Abstract
Background and objective:
As markers of sarcopenia, psoas muscle areas and indexes measured from computed tomography images have been found to predict long-term mortality in cardiothoracic as well as other surgical cohorts. Our objective was to investigate the association between psoas muscle status, taking into account muscle density in addition to area, and survival among patients undergoing open thoracic aortic reconstruction.
Methods:
This was a retrospective registry study of a total of 451 patients treated with open surgery for thoracic aortic pathology. Psoas muscle area and density were measured from preoperative computed tomography images at the L3 and L4 lumbar levels. In addition, lean psoas muscle area was calculated by averaging sex-specific values of psoas muscle area and density. The association between mortality and psoas muscle status was analyzed with adjusted Cox-regression analysis.
Results:
The median age of the study population was 63 (interquartile range (IQR): 53–70) years. The majority were male (74.7%, n = 337) and underwent elective procedures (58.1% n = 262). Surgery of the ascending aorta was carried out in 90% of the patients, and 15% (n = 67) had concomitant coronary artery bypass surgery. Aortic dissection was present in 34.6% (n = 156) patients. Median follow-up time was 4.3 years (IQR: 2.2–7.4). During the follow-up, 106 patients (23.5%) died, with 55.7% of deaths occurring within the first four postoperative weeks. Psoas muscle parameters were not associated with perioperative mortality, but significant independent associations with long-term mortality were observed for psoas muscle area, density, and lean psoas muscle area with hazard ratios (HRs) of 0.63 (95% confidence interval (CI): 0.45–0.88), 0.62 (95% CI: 0.46–0.83), and 0.47 (95% CI: 0.32–0.69), respectively (all per 1-SD increase).
Conclusions:
Psoas muscle sarcopenia status is associated with long-term mortality after open thoracic aortic surgery.
Context and Relevance
Psoas muscle parameters have been associated with postoperative outcomes after both conservative and operative treatment of thoracoabdominal aneurysm and total aortic arch repair as well as in a wide range of other cardiovascular procedures. In this study, psoas muscle area and density and lean psoas muscle area measured from computed tomography scans were independently associated with long-term mortality after open thoracic aortic reconstruction. Surgery of the thoracic aorta is associated with formidable risks. Results of this study are in line with previous findings and have value in providing measurable indices of overall prognosis by helping identify patients with risk of inferior long-term survival after open thoracic aortic surgery.
Introduction
In recent years, there has been a growing interest in investigating aging-related conditions such as frailty and sarcopenia, and their association with postinterventional survival in surgical cohorts. Sarcopenia, the involuntary decrease in skeletal muscle mass, quality, and consequently strength associated with age, has been discovered to be associated with inferior postoperative outcomes after a wide range of cardiovascular procedures1–6 and it has been estimated with different methods including muscle area and/or density measurements. Applying muscle parameters as markers of sarcopenia has proved to be both reproducible and reliable, and when utilizing routine computed tomography (CT) imaging, also convenient for clinical work without generating additional costs.7–10 Specifically, the area of psoas muscles with or without adjusting with patient height or body surface area (BSA; psoas index), has been applied in several studies.
Open thoracic aortic procedures are particularly high-risk interventions associated with considerable perioperative and postoperative mortality and morbidity, particularly in the emergency setting. Survival after surgical intervention is linked to several patient-related factors such as advanced age, previous cardiac surgery, and comorbidities such as renal dysfunction, coronary artery or valve disease, and chronic obstructive pulmonary disease. Furthermore, lower psoas muscle areas (PMAs) and indexes have been found to be independently associated with inferior postoperative outcomes after different types of open cardiothoracic surgical procedures including thoracic aortic interventions.11–16
While tools for risk prediction such as the EuroSCORE II exist, 17 CT-defined muscle parameters offer a promising yet simple method for risk evaluation in patients undergoing open thoracic aortic reconstruction. However, more research is needed to expand on current knowledge and bring muscle parameter evaluation into clinical practice. Furthermore, previous studies on the association of muscle parameters with postoperative outcomes in cardiothoracic surgery have mainly focused on muscle areas and indexes with less data on the potential effect of muscle quality. Moreover, many studies have utilized the lowest muscle area/muscle index quartiles as cutoffs for sarcopenia, which could be considered somewhat arbitrary. The purpose of this study was to investigate the association of PMA and psoas muscle density (PMD) measured from routine preoperative contrast-enhanced CT scans, as markers of sarcopenia, with postoperative survival in a contemporary cohort of patients undergoing open thoracic aortic reconstruction.
Methods
Patients
This was a retrospective registry study. The study population consisted of consecutive patients that underwent open surgery for thoracic aortic pathology at Tampere Heart Hospital between January 2007 and September 2018. We retrospectively collected the data and CT images from the Heart Center database and the Tampere University Hospital patient record database, which is linked to the continuously updated National Population Register. All suitable patients with available CT imaging of the thoracic and abdominal area from a period extending from 1 year prior to the operation to the time of the index procedure were included, and in over 90% of the patients the imaging was conducted a maximum of 6 months before the operation. Open thoracic aortic surgery encompassed the following operation types: resection of the aortic root and ascending aorta with reimplantation of coronary arteries and use of a mechanical or a biological valve prosthesis and a tube graft (Nordic Medico-Statistical Committee (NOMESCO) codes: FCA60, FCA70), repair of the aortic root with a valve sparing technique (FCA75), resection and reconstruction of the ascending aorta using a tube graft (FCA50), resection and reconstruction of the aortic arch using a tube graft and reimplantation of branches (FCB40, FCB50), resection of the descending aorta (and reimplantation of branches) (FCC60), and other repair of the thoracic or thoracoabdominal aorta (FCA96, FCD96, FCW96). During the study period, altogether 735 patients were subjected to open surgery of the thoracic at the study center. Of these, 451 (48.2%) individuals were included in the study. The main reason for exclusion was the absence of sufficient quality contrast-enhanced CT imaging within 1 year before surgery. The follow-up lasted until 2 April 2020 with no loss to follow-up.
This study was conducted adhering to the ethical principles of the Helsinki Declaration and approved by the local institutional review board with permit number R19548S. Due to the retrospective nature of this registry study, no formal patient consent was required or obtained.
Image analysis, variables, and measurements
It has been previously demonstrated that PMA and PMD can be reliably and reproducibly measured from preoperative contrast-enhanced CT images. 7 For the image analyses, we applied contrast-enhanced arterial phase aortic CT images in this study. The psoas muscle measurements were performed at the L3 and L4 lumbar levels applying the same method. Measurements were carried out by a cardiologist (21% of images) and a medical student (79% of images) after receiving proper instructions for image viewing and analysis. Axial slices for the measurements were chosen at the level, where both transverse processes were best visible, which was approximately in the middle of a vertebra. CT slice thickness varied between 0.50 and 7.0 mm. Regions of interest (ROIs), which were the left and right psoas muscles, were carefully drawn with a free-hand tool (see Fig. 1). The purpose was to outline psoas muscles individually according to the anatomical boundaries. The areas (mm2) and the densities (Hounsfield units, HU) with standard deviations were registered. CT measurements were performed with the Philips Intellivue image viewing and analysis desktop software.
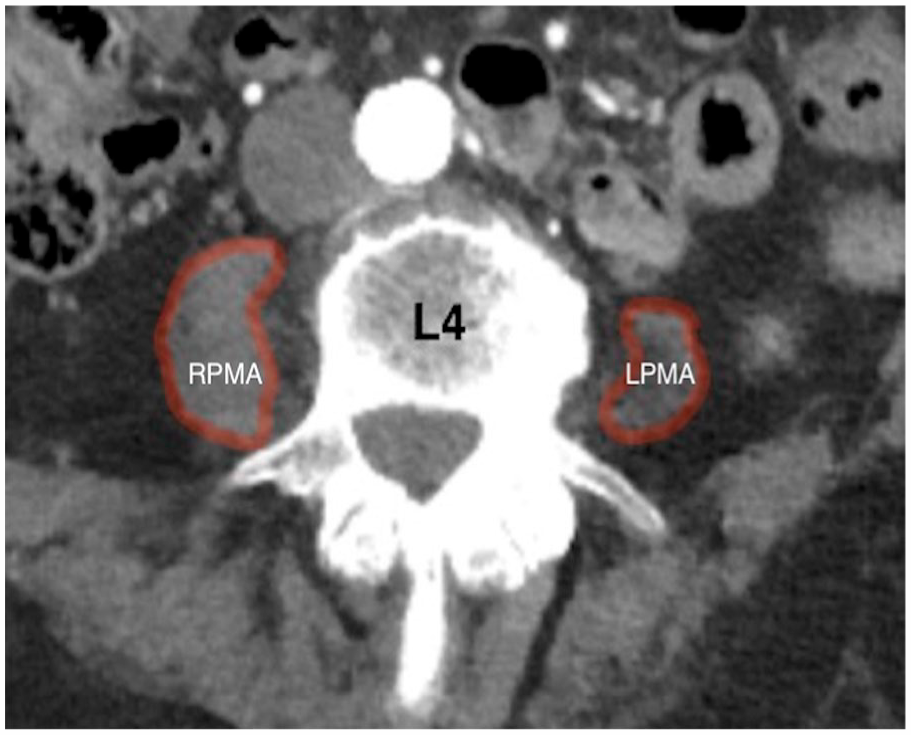
Psoas muscle area and density were measured from CT images at L4 level.
Statistical analysis
Mean PMA and PMD were used for the statistical analyses and calculated at each lumbar level (L3, L4) by applying measurements from both sides in the same axial slice: (left PMA + right PMA) / 2 for area and (left PMD + right PMD) / 2 for density, respectively. Adjusted linear regression analysis was used to determine the most significant factors associated with PMA and PMD. Due to the strong correlation between PMA and PMD (Pearson r = 0.344 (p < 1.0 × 10−12), the two parameters were analyzed separately in all multivariable models (unless otherwise stated). The results of multivariable models are presented only for significant factors in the models and the variables were filtered by a backward elimination. We considered the following variables for multivariable models: age, diabetes, hypertension, concomitant coronary artery disease, serum creatinine values, hemoglobin, patient sex, and different operation types (root reconstruction, ascending aortic reconstruction with a prothesis, arch reconstruction, descending aortic reconstruction, replacement of the aortic valve). Survival analyses were primarily performed with both L3 and L4 level psoas muscle variables and since the results were nearly equal in significance, only the results for the L4 level parameters are presented. For survival analysis, absolute values of PMA and PMD were standardized to sex-specific values and centered to mean (Z-values). In order to combine the information of muscle surface area and muscle density, mean lean psoas muscle area (LPMA) was calculated by averaging sex-specific values of PMA and PMD (the sum of sex-specific Z-values for PMA and PMD divided by 2). This methodology merges the information contained by these correlating variables into single variable depicting general muscle status by using information of both muscle size and density.
For all sex-specific and mean-centered variables, the value zero (0) denotes the same as the sex-specific mean value in this patient population and values of 1 or –1 denote one standard deviation higher or lower values, respectively, when compared to the population mean. Due to this Z-transformation of PMA, PMD, and LPMA-values, all the resulting hazard (HR) and odds ratio (OR) values obtained from the analyses correspond to a 1-SD increase in these parameter values.
Cox-regression analysis was applied to study the association between muscle parameters and the risk of death during follow-up. Schöenfeld residuals were used to check for possible violations with the proportional hazard assumption for all the variables used in the models. Subsequently, PMA, PMD, LPMA, and the urgency classification of the operations were discovered to violate this assumption (p < 0.01 for correlation with survival times). For this reason, the association of these parameters with both perioperative and long-term mortality was tested separately. Perioperative mortality was defined by deaths occurring within the first 28 postoperative days and long-term mortality by deaths occurring after the first 28 days. For perioperative mortality, binary logistic regression analysis was applied in the analyses. The results of multivariable models are presented only for significant factors in the models and the variables were filtered by a backward elimination. The threshold for statistical significance was set to a p-value of 0.05. Statistical analyses were performed with SPSS version 26 for Mac OS X.
Results
Patient demographics and risk factors
The study population consisted of 451 patients, of which 74.7% (n = 337) were men. Most of the operations were elective (n = 262, 58.1%), and approximately one-third of the procedures (33.9%, n = 153) were performed in an emergency setting. The remaining 8% of the operations were classified as urgent (see Table 1). EuroSCORE II was available for 302 patients. Mean score was 4.1 for elective patients (n = 186), 11.0 for urgent patients (n = 25), and 10.8 for emergency patients (n = 91).
General characteristics of the study population.
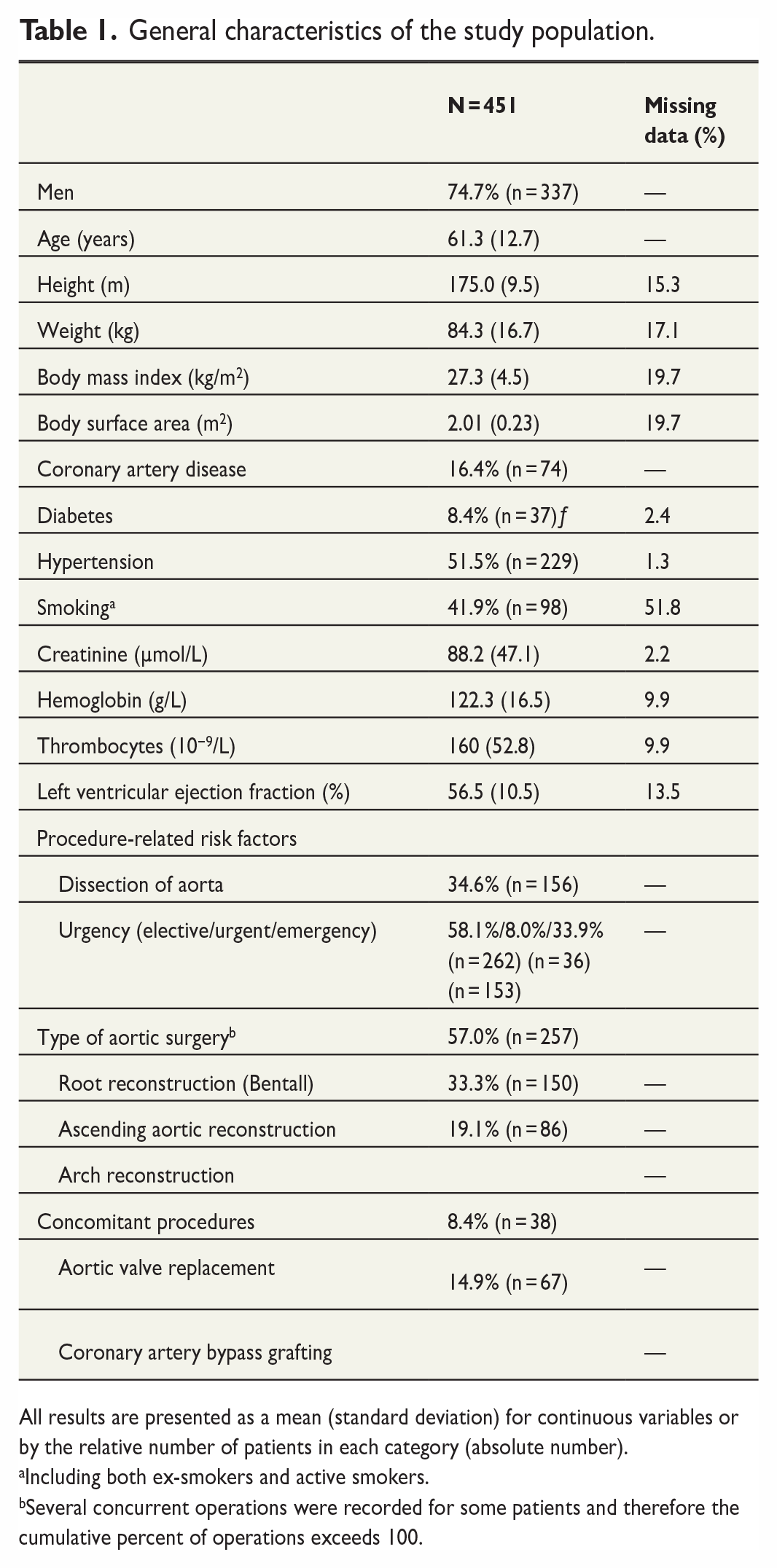
All results are presented as a mean (standard deviation) for continuous variables or by the relative number of patients in each category (absolute number).
Including both ex-smokers and active smokers.
Several concurrent operations were recorded for some patients and therefore the cumulative percent of operations exceeds 100.
The most common operation type (57.0%) was reconstruction of the aortic root and reimplantation of the coronary arteries (Bentall-deBono; see Table 1). A total of 14.9% (n = 67) of patients underwent concomitant coronary artery bypass grafting with the number of distal anastomoses varying from one to five and 8.4% had simultaneous aortic valve replacement using a mechanical or a biological prosthesis (without resection of the aortic root but with some other intervention to the aorta; see Table 1). The 6% of operations not listed in Table 1 included repair of the aortic root with a valve sparing technique (1.6%); resection of the descending aorta and reimplantation of branches (1.6%); and other repair of the ascending, thoracic, or thoracoabdominal aorta (2.2%). In two occasions, the preplanned aortic intervention was converted to a more sparing operation at the time of the surgery based on the clinical judgment of the operating surgeon (one patient underwent only isolated aortic valve replacement and another patient underwent only a decompression and drainage of the pericardium).
The median follow-up time was 4.3 years (interquartile range (IQR): 2.2–7.4). During the follow-up, 106 patients (23.5%) died, with 44.3% of the deaths occurring after the first 28 postoperative days (long-term mortality). Excluding patients who had died perioperatively, the median follow-up time was 4.9 years (IQR: 3.2–7.9). Perioperative mortality, specified as deaths during the primary hospitalization period or within 4 weeks after the operation, was 13.1% (n = 59/451), with 72.9% (n = 43) of the deaths occurring during the first postoperative week. Perioperative mortality among patients undergoing urgent or emergency operations was 25.4% (n = 48/189) and 4.2% in patients undergoing elective operations (n = 11/262).
Factors associated with psoas muscle parameters and their distribution
PMA measurements followed a normal distribution within the cohort (Figs 2 and 3) with significantly higher values measured for men when compared to women (mean PMA 1363 cm2 (SD 284) vs 859 cm2 (SD 189), p < 1.0 × 10−52). Age and BSA correlated highly significantly with PMA values (Pearson’s r = −0.436 for age and 0.549 for BSA, p < 1.0 × 10−21 for both). Age, BSA, and sex were also the only factors significantly associated with PMA in adjusted linear regression analysis with men displaying approximately 363 cm2 higher PMA values compared to women (SE 33.3, p < 1.0 × 10−23). Adjusted effect estimates for a one decade increase in age and for a 0.1 unit increase in BSA were: –63 cm2 (SE 10) for age and 36 (SE 6.4) for BSA (p < 1.0 × 10−7 for both), respectively. After correcting for age, sex, and BSA, there was no significant difference in PMA between patients treated electively, urgently, or in an emergency setting or between individuals undergoing different operation types (p > 0.1 for all comparisons). EuroSCORE II had a weak correlation with PMA among men (Spearman’s rho = −0.160, p = 0.029) but not among women (Spearman’s rho = −0.019, p = 0.876). After linear regression analysis with age and EuroSCORE II, there was no significant accociation with PMA and EuroSCORE II.
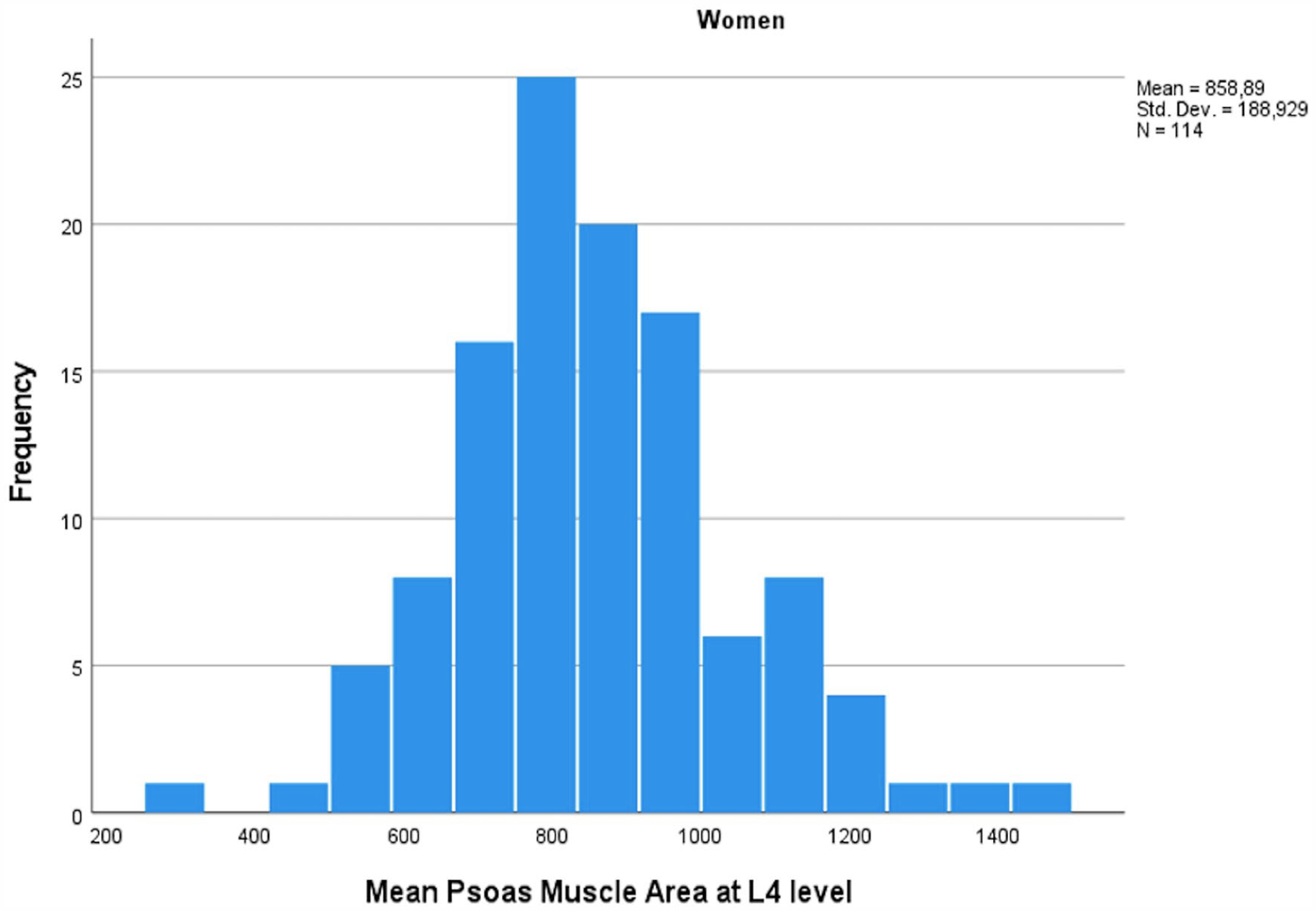
Mean psoas muscle area among women in mm2 measured at L4 level.
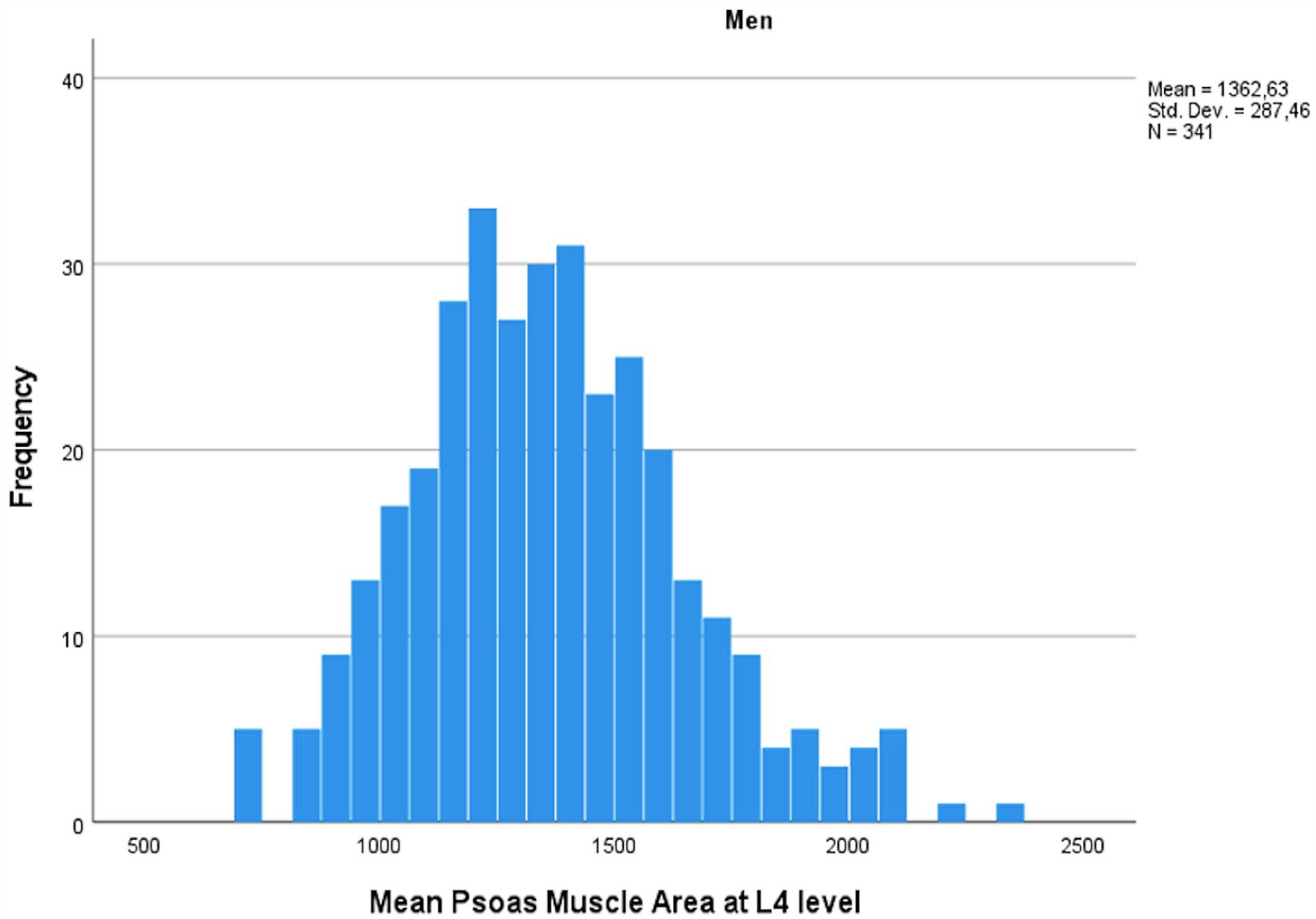
Mean psoas muscle area among men in mm2 measured at L4 level.
PMD also followed a normal distribution within the cohort (Figs 4 and 5). In simple correlation analysis, age (r = –0.366, p < 1 × 10−15), female sex (r = −0.163, p = 0.001), body height (r = 0.153, p = 0.003), BMI (r = −0.126, p = 0.018), and smoking (r = −0.145, p = 0.029) correlated with PMD. In multivariable analysis, the most significant predictors of PMD were age (one decade increase in age corresponding to a –3.44 difference in HU, p = 1 × 10−11), sex (women having –3.78 lower HU values compared to men, p = 0.016), BSA (0.1 unit increase in BSA corresponding to a –0.84 lower HU value, p = 0.007), and smoking (–3.16 lower HU values in smokers vs non-smokers, p = 0.007).
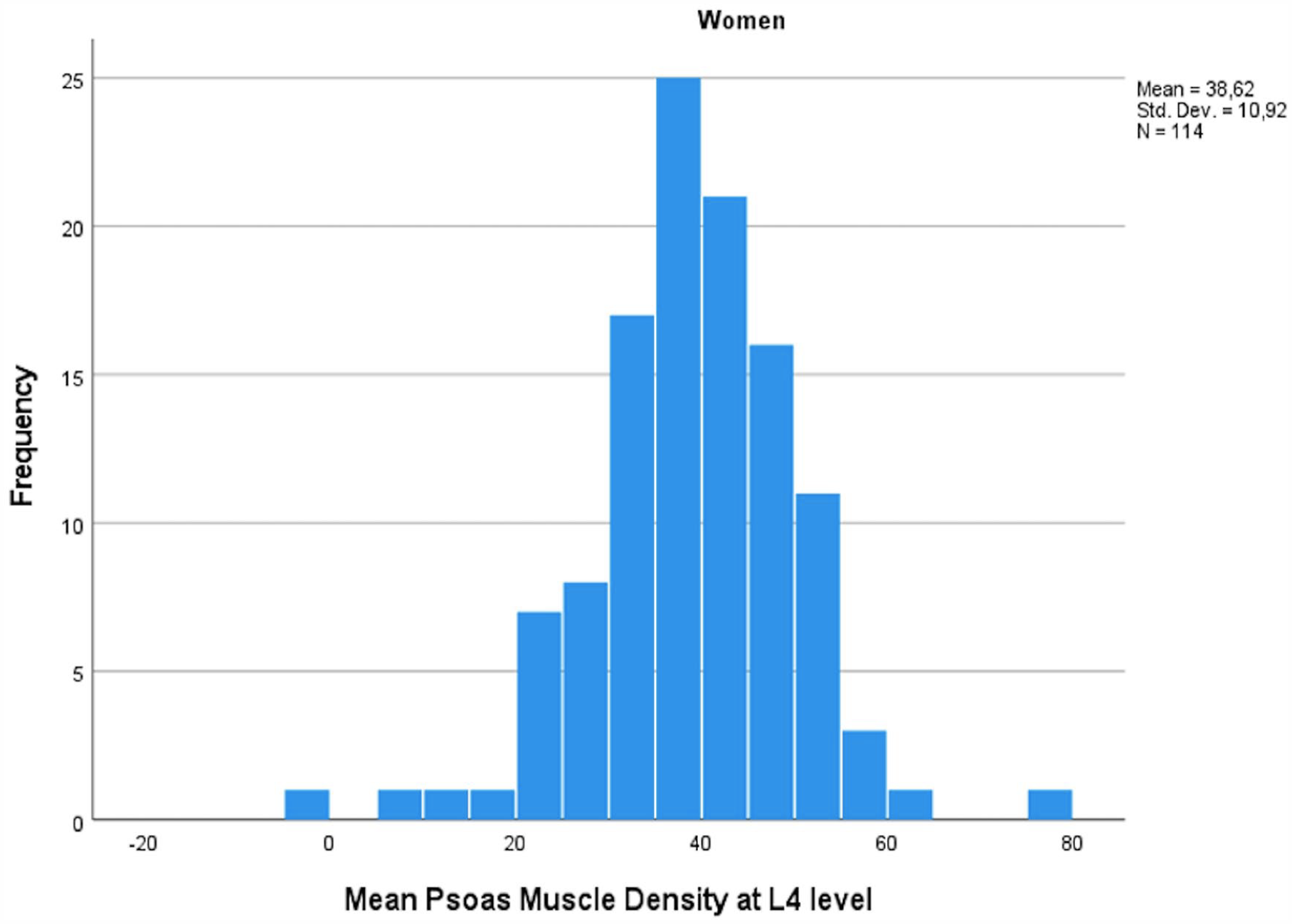
Mean psoas muscle density among women in HU measured at L4 level.
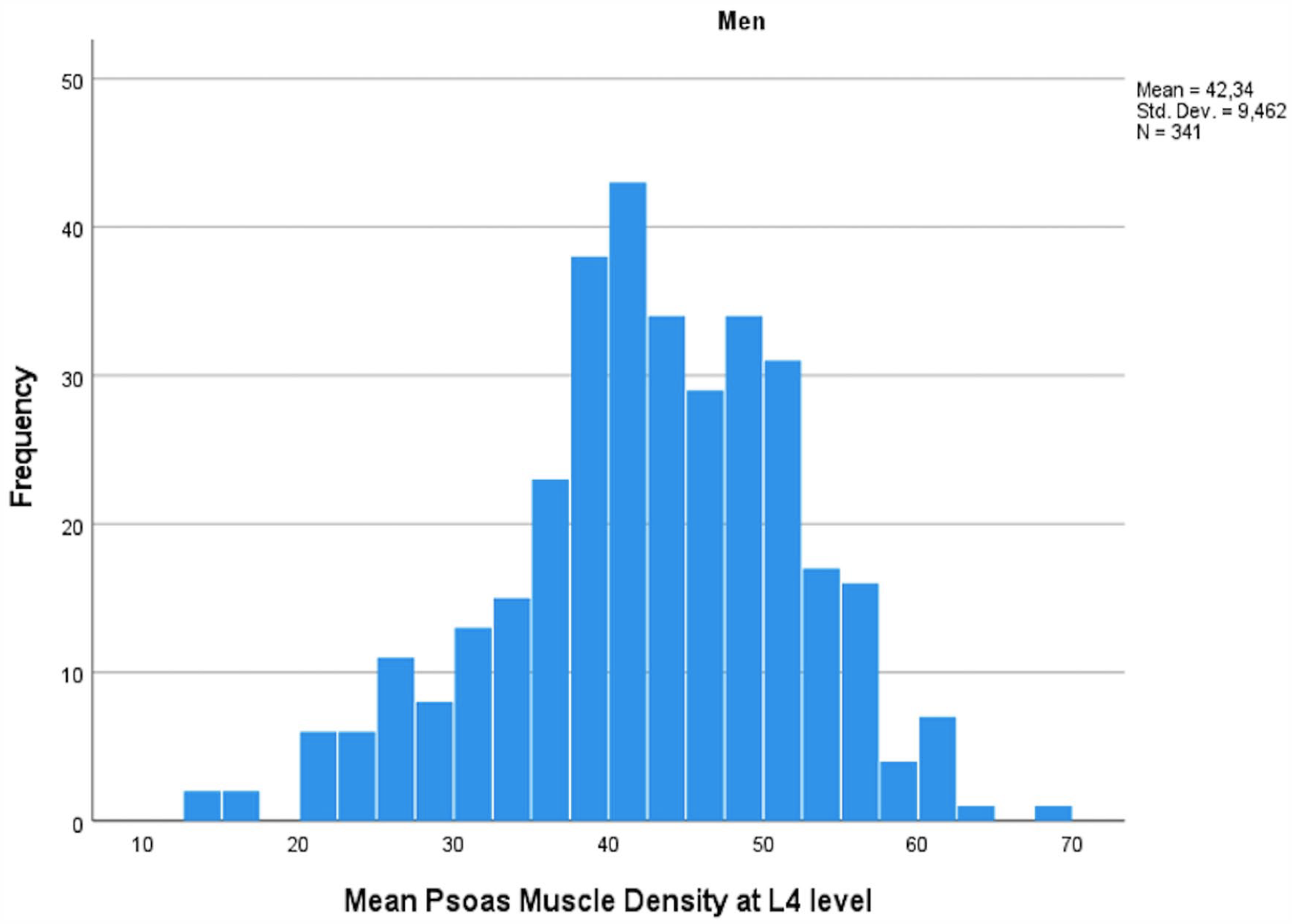
Mean psoas muscle density among men in HU measured at L4 level.
The association between muscle parameters and perioperative mortality
Age (OR: 1.07 per 1 year increase in age, 95% confidence interval (CI): 1.03–1.09, p = 0.0001), serum creatinine level (OR: 1.52 per 1-SD increase, 95% CI: 1.09–2.13, p = 0.014), and operation urgency (OR: 6.12 for operations performed for acute indications, that is, urgent or emergency operations, 95% CI: 2.99–12.52, p < 0.0001) were found to be associated with perioperative mortality in multivariable analyses. In comparison, no association of the investigated psoas muscle parameters and perioperative mortality could be demonstrated (see Table 2).
Association of muscle parameters with perioperative mortality (n = 59 deaths) after adjusting for significant risk factors (age, serum creatinine level, operation urgency) among 451 patients undergoing open surgery for aortic pathology.
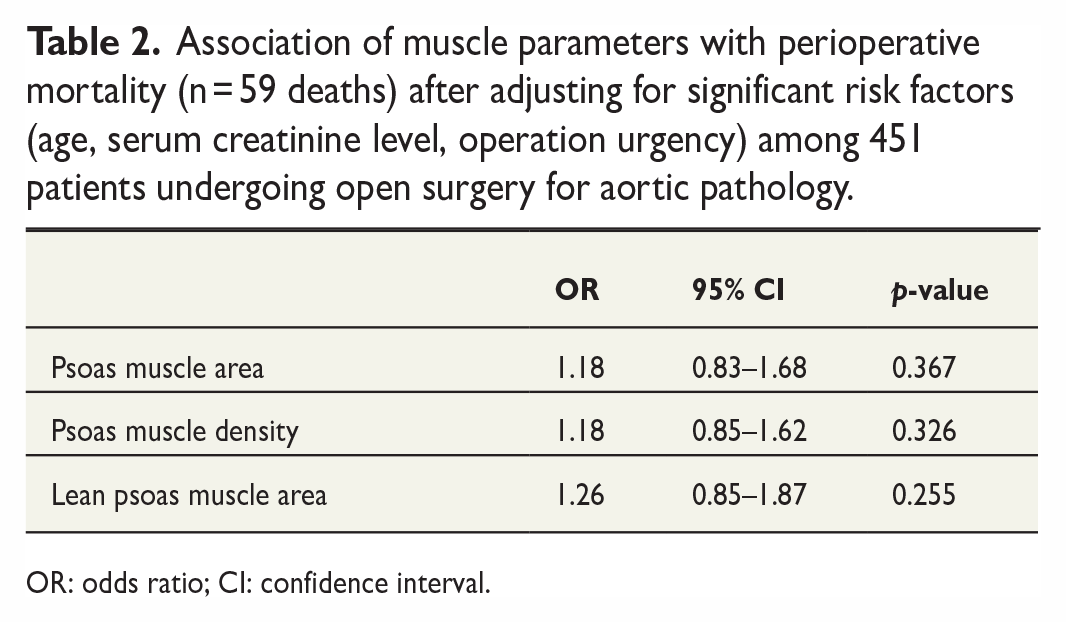
OR: odds ratio; CI: confidence interval.
The association between muscle parameters and long-term mortality
Long-term mortality was analyzed in the cohort (n = 392) of patients surviving beyond the first 28 postoperative days. In multivariable analysis, the most significant factors associated with long-term mortality were age (HR: 1.05 per 1 year increase in age, 95% CI: 1.02–1.08, p = 0.001), reconstruction of the descending aorta (HR: 9.45, 2.12–42.15, p = 0.003), and aortic dissection as the indication for the operation (2.41, 95% CI: 1.33–4.36, p = 0.004). The associations of PMA, PMD, and LPMA at the L4 level with long-term mortality were significant despite adjusting for these factors (HR: 0.63, 95% CI: 0.44–0.89, p = 0.008 for PMA; HR: 0.62, 95% CI: 0.46–0.83, p = 0.002 for PMD; and HR: 0.53, 95% CI: 0.37–0.74, p = 0.0002 for LPMA; see Table 3). HRs for psoas muscle parameters are presented per 1-SD increase: mean PMA 1363 cm2 (SD 284) for men and 859 cm2 (SD 189) for women, mean PMD 42.3 HU (SD 9.46) for men and 38.6 HU (SD 10.9) for women. If both PMA and PMD were introduced to the same model, they still persisted as significant risk factors despite the strong correlation between these two (r = 0.34, p = 1 × 10−11; HR: 0.69, 95% CI: 0.49–0.99, p = 0.041 for PMA and HR: 0.68, 95% CI: 0.50–0.91, p = 0.010 for PMD) Similarly, in the subgroup of patients with available data on BSA (overall n = 309, with n = 36 deaths), PMA indexed to BSA was significantly associated with long-term mortality (HR: 0.52, 95% CI: 0.32–0.85, p = 0.009; analysis further adjusted with sex, p = 0.033).
Factors associated with long-term mortality in multivariable models (deaths occurring after the first 28 postoperative days) among patients still alive after first four weeks of open surgery for aortic pathology (n = 392).
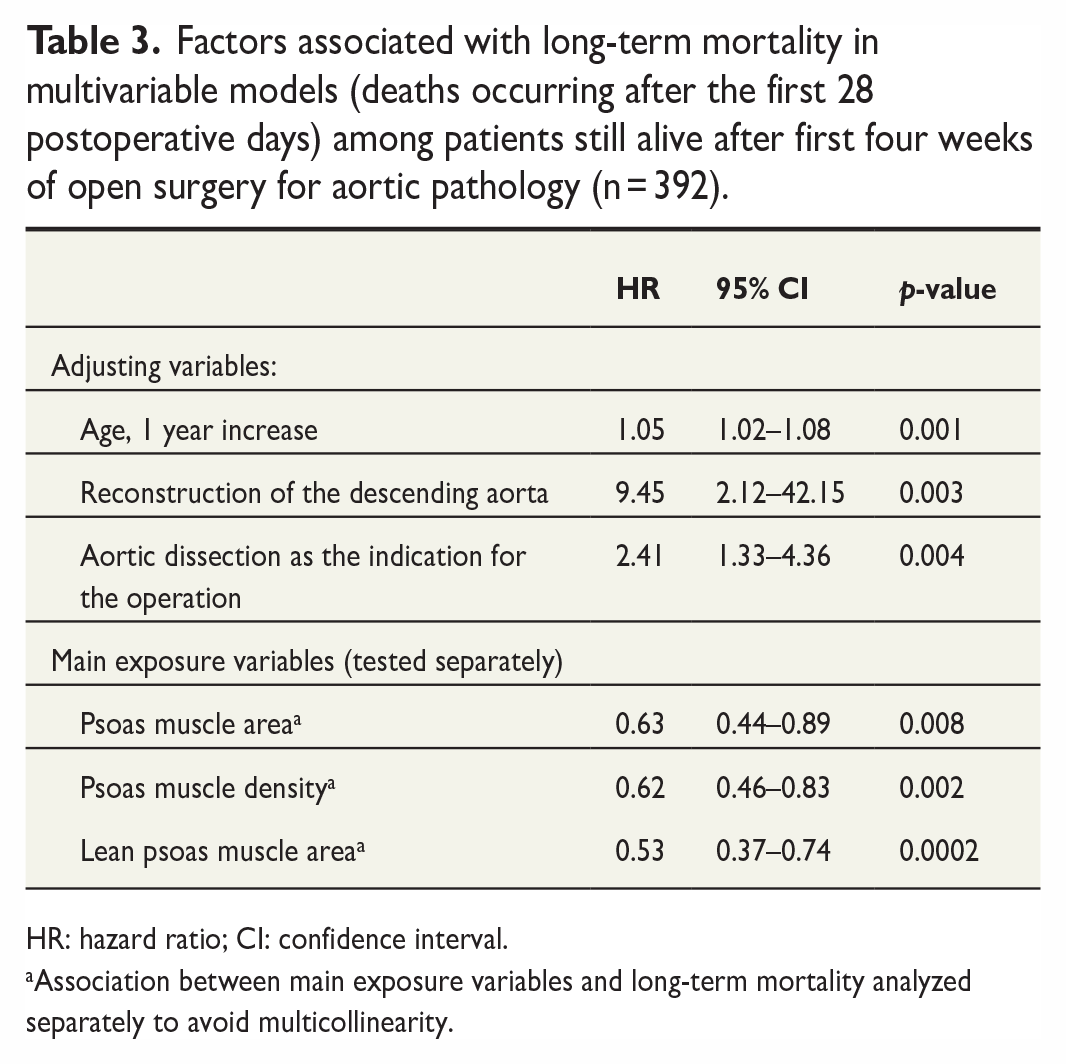
HR: hazard ratio; CI: confidence interval.
Association between main exposure variables and long-term mortality analyzed separately to avoid multicollinearity.
Discussion
In this study, we showed that in addition to age, sex, primary reconstruction of the descending aorta, and aortic dissection as an indication for surgery, both PMA and PMD (at the L4 level) were independently associated with long-term mortality after open thoracic aortic surgery. This effect was also found in a more profound manner for LPMA and persisted when PMA values were indexed to BSA.
The lack of association of the psoas muscle parameters with 28-day postoperative mortality can be explained by the different nature of short-term and long-term mortality in this cohort. Short-term postoperative mortality is usually related to acute situations in which the outcome is strongly influenced by factors such as the extent of the hemodynamic catastrophe and how promptly it is treated, the management of the systemic stress response, and how essential intensive care is provided. When the acute situation has been resolved, other factors will determine the patients’ future survival. Psoas muscle measurements are not subject to acute changes in body status or effort. PMA and PMD are rather affected by nutrition and physical exercise and therefore reflect potential for good recovery in the long term. By excluding early deaths within 28 days, a significant change in the results was observed.
Specifically, we noted that low LPMA demonstrated the strongest association with long-term mortality. Our results are in line with those presented in earlier studies. Previously, Ghaffarian et al. 1 found lower total muscle area at the L3 level to be associated with inferior long-term survival in both conservatively and operatively treated patients with thoracoabdominal aneurysms, although another study on patients undergoing open thoracoabdominal aneurysm repair was unable to demonstrate an association between the psoas index as a marker of sarcopenia, and survival despite discovering a link between sarcopenia and delayed persistent postoperative paraplegia. 18 Furthermore, lower psoas index has been ascertained to be associated with higher overall mortality after total aortic arch repair 3 and adverse events as well as long-term mortality after open and endovascular treatment of the descending aorta.2,4 Similar findings have also been observed in patients undergoing off-pump coronary artery bypass surgery and elective open heart valve surgery, where sarcopenia independently predicted inferior long-term survival along with major adverse cardiac and cerebrovascular events in the latter cohort.19,20 In addition, sarcopenia has been shown to be linked to prolonged postoperative hospitalization in patients undergoing cardiac surgery, and it was found that treating sarcopenia and improving patients’ nutritional status had a positive effect on shortening the postoperative hospital stay. 21
In this study, psoas muscle parameters were not associated with the traditionally used risk assessment method EuroSCORE II. 17 This is logical, since the EuroSCORE II evaluates the risk of in-hospital or short-term mortality, whereas the psoas parameters are more apt in assessment of long-term mortality. Based on our results, psoas muscle parameters are therefore unlikely to yield much benefit for prediction of immediate postoperative risk, which is heavily influenced by acute factors. However, evaluation of long-term risk is equally important, especially when considering elective surgery for the elderly for asymptomatic conditions such as dilatation of the ascending aorta. The results from this study show psoas muscle status to be clearly and independently associated with long-term mortality even when accounting for other significant risk factors in the analyses. Kärkkäinen et al. 22 recently used a cutoff of 90 days for early mortality in a similar study on patients undergoing complex endovascular aortic repair. The time point was chosen due to the low expected incidence in a cohort of elective patients. In this study, a cutoff of 28 days was applied based on the significant change in mortality at this time point, potentially reflecting the mixed nature of the cohort consisting of both elective and acute cases.
To date, several studies have primarily examined the association of PMA or psoas index, and in some instances, PMD with patient outcome. There are, however, some inherent challenges when analyzing muscle parameters. First, area and density measurements alone do not describe the muscle status or evaluate frailty comprehensively enough. For example, PMA alone can be misleading, since it does not take the proportion of intramuscular adiposity brought on by aging (and possibly smoking) into account, whereas PMD does not consider muscle volume. Second, PMA and PMD values correlate with each other, whereby multivariable analyses need to be planned carefully. In this study, these challenges were addressed by investigating PMA, PMD, and LPMA, and by analyzing these parameters separately in all multivariable models. Based on our results and previous findings, evaluating preoperative psoas muscle parameters shows great promise in helping identify patients at risk of higher long-term mortality after cardiothoracic surgery and who may benefit from preoperative optimization, such as diet transformation or physical exercise. This study was not designed/powered to define specific cutoff values for sarcopenia, but in the future, it would be worthwhile to investigate the optimal cutoff points for muscle parameter values in cohorts undergoing cardiovascular surgery. Potential methods for this could entail, for example, determining the psoas muscle parameter distributions of healthy adults and comparing them to older individuals, potentially including muscle strength testing, and/or a sensitivity analysis of the predictive power of muscle parameter values in an appropriately powered cohort. After standardizing cutoff points, measurements and risk analysis could perhaps even be automatized and carried out by an artificial intelligence (AI)-based software.
This single-center retrospective study must be viewed within the context of its limitations. First, some patients undergoing open thoracic aortic repair lacked sufficient-quality contrast-enhanced CT-imaging within a time frame of 1 year prior to surgery, which may have caused some selection bias. Second, CT images taken 6 months or earlier prior to the operation were used in some (under 10%) of patients and it is possible that a moderate change in muscle mass or quality could have occurred during that time. Third, it could be argued that the study population was somewhat heterogenous with regards to the variety and urgency of procedures performed. However, these factors were adjusted for in the multivariable analyses. It is important to point out that the long-term mortality of patients who underwent emergency procedures was not higher than that of electively operated patients, if they only survived the emergency procedure itself. That is why we divided the follow-up into perioperative and long-term mortality to facilitate the interpretation of the results. If we had to remove all those who had to undergo an emergency procedure, the size of our material would be unfortunately limited. Fourth, since the Tampere Heart Hospital is a tertiary referral center for a large catchment area and preoperative treatment as well as postoperative follow-up are conducted at smaller district hospitals for a proportion of patients, some patient data may not have been available in the study center’s own registry. In addition, weight or height were not measured in most emergency patients, which is why BSA could not be calculated for these individuals. However, we were able to use sensitivity analyses to demonstrate that LPMA serves as a valid and equally effective indicator as the indexed BSA value. We were also unable to obtain information on the causes of deaths and could therefore not evaluate cardiovascular mortality separately. Finally, we lack data on possible changes in patients’ medications during the study period, some of which may have affected long-term outcomes.
In conclusion, psoas muscle sarcopenia status measured from routine preoperative contrast-enhanced CT images is associated with long-term mortality after open thoracic aortic surgery. While further research is called for, psoas muscle measurements could potentially be applied in patient selection for surgical treatment in the future, as well as in optimizing risk factor management via dietary interventions and/or exercise therapy in similar cohorts.
Footnotes
Acknowledgements
We thank Juha Hautalahti MD, PhD for his valuable comments on the article.
Author contributions
S.V. contributed to data acquisition, writing of an original draft, review and editing of the article. S.K. contributed to writing of an original draft, review and editing of the article. I.L., N.K., T.S., and J.L. were involved in review and editing of the article. N.O. contributed to the study concept, study design, review and editing of the article, and supervision. J.H. contributed to the study concept, study design, data acquisition, statistical analysis, writing of an original draft, review and editing of the article, and supervision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Competitive State Research Financing of the Expert Responsibility area of Tampere University Hospital (grant nos. VTR9X040, VTRZ60081).
