Abstract
Sarcopenia is thought to be a marker for underlying frailty and malnutrition, contributing to poor functional status and suboptimal healing postoperatively. We aimed to complete an updated systematic review and meta-analysis evaluating the impact of sarcopenia on short- and long-term outcomes following colorectal cancer surgery. We searched MEDLINE, Embase, and CENTRAL up to September 2023. Studies that compared sarcopenic and non-sarcopenic patients’ short- and long-term outcomes following curative intent elective surgery for colorectal cancer were included. The main outcomes included postoperative morbidity, postoperative mortality, and length of stay (LOS), among others. Inverse variance random effects meta-analyses was performed. Risk of bias was assessed with Cochrane tools. Certainty of evidence was assessed with GRADE. After screening 215 studies, we included 40 non-randomized studies, totalling 13,422 patients, of which 5,432 (40.4%) were classified as sarcopenic. Across 27 studies, patients with sarcopenia were more likely to experience 30-day postoperative morbidity (40% vs 33%, RR 1.30, 95% CI 1.12-1.50, P < 0.01, I2 79%). The mean LOS was 1.46 days longer for sarcopenic patients (26 studies, 95% CI 0.85-2.07, P < 0.01, I2 82%). Upon pooling data from 13 studies, sarcopenic patients had increased risk of 30-day postoperative mortality (2.8% vs 1.0%, RR 2.74, 95% CI 1.63-4.62, P < 0.01, I2 0%). The findings from this systematic review suggest with low to very-low certainty evidence that in patients who are undergoing curative intent surgery for colorectal cancer, preoperative sarcopenia is associated with poor postoperative outcomes.
Introduction
Colorectal cancer is the third most common cancer in the world, with 1.9 million cases diagnosed per year worldwide. 1 Despite recent advances in neoadjuvant, endoscopic, and surgical endoluminal therapies, oncologic resection remains the mainstay for definitive care of curative colorectal cancer. Importantly, colorectal cancer is often diagnosed in patients of advanced age (ie, median age of 68 in women and 65 in men) 2 ; therefore, most patients have medical comorbidities at the time of their colorectal cancer surgery.
In addition to comorbidities, an important metric in the oncologic patient population is sarcopenia, which can be an indicator of frailty and malnutrition. Sarcopenia has been defined as a progressive skeletal muscle disorder that is associated with falls, disability, and even mortality. 3 Sarcopenia can be diagnosed based on clinical tests of muscle strength (eg, grip strength or chair rise test) or quantitative tests of muscle mass on imaging (eg, computed tomography (CT) and magnetic resonance imaging (MRI)). 3 It has been cited that between 12 and 60% of colorectal cancer patients are sarcopenic. 4 Multiple studies have shown that this is associated with worsened postoperative outcomes,4-7 which may then cause delay in adjuvant chemotherapy initiation and subsequently worse long-term oncologic outcomes, such as disease-free survival (DFS) and overall survival (OS).8-12 A recent meta-analysis by Trejo-Avila et al 9 demonstrated increases in postoperative complications, mortality, and length of stay (LOS) in patients with sarcopenia. Moreover, they demonstrated decreased DFS and OS in patients with sarcopenia following colorectal cancer surgery. 9
There have been multiple observational studies published since the most up to date data synthesis.13-17 These studies have added important data regarding surgical outcomes of colorectal cancer patients with sarcopenia. Our aim is to update previous reviews and meta-analyses to compare postoperative complication rates and survival outcomes between patients with and without sarcopenia undergoing surgery for colorectal cancer. We hypothesize that preoperative sarcopenia will be associated with worsened short- and long-term outcomes for these patients.
Methods
This systematic review and meta-analysis is reported in accordance to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) (Appendixes 1-2). The study protocol (CRD42023493416) was registered on the International Prospective Register of Systematic Reviews (PROSPERO) a priori. This study did not require local ethics approval.
Search Strategy
The databases Medline, EMBASE, and Cochrane Central Register of Controlled Trials (CENTRAL) were searched over the period from database inception through September 2023. The search was devised and executed by a medical research librarian with input from study investigators. Search terms included “sarcopenia,” “muscle atrophy,” “colorectal adenocarcinoma,” “colorectal surgery,” and more (complete search strategy available in Appendixes 3 and 4). To ensure all relevant articles were included, the references of studies meeting inclusion criteria were searched manually.
Eligibility Criteria
Articles were eligible for inclusion if they were observational studies comparing adult patients (greater than 18 years of age) with or without sarcopenia undergoing elective colorectal cancer surgery with curative intent. Studies containing data regarding one or more short-term postoperative outcome (ie, 30-day morbidity, 30-day mortality, LOS, specific complications, re-operation, or readmission) were included. Studies were excluded if they evaluated patients undergoing emergency surgery, as when compared to elective surgical patients, this population confers a high risk profile and may add significant heterogeneity. Single-armed, non-comparative studies were also excluded, as well as systematic reviews, editorials, meta-analyses, and any study not reporting primary data.
Study Selection
Four reviewers (SK, VS, MA, and EH) independently evaluated the searched titles and abstracts using a standardized, pilot-tested form on Covidence©. Any discrepancies that arose during title and abstract screening phases were resolved by inclusion of the study. The same four reviewers completed full-text screening. Discrepancies were resolved by consensus between the reviewers during the full-text screening phase. An alternate reviewer was consulted in the case of persisting disagreement.
Data Collection
Four reviewers (SK, VS, MA, and EH) independently conducted data extraction into a data collection form designed a priori and pilot-tested on Microsoft Excel©. The extracted data included study characteristics (eg, author, year of publication, and location of study), patient demographics (eg, age, gender, body mass index [BMI], comorbidities, and sarcopenia status), disease characteristics (eg, colorectal cancer location and stage of colorectal cancer), treatment characteristics (eg, operation type and receipt of neoadjuvant or adjuvant chemotherapy), postoperative complications (eg, 30-day postoperative morbidity, 30-day postoperative mortality, system-specific morbidity, anastomotic leak, and surgical site infection (SSI)), postoperative LOS, reoperation, readmission, and long-term oncologic outcomes (eg, 5-year OS and 5-year DFS),.
Outcomes Assessed
The main outcomes defined a priori included 30-day postoperative mortality, 30-day postoperative morbidity, anastomotic leak, SSI, postoperative LOS, 30-day readmission, 30-day reoperation, 5-year OS, and 5-year DFS. 30-day postoperative mortality was defined as any documented death within 30 days of the index operation. 30-day postoperative morbidity was defined according to each individual study and included any documented deviation from the expected postoperative course. If studies did not report 30-day postoperative morbidity as a pooled outcome, it was reported as missing. Postoperative LOS was defined as the number of days from the index operation to the time the patient left an acute care bed. Anastomotic leak was defined as communication between the intraluminal and extraluminal compartments at the site of surgical anastomosis, which results from defect of the intestinal wall. 18 Surgical site infection was defined according to the Centre for Disease Control and Prevention classification of SSIs. 19 If studies reported overall SSI as a pooled outcome, this was extracted preferentially; studies that reported organ or space SSI, deep incisional SSI, and superficial incisional SSI were extracted and used to represent the event rate. 30-day readmission was defined as readmission to hospital reported by each included study within 30 days of the index surgery. For this review, reoperation rate was defined as any unanticipated return to the operating room following the index procedure and within 30 days of the index surgery. 5-year OS and DFS data were defined as the percentage of patients alive vs alive and without evidence of disease at 5 years following their index surgery, respectively. This data was extracted from tables and text, and in the event that this data was not reported in those forms, they were extracted from estimations of Kaplan-Meier curves.
Risk of Bias Assessment and Certainty of Evidence
Risk of bias for observational studies was assessed using the Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) assessment tool. 20 Four reviewers (SK, VS, MA, and EH) assessed the risk of bias and certainty of evidence independently. Any discrepancies were discussed among the reviewers to reach consensus.
Certainty of evidence for estimates derived from meta-analyses was assessed by Grading of Recommendations, Assessment, Development, and Evaluation (GRADE). 21 As recommended by the Cochrane Collaborative, the GRADE results were collated in a summary of findings table. The GRADEpro software was used for calculations and organization of results into a summary of findings table. 22
Statistical Analysis
All statistical analyses were performed using Stata version 18 (StataCorp, College, TX) and DataParty (Hamilton, ON, Canada). A meta-analysis was performed using an inverse variance random effects model for all comparative data. The threshold for statistical significance was set a priori at P < 0.05. Pooled effect estimates for binary outcomes were estimated with risk ratios (RRs) along with their respective 95% confidence intervals (CIs). Pooled effect estimates for continuous outcomes were estimated with mean differences (MDs) along with their respective 95% CI. Mean and standard deviation (SD) were estimated for studies that only reported median and range or interquartile range (IQR) using the method described by Wan et al. 23 Any missing SD data were then calculated according to the prognostic method. 24 Assessment of the between-study heterogeneity was done using the I2 statistic, with I2 greater than 40% being considered as a sign of considerable heterogeneity. 25 Publication and reporting bias in meta-analyzed outcomes were assessed with funnel plots when data from more than 10 studies were included in the analysis. 26 Egger’s test was used to assess for publication bias. 27 Heterogeneity was explored through the following a priori subgroup analyses: (1) geographic location; (2) operative approach (ie, more than 50% laparoscopic vs less than 50% laparoscopic); and (3) age (mean age <65, ≥65, or <65 non-sarcopenic, ≥65 sarcopenic). A systematic narrative summary was provided for each outcome.
Results
Study Characteristics
After screening 215 studies, 40 non-randomized studies,5,7,15,28-64 totalling 13,422 patients were included. Of these patients, 5432 (40.4%) were classified as sarcopenic. A PRISMA flow diagram of the study selection process is illustrated in Figure 1. Included studies were conducted between 2012 and 2023. Detailed demographic data for each of the included studies is shown in Supplemental Table 1. PRISMA diagram of included studies From: Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. doi: 10.1136/bmj.n71.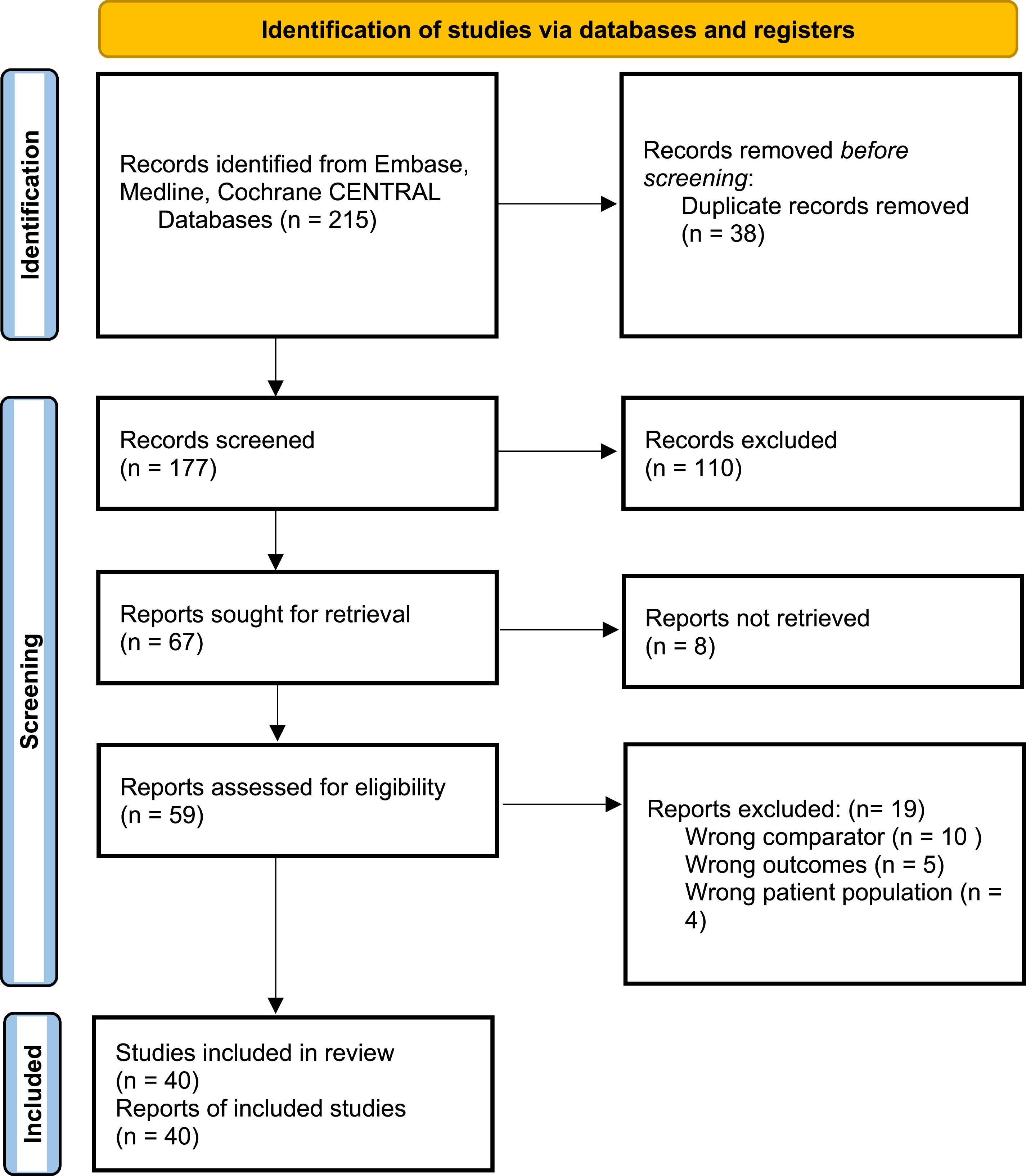
Disease Characteristics and Treatment
Features of disease and treatment course including cancer location, stage, neoadjuvant or adjuvant treatment, and surgery type are included in Supplemental Table 2. Regarding operative approach, 12 studies (30%) reported data on procedure performed, of which right hemicolectomy was the most commonly performed procedure (n = 1,518, 38%). Studies varied in their determination of sarcopenia. The majority of studies (36/40, 90%) used smooth muscle index (SMI) measured from the psoas muscle at the level of L3 in preoperative computed topography (CT) scans. Studies chose the SMI value which defines sarcopenia based on lowest quartile or tertile verses pre-set values based on prior literature in all cases except three.34,40,45
Immediate Postoperative Outcomes
Short-Term Postoperative Outcomes in Sarcopenic and Non-sarcopenic Patients Following Colorectal Cancer Surgery.
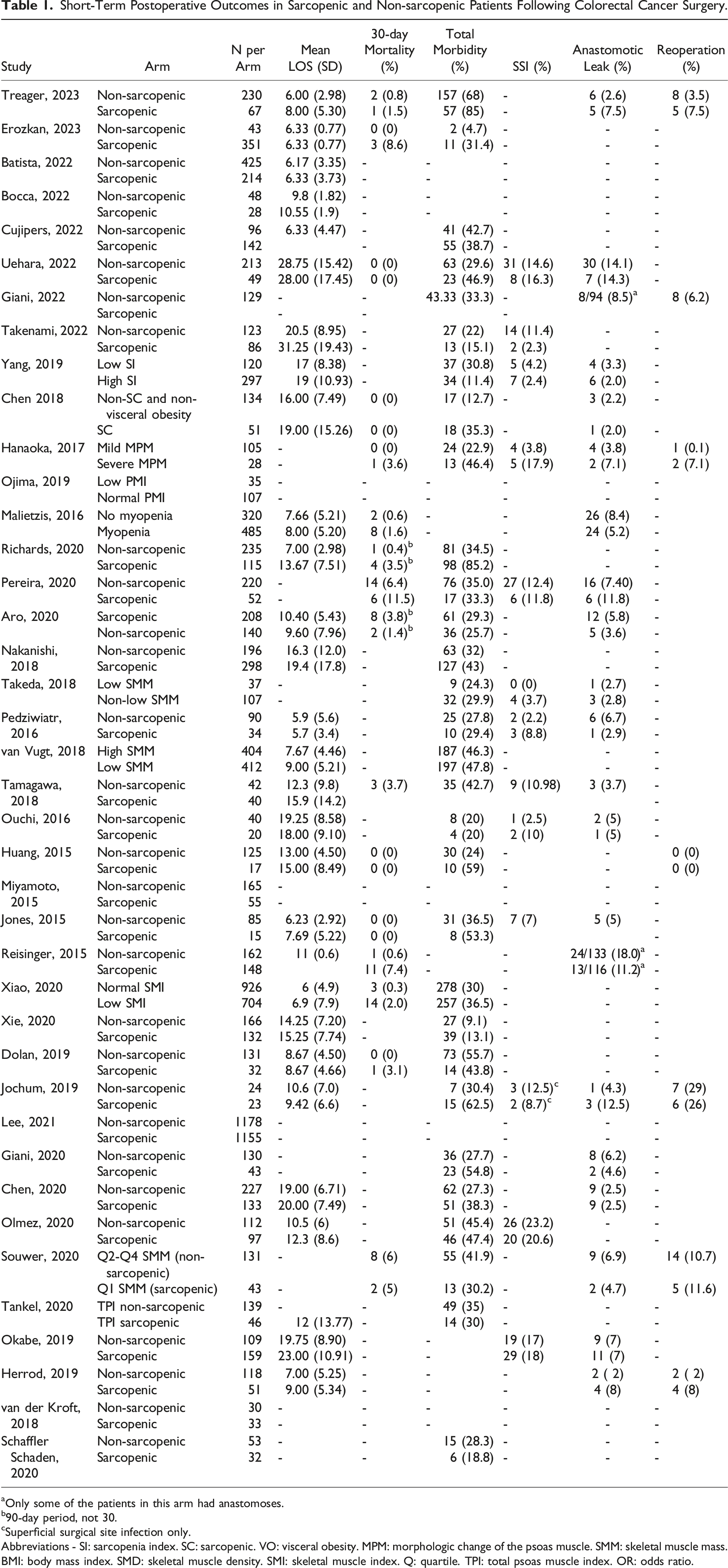
aOnly some of the patients in this arm had anastomoses.
b90-day period, not 30.
cSuperficial surgical site infection only.
Abbreviations - SI: sarcopenia index. SC: sarcopenic. VO: visceral obesity. MPM: morphologic change of the psoas muscle. SMM: skeletal muscle mass. BMI: body mass index. SMD: skeletal muscle density. SMI: skeletal muscle index. Q: quartile. TPI: total psoas muscle index. OR: odds ratio.
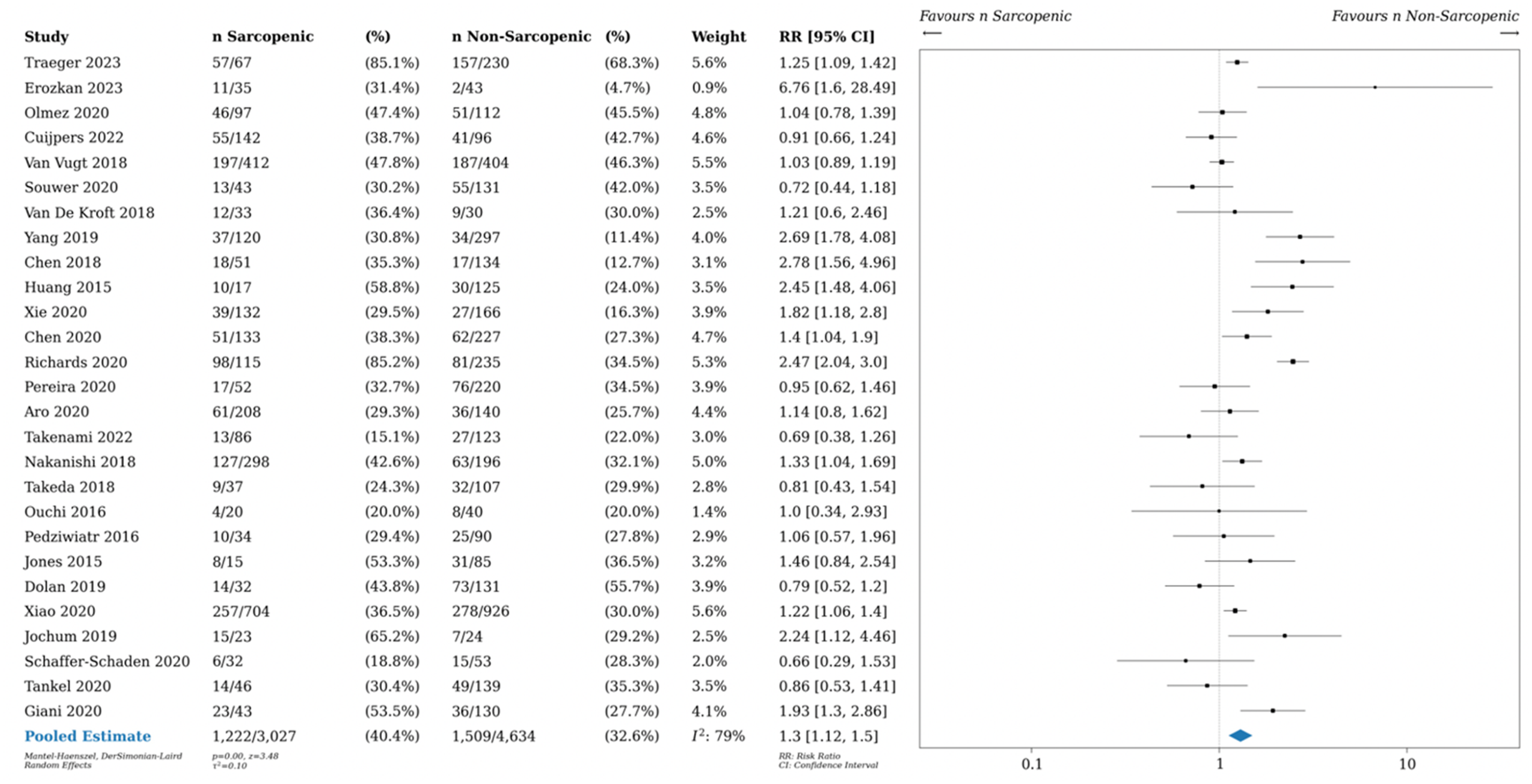
Postoperative morbidity in sarcopenic vs non-sarcopenic patients. Relative risk (RR) shown.
Twenty-six studies (63%) included data on LOS, with comparisons for both patients with and without sarcopenia. The mean difference (MD) in postoperative LOS was 1.46 days longer for patients with sarcopenia (26 studies, 95% CI 0.85-2.07, P < 0.01, I2 82%)), as shown in Figure 3. There was no significant subgroup interaction according to geographic study location (Supplemental Figure 4, X2 = 6.40, P = 0.09, I2 = 53.1%). There was no observed subgroup interaction according to operative approach (Supplemental Figure 5, X2 = 3.08, P = 0.08, I2 = 67.5%) or age (Supplemental Figure 6, X2 = 7.03, P = 0.07, I2 = 57.4%). Length of stay (LOS) postoperatively for sarcopenic vs non-sarcopenic patients. Length of stay with standard deviation (SD), as well as mean difference (MD) shown.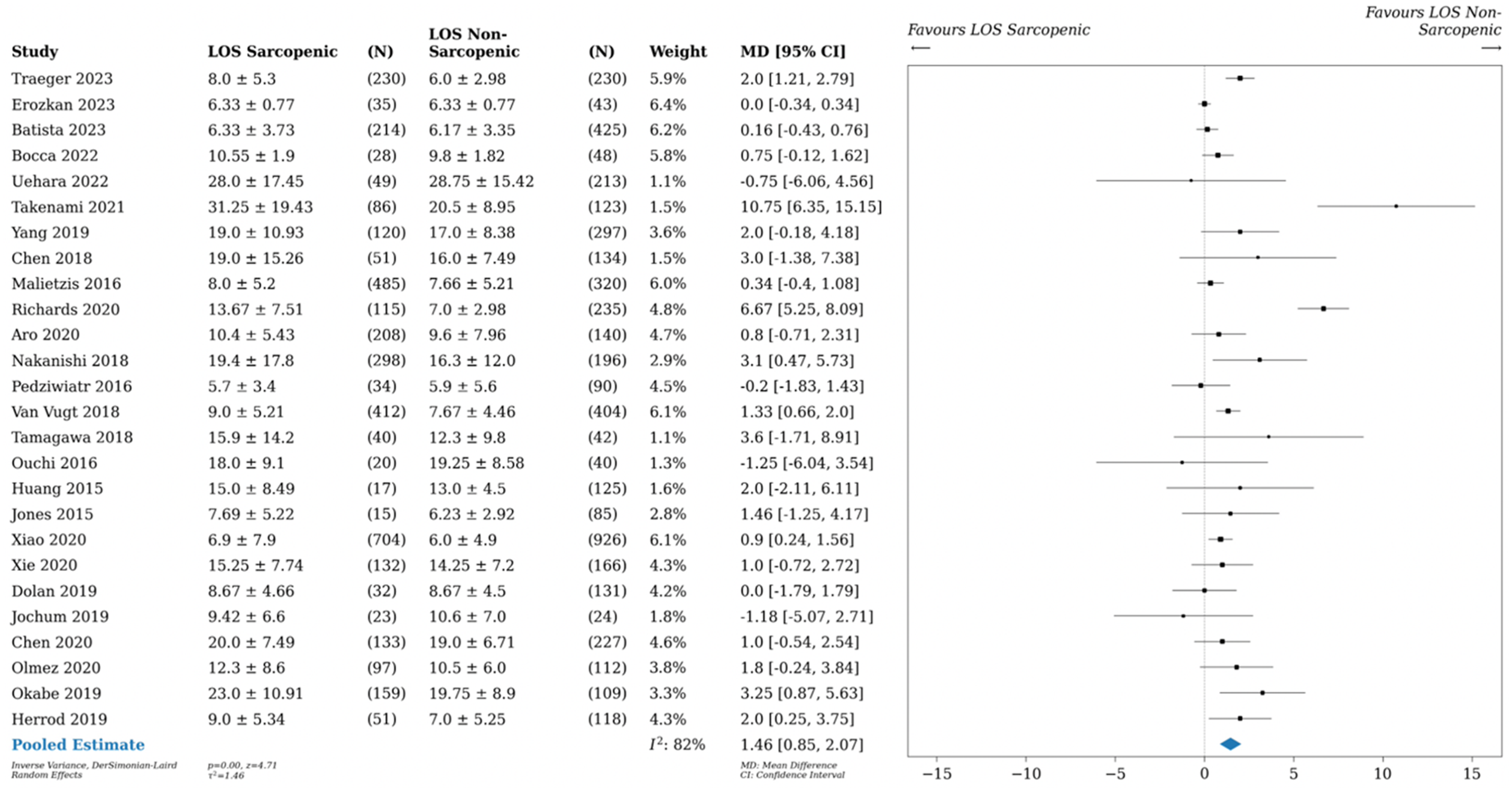
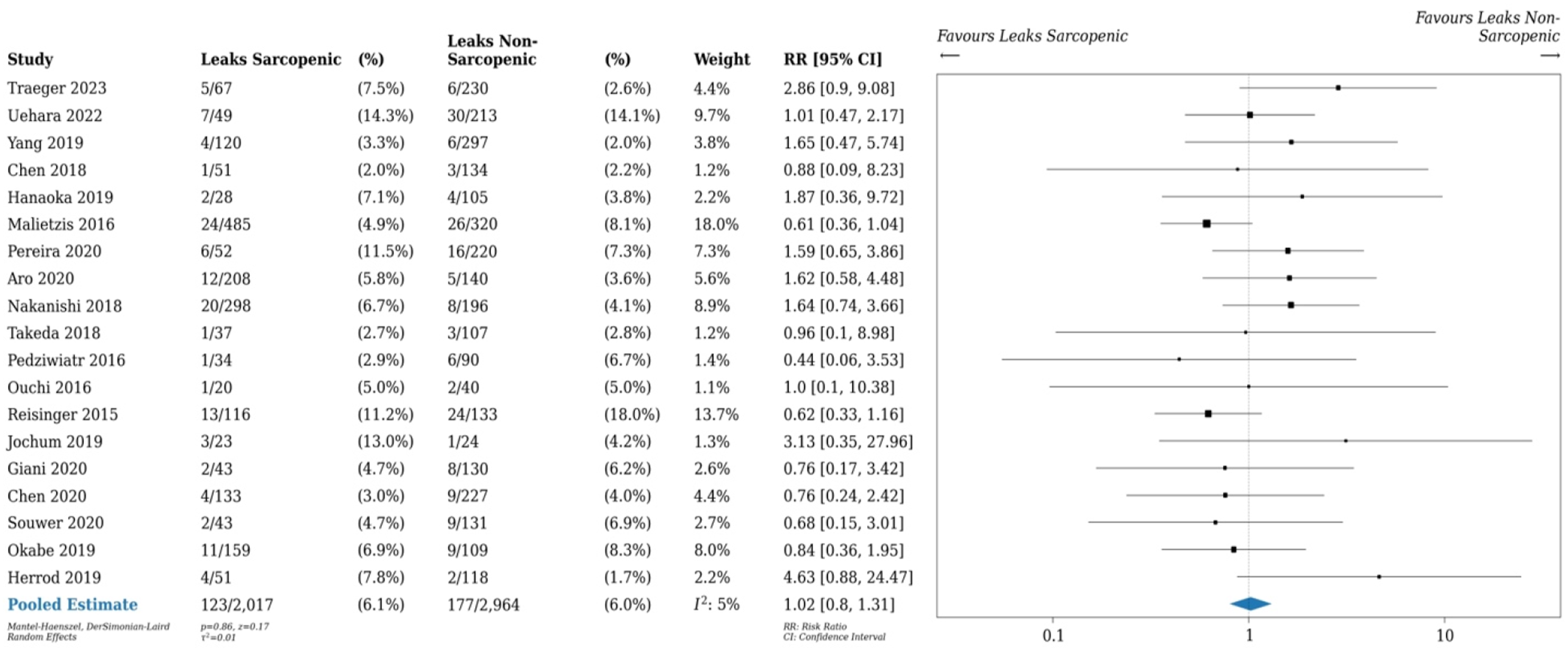
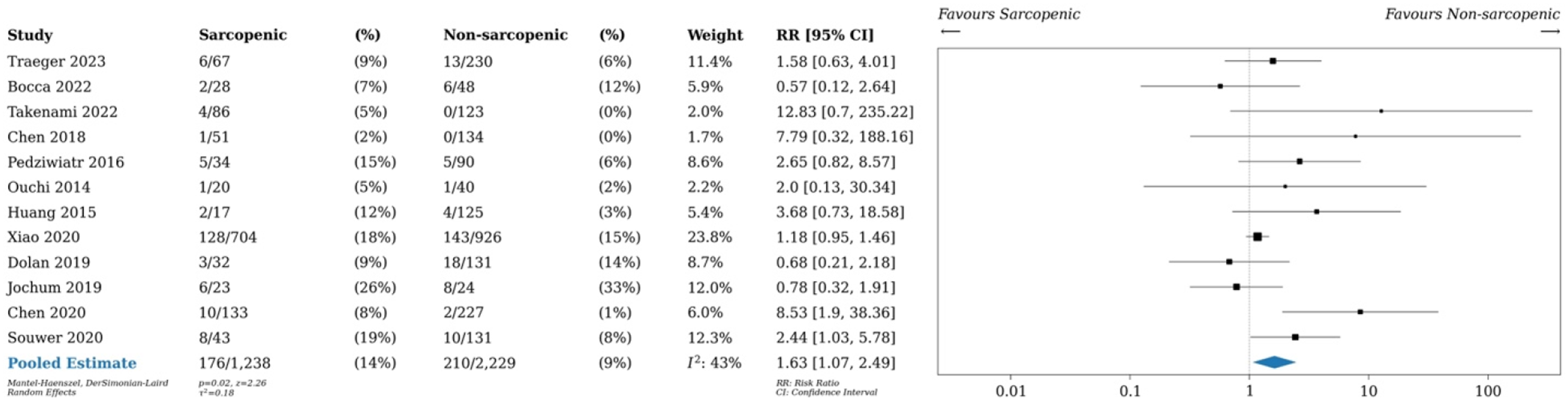
Upon pooling data from 13 studies, patients with sarcopenia had increased risk of 30-day postoperative mortality (2.8% vs 1.0%, RR 2.74, 95% CI 1.63-4.62, P < 0.01, I2 0%), as seen in Figure 4. Pooled data of 19 studies with data on anastomotic leaks showed that there was no difference in risk of anastomotic leak between patients with and without sarcopenia (6.1% vs 6.0%, RR 1.01, 95% CI 0.80-1.31, P = 0.86, I2 5%), as seen in Figure 5. As shown in Figure 6, patients with sarcopenia also were more likely to be readmitted within 30 days of surgery (12 studies, RR 1.63 95% CI 1.07-2.49, P = 0.02, I2 43%),). 30-day postoperative mortality rates in sarcopenic vs non-sarcopenic patients. Relative risk (RR) shown. Anastomotic leak rate in sarcopenic vs non-sarcopenic patients. Relative risk (RR) shown. 30-day readmission rates in sarcopenic vs non-sarcopenic patients. Relative risk (RR) as shown.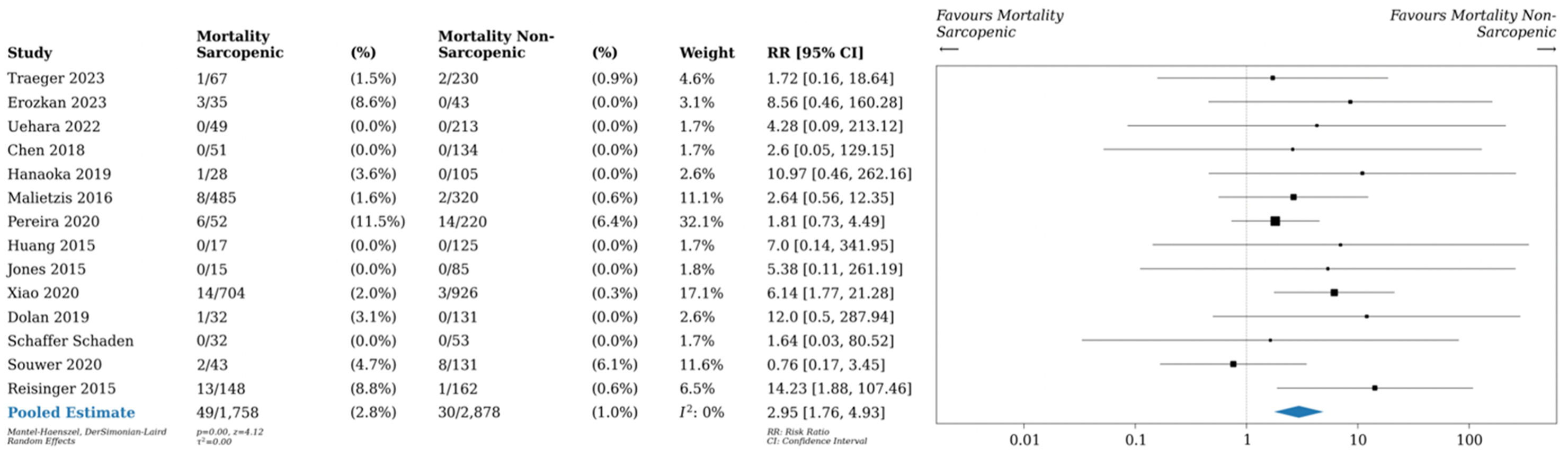
No statistically significant reporting biases were detected using Egger’s test for all of the above outcomes (Supplemental Figure 7).
Long-Term Outcomes
Long-Term Outcomes in Sarcopenic vs Non-sarcopenic Patients Following Colorectal Cancer Surgery.
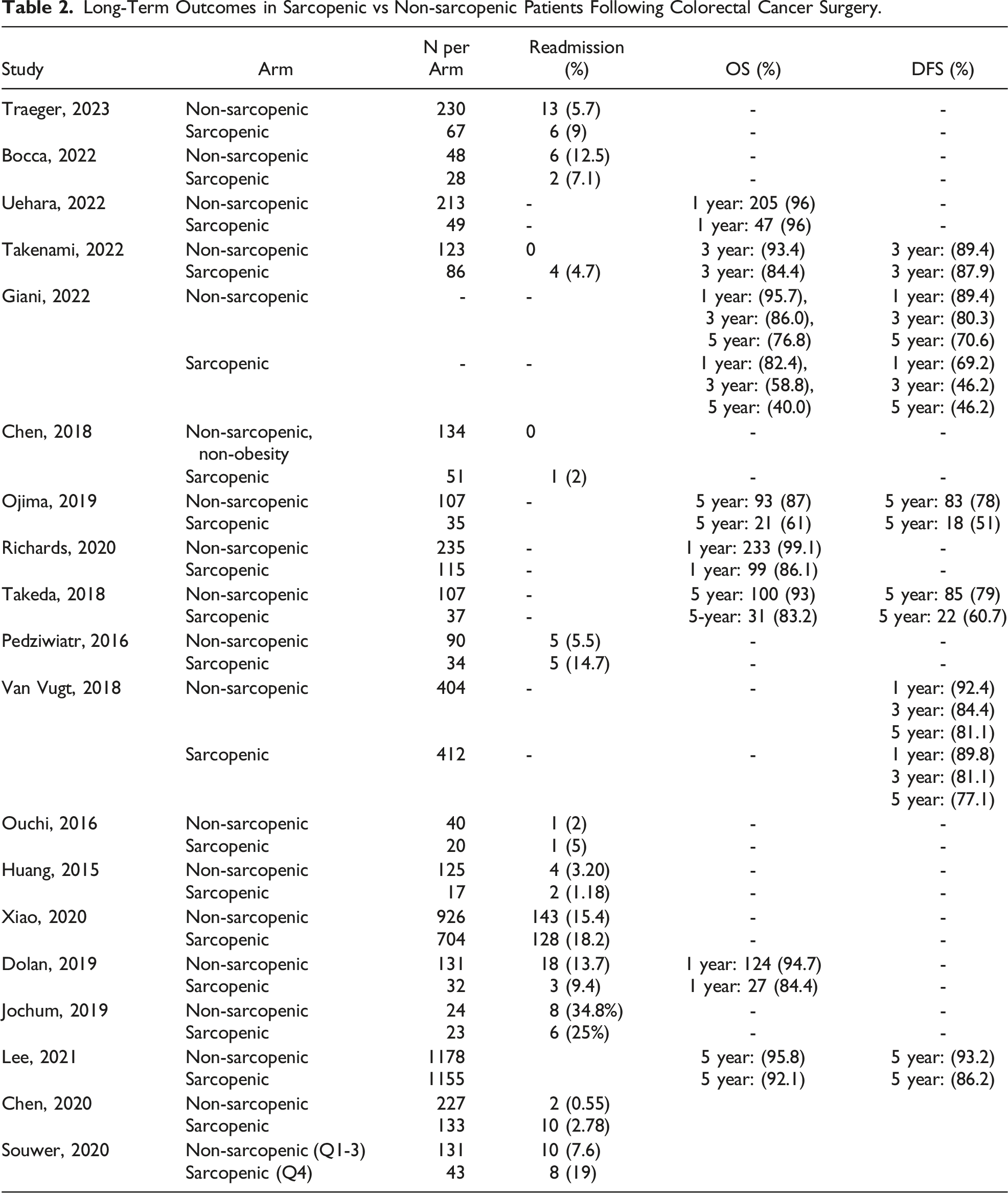
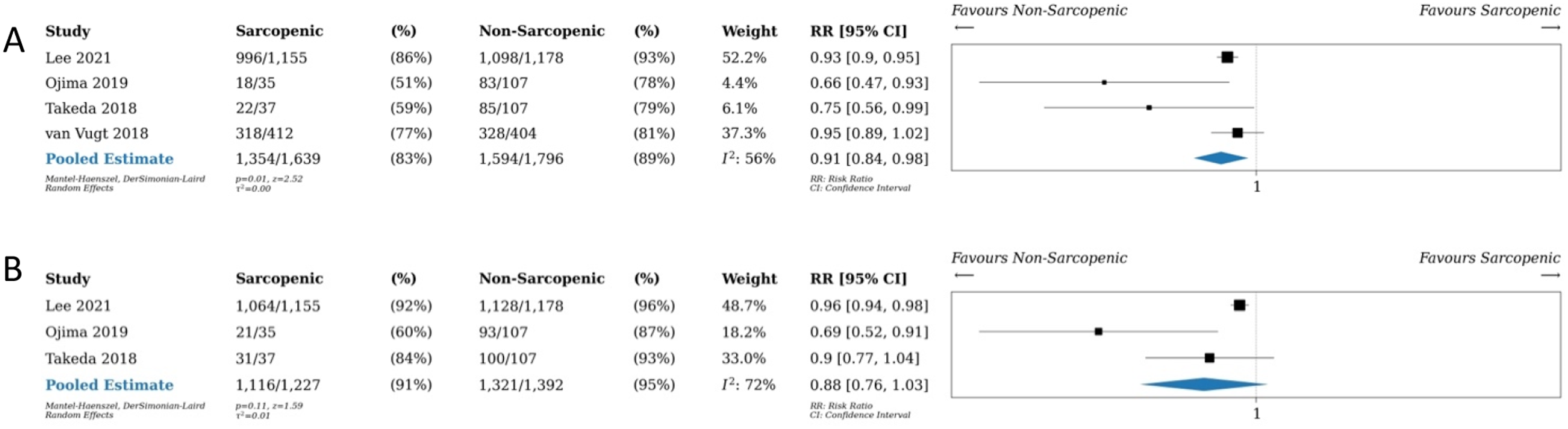
5-year outcomes in sarcopenic vs non-sarcopenic patients including (a) disease-free survival and (b) overall survival (OS). Relative risk (RR) as shown.
Risk of Bias
Risk of bias for observational studies was assessed using the ROBINS-I. The results of this analysis can be seen in Supplemental Figure 8.
Certainty of Evidence
Certainty of evidence for estimates derived from meta-analyses was assessed by GRADE (see Supplemental Figure 9 for further information).
Discussion
Sarcopenia has been associated with poor postoperative outcomes in many fields, especially as it relates to oncology. 65 This review and meta-analysis analyzes the postoperative short- and long-term outcomes in patients with sarcopenia compared to those without sarcopenia undergoing surgery for colorectal cancer, expanding and updating on previous reviews.9,10 Low to very-low certainty evidence demonstrated that there was an increase in 30-day postoperative morbidity (40 vs 33%, RR 1.3, 95% CI 1.12-1.50, P < 0.01) and 30-day postoperative mortality rate (3% vs 1%, RR 2.74, 95% CI 1.63-4.62, P < 0.01), as well as a 1.46 day longer LOS for patients with sarcopenia (26 studies, 95% CI 0.85-2.07, P < 0.01). Patients with sarcopenia also had lower 5-year DFS (4 studies, RR 0.91, 95% CI 0.84-0.98, P = 0.01), but there was no significant difference in 5-year OS (3 studies, RR 0.88, 95% CI 0.76-1.03, P = 0.11).
Previous studies and meta-analyses have shown an association between sarcopenia and adverse postoperative outcomes in gastrointestinal oncology patients,66-68 and colorectal cancer patients in particular.9,69,70 Our study further confirms the increase in morbidity and mortality, as well as longer LOS and poorer long-term oncologic outcomes. With the median age of diagnosis of colorectal cancer being 68 in women and 65 in men, 2 many patients are elderly at the time of surgery. With increasing age naturally comes decreasing muscle mass, and the prevalence of sarcopenia becomes greater. An estimated 5%-13% of those age 60-70 and 11%-50% of those 80 years and older have sarcopenia in the general population. 71 These estimates are likely even higher in the colorectal cancer population, ranging from 12% to 60% depending on the study. 10 Thus, the detrimental impact of sarcopenia on operative outcomes in colorectal cancer is highly pervasive, and it is a preoperative prognostic indicator that clinicians must be increasingly aware of and competent at managing.
In addition to the impact of sarcopenia on surgical outcomes, it may also impact how patients tolerate other aspects of multi-modal care. In rectal cancer in particular, where neoadjuvant therapies are becoming increasingly common, 72 sarcopenia can increase treatment-related complications. 73 Meta-analysis of the colorectal cancer patient population has shown that patients with sarcopenia are more likely to have premature termination of chemotherapy treatment with FOLFOX. 73 Specifically, observational data has shown higher rates of multiple systemic complications, such as neutropenia, neuropathy, thrombocytopenia, mucositis, and others in patients with sarcopenia. 74 Although there are limited data, chemotherapeutic agents are also thought to cause or worsen sarcopenia, 75 which this and other studies have not accounted for when using pre-treatment CT scans for sarcopenia analysis. Thus, as contemporary neoadjuvant therapies become more ubiquitous in clinical practice, the prevalence and impact of sarcopenia on perioperative outcomes may heighten.
The relationship between sarcopenia and adverse outcomes highlights a need to attempt to modify this risk factor through prehabilitation. Prehabilitation may include a variety of interventions, including exercise, nutrition, patient education, and geriatric specialist assessment. 76 Randomized studies have shown that prehabilitation can significantly improve objective measures such as peak volume of oxygen consumption, 77 6-minute walk test, 78 and muscle mass 79 in patients undergoing neoadjuvant therapy before rectal cancer resection when compared to a control group without prehabilitation. A recent RCT of 251 patients with colon or rectal cancer showed that prehabilitation with a three times weekly exercise program, nutritional intervention, psychological support, and smoking cessation led to significant reduction in severe postoperative complications as well as medical complications in colorectal cancer patients. 80 However, meta-analysis of the many RCTs studying prehabilitation has not yet shown significant decreases in postoperative complications. 81
This review and meta-analysis has strengths such as the number of included studies and patients, the comprehensive risk of bias assessment, and GRADE assessment. However, the study limitations include low to very-low certainty of evidence, inclusion of only non-randomized data, and a heterogeneity of patients, pathologies, and methods of sarcopenia assessment between studies. Although almost all included studies used analysis of psoas muscle at the L3 level on CT imaging to diagnose sarcopenia, there were differences among studies’ choice of cutoff values (pre-determined cutoffs vs quartiles) or methods of psoas analysis (short to long axis ratio vs psoas area). This heterogeneity among studies induces uncertainty in our findings and limited the statistical power of certain data analyses. Regarding long-term outcomes, few studies reported 5-year OS and DFS, limiting our statistical power and decreasing the confidence in our effect estimates. The GRADE assessment of the overall quality of evidence was significantly impacted by indirectness and inconsistency. It may still be difficult to ascertain the degree to which sarcopenia impact short- and long-term colorectal surgery postoperative outcomes given the low to very-low certainty of evidence in this meta-analysis. Nonetheless, an RCT aimed at assessing sarcopenia is not possible, and thus, we will have to rely on these meta-analyses of observational data to inform our clinical practices. Lastly, the observational nature of the included data places these findings at risk of selection and confounding bias.
Overall, the presence of preoperative sarcopenia in patients undergoing surgical resection for colorectal cancer is associated with poorer short- and long-term outcomes, including postoperative morbidity and mortality, as well as long-term oncologic outcomes. These data are in keeping with previous systematic reviews and meta-analyses evaluating sarcopenia in the perioperative period. The significant negative impacts associated with sarcopenia should prompt clinicians to further study, develop, and implement prehabilitation programs aimed at building muscle mass and preventing exacerbation of sarcopenia in the preoperative period.
Supplemental Material
Supplemental Material - The Impact of Sarcopenia on Postoperative Outcomes in Colorectal Cancer Surgery: An Updated Systematic Review and Meta-Analysis
Supplemental Material for The Impact of Sarcopenia on Postoperative Outcomes in Colorectal Cancer Surgery: An Updated Systematic Review and Meta-Analysis by Sara Keshavjee, Tyler Mckechnie, Victoria Shi, Muhammad Abbas, Elena Huang, Nalin Amin, Dennis Hong, and Cagla Eskicioglu in The American Surgeon™.
Supplemental Material
Supplemental Material - The Impact of Sarcopenia on Postoperative Outcomes in Colorectal Cancer Surgery: An Updated Systematic Review and Meta-Analysis
Supplemental Material for The Impact of Sarcopenia on Postoperative Outcomes in Colorectal Cancer Surgery: An Updated Systematic Review and Meta-Analysis by Sara Keshavjee, Tyler Mckechnie, Victoria Shi, Muhammad Abbas, Elena Huang, Nalin Amin, Dennis Hong, and Cagla Eskicioglu in The American Surgeon™.
Footnotes
Author’s Contributions
Conception and design of the study: all authors; acquisition of data: Keshavjee, Shi, Abbas, Huang, and McKechnie; analysis and interpretation of data: all authors; drafting and revision of the manuscript: all authors; approval of the final version of the manuscript: all authors; and agreement to be accountable for all aspects of the work: all authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Prospero Registration Number
CRD42023493416.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
