Abstract
Background:
Biomaterials are routinely used in orthopedic surgery to fill bone defects, improve bone healing, and as degradable fixation material. A wide range of materials are currently in use, and the materials are chosen according to their bioactive properties. Osteoinductive materials stimulate bone healing by promoting osteogenesis. Osteoconductive materials facilitate bone growth on the surface of the material. Despite the many materials in use and an increasing number of published studies, randomized controlled trials on the subject are scarce.
Methods:
This review aims to summarize the history of biodegradable biomaterials and also the published level I evidence currently available on orthopedic biomaterials.
Results:
Most of the studies have been superiority trials with non-significant differences compared to conventional treatment options, confirming that several biomaterials are suitable treatment options for multiple indications including bone and/or tendon fixation, filling bone defects, and spinal fusion. Biomaterials help to avoid donor site complications associated with autogenous bone grafts and often eliminate the need for implant removal. However, the surgical technique may in some cases be more demanding than with conventional methods. Careful consideration of the pros and cons is therefore recommended in clinical practice.
Conclusion:
Biodegradable biomaterials complement the range of available treatment options in several fields of orthopedic surgery. However, some biomaterials performed worse than expected and were not recommended for clinical use, emphasizing the need for high-quality randomized trials. It is also noteworthy that several trials included only a limited number of patients, rendering the interpretation of the results of these underpowered studies challenging.
Introduction
The European Society for Biomaterials for the first time in 1987 defined a biomaterial as “a non-viable material used in a medical device, intended to interact with biological systems.” 1 Both biomaterials and the definition have developed since then and now encompass several “new biomaterials” such as drug and gene delivery systems, tissue engineering, and microelectronic devices. More than any other field of medicine, orthopedic surgery has been revolutionized by the development of biomaterials, beginning with biocompatible alloys for fracture care.
More than 17,000 scientific articles can currently be found on PubMed using only the keywords “biomaterials” and “orthopedics.” Also, the number of published papers appears to be increasing every year due to technological advances and wide interest among clinical specialists, researchers, and the medical industry. However, only a fraction of these published articles report randomized clinical trials with level I evidence. Randomized clinical trials reduce bias, control confounding, and provide a tool with which to examine the effectiveness of new treatments or interventions which with any other study design would be impossible. 2
Biodegradable biomaterials are designed to degrade over a predetermined period after implantation, whereas non-degradable biomaterials are permanent. In this review, we aim to summarize the development of biomaterials and their applications in orthopedic surgery concentrating on biodegradable biomaterials. We also summarize the currently available level I evidence on biodegradable biomaterials in orthopedic surgery.
Background and applications
The evolution and development of biomaterials can be divided into three different generations. 3 The first generation, consisting of biocompatible and bioinert stainless steel and cobalt-chromium, was introduced by the 1940s, and the first successful total hip prosthesis was developed in the late 1950s. 4 Cobalt-chromium is a metal alloy of cobalt and chromium characterized by high strength and resistance to oxidation. Metallic materials were later complemented by polymers and ceramic materials, and the first-generation biomaterials continue to the present day to be widely used in traumatology and joint replacement surgery. Polymers, by definition, are any natural or synthetic substances composed of very large molecules (macromolecules). The second generation, which became available between 1980 and 2000, was defined by its ability to interact purposefully with the biological environment. As opposed to the bioinert first generation, these materials were either bioactive or biodegradable or both. The term “bioactive” means that the implant has a biological effect on living tissue. This generation consisted of bioactive ceramics (e.g. bioactive glass and hydroxyapatite (HA)) as well as several polymer compounds, which are widely used in absorbable suture materials. Third-generation biomaterials, introduced by the 2000s, are designed to stimulate specific cellular responses at the molecular level, like cell proliferation, differentiation, and extracellular matrix production and organization, 3 for example, tissue-engineered three-dimensional structures providing temporary support while stimulating cell invasion, attachment, and proliferation. 5 The aim of these is to grow progenitor cells on a resorbable scaffold outside the body before implantation to replace damaged tissues. Clinical applications include repair of cartilage, the vascular system, and the skin. 3
Biomaterials can moreover be classified according to their bioactive properties. It is essential for every clinician to understand these basic principles when selecting the optimal implant material. Osteoinductive materials, such as bone morphogenetic protein (BMP) and bioactive glass, induce osteogenesis by recruiting mesenchymal stem cells which develop into preosteoblasts, thereby stimulating bone healing. Osteoconductive materials, like tricalcium phosphate and HA, on the contrary, act as scaffolds by enabling/facilitating bone growth on the surface leading to the stable anchoring of the implant called osseointegration. 6
Biodegradable biomaterials are widely used in orthopedic surgery and their applications include fixation materials, filling materials, and temporary support structures. Biodegradable fixation materials aim to avoid biomechanical complications, which are occasionally encountered with their non-resorbable counterparts, possibly necessitating implant removal. Biodegradable filling materials, on the contrary, aim to provide an alternative to autografting or allograft, thereby minimizing harvesting site morbidity and/or the risk of transmission of infectious complications. In principle, biodegradable support structures serve the same purpose. The following sections summarize the currently available level I evidence from published randomized clinical trials of biodegradable biomaterials and their different clinical applications.
Current evidence
Biomaterials for fixation
Rokkanen et al. randomized 44 patients with a displaced fracture of the ankle to two groups: one group was treated with conventional metallic implants and the other with biodegradable polylactide-glycolide copolymer implants. There were no differences between the two groups in the early results, and bone union was achieved in all but two patients (one in each group). Due to the small number of patients, no statistical analyses were feasible. The biodegradable fixation method was advantageous because it avoided the removal procedure associated with metallic implants. 7
Several studies on bioabsorbable screws for anterior cruciate ligament (ACL) reconstruction have been presented8
–10 including one meta-analysis.
11
The screw materials included poly-
The use of biodegradable fixation devices has been studied in pediatric forearm fractures, fractures of the patella, hand surgery, and fixation of femoral head osteonecrosis. Korhonen et al. 13 compared biodegradable polylactide-co-glycolide (PLGA) nails to elastic stable intramedullary titanium nails in 35 pediatric patients (aged 5–15 years) with forearm fractures and reported equivalent clinical results regarding the range of motion in 2-year follow-up. However, there were two implant failures among 19 patients in the PLGA nail group (vs 0/16 in the titanium nail group), and the authors speculated that implant stability needs further study, especially in older children. Also, the operative technique is more challenging (i.e. requiring opening of the intramedullary cavity before insertion of the final implant) than using traditional titanium nails. A minimum 4-year long-term follow-up showed no subsequent complications and almost total degradation of all intramedullary implants. 14 Chen et al. 15 compared biodegradable self-reinforced polyglycolide (SR-PGA)/self-reinforced PLLA tension band to metallic tension band in 38 adults with transverse patellar fractures with two or three fragments. Radiographic and clinical results were comparable in 2-year follow-up, and the authors concluded that biodegradable tension band is suitable for the treatment of patellar fractures whereas metallic implants often require removal. Jensen et al. 16 studied biodegradable polymer poly-p-dioxanone (PDS) versus standard Kirschner wires in the fixation of fractures, arthrodeses, and osteotomies in the hand in 23 adults. After 6-month follow-up, there were no differences between the groups and the authors concluded that PDS pins provide effective fixation and can reduce the number of additional procedures with no need for implant removal. There were, respectively, 3/11 and 2/12 non-unions in the biodegradable pin and Kirschner wire groups. Zhao et al. 17 studied the treatment of osteonecrosis of the femoral head with vascularized bone grafting with or without biodegradable magnesium (Mg) screw fixation. In their pilot randomized controlled trial (RCT) with 12-month follow-up of 48 patients, they reported no adverse effects, and Mg screw fixation yielded improved Harris Hip Scores compared to no fixation, making this a potential fixation device for bone grafts.
Filling materials/bone substitutes
Bone substitutes in benign bone tumors
Several different types of bioactive glasses have been developed for bone graft substitutes. 18 However, published level I evidence has only been presented on one of these (BG-S53P4). Lindfors et al. studied BG-S53P4 in benign adult bone tumors in 25 patients. In their 3-year 19 and subsequent 14-year 20 follow-up, bioactive glass yielded results comparable to those of autografting and only one patient in each group required reoperation due to growing residual cyst. There were no adverse events and the authors concluded that bioactive glass is a safe and well-tolerated bone substitute with good long-term results. Aro et al. 21 reported 10-year outcomes of 49 adults with benign bone tumors randomized for treatment with either BG-S53P4 or auto/allograft. BG granules and autologous and allogeneic bone grafts resulted in similar overall rates of treatment success. Syvänen et al. 22 studied the outcomes of 51 pediatric patients with benign bone tumors randomized for treatment with either BG-S53P4 or allograft and reported comparable results in 2-year clinical and MRI follow-up with possible reduction in the size of recurring lesions in the bioactive glass group. All studies reported residual glass granules in long-term follow-up imaging, but no adverse events. It appears that BG-S53P4 is a viable alternative for standard bone grafts in benign bone tumors with the advantage of avoiding harvesting site morbidity.
Nazarova et al. 23 compared calcium orthophosphate–based bioceramic granules (HA (Ca10(PO4)6(OH)2) and collagen) with autogenous bone graft and curettage only in 200 adults with enchondroma. They reported equal outcomes in all groups but observed that autogenous bone graft may increase the risk of tumor recurrence (6/100 with autograft vs 1/100 with bioceramic granules and 1/56 with curettage only). The control group with curettage only consisted solely of hand enchondromas as larger defects require filling according to the hospital protocol. Wang et al. 24 studied beta-tricalcium phosphate (β-TCP) versus allogenous bone graft in simple bone cysts in 95 patients with mean 28-month follow-up. They reported comparable clinical and radiographical findings in both groups and concluded that β-TCP is a reliable and safe bone substitute for lacunar bone defects.
Bone substitutes in osteotomies
Lee et al. 25 compared bone union progression after open-wedge high tibial osteotomy tricalcium phosphate (TCP) versus allogenous bone chips in 54 patients in 1-year follow-up. TCP granules achieved similar bone union rates and clinical results at each time point (6 weeks, 3 months, 6 months, and 12 months) compared to allograft (grade IV union 13/27 with TCP and 12/27 with allograft at 12 months), and the authors concluded that it can be used as a bone substitute in tibial open-wedge osteotomies. On the contrary, Lind-Hansen et al. 26 also studied the outcomes of treatment with different bone substitutes (injectable and osteoconductive calcium phosphate cement with initial high compressive strength) on open-wedge tibial osteotomy and found it to be equivalent to autograft regarding bone healing but resulted in lower quality of life scores at 2 years postoperatively. They concluded that stable implant and local autografting would be sufficient. A randomized trial by Gouin et al. 27 with biphasic calcium-phosphate (MBCaPh) ceramic spacer versus autograft in open-wedge tibial osteotomy was discontinued after 40 patients due to a significant number of adverse events (loss of correction 27% vs 5%) with ceramic spacer. Also, bone union was achieved sooner (2.6 vs 5.8 months) with autograft.
Bone substitutes in traumatology
Chapman et al. 28 studied the use of collagen-calcium phosphate ceramic graft material versus autogenous bone graft in the treatment of acute long bone fractures necessitating bone grafting based on the judgment of the operating surgeon. The multicenter study included 213 adults with 2-year clinical and radiological follow-up with respective fracture healing rates of 97% and 98% at final follow-up with collagen-ceramic graft and autograft. The authors concluded that the use of collagen-ceramic-marrow composite graft material is justified as regards safety, efficacy, and elimination of harvesting site morbidity.
Pernaa et al. 29 compared bioactive glass (BG-S53P4) to autograft in depressed tibial plateau fractures in 29 patients. Long-term clinical and radiographic results were comparable, and the authors concluded that BG-S53P4 can be used in depressed tibial plateau fractures. Hofmann et al. 30 also studied the treatment of bone defects in tibial plateau fractures and compared the outcomes of 135 patients treated with either autologous iliac bone graft or bioresorbable HA and calcium sulfate cement. Clinical, patient-reported, and radiographic outcomes were comparable in both groups at each timepoint during their 6-month follow-up.
Bone morphogenetic protein 2 (BMP-2) is an osteoinductive and osteoconductive grafting material frequently used in spinal fusion. BMP-2 is implanted using a carrier material. Govender et al. 31 investigated the additional value of local recombinant human bone morphogenetic protein 2 (rhBMP-2) on the outcomes of open tibial fractures in a randomized clinical trial. Four hundred and fifty adult patients with an open tibial fracture were randomized to receive either standard care (intramedullary nail fixation and routine soft-tissue management (the control group)), standard care and an implant containing 0.75 mg/mL of recombinant human BMP-2 (total dose of 6 mg), or standard care and an implant containing 1.50 mg/mL of BMP-2 (total dose of 12 mg). BMP-2 applied on an absorbable collagen sponge was placed over the fracture at the time of definitive wound closure. Using locally 1.50 mg/mL of BMP-2 significantly accelerated fracture and wound healing and reduced the infection rate in these patients.
Bone substitutes in spine surgery
Posterior spinal fusion
Glassman et al. 32 compared human BMP-2 with compression resistant matrix (rhBMP-2/CRM) and autologous iliac crest bone graft in patients undergoing instrumented posterolateral fusion for single-level degenerative lumbar disease. The grade of the fusion was assessed with CT scans. The study reported significantly more rapid fusion in the BMP-2 group than in the iliac crest group at 1-year follow-up. Cho et al. 33 compared Escherichia coli–derived rhBMP-2 with HA carrier and autogenous iliac crest graft in patients undergoing single-level lumbar or lumbosacral posterior fusion. The fusion rate was assessed with CT scans at 12 and 24 weeks, and patient-reported outcome scores were collected. Outcomes were similar between the groups at 24-week follow-up.
Korovessis et al. 34 conducted a comparative study to evaluate the effectiveness of three different treatments: coralline HA alone, iliac bone graft alone, and a combination of these in patients undergoing posterior spinal fusion for degenerative lumbar stenosis. Fusion was found to have been achieved at 12-month follow-up in CT scans in all groups. However, HA was found to be unsuitable for intertransverse posterolateral fusion due to the need for a large bleeding bone surface.
Kanayama et al. 35 compared osteogenic protein-1 (OP-1) to allograft in patients undergoing posterolateral lumbar fusion for degenerative spondylolisthesis. OP-1 is an osteoconductive material with limited osteoinductive properties. The results showed a radiographic fusion rate of seven out of nine OP-1 patients and nine out of ten control patients, with surgical exploration revealing solid fusion in four out of seven OP-1 and seven out of nine control patients. The authors concluded that while OP-1 reliably stimulated new bone formation without the need for autogenous bone graft, the fusion rate was not satisfactory, suggesting that modifications to the surgical approach or carrier of the OP-1 device may be necessary for broader application in instrumented posterolateral lumbar fusion.
Ransford et al. 36 assessed the use of synthetic porous ceramic (Triosite) as a bone graft substitute in posterior spinal fusion for idiopathic scoliosis. Patients were allocated to receive either autograft (171 patients) or Triosite blocks (170 patients). Postoperative assessments were conducted for up to 18 months. The rate of deformity correction was similar in both groups, and pain levels were comparable between the groups. Complications such as delayed healing, infection, and pain at the donor site were more prevalent in the autograft group. Hematological and serum biochemistry results were normal, and there were no adverse events or allergenicity related to Triosite. Histological findings demonstrated the ability of Triosite to scaffold new bone formation and integrate into the fusion mass. The authors suggested that Triosite is a safe and effective substitute for autograft in posterior spinal fusion for idiopathic scoliosis.
Dai et al. 37 reported the efficacy of β-TCP (beta-tricalcium phosphate) combined with local autograft to be comparable to that of autologous iliac crest bone graft in single-level instrumented posterolateral lumbar fusion. After 3 year follow-up, both treatment groups showed successful radiographic fusion, with no significant differences in clinical recovery rates or quality of life improvement. However, patients in the autograft group experienced donor site pain, although this decreased over time. Based on these findings, the authors recommend using β-TCP as a bone graft substitute in instrumented posterolateral fusion of the lumbar spine to eliminate the need for iliac crest bone graft harvesting.
Lehr et al. 38 compared the rate of fusion between 1 and 2 years after instrumented thoracolumbar posterolateral fusion with a standalone ceramic bone graft substitute or autograft. The rate of fusion was significantly higher at 2-year follow-up in both groups, with no difference between the groups. The continuous development of new bone was only detected among the facet joints rather than in the intertransverse region. This strongly suggests that the process is primarily due to immobilization rather than grafting.
In a study by Jacobsen et al., 39 non-instrumented posterolateral fusion was performed on elderly patients with poor bone stock and instability. The study compared the use of a synthetic 15 amino acid residue (ABM/P-15) to femoral head allograft. The ABM/P-15 group achieved a significantly higher fusion rate (50%) than in the allograft group (20%) according to CT scans. However, there were no significant differences between the two groups in patient reported outcomes.
Interbody fusion
Putzier et al. 40 compared autologous iliac crest graft and autologous periosteal cells in a fibrin/polyglactin-poly-p-dioxanone (PGLA-PPD) fleece in patients undergoing single-level interbody fusion for degenerative spinal disease. After 6 and 9 months, patients in the PGLA-PPD group achieved a significantly higher fusion rate than those in the control group. After 12 months, both groups exhibited high fusion rates (80% in the iliac crest graft and 90% in the PGLA-PPD group), with no complications observed.
Jiya et al.41,42 compared resorbable poly-
A study by Michielsen et al. 43 evaluated the clinical and radiographic effects of recombinant human rhBMP-2 compared to autologous bone graft in patients undergoing single-level posterior lumbar interbody arthrodesis with PEEK cages. End-plate resorption, osteolysis, and ectopic bone formation occurred in the rhBMP-2 group. However, there were no differences in the fusion rates or clinical outcomes between the groups at 1-year follow-up.
Arnold et al. 44 compared 15-amino-acid polypeptide (P-15) to autograft in patients undergoing interbody fusion after cervical discectomy. The fusion rates (97% in P-15% and 94% in the control group) and clinical outcomes were comparable between the groups at 2-year follow-up.
Rickert et al. 45 evaluated the efficacy of a nanocrystalline HA embedded in a silica gel matrix (NH-SiO2) compared to homologous bone as cage filling in instrumented anterior lumbar interbody fusion. The results showed no significant differences between the NH-SiO2 group and the homologous bone group in terms of functional, radiologic, and clinical outcomes at 12-month follow-up.
Kersten et al. 46 compared the performance of silicon nitride (Si3N4) cages to PEEK cages in lumbar interbody fusion procedures. Both groups showed significant clinical improvements, with no statistically significant differences in clinical and radiographic outcomes between the two cage materials. However, the Si3N4 group had longer operative times and higher blood loss. There was a slightly higher incidence of complications and revisions in the Si3N4 group, although this was not statistically significant. The study did not provide sufficient evidence to permit the conclusion that Si3N4 cages were non-inferior to PEEK cages.
BMP-2
Safety concerns have been expressed regarding rhBMP-2 in spinal surgery. Greater risks for adverse events like implant displacement (osteolysis), infection, radiculitis, ectopic bone formation, and even risk of new malignancy have been reported. 47 However, the existing data are inconsistent, and the increased risk for adverse events has not been confirmed in all studies. 48 A recent report has confirmed that off-label use of rhBMP-2 continues to be relatively common among spine surgeons. 49
Other applications for bone substitutes
McKee et al. 50 compared the effectiveness of an antibiotic-impregnated bioabsorbable bone substitute (alpha hemihydrate calcium sulfate) in non-hematogenous long bone osteomyelitis and/or infected non-union compared to standard antibiotic-impregnated cement beads in 30 patients. Clinical results regarding infection eradication and achieving union were similar, and bioabsorbable bone substitute was associated with fewer reoperations.
Daniels et al. 51 randomized 104 patients requiring ankle of hindfoot arthrodesis for either autograft or recombinant human platelet–derived growth factor BB homodimer (rhPDGF-BB) combined with β-TCP-collagen treatment. Clinical and radiological outcomes after 1-year follow-up were similar with 68.1% versus 68.4% osseous bridging and 84.8% versus 90.7% clinical healing with rhPDGF-BB/β-TCP-collagen and autograft, respectively. The authors concluded that rhPDGF-BB/β-TCP-collagen is as effective as autograft with less pain and morbidity than autograft.
Kasai et al. 52 reported successful application of apatite- and wollastonite-containing glass ceramic (AWGC) granules in posterolateral lumbar fusion in 35 patients with three different mixture ratios (2:1, 1:1, and 1:2) of allograft and bioactive glass. The fusion rate was approximately 80% in all three groups, and the authors concluded that lumbar posterolateral fusion can be successfully achieved with even higher mixture ratios of bioactive glass.
Other applications for biomaterials
Nanostructured collagen-hydroxyapatite (coll-HA) multilayer scaffold can be used for osteochondral lesions, and Kon et al. 53 compared its efficacy in microfractures in their randomized study on 100 patients. Both clinical and MRI results were comparable, and the authors concluded that biomimetic implant can be considered a suitable option for the treatment of osteochondral lesion.
Biodegradable cement restrictor made of a compound of polyethylene oxide terephthalate and polybutylene terephthalate performed poorly in a randomized study on 70 patients. 54 Due to high risk of cement leakage (13 in 34 vs 3 in 35) and plug displacement (8 in 34 vs 0 in 35), the authors do not recommend using biodegradable cement restrictor.
Discussion
According to the present review, there are several biomaterials, some with multiple applications, supported by level I evidence of similarity compared to traditional treatment (Table 1). On the contrary, some of the materials performed worse than expected and could not be recommended for clinical use, thereby highlighting the importance of high-quality randomized clinical trials.
Summary of randomized clinical trials on orthopedic biomaterials.
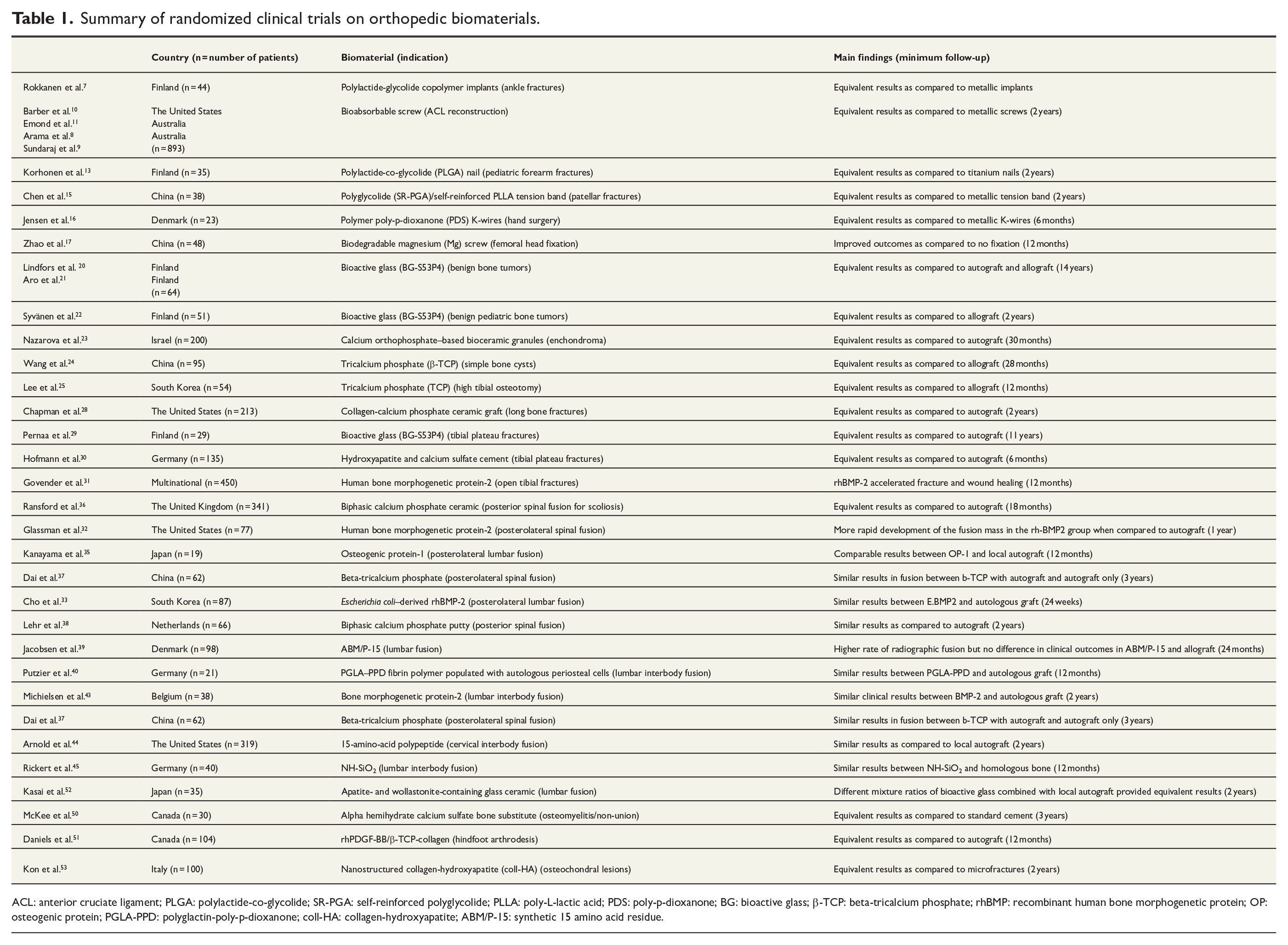
ACL: anterior cruciate ligament; PLGA: polylactide-co-glycolide; SR-PGA: self-reinforced polyglycolide; PLLA: poly-L-lactic acid; PDS: poly-p-dioxanone; BG: bioactive glass; β-TCP: beta-tricalcium phosphate; rhBMP: recombinant human bone morphogenetic protein; OP: osteogenic protein; PGLA-PPD: polyglactin-poly-p-dioxanone; coll-HA: collagen-hydroxyapatite; ABM/P-15: synthetic 15 amino acid residue.
In clinical practice, the pros and cons of biomaterials must be considered for each individual patient. Biodegradable fixation materials often eliminate the need for implant removal. However, the operative technique may be more challenging and time-consuming, not to mention minor concerns regarding implant stability. 13 From a clinical perspective, this may result in a need for additional external support such as a cast, despite the surgical fixation. As bone substitutes, the benefits of biomaterials are related to reduced harvesting site morbidity compared to autogenous bone graft. Autograft is moreover available in only limited supply, especially in children. Then again, challenges due to hard mixture of biomaterial and newly formed bone in reoperation have been reported. 22
Bone grafts are routinely used in spine surgery to achieve rigid fusion between the vertebrae. A frequently used technique is posterior spinal fusion with or without instrumentation, in which bone grafts are applied to the transverse processes to fuse the adjacent vertebrae together. In some cases, interbody fusion is performed in which the intervertebral disk is removed, and the graft material inserted between the vertebral bodies. Any restricted area of decorticated vertebral bony elements poses a challenge to the grafting material. Osteoinductive materials may improve bone healing on decorticated elements. However, excessive soft-tissue ossification in the spine may present with complications, such as foraminal stenosis. 43
None of the randomized clinical trials included in this review assessed the cost-effectiveness of biomaterials. However, Thaci et al. 55 demonstrated that 15-aminoacid polypeptide bone substitute significantly reduced costs compared to autograft in cervical discectomy and fusion. Nevertheless, the evidence on the cost-effectiveness of biomaterials on orthopedic surgery is limited and warrants further studies.
Conclusion
There is evidence of the effectiveness and safety of biomaterials in orthopedic surgery. However, it must be noted that many of the studies included in the review were underpowered for drawing firm conclusions of effectiveness as compared to traditional treatment methods. Furthermore, none of the studies assessed the actual requirement for bone graft, and the outcomes were compared between biomaterials and traditional grafts only. It appears that biomaterials help to avoid donor site morbidity associated with allogenous bone grafts, such as chronic pain and infection. Biomaterials can be chosen according to their bioactivity properties to address specific requirements.
Footnotes
Author contributions
I.H., A.R., and J.-J.S. contributed to the study conception and design. A.R. and A.J.S. contributed to the acquisition of data. All authors contributed to the analysis and interpretation of data. A.R. and A.J.S. contributed to the drafting of manuscript. All authors contributed to critical revision.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Scientific funding has been received from Medtronic, Stryker, Cerapedics, Nuvasive, and Bonalive to institution. I.H. works as a consultant for Medtronic, Bonalive, and Globus. I.H. and J.-J.S. are the section editors of the Scandinavian Journal of Surgery.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A.R. has received research grants from the Päivikki and Sakari Sohlberg Foundation and I.H. from the University of Helsinki, Research Foundation to Injuries and Insurances as well as from the Pediatric Research Foundation and Finska Läkaresällskapet.
