Abstract
Background and objective:
The incidence of early-onset colorectal cancer among the young (<50 years) has been reported to have risen in last decades. This retrospective study aimed to investigate the characteristics of early-onset rectal cancers (EO-RCs) and potential changes in proportion of EO-RCs, and further to report the mortality and recurrence rates of EO-RCs.
Methods:
In the years 2007–2021, 2557 rectal cancer (RC) patients were operated in Helsinki University Hospital and of them 147 were 18–49 years old. Cumulative overall survival (OS), disease-specific survival, and disease-free survival were calculated using the Kaplan–Meier analysis.
Results:
The percentual amount of the EO-RCs varied between 2.5% and 11.3% annually and there was no perceivable trend. Majority were adenocarcinomas (98.7%), of which 8.8% were mucinous. Predisposing factors such as Lynch syndrome, polyposis, or ulcerative colitis were seen in 26 patients (17.7%) and in 10 of 22 patients (45.5%) under 35 years. The cumulative 5-year OS was 91.9% in stage I, 93.3% in stage II, 86.7% in stage III, and 50.0% in stage IV disease. Metastatic recurrence was found in 22 cases (18.4%) and local recurrence in 8 patients (5.4%) and 6 patients had both.
Conclusions:
In our cohort of 147 EO-RC patients, OS was good and the clinical course did not seem to differ much from the course of RC in general population.
Context and Relevance
This retrospective study of 147 young (<50 years) patients concentrates specifically on early-onset cancer of the rectum. All patients were operated in the same high-volume unit. In comparison with register studies, this article gives a real-life description of the patients’ overall and disease-free survival. Follow-up time is also relatively long. In our study population, overall survival is good and the clinical behavior of the early-onset rectal cancer does not seem to differ much from the rectal cancer in general population.
Introduction
The incidence of colorectal cancer (CRC) among the young (<50 years) has risen during the last decades both in the United States and in Europe. The rise has been most prominent for rectal cancer (RC) reaching up to a 3.2% annual rise among the youngest (20–29 years).1,2 The rate of increase in the young has varied between different countries. In Europe, the incidence of colon cancer seems to have risen more than the incidence of RC. Vuik et al. 3 reported the annual rate of increase for early-onset colorectal cancer (EO-CRC) to be 1.6–9.3% versus 0–3.5% for early-onset rectal cancer (EO-RC). The Finnish Cancer Registry has reported a steady increase in the incidence of colon cancer, while the incidence rate for RC has not markedly increased. At the same time cancer-related mortality of RC is declining, and the 5-year disease-specific survival (DSS) for the period 2006–2015 was 62.3% compared to 53.6% in the previous decade.4,5 Incidence among young patients was not reported separately.
The causes for the increased incidence among the young are thought to be multifactorial.6,7 Hereditary cancer syndromes are overrepresented in the EO-CRC patients compared to older patients. Tumors secondary to hereditary mutations have been reported to account for up to 16% of EO-CRC. Lynch syndrome (LS) alone accounting for 8.4% of them. 8 Inflammatory bowel syndromes, ulcerative colitis (UC) and Crohn’s disease, are other risk factors for EO-CRC. 9 In addition, many EO-CRC patients have a familial background of CRC, even though no hereditary mutation has been recognized. The risk of CRC is 2-fold in first-degree relatives of CRC patients. 10
The incidence of hereditary cancers is not expected to have risen. Therefore, the rise in incidence is assumed to be in the sporadic percentage of the patients. Over 80% of the EO-CRC patients do not have hereditary cancer syndrome or other underlying cause. 11 The risk factors associated with sporadic EO-CRC seem to be the same as for CRC in general 7 which include the consumption of alcohol, tobacco, or processed red meat, obesity, and low-fiber diet. 6
As survival rates of CRC patients in general have improved, the data on EO-CRC patients have varied. Mauri et al. noted in their review article that the survival of CRC patients with very early onset (<35 years) is worse than for patients over 50 years. However, the data on survival are conflicting. When comparing patients under and over 50 years of age, the included studies found either no significant difference in prognosis and overall survival (OS) or a significantly better survival for patients under 50 years of age. 12 Tumor biology is thought to be the main reason for the inferior survival of very EO-CRC patients. EO-CRC seems to present with more aggressive tumor biology (mucinous or signet-cell histology, high-grade adenocarcinoma) and often at a more advanced stage at the time of diagnosis.12–14 The latter may be due to the low suspicion of cancer in the young population.
The present study aimed to answer some of these questions in a cohort of consecutive RC patients operated in a high-volume center. The primary aim was to study whether the percentage of EO-RC patients has risen during the study period and which factors could have been affecting the possible change: tumor-specific factors or molecular alterations. Secondary aims were to examine if the OS, cancer-specific survival, and disease-free survival (DFS) were comparable to RC patients in general. The subgroup of LS patients was also investigated regarding survival and recurrences, because LS patients are considered to have better prognosis than sporadic CRC patients. 15
Patients and methods
We conducted a retrospective search of a patient cohort consecutively operated for RC at Helsinki University Hospital during the time period 2007–2021. This unit has long been a tertiary referral center for hereditary CRC syndrome patients in the southern Finland and has 2.2 million residents in the specific catchment area. The centralization of RC treatment to university hospitals by legislation has also increased the number of sporadic RC patients treated in this unit from 2018 onward. Patients were identified by a search in the database of colorectal units using the 10th revision of the International Classification of Diseases diagnostic code (C20) for RC and procedural codes for rectal resections or procedures. Patients with the age of 18–49 years at the time of the operation were included. Very EO-RC (<35 years) group was compared with the older patients (35–49 years). Patients’ clinical and family history, when available in electronical patient records Clinical variables such as height, weight, American Society of Anesthesiologists (ASA) class, tobacco, and alcohol consumption were collected from the electronic database. Known risk factors for EO-CRC were also recorded, including inflammatory bowel disease, LS, and polyposis syndromes. The family history was considered positive, if patients had mentioned ⩾1 first-degree relative had CRC or if ⩾2 relatives had had CRC or other LS-related cancers. Possible preoperative and adjuvant treatment and type of operation were recorded. Radiological findings were collected from the computed tomography (CT) and magnetic resonance imaging (MRI) reports.
Tumor data were collected from the pathology reports and included histology, lymphovascular invasion, and nervous invasion. The tumor-node-metastases (TNM) classification of the American Joint Committee on Cancer 7th edition was used in stage classification. Tumor grade was reported according to World Health Organizations Classification of Tumours of the Digestive System 4th edition 2010. Tumor budding was extracted from the pathology reports but has been reported only since 2018. Microsatellite instability (MSI) and CRC-mutation search reports were registered if conducted. The follow-up information on local recurrences, metastases, and deaths including causes of death was collected from the electronic patient records and from the Finnish Population Register Center until the end of 2022. The follow-up was arranged for 5 years postoperatively. The protocol included laboratory tests (carcinoembryogenic antigen and hemoglobin) and clinical examinations and endoscopy for all until 5 years. Whole body CTs and lower abdominal MRIs were ordered regularly for patients with an increased risk of local recurrence or distant metastases until 3 years (including patients with primarily locally advanced tumors and/or patients with preoperative long-course chemoradiotherapy). Patients with hereditary CRC syndromes are included in a routine lifetime follow-up at our unit.
Molecular analysis
To investigate MSI status, immunohistochemistry (IHC) or polymerase chain reaction testing was used. Routine systematic IHC testing for MSI was introduced for all CRC specimens in 2018. Next-generation sequencing was used to analyze the cancer tissue first and if there were pathologic mutations, germline mutations were then analyzed in clinical genetics laboratory with a CRC blood test packet, which identifies 12 most common genetic mutations (APC, BMPR1A, MLH1, MLH3, MSH2, MSH6, MUTYH, PMS2, POLD1, POLE, SMAD4, STK11).
Statistical analysis
Patient and tumor characteristics were described using frequency and percentage for categorical variables, and median and range for continuous variables. OS, DSS, and DFS were calculated with Kaplan–Meier analysis, and the survival differences were compared with log-rank test. In the analysis, p < 0.05 was considered statistically significant. The statistical analysis was conducted using SPSS 25 software.
Results
Patient characteristics
Among 2557 patients operated for RC at Helsinki University Hospital between 2007 and 2021, there were 147 (5.7%) patients under 50 years of age at the time of the operation. The proportion of the EO-RCs varied between 2.5% and 11.3% annually (median 5.9%) and there was no perceivable trend (Fig. 1).
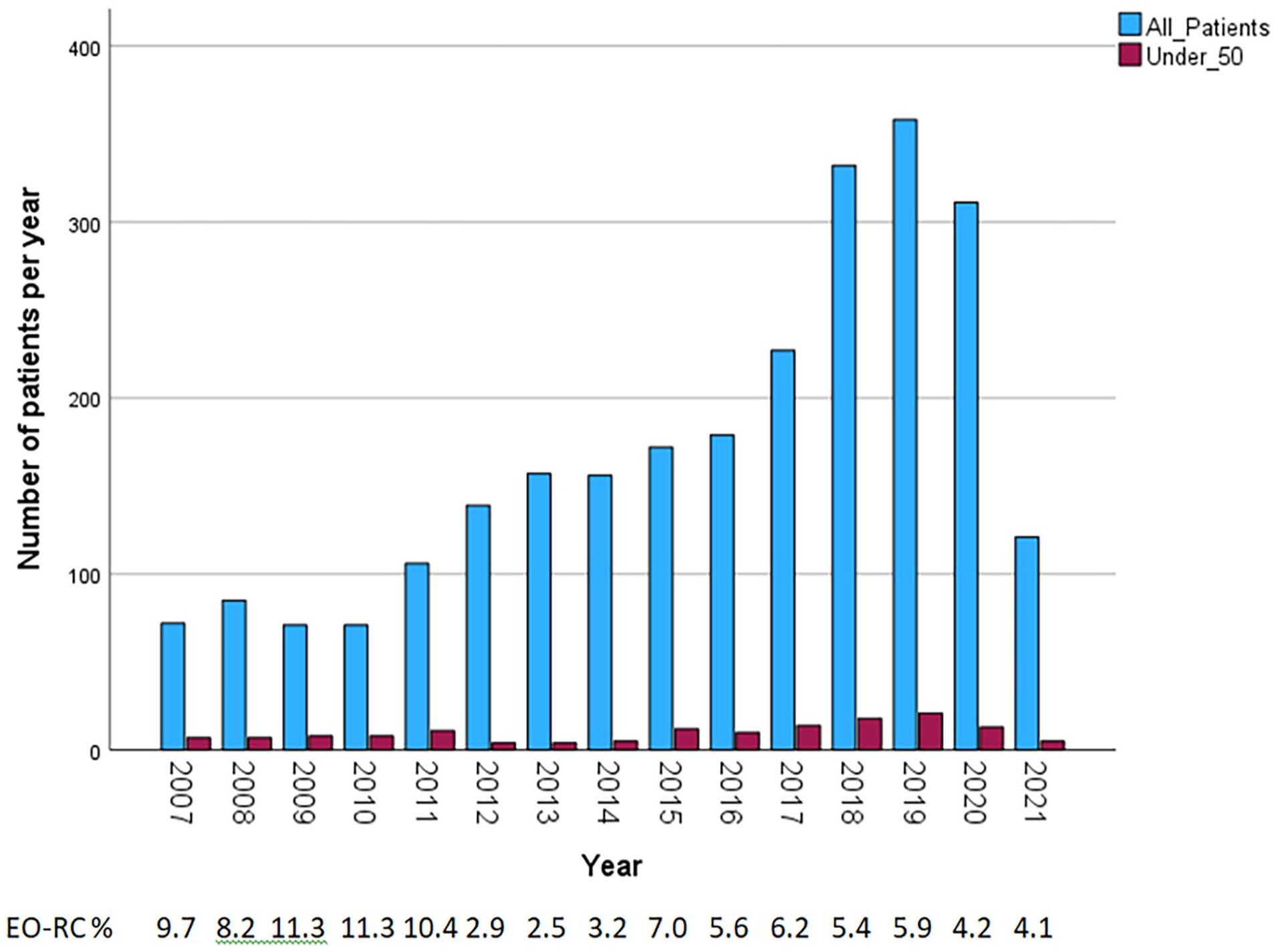
EO-RCs per year.
The median age at the time of the operation was 45.2 years (range 23.8–49.9; Table 1). Twenty-two patients (15.0%) under 35 years of age were classified as very EO-RCs. Seventy-eight (53.1%) were male. A total of 121 (82.3%) were ASA class I and II. Tobacco smoking was reported by 31 (21.1%). Alcohol consumption (over 1 dose per week) was reported by 34 (24.1%).
Patient characteristics (n = 147).
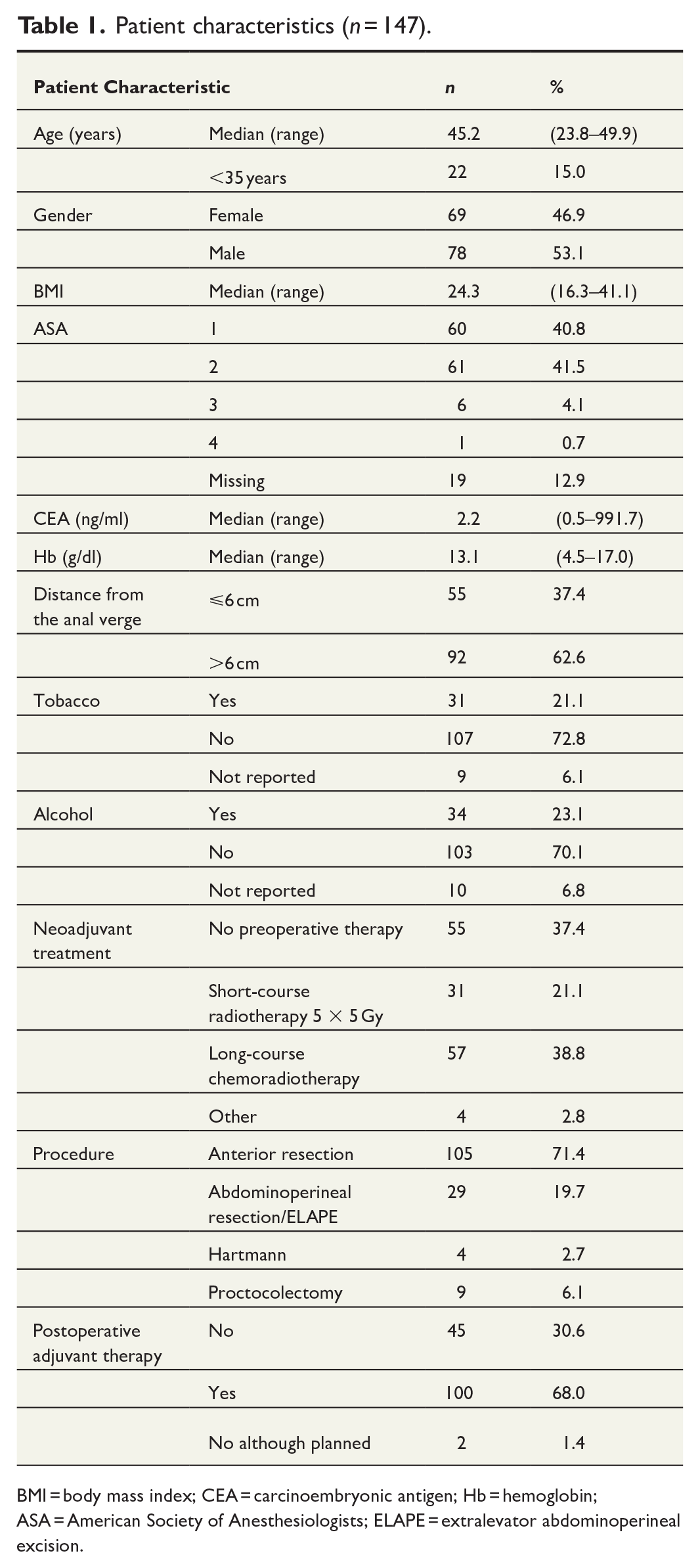
BMI = body mass index; CEA = carcinoembryonic antigen; Hb = hemoglobin; ASA = American Society of Anesthesiologists; ELAPE = extralevator abdominoperineal excision.
Altogether 105 patients (71.4%) underwent anterior resection, and 92 (62.6%) had preoperative neoadjuvant treatment. One hundred patients (68.0%) received adjuvant therapy, usually oxaliplatin and capecitabine for 3–6 months (Table 1).
Tumor characteristics
Histology of the tumors was adenocarcinoma in 145 (98.7%) patients. Of these, 13 (8.8% of all tumors) were mucinous adenocarcinomas. Two (1.4%) were neuroendocrine carcinomas (Table 2).
Tumor characteristics (n = 147).
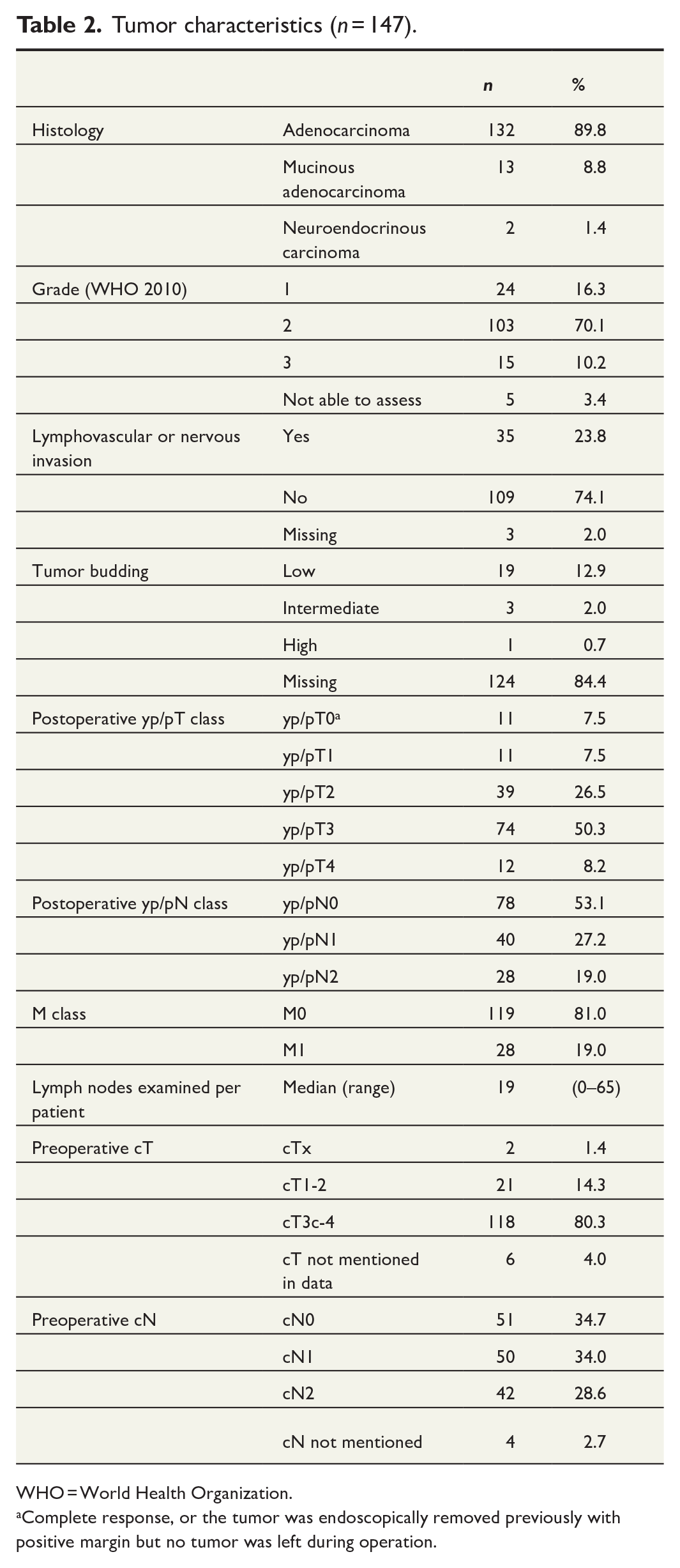
WHO = World Health Organization.
Complete response, or the tumor was endoscopically removed previously with positive margin but no tumor was left during operation.
The tumors were grade 1 (well differentiated) in 24 patients (16.3%), grade 2 (moderately differentiated) in 103 (70.1%), and grade 3 (poorly differentiated) in 15 (10.2%) patients. The grade was not defined in five cases. In 35 tumors (23.8%), invasion to vessels or nerves was detected.
The histopathological stage of the primary tumor was yp/pT1 in 11 cases (7.5%), yp/pT2 in 39 (26.5%), yp/pT3 in 74 (50.3%), and yp/pT4 in 12 (8.2%). In nine patients, the primary tumor could not be found at histopathology due to complete response after chemoradiotherapy (ypT0) and in one patient due to previous endoscopic removal. In one patient, the primary tumor was not removed because of carcinomatosis. Lymph node status was yp/pN0 in 78 (53.1%) patients, yp/pN1 in 40 (27.2%), and yp/pN2 in 28 (19.0%). A median of 19 lymph nodes per patient were examined (0–65).
Predisposing conditions for CRC
Family history of CRC was reported by 23 patients (15.3%; Table 3). A predisposing condition such as LS, polyposis syndromes, or UC was found in 26 patients (17.7%). Only six of these patients were aware of the predisposing condition (three LS, two familial adenomatous polyposis (FAP), one UC). Of the very EO-RCs (<35 years), 10 out of 22 patients (45.5%) were found to have a predisposing condition. Fifteen out of 147 EO-RCs (10.2%) were microsatellite instable (MSI-high [MSI-H]) and 12 (8.2%) of the MSI-H patients were diagnosed with LS either before or after the operation.
Predisposing conditions for CRC and molecular characteristics of the tumors (n = 147).
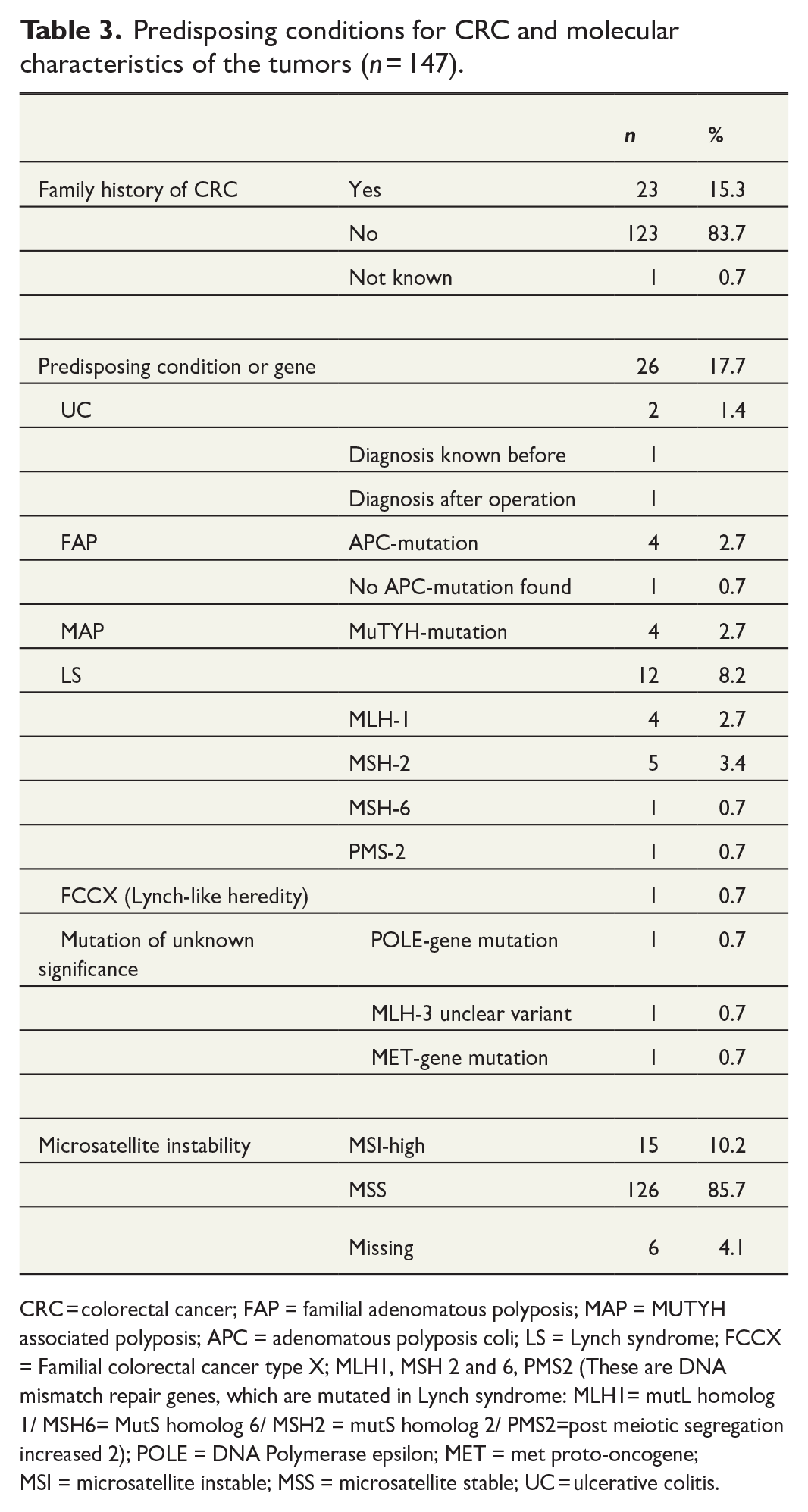
CRC = colorectal cancer; FAP = familial adenomatous polyposis; MAP = MUTYH associated polyposis; APC = adenomatous polyposis coli; LS = Lynch syndrome; FCCX = Familial colorectal cancer type X; MLH1, MSH 2 and 6, PMS2 (These are DNA mismatch repair genes, which are mutated in Lynch syndrome: MLH1= mutL homolog 1/ MSH6= MutS homolog 6/ MSH2 = mutS homolog 2/ PMS2=post meiotic segregation increased 2); POLE = DNA Polymerase epsilon; MET = met proto-oncogene; MSI = microsatellite instable; MSS = microsatellite stable; UC = ulcerative colitis.
Stage, recurrence (DFS), and survival (DSS, OS)
A total of 28 patients (19%) had synchronous metastases at the time of the diagnosis or surgery. Based on pathologic TNM of surgical specimen, 37 (25.2%) had stage I, 30 (20.4%) had stage II, and 45 (30.6%) had stage III disease. Seven patients (4.8%) had pathologic complete response (pCR, ypT0N0) after long-course chemoradiotherapy. During the follow-up, 29 (19.7%) patients died, 17 (58.6%) of whom already had stage IV disease at the time of surgery. Twenty-six (17.8%) deaths were considered to be cancer related. The cumulative 5-year OS was 91.9% for stage I, 93.3% for stage II, 86.7% for stage III, and 50.0% for stage IV (Fig. 2). Cumulative DSS at 5 years was 97.2% in stage I, 93.3% in stage II, 86.7% in stage III, and 50.0% in stage IV (Fig. 2).
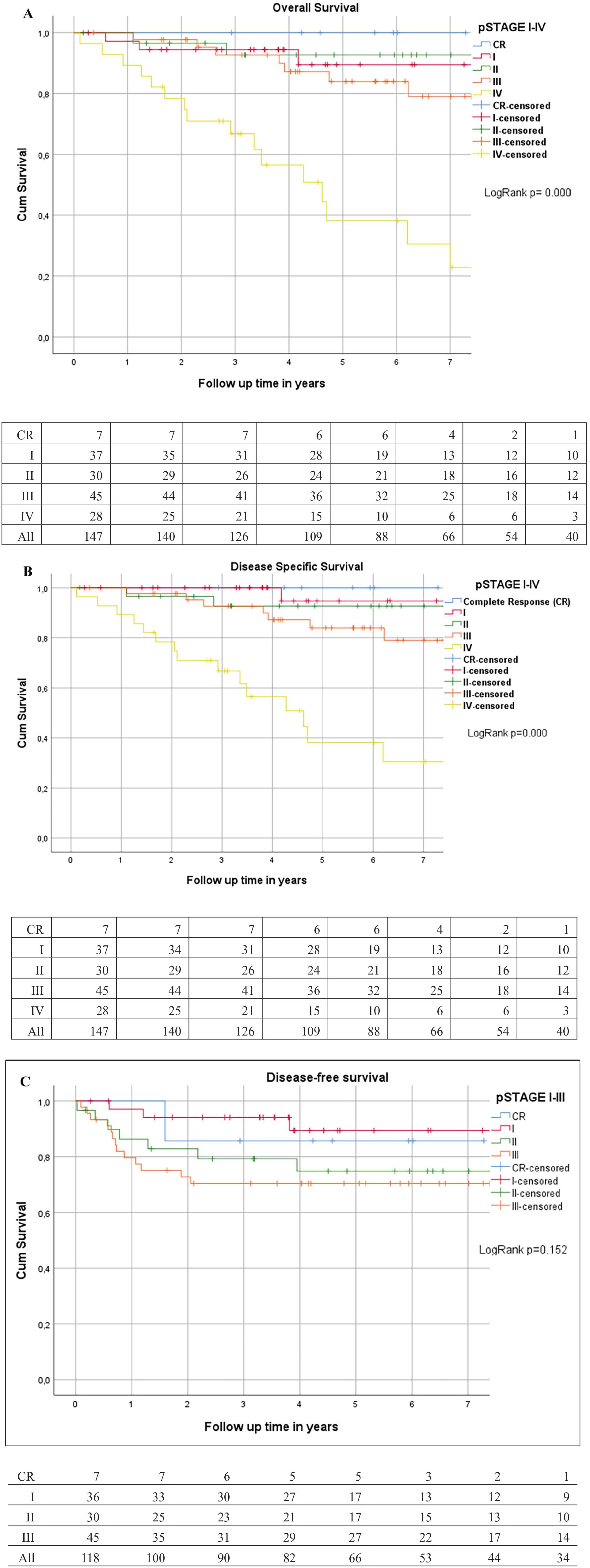
OS, DSS, and DFS. (A) OS stages I–IV (n = 147). (B) DSS stages I–IV (n = 147). (C) DFS stages I–III (n = 118).
Altogether 24 (20.2%) of the 119 patients with stages I–III or pCR at the time of surgery had recurrences. Twenty-two (18.4%) had distant metastases, eight (5.4%) had local recurrence, and six of these were diagnosed with both distant and local recurrences simultaneously. Cumulative DFS is shown in Fig. 2C.
We also compared the very EO-RC cohort (<35 years) to the 35–49 years old patients. There were 22 (15.0%) patients in the very EO-RC cohort. Six (27.3%) patients of those had metastatic stage IV disease at the time of surgery, compared to 19 (15%) stage IV patients in the 35–49 years group. Four patients in the very EO-RC cohort died within 5 years after surgery, all of whom had metastatic disease at the time of diagnosis.
When comparing OS, DSS, and DFS of the very EO-RC patients with stages I–III disease or pCR, to the 35- to 49-year-old cohort, no statistically significant difference was noticed (Fig. 3). The cumulative 5-year OS in the very EO-RC cohort and EO-RC patients was 100% versus 89.3%, 5-year DSS was 100% versus 92.2%, and 5-year DFS was 75.0% versus 80.4%, respectively. Five-year OS for LS patients was 93.3%, 5-year DSS was 93.3%, and 5-year DFS was 90.0% (Fig. 4).
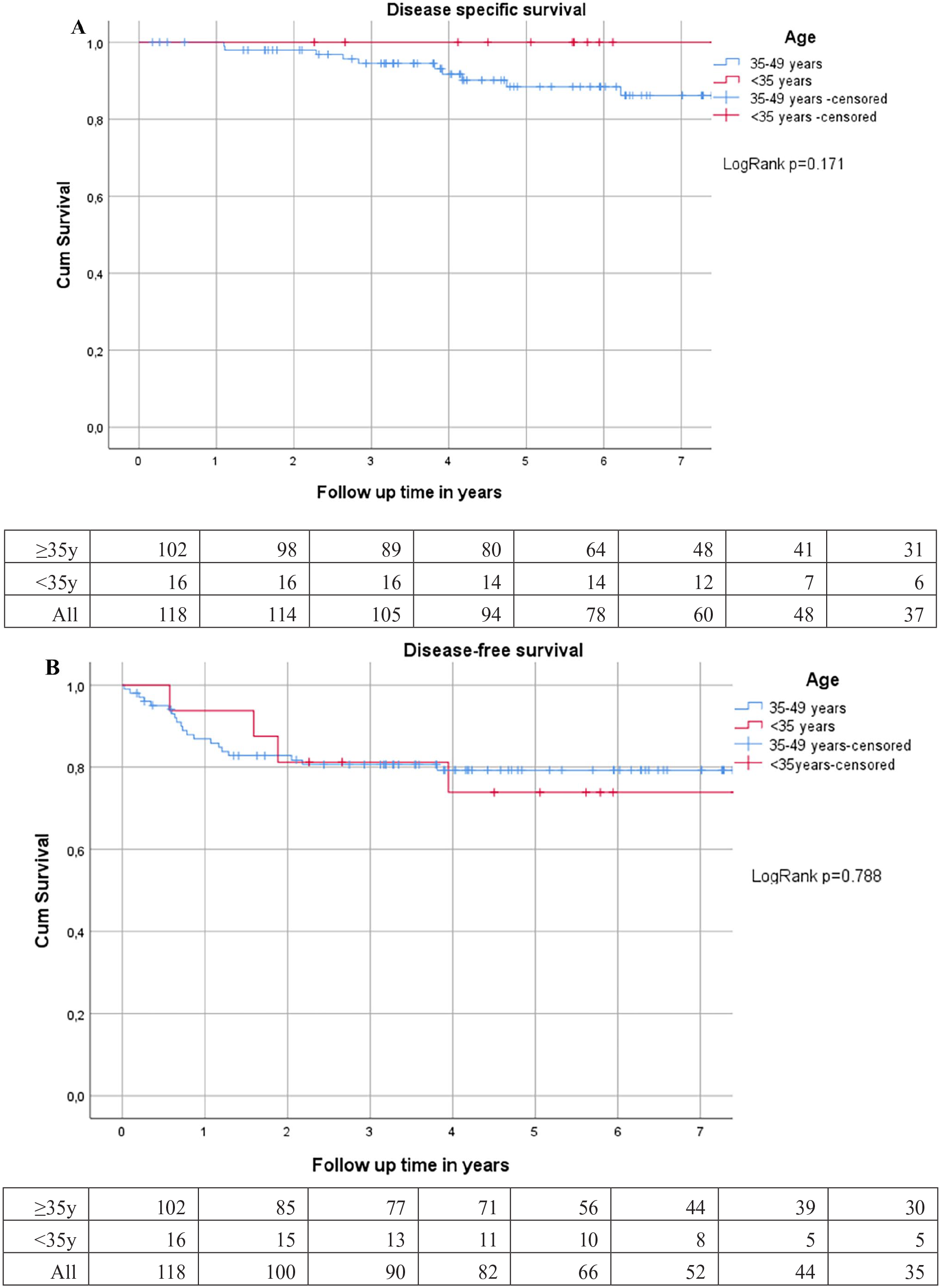
DSS and DFS of stages I–III very young patients (n = 16) in comparison to the 35–49 years group (n = 102). (A) DSS stages I–III. (B) DFS stages I and II. DSS = disease-specific survival; DFS = disease-free survival.
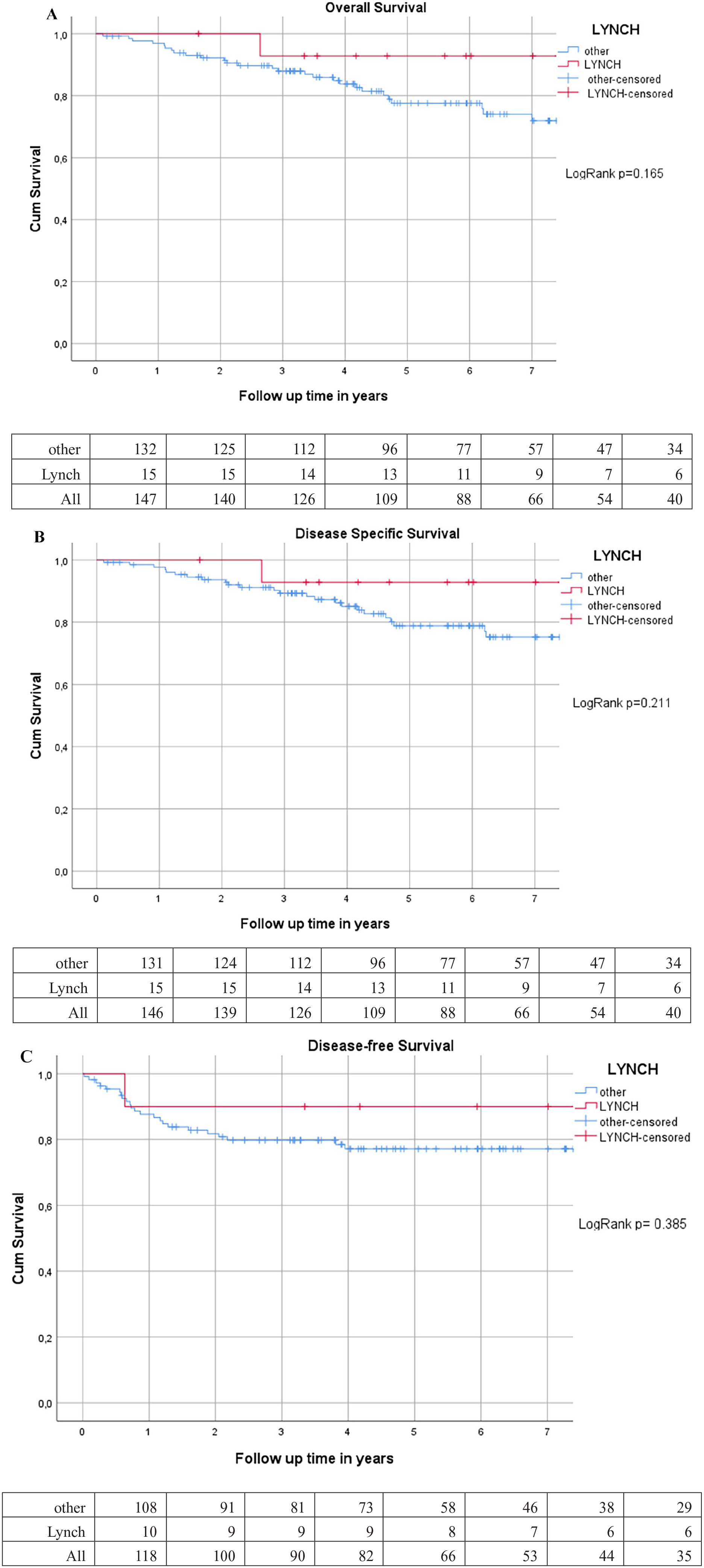
LS patients (n = 15) versus other EO-RC (n = 132). (A) OS stages I–IV. (B) DSS stages I–IV. (C) DFS stages I–III LS (n = 10) versus other EO-RC (n = 108). OS = overall survival; DSS = disease-specific survival; DFS = disease-free survival; EO-RC = early-onset rectal cancer; LS = Lynch syndrome.
Discussion
This retrospective cohort study investigated the occurrence, clinical features, and survival of EO-RC operated in a high-volume center in Finland. The percentage of EO-RCs did not significantly change during the 14 years study period. OS of EO-RC patients and the distribution of the stages are comparable to earlier reports. Even though cancer has metastasized, these patients live quite long with the disease. About 10% of EO-RCs were poorly differentiated and 8% were mucinous adenocarcinomas which is comparable to the average RC population. There were two neuroendocrine carcinomas, which were included because their neoadjuvant and surgical treatment is the same as in adenocarcinomas.
Fifteen percent of the patients reported a family history of CRC, but only half of them were found to have an underlying predisposing condition. Altogether almost one-fifth of the 147 patients (17.7%) turned out to have an underlying predisposing condition.
There is conflicting data whether the internationally observed rise in CRC incidence is owed more to colon or to RCs.1,2,3,16 In this cohort of RC patients operated during 14.5 years, the proportion of EO-RC patients treated with surgery was unchanged over time. However, the operative treatment of hereditary cancer syndromes which are more common in EO-RC was centralized to university hospitals throughout the whole study period. The centralization of sporadic RC treatment in Finland since 2018 has led to an increase of sporadic cancers treated at our institution. This may in fact have concealed an actual increase of the proportion of patients treated for EO-RC.
It is self-evident that the proportion of hereditary predisposing syndromes in younger patients is higher than in older cancer patients, especially in very EO-RC patients. In our study, almost half of the very EO-RC patients had some underlying predisposing condition and 40.9% had a hereditary syndrome. This is in accordance with previously published data. 17 Many of the EO-RC patients in our cohort were probands, meaning that their cancer diagnosis led to the diagnosis of a previously unknown hereditary cancer syndrome in their family. Only 23.1% of the very EO-RC patients with predisposing conditions reported a family history or knew about their own genetic predisposition beforehand, which is in line with a previously published study reporting that 19% of the hereditary syndromes diagnosed in very EO-RC patients did not have a family history of the disease. 17 Even though the proportion of patients with known underlying causes is higher in the young, it is noteworthy that still over 80% of RCs in young patients were sporadic.
The proportion MSI-H tumors was with 10.2% approximately the same as reported previously in RCs. 18 The proportion of LS patients was 8.2%, which is higher than in the general population, where it has been reported to be 3%. 19 Pearlman et al. 8 have reported a similar percentage of LS in EO-CRC patients. In our cohort of LS patients, the mutations most frequently found were either MLH-1 or MSH-2. This is in concordance with the fact that MLH-1 mutation is the main cause of LS in the Finnish population. 20 Globally, MSH-2 and MLH-1 mutations constitute 76% of the mutations causing LS, MSH-6 and PMS-2 being rarer. 21 In previous studies, the prognosis of LS patients was found to be better than that of other CRC patients. 15 However, one main reason for this may be that many CRCs in LS patients are detected at an earlier stage than cancers of non-Lynch patients. 22 In our study, 2 of 12 LS patients (16.7%) had stage IV disease at the time of diagnosis or surgery. Although the prognosis of LS patients in our study tended to be better than in non-LS patients, the difference was not statistically significant. This might be due to the small number of LS patients.
The stage-specific OS in our cohort of EO-RC patients was at least as good as reported in RC patients in general.4,5,23 According to the American Cancer Society’s 2024 annual report, the 5-year stage-specific OS was 91% in stages I and II RC, 73% in stage III RC, and 14% in stage IV RC. In comparison, the OS in our EO-RC patients was more favorable in stage III (86.4%). Our center is a large tertiary center operating about 350 RC patients per year, which may explain the better outcome in stage III RC patients. The survival difference (5-year OS: 51.8%) in stage IV RC patients most probably is due to that our cohort represents only patients having had surgery for their primary cancer and those patients with the most advanced stage IV patients are not included.
This study included only the patients who were surgically treated and therefore we cannot draw any conclusions for the whole population of EO-RC. In our study, the very EO-RC group of patients seemed to have the disease diagnosed at a later stage than the other patients: 27.3% had stage IV disease, when only 19.0% of the whole group and 15.0% of the 35–49 years old had a stage IV disease at the time of diagnosis. However, for patients with stages I–III, the prognosis of the very EO-RC patients did seem to be at least as good as in the older group. This is in accordance with previously published results. 24 Thus, it seems that the notion of a more aggressive tumor biology in younger patients might only be owed to the often delayed cancer diagnosis in this group.
Limitations
The main limitation is the retrospective nature of this study. Furthermore, the included timespan is quite long with centralization of treatment happening during it. On the other hand, the long time span of the study enabled to include a large cohort of purely EO-RC patients and a longer follow-up period which is essential for a reliable survival analysis. In many previous EO-CRC studies, RC patients have not been analyzed separately, although the clinical picture and the treatment differ in colon and RC patients. Thus, our series of EO-RC patients only is difficult to compare with some of the existing literature. However, the study gives for its part, additional data on this subgroup.
Another possible limitation is that the genetic testing evolved through the study period. However, our unit has started the national Lynch and polyposis research registers in Finland in the 1980s and has maintained them ever since. Therefore, there should be no missing Lynch and classical FAP diagnoses. Still, genetic panels recognizing also more uncommon genetic mutations predisposing for CRC have been introduced to routine over time. Therefore, some rare mutations may have remained unrecognized at the start of the study period.
The cohort comprises of surgically treated patients only and is heterogeneous in terms of tumor stage (I–IV). Furthermore, the total number of patients is relatively small, which makes the comparison between subgoups unreliable. This of course sets limits to conclusions made from this study.
Conclusion
The clinical behavior of the EO-RC does not seem to differ much from that of RC in the general population. In this study of patients with surgically resected EO-RC, the diagnosis was often reached later among young individuals but the idea of the more aggressive disease of the young may not be correct, when treatment is timely.
Footnotes
Author contributions
All the authors have given substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work. All authors have participated in drafting the work or revising it critically for important intellectual content. All authors have given their final approval of the version to be published.
I, the designated corresponding author of the above article submitted for consideration of publication, hereby confirm that:
1. All named authors agree to the submission of the article to Scandinavian Journal of Surgery.
2. All authors who qualify for authorship under the criteria listed above have been named on the article.
3. The article is not currently under review by another journal.
4. The article has not been accepted for publication elsewhere.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics and patient consent
The Ethics Committee approval was waived due to the retrospective nature of the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
