Abstract
Background and aim:
The purpose of this study was to evaluate clinical experiences and cost-effectiveness by comparing robot-assisted surgery with laparoscopic- or open surgery for pelvic and renal operations.
Methods:
A narrative review was carried out.
Results:
When using robotic-assisted surgery, oncological and functional results are similar to after laparoscopic or open surgery. One exception may be a shorter survival in cancer of the cervix uteri. In addition, postoperative complications after robotic-assisted surgery are similar, bleeding and transfusion needs are less, and the hospital stay is shorter but the preparation of the operating theater before and after surgery and the operation times are longer. Finally, robot-assisted surgery has, in several studies, been reported to be not cost-effective primarily due to high investment costs. However, more recent studies provide improved cost-effectiveness estimates due to more effective preparation of the operating theater before surgery, improved surgeon experience, and decreased investment costs.
Conclusions:
Complications and functional and oncological outcomes after robot-assisted surgery are similar to open surgery and laparoscopic surgery. The cost-effectiveness of robot-assisted surgery is likely to equal or surpass the alternatives.
Introduction
The use of robot-assisted surgery (RAS) has rapidly increased since the beginning of the 2000s.1–5 Since the introduction of da Vinci instrumentation in 2001, 6 the number of RAS procedures has increased exponentially, for example, eight-fold in Michigan from 2012 to 2017. 3 Among 15,576 elective proctectomies during 2009–2015, the percentage of robotic-assisted proctectomies increased from 2% to 22%. 7 The increase in use has been even more pronounced in surgery for inguinal hernia. 3 The number of publications on RAS indexed on PubMed was less than 10 each year before 2000 but more than 700 after 2017. 6
A major upside of robotic surgery is the increased dexterity of the laparoscopic instruments, allowing for more complicated movements and dissection. Its main advantages are better orientation in the three-dimensional image of the operating area and better manual precision and ergonomics for the surgeon. 8 The benefits are most significant when operating in narrow spaces. However, investment and operating costs are high, and there is still uncertainty regarding its clinical results as well as its cost-effectiveness. The first two decades of RAS have been dominated by a single surgical robotic company which is currently facing increased competition.
As resources are scarce, there is always a need to prioritize between treatments in healthcare. Prioritization in healthcare should evidently be based on the effect of the treatment but also include cost-effectiveness as a criterion. This is because there always is an opportunity cost when resources are used, that is, the same resources cannot be used for an alternative treatment. 9
The purpose of this paper was to undertake a narrative review to evaluate clinical experiences and cost-effectiveness when using RAS compared to laparoscopic surgery (LS) or open surgery (OS) in pelvic and renal surgery.
Methods
The selection criteria (PICO Patient group, Intervention, Comparator, Outcomes) used in the literature search strategies are shown in Table 1. A narrative review was carried out in line with PRISMA. 10
Selection criteria of accepted studies (PICO).

The results of a search in PubMed of publications until March 7, 2021, were published in Swedish. 11 These results were used as a publication base and supplemented with a new literature search in PubMed covering the entire year 2021 and the year 2022 until August 23. These additional publications were assessed according to title and abstract by two independent reviewers in parallel and selected according to the PICO presented. Publications selected by at least one reviewer were retrieved in full text for further assessment. Disagreements were discussed until a consensus was reached. Each reviewer independently critically appraised the risk of bias in the studies, and discrepancies were discussed until a consensus was reached.
Results
The updated search in 2022 found 357 additional publications. Sixty-one publications selected by at least one reviewer were retrieved in full text for further assessment. Thirty publications in addition to the ones included in our earlier 2022 study 11 were included in the present study.
Clinical findings
Gynecological surgery
A Cochrane report, 12 published in 2019, based on 1016 patients from 12 randomized and controlled trials (RCTs), concluded that it was difficult to state that complications occurred less frequently with RAS compared with conventional LS due to the studies being primarily of low quality. Compared with OS, RAS had a 46-min longer operating time, but the stay in the hospital was 2 days shorter. Survival data were not presented.
A systematic review published in 2021 regarding early cervical cancer showed that minimally invasive (RAS and LS) hysterectomy was associated with reduced intraoperative morbidity, lower blood loss, and improved convalescence compared with OS. 13 Another systematic review in 2021 comparing RAS with LS for various gynecological operations found a decrease in hospital stay after RAS. 14
In studies of tissue damage and markers for inflammatory response,15,16 the perioperative cortisol levels were lower during hysterectomy with RAS than during OS, and recovery was faster after RAS than after OS.
A population-based retrospective study from 2021 with more than 5000 patients with endometrial cancer showed that postoperative complications, hospital stay, and costs were more favorable after RAS and LS than after OS. 17
Randomized studies regarding postoperative complications after removing lymph nodes have compared robot-assisted hysterectomy with OS without detecting any differences. The quality of life (QoL) after 12 months was also examined in the same 113 patient cohorts without showing any difference between the two types of surgery.18,19 In other studies, recovery after hysterectomy due to early endometrial cancer was 2 weeks shorter after RAS compared to after OS, while pain relief and duration of care did not differ.20,21 Another study published in 2018, compared RAS or LS with open radical hysterectomy for cervix cancer in a total of 536 patients. There was no difference regarding complications during or after the operation when comparing the surgical methods. However, RAS was not reported separately. 22
One RCT from the United States in 2019 23 included 319 patients with cervix cancer in the minimal-invasive group (LS n = 269, RAS n = 50) and 312 in the OS group. The risk of recurrence after LS or RAS was four times higher, and the risk of death was six times higher than after OS. In the minimally invasive group, where LS dominated (84%), recurrence and death were significantly more common (71% higher risk) than in OS. The study was discontinued after 4.5 years when 96.5% of the patients in the group who underwent OS were alive compared with 86.0% after LS or RAS. This difference could not be explained by differences in tumor stage or by the experience of the operating surgeons.
A review article from 2020 24 reported survival in 9499 patients, of whom 51% underwent open hysterectomy, and the remainder underwent RAS or LS (RAS n = 2675, LS n = 2009). If concluded that among patients undergoing radical hysterectomy for early-stage cervical cancer, minimally invasive radical hysterectomy was associated with an elevated risk of recurrence and death compared with open surgery.
A systematic review of early-stage cervix cancer, based on 39 nonrandomized studies and one RCT, showed that LS and RAS were associated with a higher risk of intraoperative complications compared to open surgery but with a lower frequency of postoperative complications. 25 More recently, a narrative literature review showed that minimally invasive surgery was associated with statistically significant lower survival. 26
In hysterectomy, the operation time has been reported to be approximately 1 h longer for RAS than for LS, published in 2018. 27 The learning curve for RAS was, however, not analyzed.
The findings regarding gynecological surgery are concluded in Table 2.
Schematic summary of the pros and cons of robot-assisted surgery in pelvic and renal surgery.
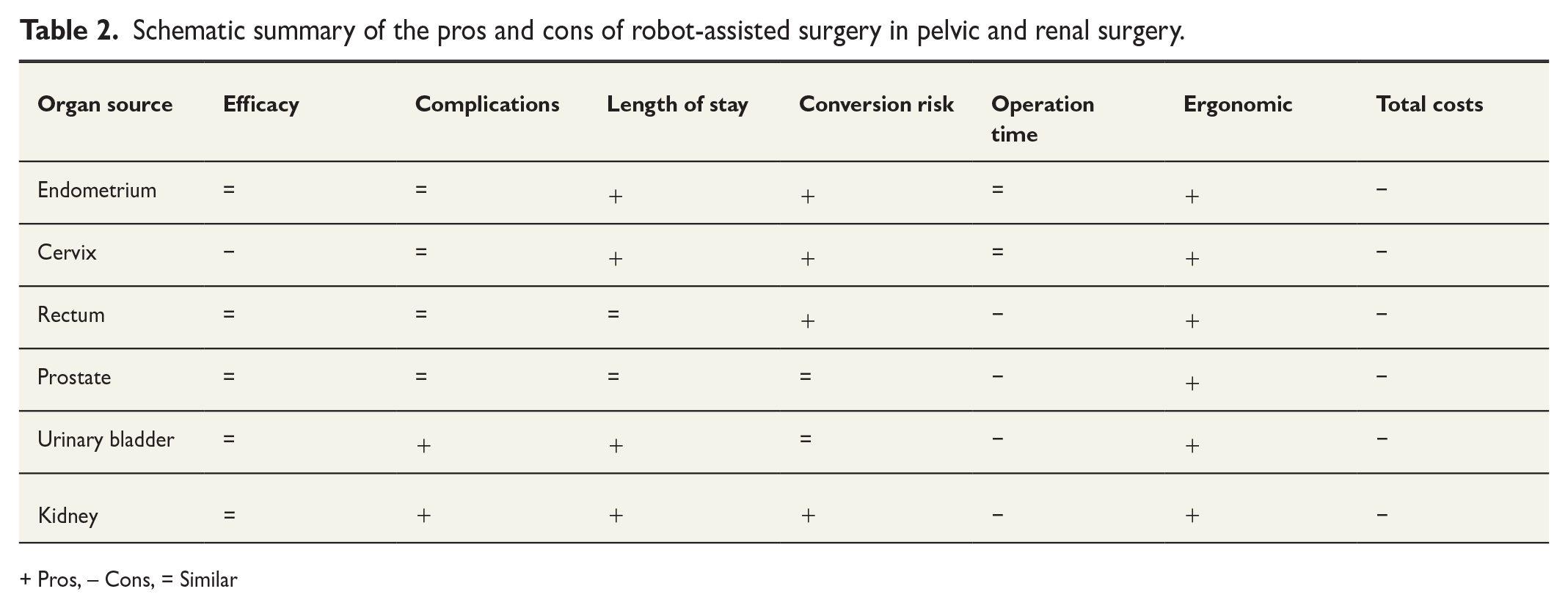
+ Pros, – Cons, = Similar
Prostate cancer
A Cochrane report from 2017 28 based on two RCTs with 446 patients operated for cancer of the prostate using either RAS, LS, or OS, with reported outcomes after 3 months. No account was taken for the surgeons’ experience with the three interventions. The hospital stay was shorter, and the need for transfusion was lower after RAS. Still, there were no differences in postoperative complications, mortality, oncological outcome, or QoL related to the urinary tract or recovery of sexual function. 28
One RCT published in 2021 comprising 782 patients compared open radical prostatectomy with either RAS or LS. At 3 months, postoperative urinary continence occurred in 54% after RAS and 46% after LS (p = 0.027). RAS had a significant benefit regarding early recovery of potency. The oncological and morbidity outcomes were similar. 29
In a systematic review and meta-analysis published in 2017, 30 two RCTs and 28 observational studies were analyzed regarding biochemical recurrence after RAS and LS respectively. There were no differences in microscopic radicality. The risks of biochemical recurrence, postoperative complications, and urinary incontinence were lower after RAS, and the potency was better.
In an RCT from Australia published in 2018, 31 RAS (n = 150) was compared with open retropubic prostatectomy (n = 146). After 24 months, the patient groups had no differences in functional results. In another review from 2016 32 based on 61 included observational studies, there was no difference in the radicality at microscopic examination. Still, the finding was that RAS led to fewer complications and less risk of incontinence and impotence.
The findings regarding prostate cancer are concluded in Table 2.
Cancer of the urinary bladder
A multinational randomized non-inferiority study (RAZOR, n = 302) investigated the oncological outcome after cystectomy with RAS or OS in cancer of the urinary bladder.33–35 Follow-up after 2 and 3 years showed no differences in mortality, recurrence, or QoL. 33 In a meta-analysis from 2019 including 10 studies (of which two were randomized), 36 RAS resulted in shorter time in hospital, fewer postoperative complications, more lymph nodes in surgical specimens and lower mortality. In two other systematic reviews,37,38 there were no differences between RAS, LS, or OS in terms of radicality, complications, and QoL. For RAS, the operation time was longer, the bleeding less and the time in the hospital shorter.
Vejlgaard et al. 39 reported no difference in QoL between RAS, LS, and OS. In addition, a recently published RCT 40 with 169 patients treated with RAS and the same number with OS showed a significant increase in days alive after RAS.
A randomized study of cystectomy published in 2015 compared RAS (n = 60) with OS (n = 58). There was no difference in perioperative complications, and the duration of treatment was 8 days for both types of surgery. The RAS group had a longer operating time but less bleeding. In a later study reporting 2–6 years follow-up, there was no difference in recurrence or cancer-specific mortality.41,42
Another systematic review based on five RCTs and 49 observational studies compared the incidence of complications over 90 days between RAS (n = 6500) and OS (n = 23,197). Generally, the operation times were longer (61–127 min) in robot-assisted cystectomy compared with OS.32,36,37,42–44 The presence of all severe complications did not differ between the two groups, but postoperative mortality was lower in the robot group, operation time was longer, mild complications were fewer, and hospital time shorter. 45 Another RCT from Italy reported lower perioperative transfusion rates in RAS compared with OS. Perioperative complications, hospital stay and QoL were considered comparable between the groups. 46
The current literature does not support differences between RAS, LS, and OS. The findings regarding cancer of the urinary bladder are concluded in Table 2.
Rectal cancer
ROLARR is a large multinational, randomized study published in 2017 47 comparing RAS (n = 237) with LS (n = 234) for conversion to OS in rectal cancer. The responsible surgeon had performed at least ten operations to include patients in the study. Conversion occurred in 8.1% in RAS and 12.2% in LS, a nonsignificant difference. In addition, there was no difference in microscopic radicality or postoperative complications. The study showed no advantages for RAS in rectal cancer over LS. 47 The learning effect can be a disruptive factor in randomized trials. When this was corrected in a follow-up study, it was shown that RAS reduced the risk of conversion. 48
In a randomized study from South Korea published in 2018, radicality in the surgical specimen after RAS (n = 73) was compared with LS (n = 66) for rectal cancer. There was no difference between the groups regarding radicality to the sides, resection margin, or the number of lymph nodes in the surgical specimens. Nor were there any differences in complications, postoperative bowel function, or QoL. The operation time for RAS was almost 2 h longer (339 vs 228 min), but the bleeding was less (50 vs 100 mL). After 12 months, sexual function was better in the group operated on with RAS. 49
A systematic review from 2019 of rectal cancer surgery that included eight randomized studies with 1305 patients analyzed the need for conversion to open surgery and oncological outcome. At RAS, 5.7% were converted to OS, and at LS, 11.9%, which was a significant difference. The operation time was shorter in LS. 50
A controlled study from Denmark compared the need for analgesic treatment during and after surgery for rectal cancer using RAS (n = 26) or LS (n = 25). The use of opioids was less using RAS (p = 0.001). After the operation, however, there was no difference between the groups in need for pain relief, despite a significant difference in conversion to OS (1 vs 10 patients). The operating times did not differ. 51
In a systematic review and meta-analysis from 2018 based on 334 RAS and 317 LS for rectal cancer, there were no obvious differences in primary oncological results. The conversion rate was lower after RAS, but the operation time was more extended. 52 Another review from 2019 based on 17 systematic reviews reported a lower conversion rate after RAS, and no other certain differences. 53
The findings regarding rectal cancer are concluded in Table 2.
Renal cancer
In a systematic review published in 2019 based on 34 studies of partial nephrectomy, 19,638 patients operated with RAS were compared with 41,170 who underwent OS. RAS resulted in less bleeding, fewer postoperative complications, shorter hospital stays, and fewer readmissions. 54
A retrospective case study with matched groups (n = 52 × 2) compared RAS with OS in partial nephrectomy. The operation time for RAS was 174 min (130–210), and for OS, 120 min (100–140). Postoperative complications occurred in 8% after RAS and 21% after OS. 55
A retrospective multi-center study of nephrectomy evaluated 404 patients operated on with RAS and 537 patients with LS. The RAS group had a longer operation time but a shorter period of stay in the hospital. 56 In another multi-center study comparing surgical procedures for tumors larger than 7 cm (T 2), no difference was found in oncological outcomes measured as 5-year survival. 57
The findings regarding renal cancer are concluded in Table 2.
Cost-effectiveness findings
Several of the included studies investigated the costs or cost-effectiveness of RAS as a primary endpoint or in secondary analyses. Most such studies show that costs increase with RAS, although it has also been reported that a shorter hospital stay partly compensates for this.58–61
Bai et al. 58 performed a systematic review, published in 2022, with the incremental cost-effectiveness ratio as the outcome of interest. It included 31 studies where most of them (81%) found that RAS was cost-effective compared with OS for prostatectomy using a long-time horizon (10 years or lifetime). Inconsistent evidence was found for the cost-effectiveness of RAS versus LS and (chemo)radiotherapy.
An analysis of perioperative outcomes and hospitalization cost for treatment of benign prostatic hyperplasia after OS and RAS were presented by Bhanvadia et al. 62 RAS had significantly less perioperative complication rates with shorter LOS and likely discharge to home. However, total hospitalization costs remained higher. In their cost model, it was found that the length of stay would need to be 3–4 days shorter with RAS to offset the surgical costs of the robot.
A simulation model for the estimation of the cost-effectiveness of RAS compared with OS and LS was created by Faria et al. 63 The model was used for the simulation of a 20-year horizon. RAS was more expensive but more effective than both OS and LS. It was concluded that RAS is cost-effective even when considering willingness-to-pay thresholds lower than the traditionally used ones.
Kosa et al. 64 made a prospective comparison of costs between RAS, LS, and OS in endometrial cancer in women with severe obesity. There was no difference in overall costs between the three surgical modalities in patients with BMI ⩾ 40.
Labban et al. 65 created a simulation model in which 65-year-old men who underwent radical prostatectomy for localized prostate cancer were followed up for 10 years after either RAS, OS, or LS. They found that RAS has an incremental cost-effectiveness ratio lower than the willingness-to-pay threshold for patients with localized prostate cancer in the UK. Thus, it is a cost-effective surgical treatment option compared to OS and LS.
In a study from the Netherlands in 2022, RAS was cost-effective compared to LS based on data from a population-based study after 7 years of follow-up. 66 The cost-effectiveness of OS, LS, and RAS approaches to proctectomy from societal and healthcare system perspectives by Simianu et al. 67 using a decision-analytic model. The major factors influencing cost-effectiveness were the operative cost and the postoperative length of stay. LS was the most cost-effective approach. RAS was cost-effective if modest differences in costs or postoperative length of stay could be achieved.
A study from Sweden on hysterectomy 68 reported that RAS entails similar costs as LS if the robot’s purchase cost is not included. The cost increase was a total of about 20%. Higher purchase and maintenance costs and longer operating times mean that RAS is less cost-effective than LS or OS.
The market for equipment for robot-assisted surgery is increasingly competitive as new manufacturers enter. Competition in a free market is known to increase cost-effectiveness and stimulate innovations, both ultimately benefiting the patients.
Ergonomics
RAS has ergonomic benefits and reduces workload compared to LS or OS. 69 Operating surgeons have lower estimated physical exertion at RAS compared to OS, 70 less pain at RAS compared to LS or OS in radical prostatectomy, 71 and less exertion at RAS compared to LS in gynecological surgery. 72 RAS also stimulates a two-handedness, 73 an ergonomic advantage. However, some surgeons also indicate increased physical exertion associated with RAS requiring ergonomic adjustments and/or frequent breaks. 74
Discussion
Recent studies indicate that RAS is not inferior to LS or OS regarding oncologic or functional outcomes. However, one exception may be following operations for uterine cervical cancer, where survival after LS and RAS may not be as good as after OS. After RAS, there is less bleeding and the need for blood transfusion after RAS, and postoperative complications are similar to OS or LS. Furthermore, RAS offers ergonomic advantages compared to LS.
Baek et al. 75 reported that learning of RAS is shorter than for LS and OS. A publication from China states that the learning time for robot-assisted cystectomy is 50–110 procedures. 43 However, the effect of the surgical learning curve is not sufficiently studied, which probably has affected the results of RAS, as other studies have shown that the experience of the operating surgeons with RAS may have been less than at LS and OS.
The operation time is longer with RAS than with LS or OS,21,29–39,41,45 with risks of delaying and canceling other operations. 76 It is rarely detailed whether preparation and settlement time in the operation theater is included in the operation time. However, one study has shown that the operation time of prostatectomy was shorter after RAS than OS. 30
Complications were less common in RAS for cancer of the urinary bladder, but for other cancers, no difference was seen.36,45,54,77 LOS appears to be shorter after gynecological operations and cystectomy, but there was no difference after rectum or prostate cancer. The risk of conversion to open surgery in rectal cancer surgery is less in RAS than in LS.48,52,53 The QoL did not differ between the operation types.18,19,28 Studies show ergonomic benefits and reduced workload at RAS compared to the LS or OS.69–74 RAS leads through structural rationalization to centralizing specific surgical procedures, which may risk unequal access to such interventions.
The findings of all the presented studies are summarized in Table 2. In this table, the symbol “+” indicates a mainly positive relative outcome of RAS compared to LS or OS, while “−“ indicates a mainly negative relative outcome. However, these pros and cons are not suitable to be combined into a summary index, guiding decision-makers on what to do. Instead, it should be interpreted as the current best information about these aspects without data about their importance in specific decisions.
Minimally invasive surgery (RAS or LS) has been found to have several advantages compared to OS. RAS results in prolonged operation times compared to LS, but the risk of conversion to OS is less than for LS, except in prostate and urinary bladder cancer surgery. The oncological outcome for rectal and urologic cancer and renal cancer has been found not to differ except for cancer of the cervix, where OS seems to be favorable. Postoperative complications occur less often after RAS in cancer of the urinary bladder and the kidneys but are similar in other forms of cancer.
LOS has been shorter for RAS and equal for rectal and prostate cancer in most cancer types. The risk of conversion to OS is less for RAS than for LS, except in cancer surgery of the prostate and urinary bladder. The overall operation time is longer for RAS. This may, however, decrease as more centers gain extensive experience in robotic-assisted surgery. A serious drawback is that long-term follow-up studies are lacking.
Ergonomically, RAS is more favorable than LS and OS in all types of operations reported here. The costs for RAS have been shown to be higher. However, recent studies imply that they are approaching LS and OS due to decreasing investment costs and shorter LOS.
In summary, this review of current literature shows that complications and functional and oncological outcomes after RAS are similar to OS or LS. An exception may be the removal of the uterus in the case of cervical cancer. RAS is associated with the more extended operation when preparation and settlement time are appropriately included. RAS’s cost-effectiveness is likely to equal or surpass OS and LS with decreased investment costs, more effective preparation of the operating theater before surgery, and increased surgeon experience.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
