Abstract
Background:
The surgical treatment of gastric and esophageal cancer in Denmark is centralized in four specialized esophagogastric cancer (EGC) centers. Patients are referred after an esophagogastroduodenoscopy (EGD) at a secondary healthcare facility. The EGD is repeated at the specialized EGC center before determining a surgical treatment strategy. This multicenter retrospective study aimed to investigate the quality of EGDs performed at a secondary healthcare facility and evaluate the clinical value of repeated EGD at a specialized center when determining the surgical treatment strategy.
Methods:
Patients from three of the four centers, who underwent esophagectomy or gastrectomy with curative intent from 1 June 2016 to 1 May 2021, were included. EGD reports from the referral facilities and EGC centers were compared based on a predefined checklist. Furthermore, endoscopist experience, the time between examinations, and histology were registered. Finally, it was assessed whether the specialized EGD led to any substantial changes in surgical treatment. Baseline characteristics and differences in EGD reports were described and McNemar’s chi-square test was performed. A logistic regression analysis was conducted to identify risk factors for a change in surgical strategy.
Results:
The study included 953 patients who underwent both an initial EGD and EGD at referral to a specialized center. In 644 cases (68%), the information from the initial EGD was considered insufficient concerning preoperative tumor information. In 113 (12%) cases, the findings in the specialized EGD would lead to a significant alteration in the surgical strategy compared with the primary EGD.
Conclusion:
The findings suggest that repeated EGD at a specialized center is of clinical value and helps ensure proper surgical treatment for patients undergoing curative surgery for gastroesophageal cancer.
Keywords
Context and Relevance
Limited evidence exists regarding the importance of the quality of esophagogastroduodenoscopy (EGD) in the management of gastric and esophageal (gastroesophageal) cancer. This study evaluated the role of repeated EGD at a specialized center when determining a surgical strategy for gastroesophageal cancer in Denmark. The study included 953 patients and found that a significant proportion of EGD reports (68%) presented with insufficient preoperative tumor information. In 12% of the cases, the re-examination at the specialized center resulted in substantial changes in the surgical treatment strategy. This study demonstrates that repeated EGD at specialized centers adds considerable clinical value, ensuring proper surgical treatment for patients undergoing surgery for gastroesophageal cancer.
Introduction
Esophagogastroduodenoscopy (EGD) is the gold standard when diagnosing esophageal and gastric malignancies. A detailed description of location and extent along with sufficient biopsy material is required, when determining a surgical treatment strategy.1,2 In 2020, more than 1,700,000 patients were diagnosed with gastric or esophageal (gastroesophageal) cancer worldwide. 3 In Denmark, approximately 1100 patients are diagnosed annually and around 40% are referred for surgery with a 5-year survival rate of 43%–48%. 4 Since 2003, the treatment of these patients has been centralized in four tertiary, specialized esophagogastric cancer (EGC) centers across the country, leading to a substantially lowered 30-day mortality for gastric and esophageal cancer surgery, reduced from 8.2% to 2.4% and 5.5% to 1.7%, respectively.5,6 Similar results have been observed in other countries following centralization. 7
Most patients undergo an EGD at a secondary healthcare facility (public hospital or private outpatient clinic) (primary EGD). Within 14 days, a second EGD is performed upon referral to the EGC center (specialized EGD) to reassess tumor characteristics, to ensure that additional pathology, such as Barrett’s esophagus or synchronous tumors, is not missed, and to facilitate re-biopsies if necessary. Furthermore, a clinical examination and an 18F-Fluorodeoxyglucose Positron Emission Tomography/CT (18F-FDG PET/CT) are performed. The multidisciplinary team (MDT) determines the surgical and oncological strategy, based on the findings. T1aN0M0 tumors are referred for endoscopic submucosal dissection (ESD) or endoscopic mucosal resection (EMR). Patients with more deep-seated tumors are referred for curatively intended surgery (potentially with neoadjuvant therapy) or palliative treatment, depending on histopathology, TNM-stage, and performance status.1,2,8 The surgical procedures include esophagectomy, subtotal gastrectomy, or total gastrectomy depending on tumor characteristics.
Despite a low rate of adverse events, 9 many patients experience discomfort and pain during EGD.10,11 Furthermore, EGD is time-consuming and expensive for the healthcare system (approximately 1700 USD per procedure in Denmark). 12 In addition, the quality of EGD reports varies significantly, possibly due to the absence of standardized guidelines for describing gastroesophageal cancer upon discovery.
In this study, we investigated the quality of EGDs performed at secondary healthcare facilities and aimed to ascertain the frequency with which subsequent EGDs at a specialized EGC center revealed significant findings that could influence surgical decision-making. Furthermore, we sought to identify potential risk factors for surgical strategy changes after specialized EGD.
Materials and methods
Patient inclusion
In this multicenter retrospective cohort study, the cohort consisted of patients who underwent surgery with a curative intent due to gastroesophageal cancer in the period 1 June 2016 to 1 May 2021. Patients with gastric cancer were included from three of four national EGC centers: Copenhagen University Hospital, Rigshospitalet (center A), Aalborg University Hospital (center B), and Aarhus University Hospital (center C). Patients with esophageal cancer from centers A and C were included in the study. Center B did not routinely perform EGDs during the study period. Instead, they recommended repeated EGDs for specific patients based on the tumor information provided in the report. As a result, patients with esophageal cancer from center B were not included.
Inclusion criteria were as follows: ⩾18 years of age, confirmed esophageal or gastric malignancy, a minimum of two EGDs performed (one before referral (primary EGD) and one at the EGC center (specialized EGD)), assessment by MDT, and surgery with a curative intent.
Patients were excluded if the specialized EGD report was unavailable, if they had not consented to share their health data for quality control, or if they had a gastrointestinal stromal tumor (GIST). Patients were followed from the primary EGD raising cancer suspicions until surgery at EGC center.
Ethics
At the time of referral to the EGC center, all patients were asked to sign a consent form, allowing their data and health information to be reviewed for quality control. The study was approved as quality assurance by the hospital manager at each center, thus institutional review board approval was not needed.
Data collection
The patient group was identified by procedure codes from local databases at each of the EGC centers. The electronic patient records were reviewed individually. Patients were divided into two groups; “esophageal/GEJ cancer” (includes Siewert 1, 2, and 3) and “gastric cancer.” Tumor information was collected, using a predefined checklist depending on tumor group (see Supplementary Table 1 available online), and was registered as present or not, regardless of correctness, along with general information on gastroscopy circumstances and patient characteristics (see section “Results”).
Subsequently, each primary EGD report was deemed sufficient or insufficient as a basis for determining a surgical strategy. Criteria for being sufficient were a malignant biopsy and an available EGD report (not all reports were available at referral to the EGC center). Additional criteria for esophageal/GEJ tumors included distance from incisors to oral tumor border and distance from incisors to anal tumor border or information on gastric involvement. For gastric tumors, additional criteria involved tumor location and information regarding involvement of the GEJ or pylorus. If one or several of the criteria were missing, the report was considered insufficient.
Finally, it was assessed whether the specialized EGD yielded tumor information that would necessitate a different surgical strategy than the plan determined based on the primary EGD. At centers B and C, a surgical strategy was suggested in the patient record at the EGC center prior to the specialized EGD; thus, the data collectors noted whether the specialized EGD resulted in a surgical strategy change. At center A, the surgical strategy following primary EGD was determined retrospectively in accordance with the Danish treatment guidelines.8,13,14 Later, it was compared with the surgical strategy following the specialized EGD (Fig. 1). If a change in the surgical strategy was solely attributed to findings in other examination modalities, it was not noted as a change in strategy. In case of uncertainty, the case was consulted with an EGC specialist. In addition, all cases from center A, where a change was noted after first assessment, were also reviewed by an EGC specialist. All changes in surgical strategy were categorized as one of the following: extended resection, change in procedure type (including change to endoscopic resection), or other. For esophageal/GEJ cancer, a difference of more than 5 cm in the reported distance from incisors to oral tumor border was categorized as a change in surgical strategy, as it would lead to a substantial increase/decrease in the resection extent.
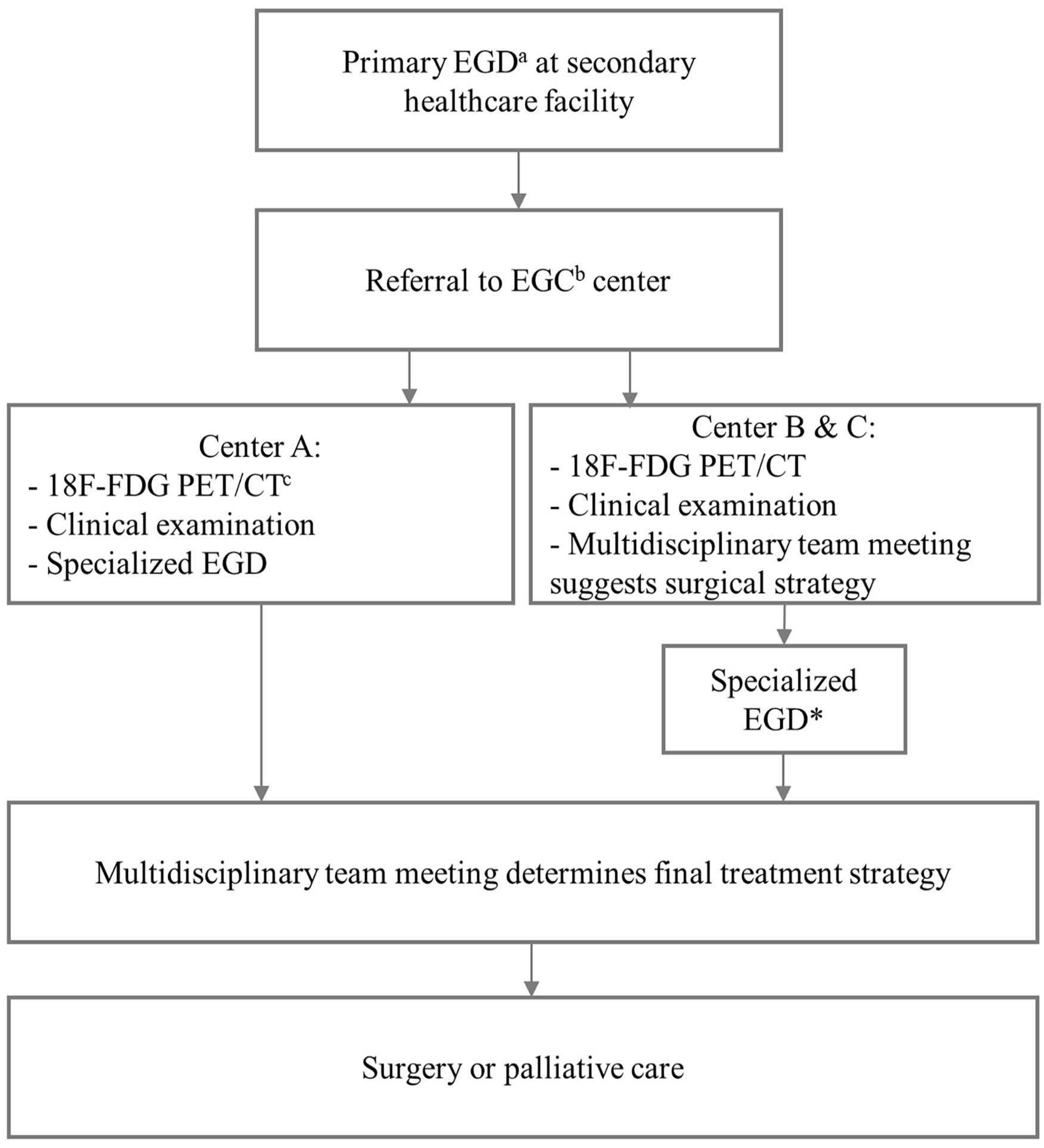
Diagnostic workup procedure prior to surgical treatment at the different EGC centers in Denmark.
Data were collected in the second half of 2021 and the first half of 2022 using Research Electronic Data Capture (REDCap 10.6.18—©2022 Vanderbilt University), hosted at Rigshospitalet.15,16
Statistical analysis
Data analysis was conducted in R (version 4.1.0). 17 Patient and EGD characteristics were described as means with minimum and maximum values or interquartile range, contents of the gastroscopies as binary categorical values (yes/no) using counts and percentages. McNemar’s chi-square test was performed for each report parameter to explore discrepancies between the primary and specialized EGD report.
Logistic regression analyses were conducted separately for esophageal/GEJ and gastric cancer, with change in surgical strategy as the outcome variable to identify potential risk factors. Results were reported as odds ratios (OR) with a 95% confidence interval (95% CI) and a p-value ⩽ 0.05 was considered statistically significant. Listwise deletion was applied to the logistic regression analysis for missing data, including patients with an unassessable case.
Strengthening the reporting of observational studies in epidemiology
The study is reported according to the strengthening the reporting of observational studies in epidemiology (STROBE) guidelines. 18
Results
A total of 1364 patients underwent surgery during the 5-year period. Among these, 953 met the inclusion criteria, with 747 having esophageal/GEJ cancer and 206 having gastric cancer (Fig. 2).
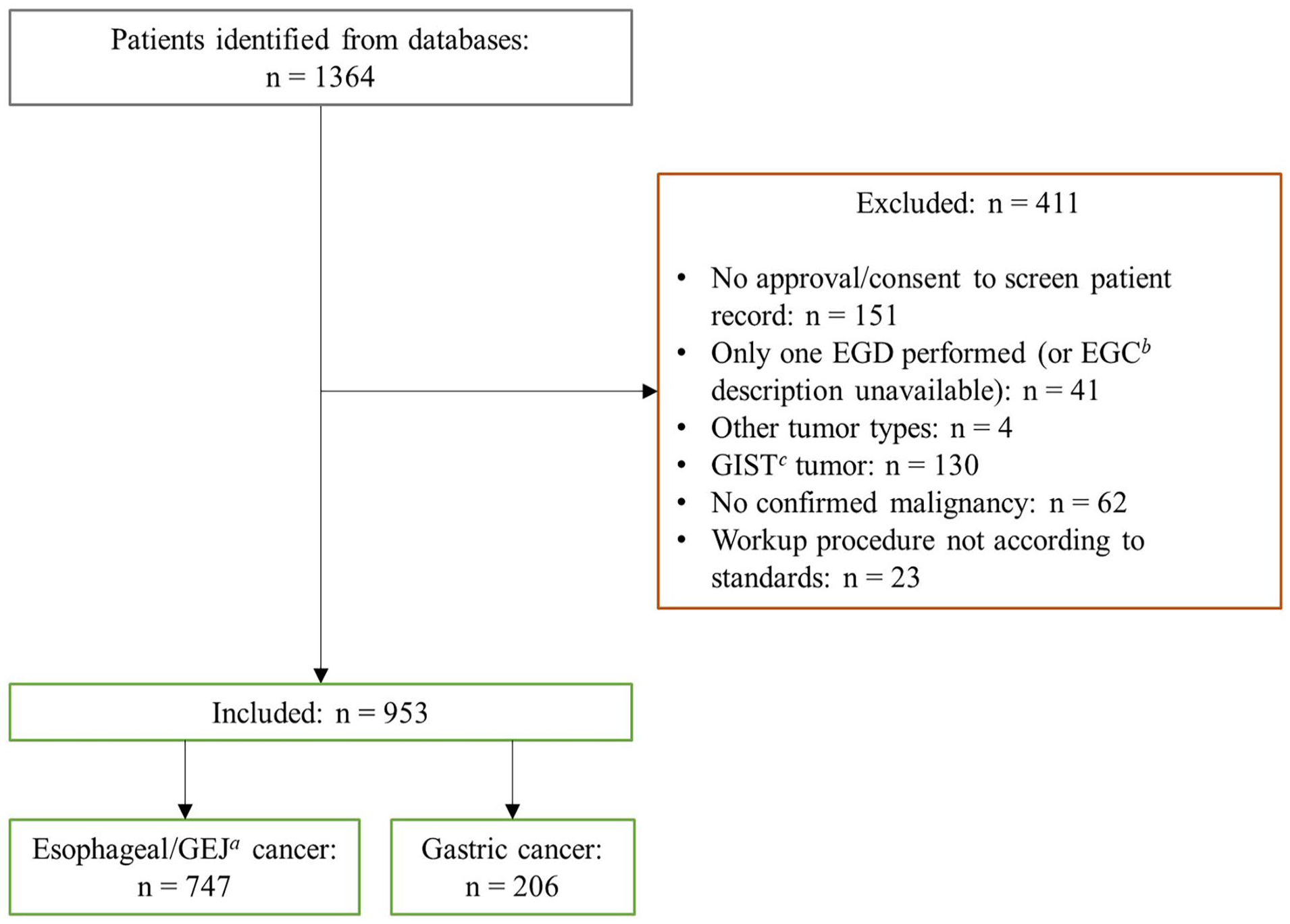
Flowchart on patient inclusion and exclusion.
Patient characteristics and EGD settings are shown in Table 1, stratified by tumor location. Table 2 presents the contents of the EGD reports including the prevalence of parameters only reported in the specialized EGD. In summary, 15% did not have biopsy-confirmed malignancy at referral to the EGC center (see Supplementary Table 2 available online for distribution among tumor types). The EGC center reports more often described conditions such as distance from incisors and gastric involvement compared to the initial reports.
Patient characteristics and EGD a circumstances.
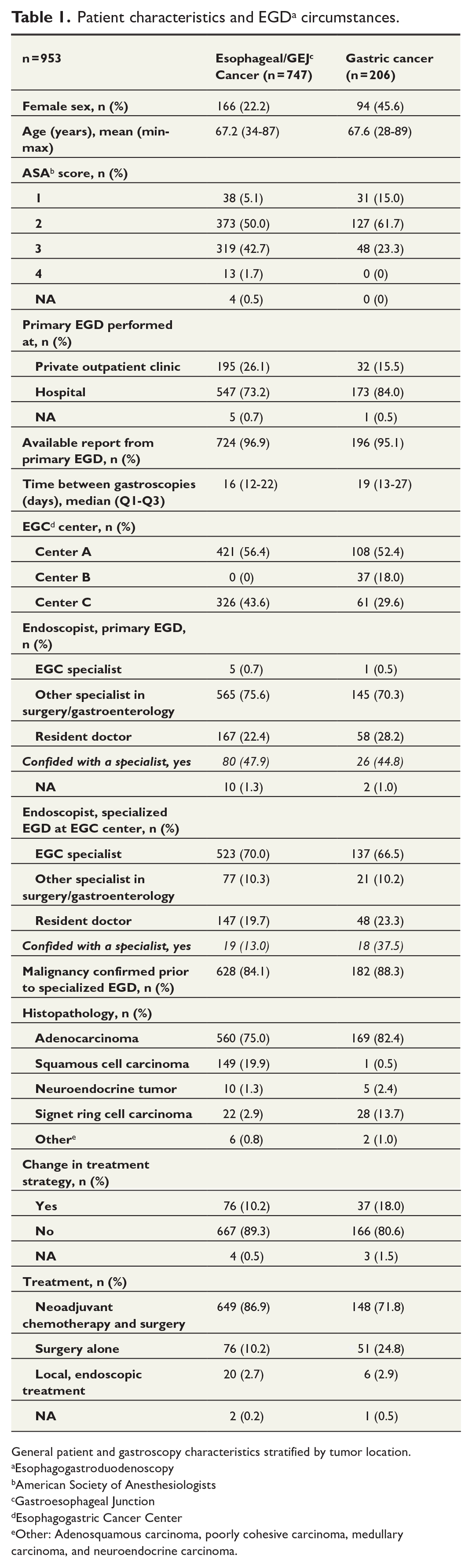
General patient and gastroscopy characteristics stratified by tumor location.
Esophagogastroduodenoscopy
American Society of Anesthesiologists
Gastroesophageal Junction
Esophagogastric Cancer Center
Other: Adenosquamous carcinoma, poorly cohesive carcinoma, medullary carcinoma, and neuroendocrine carcinoma.
EGD a report contents.
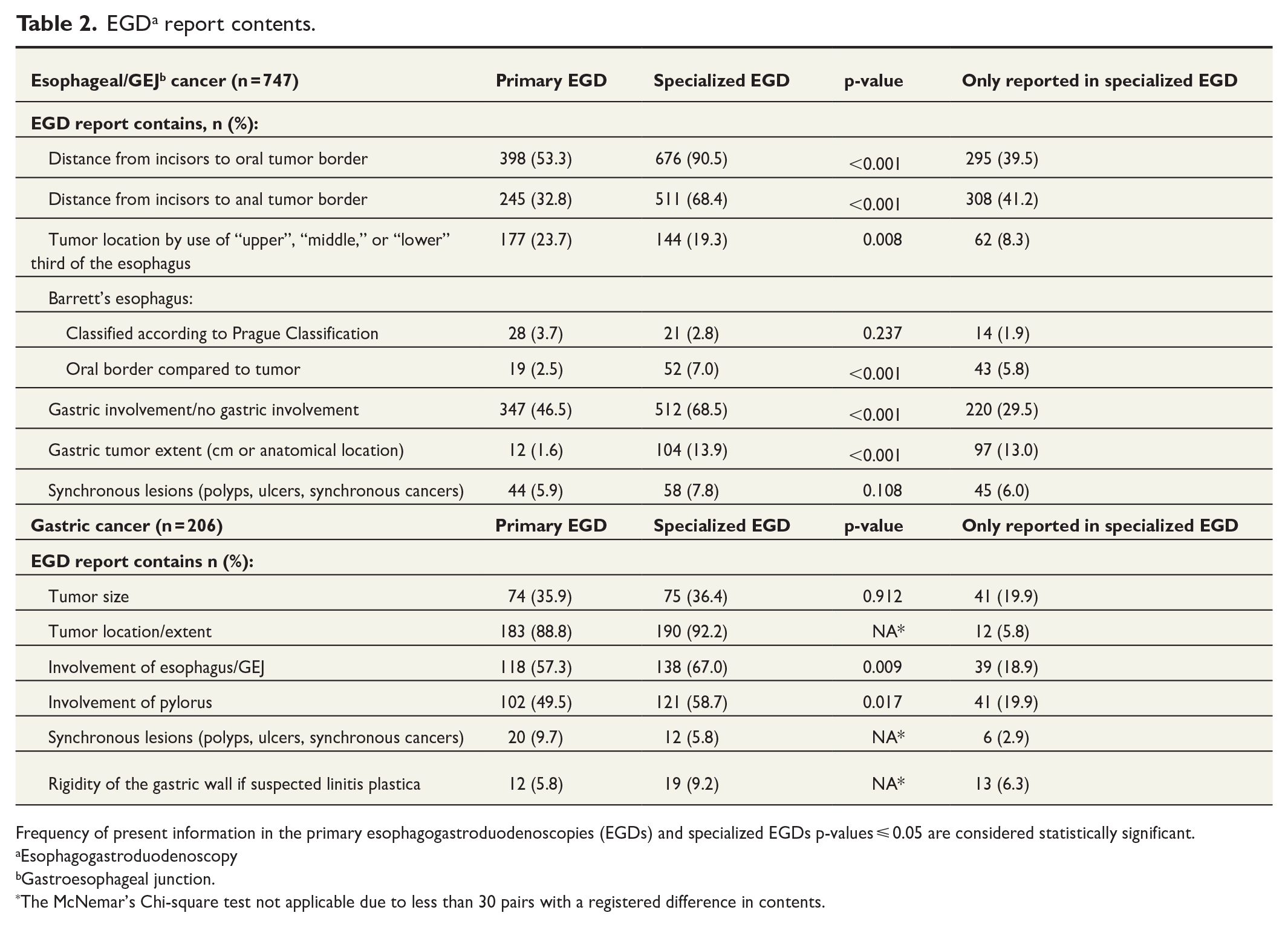
Frequency of present information in the primary esophagogastroduodenoscopies (EGDs) and specialized EGDs p-values ⩽ 0.05 are considered statistically significant.
Esophagogastroduodenoscopy
Gastroesophageal junction.
The McNemar’s Chi-square test not applicable due to less than 30 pairs with a registered difference in contents.
Fig. 3 shows the number of sufficient primary EGD reports and the number of patients with a change in surgical strategy due to specialized EGD along with the nature of the change. Of the primary EGD reports, 309 (32.4%) were considered sufficient in total. However, in 44 (14.3%) cases, a change in surgical strategy after specialized EGD was registered. The remaining 644 (67.6%) were considered insufficient with specialized EGD leading to a change in surgical strategy in 69 cases (10.8%). In seven cases, it was not possible to assess whether there was a change in the surgical strategy.
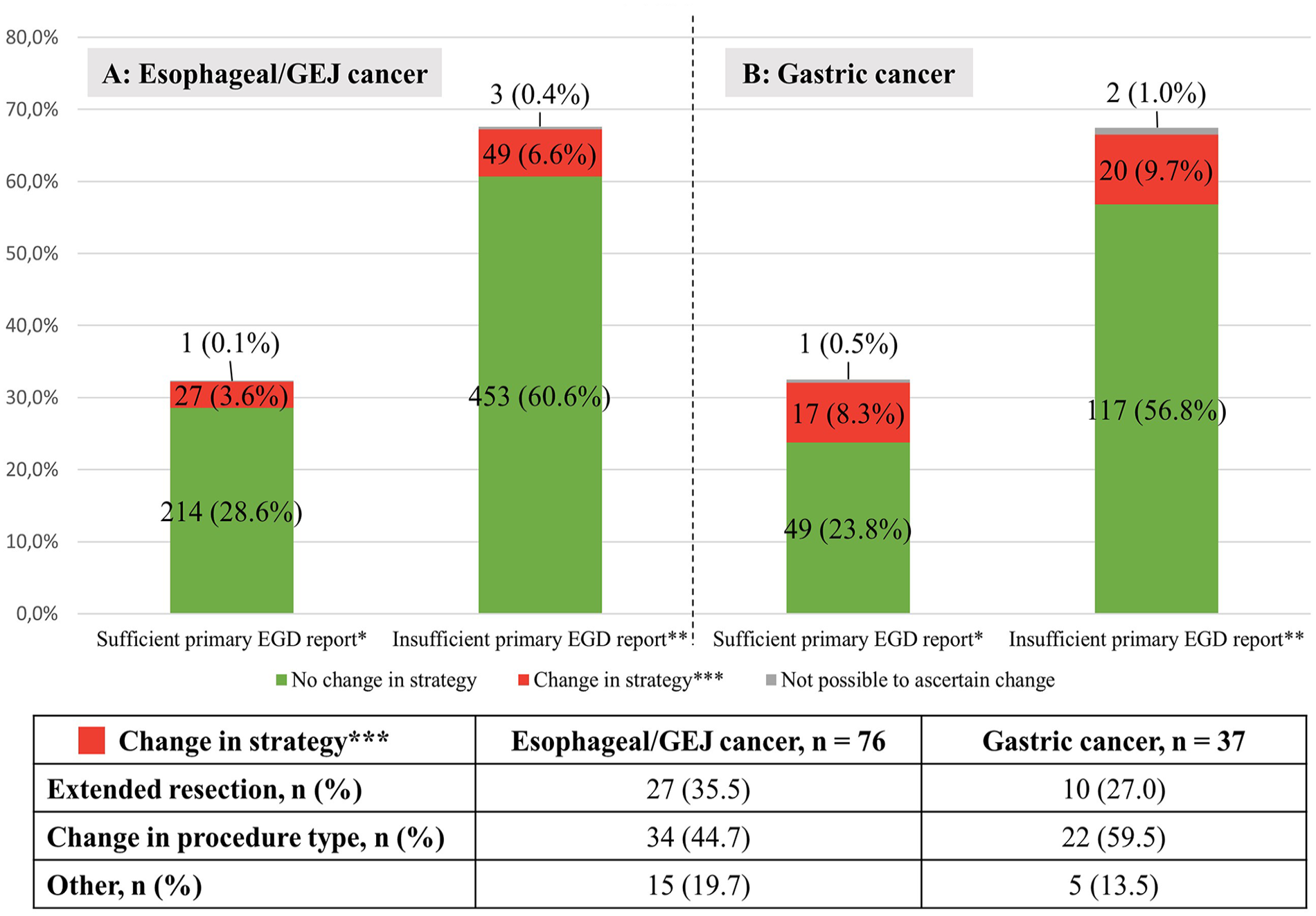
Sufficiency of primary esophagogastroduodenoscopy (EGD) reports depicted with change in surgical strategy after specialized EGD. (A) N = 747. *Sufficient primary EGD’s include (1) a biopsy with confirmed malignancy, (2) a report with information on distance from incisors to tumor, (3) distance from incisors to anal tumor border or information on gastric involvement. **Insufficient primary EGD’s are missing a report altogether or one or several pieces of abovementioned information. (B): N = 206. *Sufficient primary EGD’s include (1) a biopsy with confirmed malignancy, (2) a report with information on anatomical location of tumor, involvement of the pylorus and involvement of the gastroesophageal junction. **Insufficient primary EGD’s are either missing a report altogether or missing one or several pieces of abovementioned information. ***Change in strategy: see section “Results” for elaboration.
For patients with esophageal/GEJ cancer, new information from the specialized EGD resulted in a change in the surgical strategy in 79 (10.2%) cases. Among these, 27 (35.5%) necessitated an extended oral resection. This was primarily due to previously missed Barrett’s esophagus segments, the presence of proximal synchronous tumors, or the oral tumor border being more than 5 cm proximal to the initial tumor description. Another 34 cases (44.7%) involved a change in surgical procedure, that is, change to endoscopic approach due to a less extensive tumor than initially described, a change from esophagectomy to simultaneous total gastrectomy (or vice versa) depending on gastric involvement, or a new biopsy discovering a signet ring cell tumor also requiring a total gastrectomy. In the remaining 15 cases (19.7%), the change was categorized as “other” encompassing less extensive esophagectomies or significant modifications in the of radio/chemotherapy due to revised tumor location, as well as cases where determining a surgical strategy based on the primary EGD was not feasible due to inadequate tumor information.
Among patients with gastric cancer, the EGC gastroscopies led to a change in surgical strategy in 37 (18.0%) cases. Among these, 10 (27.0%) were categorized as an extended resection (from subtotal to a total gastrectomy), typically attributed to newly described involvement of the greater curvature. A change in surgical procedure type was reported for 22 (59.5%) cases. This included changes from gastrectomy to subtotal gastrectomy or ESD/EMR due to a less extensive tumor than initially described and less extensive resections proximally due to ascertainment of the tumor not involving the GEJ. For the remaining five cases (13.5%), determining a surgical strategy based on the primary EGD was not feasible due to inadequate tumor information.
The odds of a change in surgical strategy after specialized EGD (for patients with esophageal/GEJ cancer) was higher for other tumor types (NET, signet ring cell carcinoma, other types) compared with adenocarcinomas (odds ratio (OR) = 3.05, 95% CI = 1.29–6.65, p = 0.01). For patients with gastric cancer, no significant risk factors were found. Results are reported in Table 3.
Multivariate logistic regression analysis.
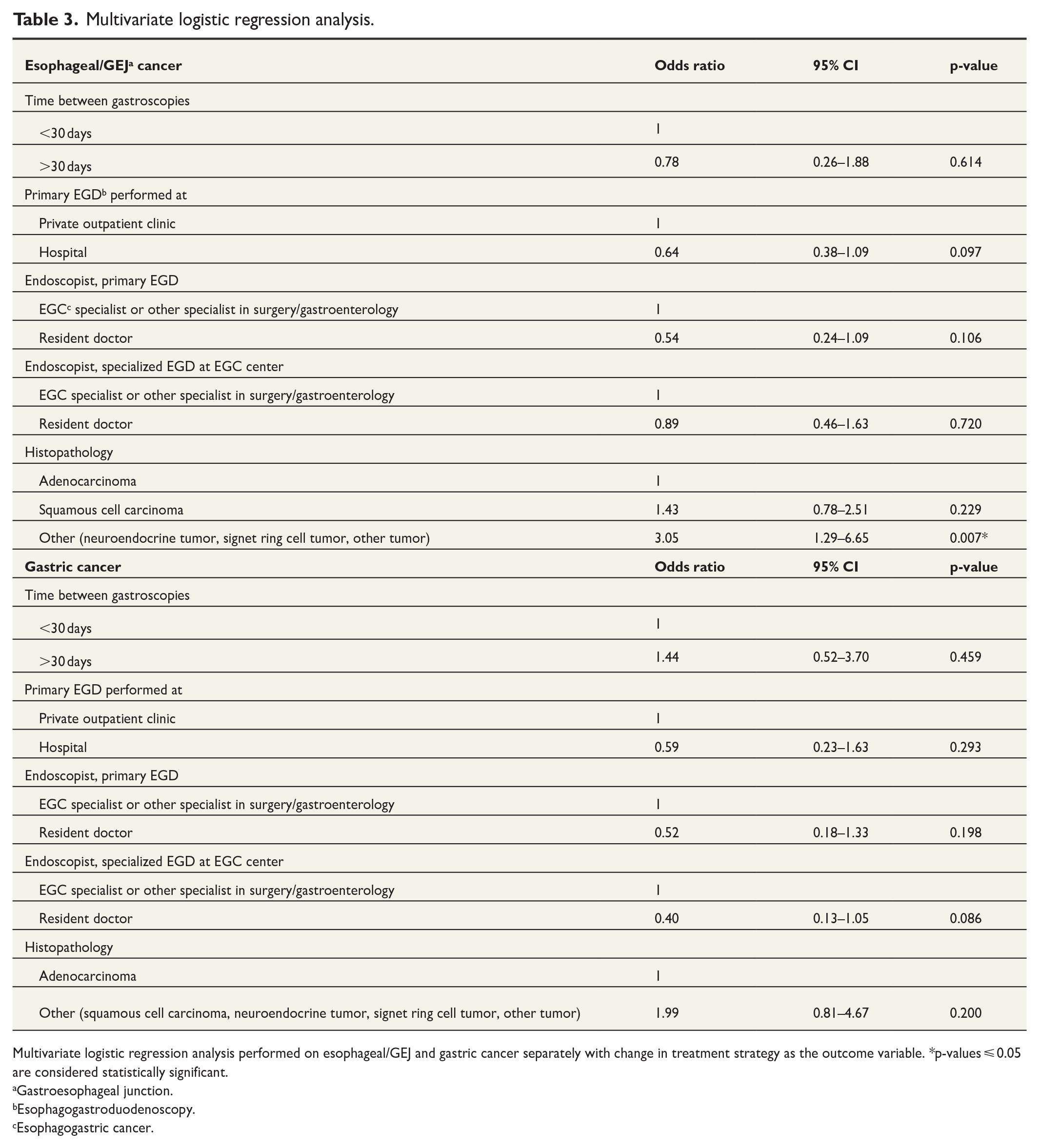
Multivariate logistic regression analysis performed on esophageal/GEJ and gastric cancer separately with change in treatment strategy as the outcome variable. *p-values ⩽ 0.05 are considered statistically significant.
Gastroesophageal junction.
Esophagogastroduodenoscopy.
Esophagogastric cancer.
Discussion
Few studies have examined the value of repeated EGD at a specialized center before gastroesophageal cancer surgery. In this multicenter retrospective study, we found that 67.6% of the cases, the information from the primary EGD reports was insufficient upon referral for surgery. Regardless of whether the primary report was sufficient or not, a subsequent EGD conducted at a specialized center led to a modification in the surgical strategy for 11.9% of cases.
A study from 2018 investigated the quality of endoscopy reports for 119 patients undergoing esophagectomy due to esophageal cancer and found that only 34% of EGD reports met their criteria for “good-quality” reports. 19 Quality was higher at the specialized institution compared to other hospitals. They did not evaluate how often low-quality reports led to inappropriate surgical treatment. Another study examining initial endoscopic descriptions of esophageal squamous cell carcinomas found that tumor extent, upper-, and lower borders were only documented in 51%, 79%, and 41% of reports, respectively, with 14% requiring a repeat EGD due to imprecise tumor description. 20 These findings align with our results.
The American Society for Gastrointestinal Endoscopy (ASGE) lists intraprocedural quality indicators, which include visualizing all relevant organs, obtaining appropriate biopsy specimens, and accurately measuring Barrett’s esophagus. 21 These parameters were considered in our assessment of sufficient EGDs. Although ASGE’s quality indicators do not explicitly include a description of malignancy location and extent, we considered this crucial, as EGD forms the basis for treatment planning.
In our study, 4% of cases revealed new findings during specialized EGD that led to extended resection. Not repeating the EGD could potentially have led to R1/R2 resections with a poorer prognosis than R0 resections.22,23 A retrospective study from 2007 examining the overall survival in relation to the resection margin of GEJ cancer found a 5-year survival rate of 47% for patients with a proximal resection margin >3.8 cm and only 29% for patients with a proximal resection margin <3.8 cm. 24 This highlights the importance of knowing the precise tumor location before surgery to secure a resection margin of at least 5 cm. In addition, patients with unresected Barrett’s esophagus segments are at risk of developing novel carcinomas, with studies showing more than a 10% risk per year if high-grade dysplasia is present.25,26
In at least 21 cases (2.3%), the specialized EGD revealed a shorter tumor segment than initially reported, leading to a less extensive removal of the esophagus or stomach. Several studies comparing total gastrectomy versus subtotal gastrectomy for distal gastric cancer have reported a higher quality of life for patients after subtotal gastrectomy.27 –29 In six cases of this study, the patients were offered an endoscopic resection instead of an esophagectomy based on novel information from the specialized EGD. For T1a tumors, an ESD is advantageous compared with an esophagectomy regarding both operation time and adverse events. However, a meta-analysis from 2015 suggests higher incidence of local cancer recurrence. 30
A logistic regression analysis was performed to identify risk factors for a change in surgical strategy after specialized EGD. The only significant risk factor found was “other tumor types” (i.e. not adenocarcinomas or squamous cell carcinomas) in the esophageal/GEJ cancer group, with an OR of 3.05 (95% CI = 1.29–6.65, p = 0.01) compared to adenocarcinomas. This may be attributed to the relative rarity of these tumor types as endoscopists are less accustomed to describing them.
The specialized EGD was considered the gold standard, and any new tumor information was considered correct. While using the EGC description as the gold standard may introduce some bias, the endoscopists at the specialized center generally have more experience than endoscopists at the secondary facilities, when it comes to providing relevant information before surgery for gastroesophageal cancer. Table 2 reveals that endoscopists at the EGC center were more likely to include the information in their reports, for most of the predefined parameters. This may be because the parameters were defined by EGC specialists who considered the information highly relevant when determining the safest surgical strategy. It is also likely that the primary focus during the primary EGD is finding a tumor, and a detailed description is not deemed necessary, with the knowledge that the EGD is routinely repeated at a specialized center. Importantly, the study results did not indicate any correlation between endoscopist experience and a change in surgery after specialized EGD.
The group of patients with insufficient EGD reports constituted 15% without a malignant biopsy upon referral to the EGC center. This biopsy is crucial when determining the appropriate oncological and surgical strategy.1,2 The group also encompassed reports with unclear tumor location. While this may not always lead to a change in the surgical plan, it remains essential to know the precise tumor location before initiating neoadjuvant chemotherapy, as some tumors cannot be macroscopically identified during surgery due to a complete response to chemotherapy. In this study, 87% received neoadjuvant chemotherapy.
One approach to address the issue of insufficient reports from the initial examination is to introduce a template for describing tumors. However, EGC is still a rare disease in the Western world, compared to, for example, colorectal cancer, which most endoscopists encounter regularly. 31 Even at the specialized centers, critical parameters were not consistently reported, emphasizing the value of centralization, and the introduction of more specific reporting guidelines. A potential disadvantage could occur if the guidelines are too inflexible and fail to include how to describe atypical tumors.
The study’s retrospective nature enabled the inclusion of a large patient cohort. While data were collected at several different centers, the study was designed by EGC specialists, and the EGD reports assessment was aligned using the same methodology across the centers. Some requirements in the predefined checklist might have contributed to an overestimation of insufficient reports. For instance, the requirement to describe the involvement of GEJ in a tumor located in the distal part of the stomach. On the contrary, a report was considered sufficient if it included tumor length even if it did not specify whether the GEJ was involved. Another limitation of the study was that a surgical strategy was not documented at center A before the specialized EGD, which could introduce potential bias. Nonetheless, the use of national guidelines on surgical approach based on endoscopic findings, likely mitigated this bias. In addition, any uncertainty regarding changes in surgical strategy, prompted consultation with an EGC specialist. Other potential limitations include not considering images attached to the EGD reports when assessing quality. Conversely, only patients who underwent surgery were included, excluding a potential group where the specialized EGD uncovered unexpected findings rendering the patients inoperable.
Follow-up was limited to the surgical procedure. Data on 5-year survival and cancer recurrence could provide additional insights into the value of repeated gastroscopies.
Finally, the study was conducted within a Danish context, following local guidelines and treatment protocols. Nevertheless, the results hold significant value due to the extensive data set and the inclusion of a substantial portion of the patient population across the country. Consequently, the findings are relevant for endoscopists and surgeons globally using similar methods when diagnosing and treating esophageal and gastric cancers. The study also highlights the value of centralization to ensure proper tumor description, and thus the highest quality specialized treatment.
Conclusion
The findings of this study suggest that a specialized EGD prior to surgery is of high value, potentially leading to a more appropriate extent of resection and possibly better patient outcomes. In 68% of cases, the initial report was considered insufficient as a basis for planning the surgical treatment and in 12% of cases, findings in the specialized EGD would lead to a significant alteration of the surgical strategy.
Supplemental Material
sj-docx-1-sjs-10.1177_14574969241242202 – Supplemental material for Assessing the benefits of repeated esophagogastroduodenoscopy at a specialized center before gastric and esophageal cancer surgery
Supplemental material, sj-docx-1-sjs-10.1177_14574969241242202 for Assessing the benefits of repeated esophagogastroduodenoscopy at a specialized center before gastric and esophageal cancer surgery by Astrid Kolind Christensen, Charlotte Egeland, Jens Bjoern Heje, Sofia Kamakh Asaad, Roberto Loprete, Trygve Ulvund Solstad, Daniel Kjaer, Sarunas Dikinis and Michael P. Achiam in Scandinavian Journal of Surgery
Supplemental Material
sj-docx-2-sjs-10.1177_14574969241242202 – Supplemental material for Assessing the benefits of repeated esophagogastroduodenoscopy at a specialized center before gastric and esophageal cancer surgery
Supplemental material, sj-docx-2-sjs-10.1177_14574969241242202 for Assessing the benefits of repeated esophagogastroduodenoscopy at a specialized center before gastric and esophageal cancer surgery by Astrid Kolind Christensen, Charlotte Egeland, Jens Bjoern Heje, Sofia Kamakh Asaad, Roberto Loprete, Trygve Ulvund Solstad, Daniel Kjaer, Sarunas Dikinis and Michael P. Achiam in Scandinavian Journal of Surgery
Footnotes
Author contributions
A.K.C., C.E., D.K., S.D., and M.P.A. contributed to conception and design. A.K.C., C.E., J.B.H., S.K.A., R.L., T.U.S., D.K., S.D., and M.P.A. contributed to analysis and interpretation of data. A.K.C. and C.E. contributed to drafting on the article. A.K.C., C.E., J.B.H., S.K.A., R.L., T.U.S., D.K., S.D., and M.P.A. contributed to critical revision of the article. A.K.C., C.E., J.B.H., S.K.A., R.L., T.U.S., D.K., S.D., and M.P.A. contributed to final approval of the article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr A.K.C., Dr S.K.A., Associate Professor S.D., Dr C.E., Dr J.B.H., Dr R.L., and Dr T.U.S. have nothing to disclose. Associate Professor M.P.A. is a chairman of the Danish Esophagogastric Cancer Group (DEGC) and has received grants from Medtronic and Arthrex. Associate Professor D.K. is a board member of the DEGC and the Danish Surgical Society.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
