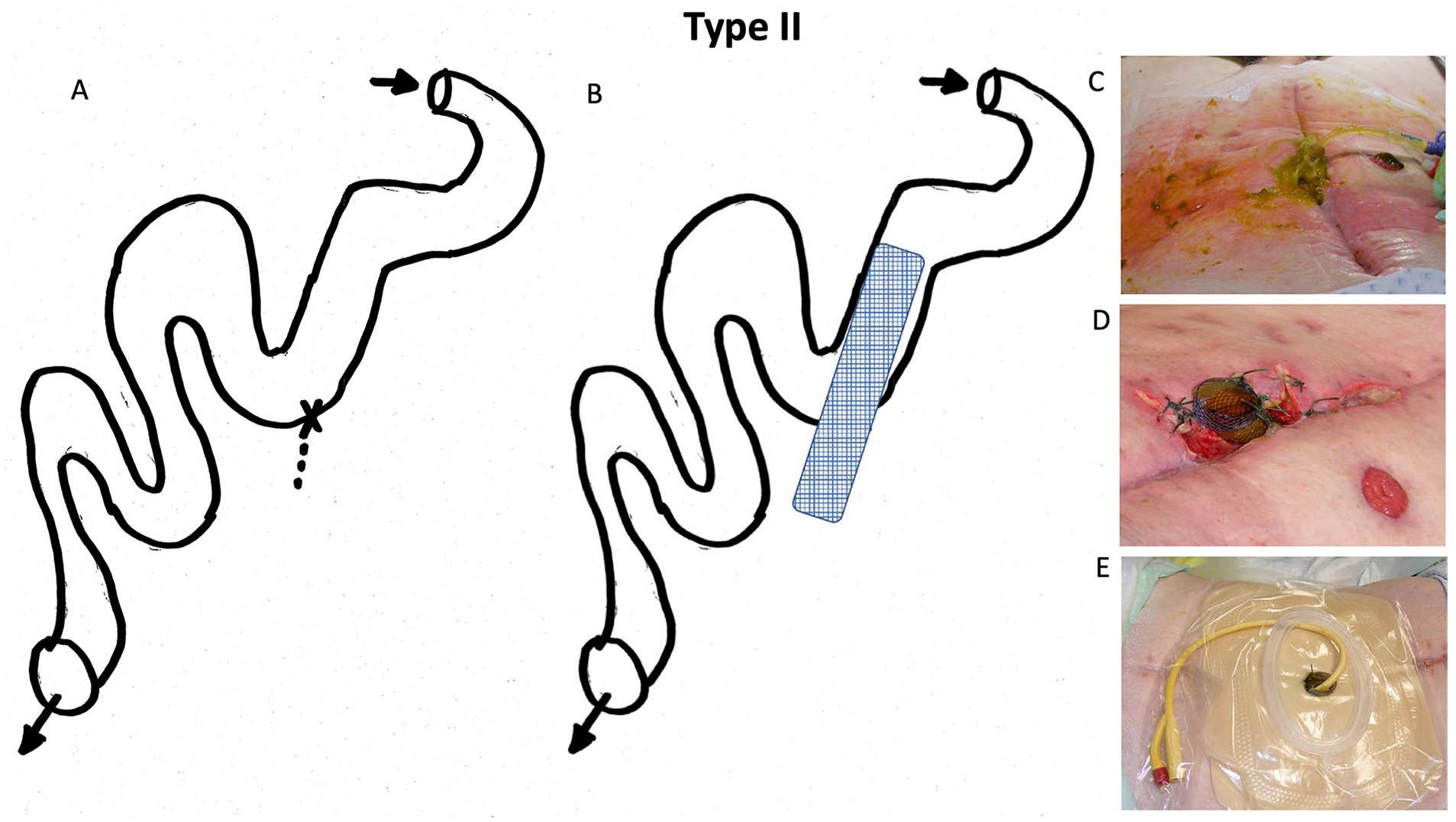
Fistula classification: Fistula Type II.
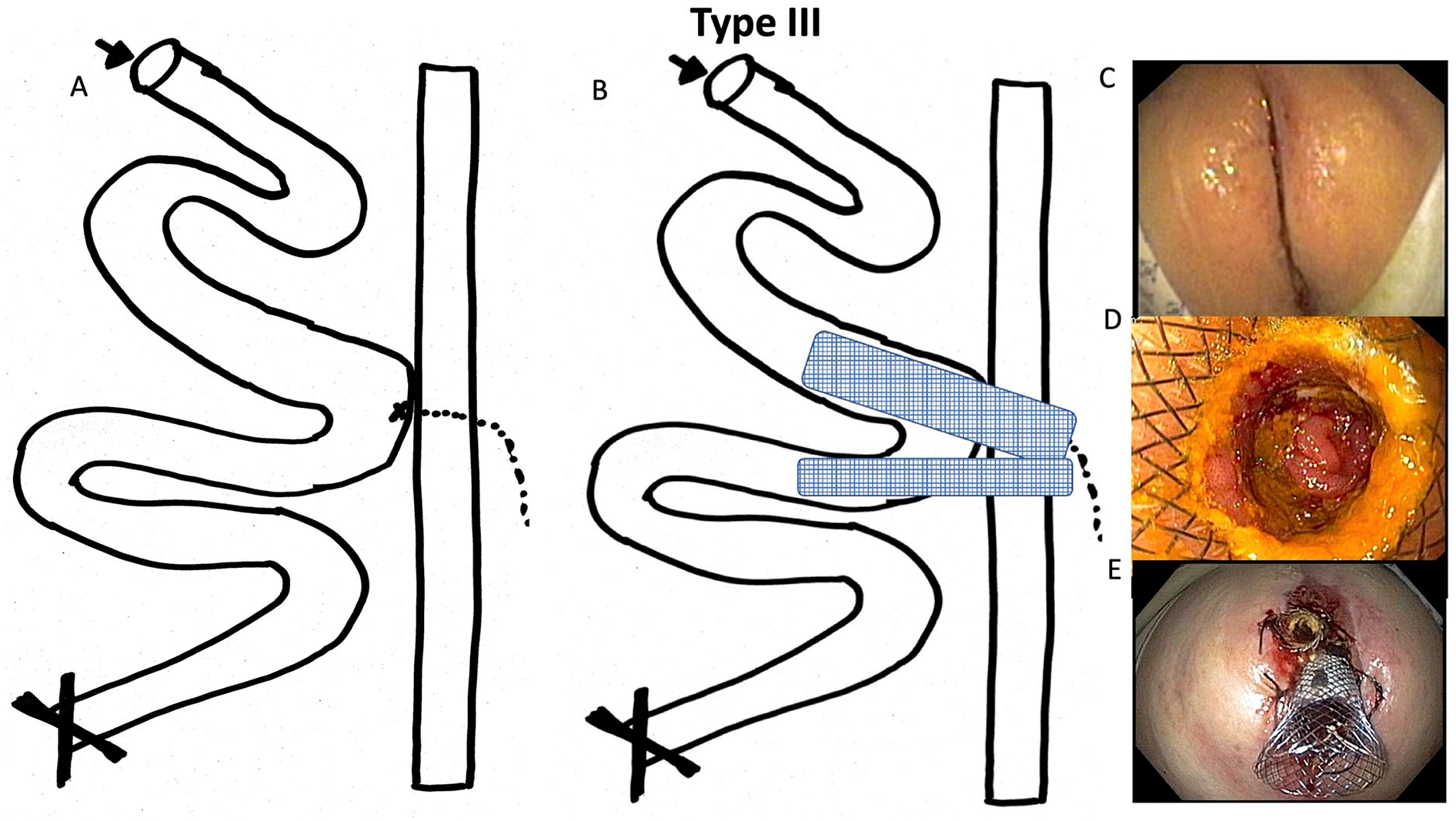
Fistula classification: Fistula Type III.
Endoscopic treatment
The anatomical status of each patient was evaluated radiologically through fistula filling, followed by endoscopic inspection using a 5.6-mm fine-caliber gastroscope (Olympus) with a 2.0-mm working channel. A standard gastroscope was used if the fistula was accessible via the ileostoma. Typically, the fistula opening served as the interventional entry point into the small intestine or fistula system. Following an evaluation of the skin, abscess/fistula system, and small intestine, a 0.035 inch guidewire (VisiGlide Guidewires, Olympus, PA, USA; Jagwire™ Guidewire, Boston Scientific, Marlborough, MA, USA) was placed approximately 30 cm deep into the small intestine under both endoscopic and radiologic guidance.
The type of stent to be implanted and the subsequent procedural approach were determined on a case-by-case basis, tailored to the anatomy of the leakage and the condition of the abdominal wall. In general, a partially covered SEMS with a shaft diameter of 20 mm and tulip diameter of 28–23 mm was selected. Stent length and diameter were individually adjusted to accommodate the patient’s natural anatomy and clinical situation. Stents such as the “Niti-S™ Esophageal Stent,” TaeWoong Medical, South Korea, or “WallFlex™ Esophageal Stents,” Boston Scientific, USA, were prepared by the manufacturers to ensure that the large-lumen tulip was positioned close to the endoscope during implantation. If necessary, stents were externally rotated 180° on the standard carrier system and reattached before implantation to mitigate the risk of stent dislocation due to high peristalsis and to facilitate better intestinal closure. This technique enabled the release of the large-lumen tulip into the small intestine, while the smaller lumen tulip end was positioned at the ostomy side. This approach aimed to enhance the anchoring of the SEMS in the small intestine, thereby reducing the likelihood of dislocation and improving sealing rates.
Endoscopic evaluations demonstrated satisfactory ingrowth of hypertrophied mucosa in the area of the uncovered portion of the stent tulip within 5–7 days in all patients, effectively preventing dislocation during the procedural course and allowing for the timely removal of external sutures after 10–14 days. Furthermore, this method anchored the uncovered part of the stent to healthy small intestine tissue. The fistulating segment of the small intestine was directed past the abscess cavity, enabling controlled discharge of intestinal contents through the covered section of the stent. In some patients, inflammatory defects between the small intestine and the external area were managed using stoma paste. The contents of the small intestine were discharged via the stent stoma in a controlled manner into an ostomy bag secured by a nursing base plate. Examples of the treatment and subsequent clinical courses are shown in Figs 1 to 3.
Primary and secondary endpoints
The primary endpoint of this study was successful clinical treatment of the fistula through endoscopic stent insertion, defined as an improvement in clinical symptoms related to the fistula (e.g. local wound management), successful bridging to later surgical revision, and restoration of enteral nutrition. The term “technical success” was referred to the successful implantation of a SEMS over the intended fistula. Treatment failure was defined as a deterioration of clinical symptoms related to the fistula and the continued inability to achieve enteral nutrition. Secondary endpoints included the feasibility of the endoscopic procedure, complications associated with the endoscopic intervention (such as stent dislocations, perforations, bleeding, and peri-interventional complications like aspiration and oxygen desaturation during sedation), as well as procedure-related, 30-day and overall mortality rates.
Consent
Written informed consent was obtained from all patients, including consent for data acquisition and analysis. All participants provided written informed consent for the publication of research details and clinical images. Institutional Review Board approval was not required for this study, since the stent used for this procedure was already approved for the management of gastrointestinal tract strictures, and no experimental materials that may require ethical approval were employed.
Statistical analyses
Descriptive statistics were calculated for all parameters. Results are presented as means ± standard deviation or as medians (interquartile range) for continuous variables. Categorical variables are presented as counts and percentages. The analyses were performed using SPSS software (SPSS Inc., Chicago, IL, USA, version 29.0).
Results
Baseline characteristics
Eight patients (five women and three men) with a median age of 59 years were indicated for stent stoma placement in the small intestine as a final therapeutic option. All patients presented with high-output fistulas of the small bowel, accompanied by abscess formation, inflammation, skin maceration, and/or small bowel stenosis, which hindered enteral nutrition. Six patients (75%) were diagnosed with advanced cancer, three of whom had peritoneal carcinomatosis. One patient had intestinal fistulas due to Crohn’s disease, while another experienced complications following multiple surgeries after a coiling and rupture of the hepatic artery.
Patients underwent a median of seven previous operations for fistula occlusion, yet none of these surgical interventions succeeded in achieving fistula closure or clinical improvement. Furthermore, none of the eight patients were effectively managed with a conventional ostomy system or dressing material. The median interval before the first endoscopic intervention was 33 days, and the median duration of stent treatment was 31.5 days. The median follow-up time was 2.7 months, ranging from 1 to 29 months until death or last contact.
The baseline characteristics of the patients are presented in Table 1.
Baseline characteristics.
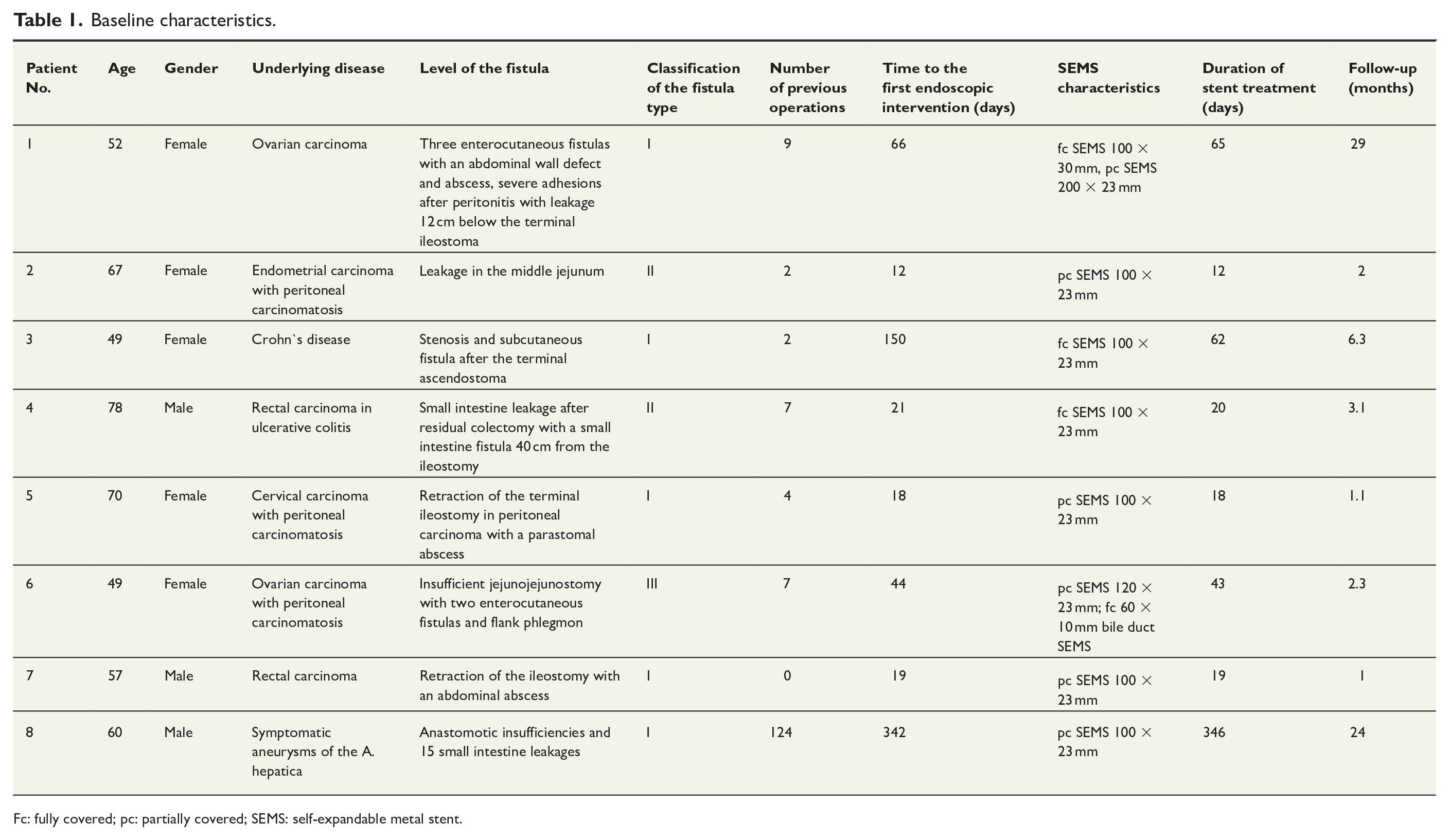
Fc: fully covered; pc: partially covered; SEMS: self-expandable metal stent.
Feasibility
The procedure was performed under analgosedation in all patients. Typically, the fistula was evaluated radiologically and endoscopically using a fine-caliber gastroscope (5.6 mm). Following stent selection, the implantation took place during a subsequent endoscopy session. A high risk of dislocation was observed after SEMS implantation in the first patient, leading to insertion of a second stent. This stent was subsequently secured to the small intestine cavity with external skin sutures during the first 14 days and anchored to the exposed small intestine with a minimum of three non-resorbable stitches. Altogether, SEMS implantation was feasible in all patients, converting the small intestinal fistula into a stent stoma (technical success rate: 100%, n = 8/8). Detailed stent characteristics are presented in Table 1.
Effectiveness
As shown in Table 2, the clinical success rate was 87.5% (n = 7/8), indicating significant clinical improvement in fistula-related symptoms, successful bridging to later surgical revision, and restoration of enteral nutrition following stent implantation. The creation of a stent stoma facilitated substantial clinical improvements, including sufficient sealing of the fistula and the ability to manage small intestine secretions. All patients (n = 8/8, 100%) experienced notable improvements in local wound management shortly after stoma creation. Prior to the establishment of the stent stoma, patients required dressing changes 2–6 times per day. Postimplantation of SEMS, the treatment transitioned to a conventional stoma system with a base plate, allowing for changes every 2–3 days. This shift improved wound care successfully addressed skin macerations and prevented further damage.
Feasibility, effectiveness and safety of the stent stoma.
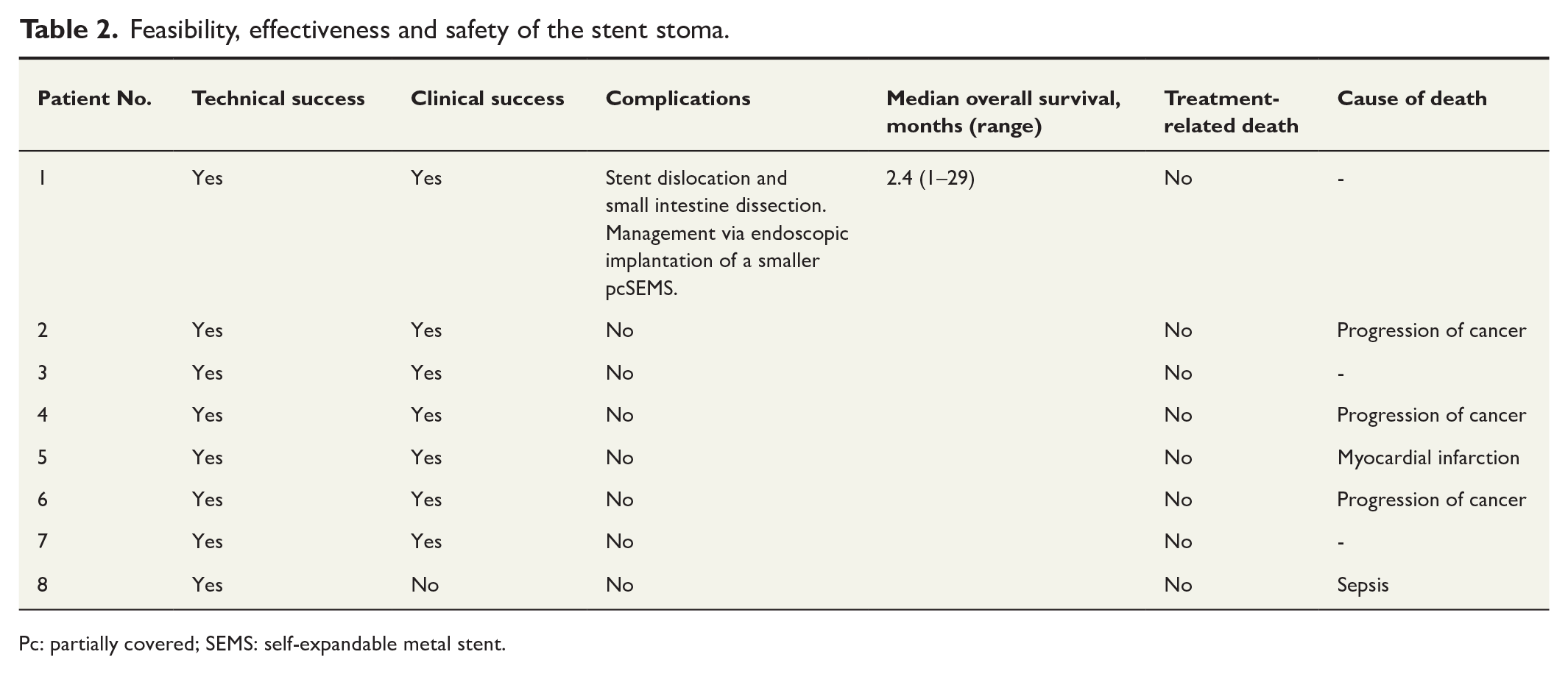
Pc: partially covered; SEMS: self-expandable metal stent.
Enteral nutrition was achieved in seven out of eight patients (n = 7/8; 87.5%). The remaining patient had a complicated clinical course following hepatic artery coiling and rupture, resulting in multiple adhesions from revision surgeries, which rendered oral nutrition impossible. This patient underwent further surgical adhesiolysis and percutaneous endoscopic gastrostomy for decompression but unfortunately did not show clinical improvement, and died from sepsis after 2 years of hospitalization.
Successful bridging to a later operation was achieved in one patient through stent insertion into the small intestine. This patient had a therapy-refractory small intestinal fistula located 40 cm above the ileostomy, extending to a midline laparotomy without further surgical therapeutic options. This procedure was performed during R0 resection via colectomy for colitis-associated rectal carcinoma. Following stent implantation, wound care improved significantly, and the remaining fistula transitioned from high-output to low-output, ultimately allowing for successful management through fistula plug occlusion and subcutaneous displacement, leading to regular wound healing.
Safety
The detailed safety data are presented in Table 2. Procedure-related complications occurred in one patient (12.5%; n = 1/8), who experienced multiple stent dislocations and complete small intestine perforation following the implantation of a fully covered stent with a proximal flare diameter of 30 mm. The stent penetrated the intestinal wall during dislocation into the fistula. Management of this complication was performed endoscopically by implanting a smaller, partially covered stent. Subsequently, the technique was modified to utilize partially covered stents secured to the small intestine using sutures for all patients.
The 30-day mortality rate for this study cohort was 0% (n = 0/8), while the overall mortality rate was 62.5% (n = 5/8), with none of the deaths attributable to treatment. Causes of death included cancer progression in three patients, myocardial infarction in one patient, and sepsis in the fifth. Three patients survived until the final follow-up. The median overall survival for the entire study cohort was 2.4 months (range 1–29 months).
Discussion
This study demonstrated the feasibility, effectiveness, and safety of endoscopic stent insertion for therapy-refractory small intestinal fistulas. The treatment options for this severe condition are often challenging and limited. While conservative therapies and surgical interventions, such as overstitching or resection of the affected small bowel, are available, surgery carries a high risk of recurrence, patient discomfort, and other potential postoperative complications.5,7 Moreover, some patients may not be eligible for surgery owing to factors like small intestinal adhesions, poor overall health, or severe inflammatory changes.
One of the most concerning complications associated with fistulas is sepsis, which can arise from intraperitoneal abscess formation or secondary skin infections due to the bacterial contents of the small bowel fistula.7,15 In these patients, addressing malnutrition, restoring blood volume, and correcting fluid and electrolyte imbalances are crucial. However, enteral nutrition is often difficult or even impossible due to ileus, sepsis, or inadequate absorptive capacity. When feasible, enteral nutrition is preferred over total parenteral nutrition, as it promotes intestinal integrity, prevents mucosal atrophy and bacterial translocation, and consequently reduces the risk of sepsis.5,16 Nonetheless, the benefits of enteral nutrition must be weighed against the potential drawbacks, such as increased fistula output and further deterioration of wounds around the fistula site. 15 In this context, SEMS implantation in small intestine fistulas represents a novel approach that may improve the quality of life for these patients and palliate their symptoms.
Stent implantation has been described in the literature as an intervention for malignant or benign stenoses, postoperative anastomotic insufficiencies, and leakages within the upper and lower gastrointestinal tract. However, studies investigating the use of metal stents specifically in the small intestine are scarce. To our knowledge, this is the first study to report the successful modification of a small intestinal fistula into a stent stoma.
In this study, the implantation of a SEMS in the small intestine under endoscopic and radiological guidance was technically feasible for all patients. Only one minor procedure-related complication was observed, which was effectively managed using conservative endoscopic measures. Overall, this endoscopic treatment method is minimally invasive and offers clear clinical benefits regarding wound care and nutritional status. This technique can serve as either a permanent palliative solution or a temporary measure to optimize the patient’s condition before proceeding with planned revisions or other curative treatments. As such, it may significantly expand the range of interventional endoscopic options available.
Our study does have some limitations, including its retrospective and non-randomized design and the relatively small patient cohort. However, there are no prospective randomized or comparative trials on this topic. Nonetheless, this new approach should be considered a less invasive interventional option for patients with therapy-refractory small intestinal fistulas who fail surgical and conservative therapies.
In summary, endoscopic stent insertion is a feasible, effective, and safe treatment option for therapy-refractory small intestinal fistulas. The creation of a stent stoma may improve the quality of life for affected patients.
Footnotes
Author contributions
M.K. performed the endoscopic procedures; contributed to conception and design of the study, to interpretation of the data, and to drafting the article; and wrote the manuscript. P.S. helped to revise and edit the manuscript. M.A. contributed to the collection of data, and helped to revise and edit the manuscript. N.M. helped to revise and edit the manuscript. T.R. helped to revise and edit the manuscript. C.C.Z. contributed to conception and design of the study; performed the analysis and interpretation of the data; and wrote the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
