Abstract
Background and objective:
The prevalence of acute cholecystitis among elderly patients is increasing. The aim of this study was to compare laparoscopic cholecystectomy (LC) to antibiotics in elderly patients with acute cholecystitis.
Methods:
A randomized multicenter clinical trial including patients over 75 years with acute calculous cholecystitis was conducted in four hospitals in Finland between January 2017 and December 2019. Patients were randomized to undergo LC or antibiotic therapy. Due to patient enrollment challenges, the trial was prematurely terminated in December 2019. To assess all eligible patients, we performed a retrospective cohort study including all patients over 75 years with acute cholecystitis during the study period. The primary outcome was morbidity. Predefined secondary outcomes included mortality, readmission rate, and length of hospital stay.
Results:
Among 42 randomized patients (LC n = 24, antibiotics n = 18, mean age 82 years, 43% women), the complication rate was 17% (n = 4/24) after cholecystectomy and 33% (n = 6/18, 5/6 patients underwent cholecystectomy due to antibiotic treatment failure) after antibiotics (p = 0.209). In the retrospective cohort (n = 630, mean age 83 years, 49% women), 37% (236/630) of the patients were treated with cholecystectomy and 63% (394/630) with antibiotics. Readmissions were less common after surgical treatment compared with antibiotics in both randomized and retrospective cohort patients (8% vs 44%, p < 0.001% and 11 vs 32%, p < 0.001, respectively). There was no 30-day mortality within the randomized trial. In the retrospective patient cohort, overall mortality was 6% (35/630).
Conclusions:
LC may be superior to antibiotic therapy for acute cholecystitis in the selected group of elderly patients with acute cholecystitis.
Keywords
Introduction
With increasingly aging populations, an increasing number of elderly patients will be referred to emergency departments due to complicated gallstone disease. While laparoscopic cholecystectomy (LC) is the gold standard treatment for acute cholecystitis,1–4 emergency cholecystectomy among elderly patients is still controversial.
Earlier studies have reported significant morbidity (24%–31%) and mortality (4%–13%) associated with acute cholecystitis in older patients.5–7 Mortality after LC in patients aged ⩾ 65 years varies between 0.8% and 3.0%, and in patients over 80 years, the mortality is further increased.3,4,8,9 This is mostly attributed to cardiac, respiratory, and septic complications.10–12 Fear of increased perioperative complications may have resulted in favoring antibiotic therapy over surgery. Antibiotic therapy can be combined with percutaneous drainage.7,13 However, percutaneous drainage has been associated with similar mortality rates as cholecystectomy.13–15 Furthermore, conservative treatment with or without percutaneous drainage does not affect the underlying gallstone disease, and subsequent complications can occur. 16 Although conservative treatment may be initially successful in 96% of cases, almost one in four patients suffer from subsequent acute cholecystitis episodes and require a new surgical referral. 16
As life expectancy increases, gallstone disease in elderly patients may constitute a substantial burden to health care systems. There are no high-quality randomized trials comparing LC to conservative treatment with antibiotics alone in elderly population with acute cholecystitis. We performed a multicenter randomized trial complemented with a retrospective cohort of all eligible patients to mitigate the limitation of the small number of randomized patients and premature study termination. The aim of the study was to compare the morbidity between LC with antibiotic therapy in patients over 75 years of age in the treatment of imaging-confirmed acute cholecystitis.
Methods
The trial was conducted in four hospitals in Finland (Kuopio, Tampere, and Turku university hospitals and Mikkeli central hospital). Patients aged 75 years or over admitted to emergency department with imaging-confirmed acute calculous cholecystitis (ultrasonography, magnetic resonance imaging, or computed tomography) were evaluated for enrollment. Patients with suspected common biliary duct stone or cholangitis, liver insufficiency, other severe illness (American Society of Anesthesiologists (ASA) > 3), previously treated cholecystitis, or incapability to give informed consent were excluded.
Patients were randomly assigned to undergo either LC or conservative treatment with antibiotics and/or radiological drainage, if needed. The randomization was carried out by means of block randomization of five opaque envelopes. We conducted a power analysis using an alpha of 0.80 to determine the necessary sample size for this study. Based on our analysis, we estimated that we would need a total of 200 patients (100 in each group) to achieve a statistical power of 0.80. Due to the challenges of an acute care setting and strict inclusion criteria, patient enrollment in the randomized trial was markedly slower than anticipated resulting in premature termination of the trial in December 2019. In order to assess the whole eligible patient cohort during the study period at the study hospitals, we retrospectively collected the data of all patients ⩾75 years presenting with acute cholecystitis to complement the small number of randomized patients. Patient demographics included age, sex, ASA classification scores, body mass index (kg/m2), pre-existing medical treatment, and C-reactive protein, white blood cell count, and liver enzymes (alkaline phosphatase, bilirubin).
Interventions
Emergency LC (<24–48 h) was performed using the standard laparoscopic technique. Conversion to open surgery or perioperative cholangiography was performed, if necessary. Operated patients received preoperative intravenous (IV) cefuroxime 1.5 g and in cases of a more severe cholecystitis, antibiotic therapy was continued postoperatively.
Patients randomized to the antibiotic group received IV cefuroxime 1.5 g three times a day until clinical recovery was observed. At discharge, the patients were prescribed 5–7 day per oral (PO) cephalexin 500 mg three times a day. Percutaneous drainage of the gallbladder (cholecystostomy) or elective delayed cholecystectomy were performed or planned only if there was clinical deterioration.
Outcome measures
The primary outcome was morbidity defined according to the Clavien–Dindo classification. 17 Surgery-associated bile duct injuries were graded according to the Strasberg classification. 18 Predefined secondary outcomes included postoperative mortality (⩽30 days), need for additional treatment (e.g. percutaneous drainage), length of hospital stay, readmissions, or later gallstone-related interventions, all of which were assessed from electronic medical charts with at least 1-year follow-up after index treatment. Pre- and post-operative quality of life was initially predefined secondary endpoints, but these were omitted during the study as the elderly patients were not capable of completing these form questionnaires sufficiently.
Data analysis
All statistical analyses were performed using SPSS Statistics version 26 for Windows (IBM Corp, Armonk, NY, USA). Values were presented as absolute values with percentage or median with minimum and maximum or with interquartile range (IQR). Two-tailed chi-square tests or Fisher’s exact tests were used to compare categorical variables, and the Student’s t-test or Mann–Whitney U-test was employed to compare continuous variables. Statistical significance was set at a p-value of less than 0.05.
Ethical aspects
The study was conducted according to the Helsinki Declaration. The study was registered in Clinical Trials.gov (NCT02972944) and approved by the Ethical committee of Kuopio University Hospital and the ethical committees of all study hospitals. No funding was received for this study. All randomized patients gave written informed consent.
Results
Trial patient flow for the randomized clinical trial (RCT) is shown in Figure 1. A total of 237 patients were evaluated for enrollment. Of these, 42 patients (mean age 82 years, 43% women) underwent randomization with 24 patients randomized to LC and 18 patients to antibiotic therapy. The baseline characteristics of the randomized patients are presented in Table 1. There were no significant differences in baseline characteristics between study groups. The retrospective cohort of the randomized trial enrollment period included 630 patients over 75 years of age with imaging-confirmed acute cholecystitis (see Table 2 for baseline demographics). In total, 236 (37%) patients were treated with surgery and 394 (63%) with antibiotics or percutaneous drainage, if necessary. In the retrospective cohort, patients undergoing antibiotic therapy were older (p < 0.001), had higher ASA grades (p = 0.045), and prevalence of chronic cardiovascular (p = 0.007) or neurological diseases (p < 0.001) compared with patients undergoing surgery. The median follow-up time of RCT patients was 29 (IQR = 23–36) months, and the median follow-up time of patients in the retrospective cohort was 28 (IQR = 20–37) months.
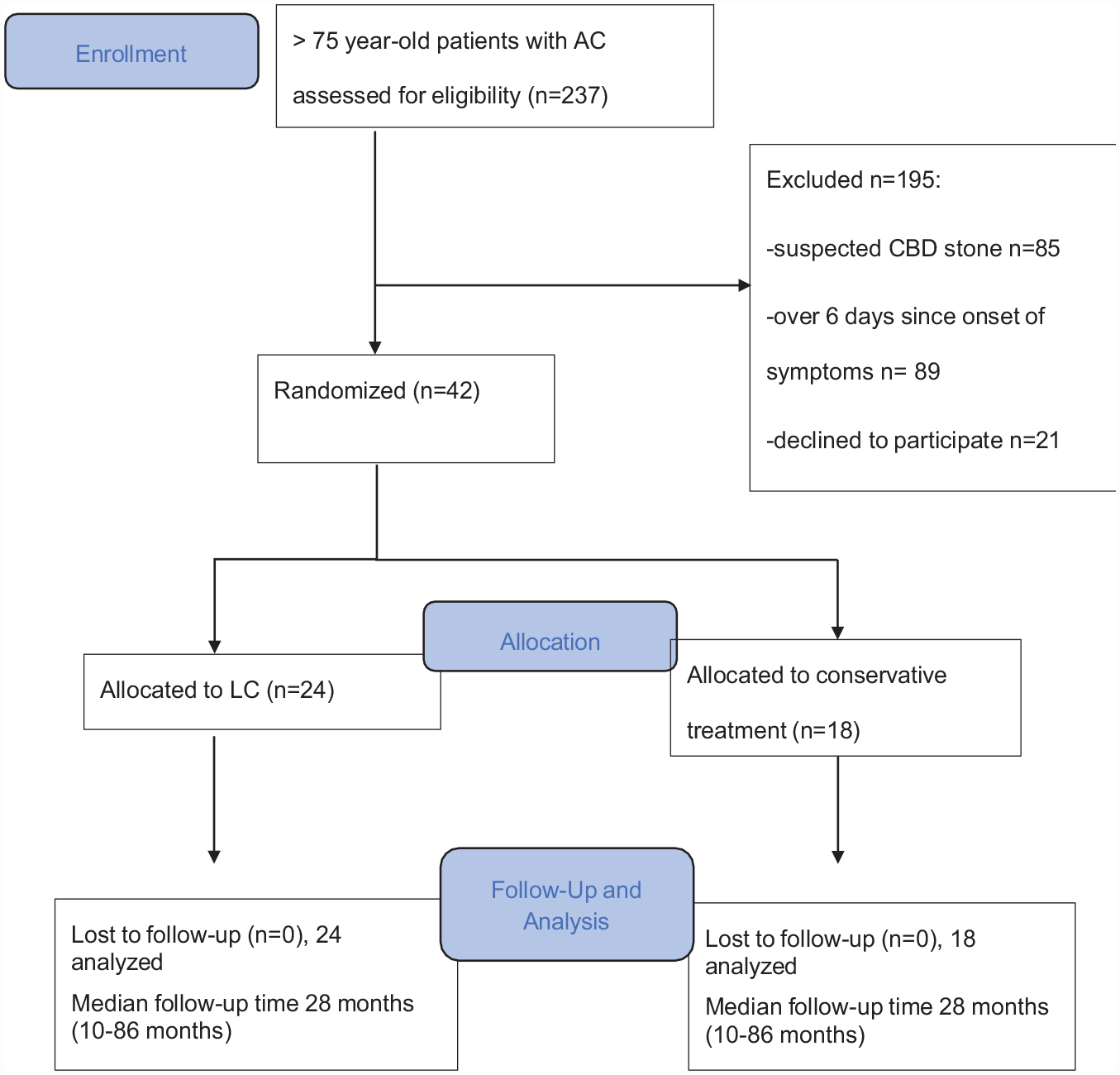
Patient flow in the randomized trial.
Baseline patient demographics of the randomized trial (n = 42).
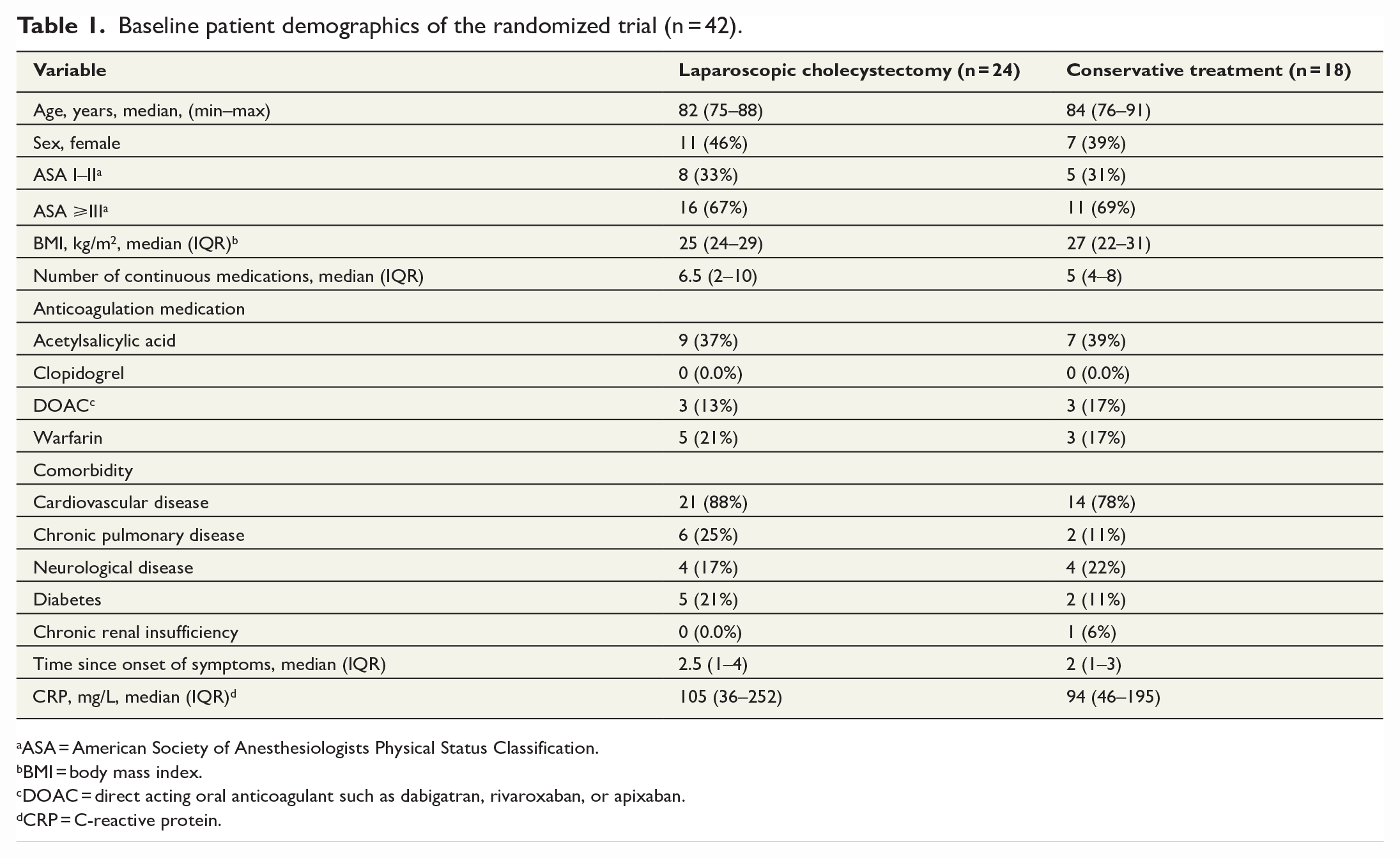
aASA = American Society of Anesthesiologists Physical Status Classification.
bBMI = body mass index.
cDOAC = direct acting oral anticoagulant such as dabigatran, rivaroxaban, or apixaban.
dCRP = C-reactive protein.
Baseline demographics of the data of the retrospective cohort (n = 630), all patients over 75 years of age with imaging-confirmed acute cholecystitis between January 2017 and December 2019.
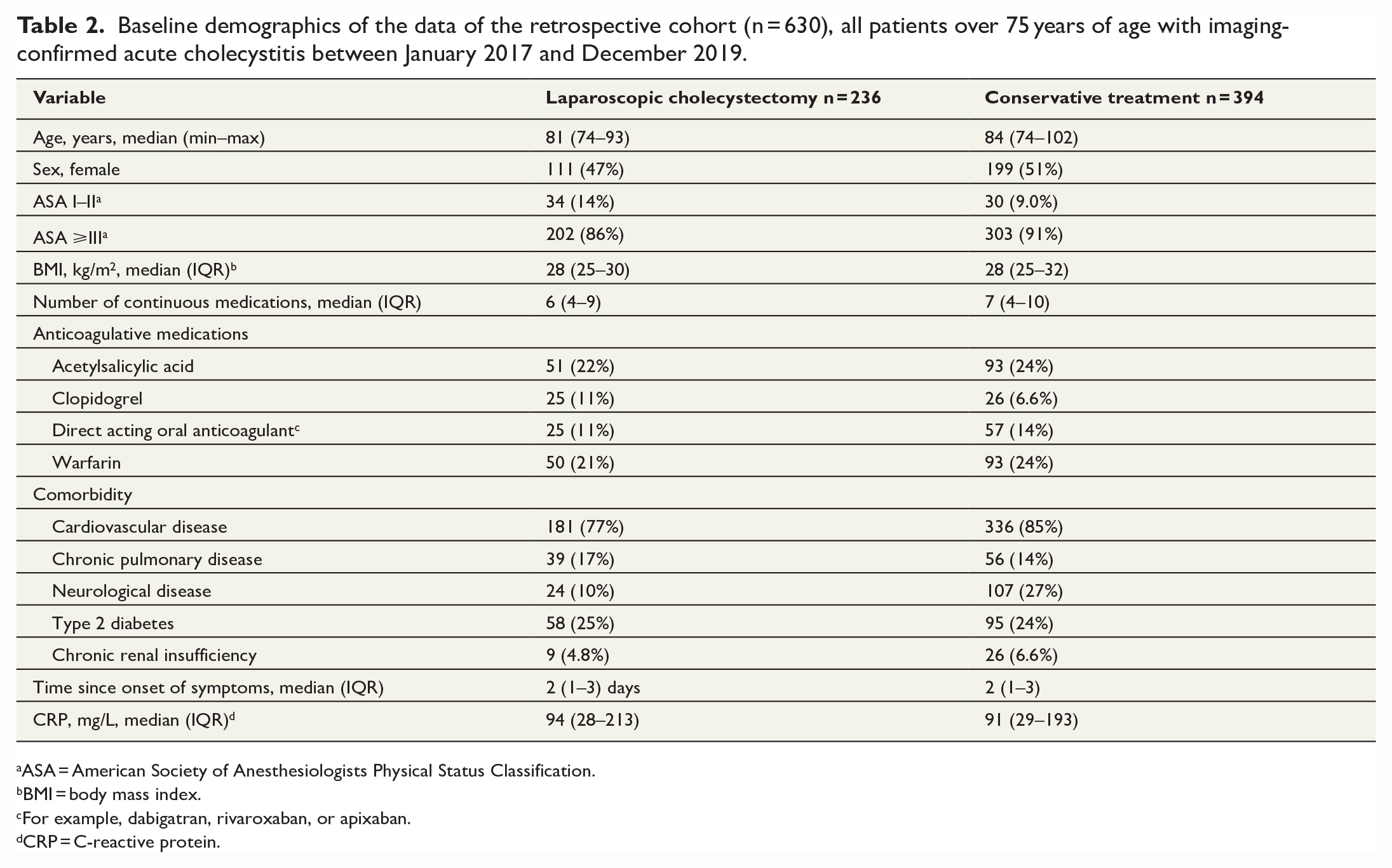
ASA = American Society of Anesthesiologists Physical Status Classification.
BMI = body mass index.
For example, dabigatran, rivaroxaban, or apixaban.
CRP = C-reactive protein.
The complication rate within the randomized trial was 17% (n = 4/24) after cholecystectomy and 33% (n = 6/18) after antibiotics (p = 0.209) (complications are presented in detail in Table 3). Numbers of readmissions (44% vs 8.3%, p = 0.009) and reinterventions (56% vs 8.3%, p < 0.001) were higher in the antibiotic group (Table 3). There was no 30-day mortality or need for intensive care unit (ICU) care in the randomized trial patients. Both length of hospital stay (5 days after LC and 4 days after antibiotics) and cumulative length of hospital stay (5 days vs 5.5 days, respectively) were similar between the study groups. Patients who initially had emergency cholecystectomy were less likely to have readmissions (2/24, 8.3%) compared to patients initially treated with antibiotics (8/18, 44%) (p = 0.009).
Morbidity of the randomized trial patients.
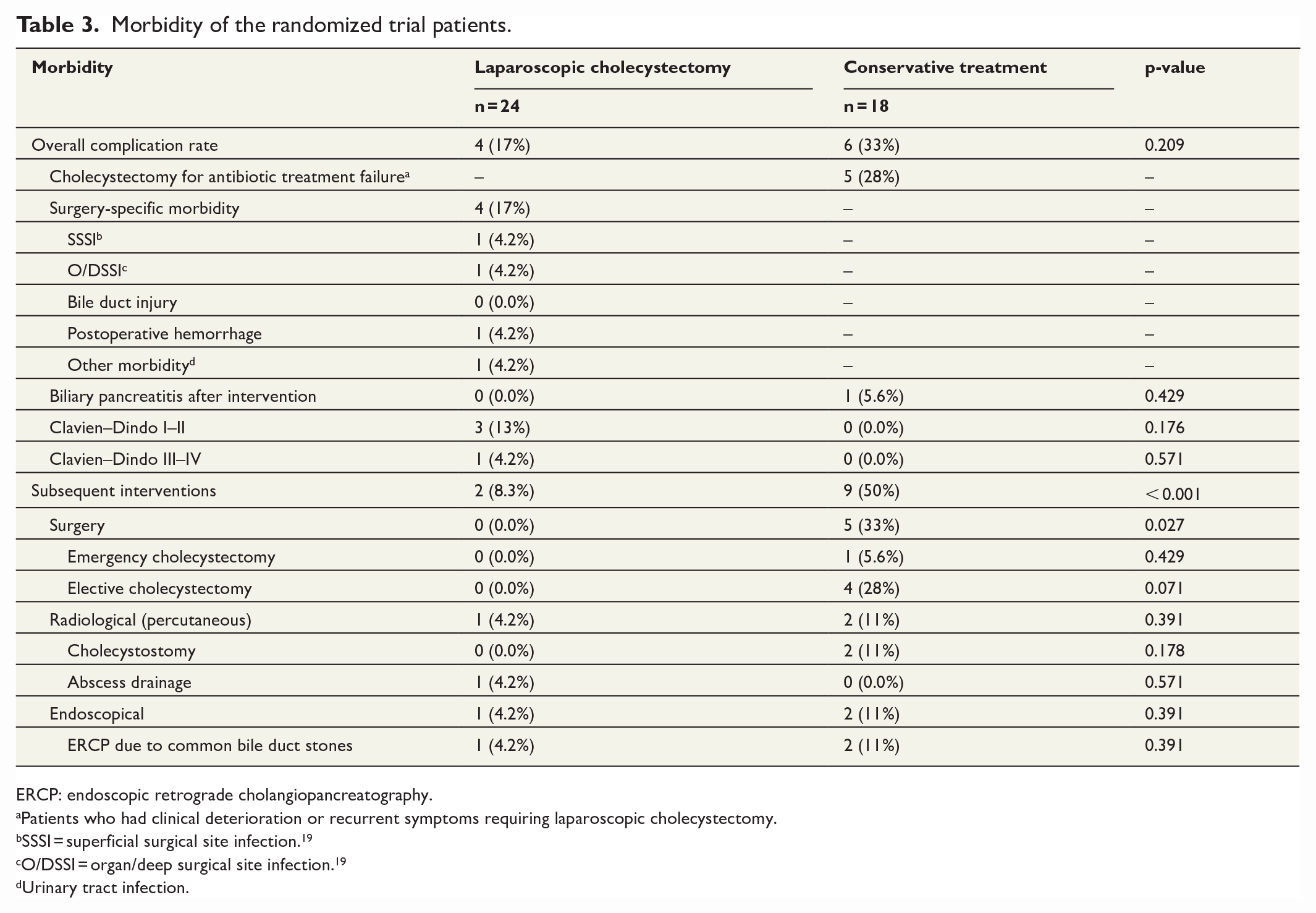
ERCP: endoscopic retrograde cholangiopancreatography.
Patients who had clinical deterioration or recurrent symptoms requiring laparoscopic cholecystectomy.
SSSI = superficial surgical site infection. 19
O/DSSI = organ/deep surgical site infection. 19
Urinary tract infection.
Outcomes of the retrospective cohort are presented in Table 4. Patients treated with antibiotics had more readmissions (33% vs 11%, p < 0.001), higher 30-day mortality (7.1% vs 3%, p = 0.028), and more reinterventions (50% vs 4.6%, p < 0.001) than those undergoing LC. Antibiotic treatment failed in 52 patients (13%), who were then treated with surgery. Percutaneous drainage was necessary in 94 patients (24%) in addition to antibiotic therapy. There were 10 (4.2%) bile duct injuries in the surgically treated patients, all of which were classified as Strasberg A. Conversion rate to open cholecystectomy was 18% (n = 39).
Outcomes of the retrospective cohort patients: ⩾75-year-old patients with acute cholecystitis (n = 630).
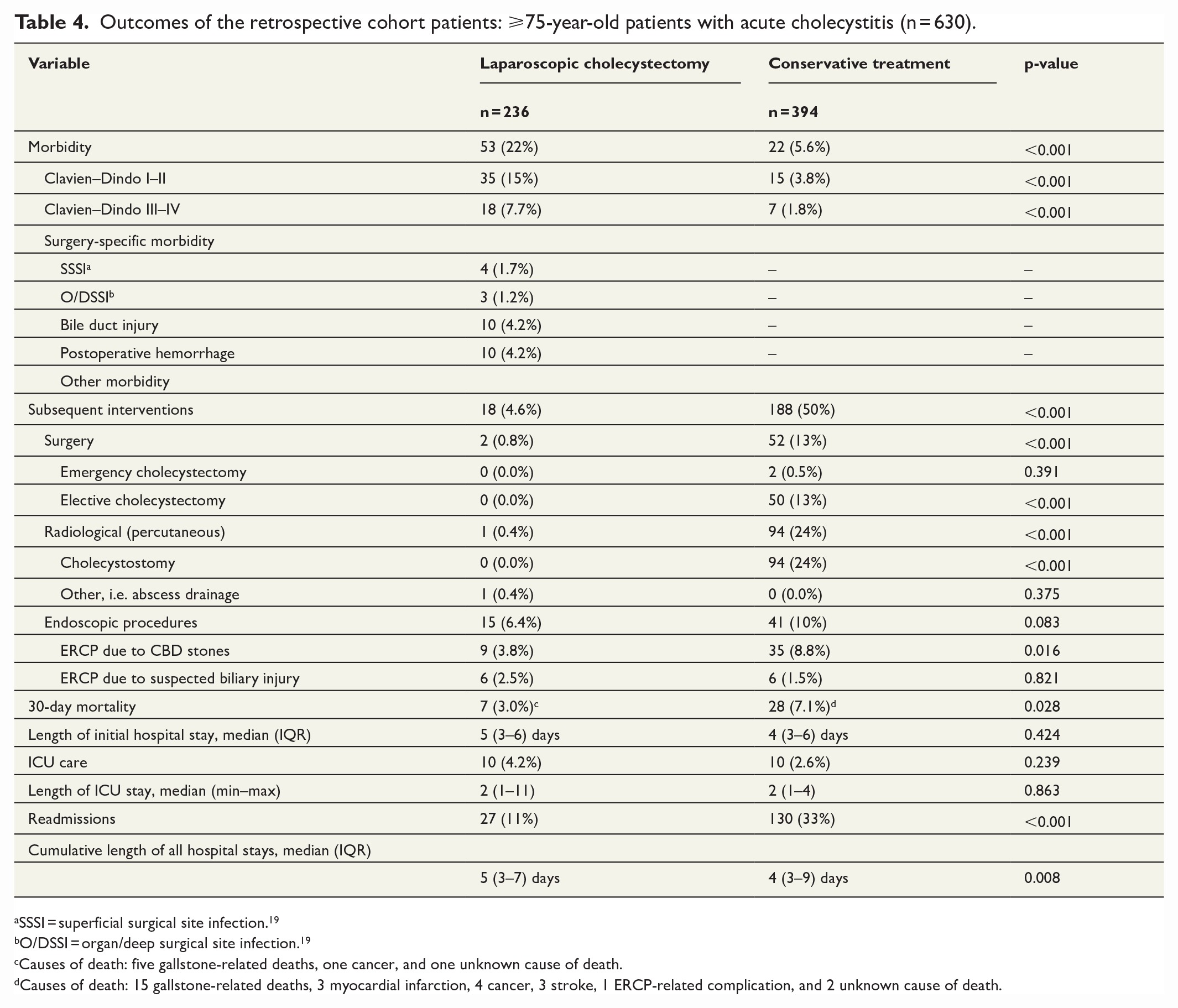
SSSI = superficial surgical site infection. 19
O/DSSI = organ/deep surgical site infection. 19
Causes of death: five gallstone-related deaths, one cancer, and one unknown cause of death.
Causes of death: 15 gallstone-related deaths, 3 myocardial infarction, 4 cancer, 3 stroke, 1 ERCP-related complication, and 2 unknown cause of death.
Discussion
In this pilot-type RCT, in patients over 75 years of age with imaging-confirmed acute cholecystitis, there were no differences in the complication rates between LC and antibiotic treatment. LC was associated with significantly lower rate of readmissions and reinterventions compared to antibiotic treatment, corroborating the findings of earlier studies showing emergency cholecystectomy for acute cholecystitis to reduce costs, readmissions, and length of hospital stay.1–4
Older age is associated with increased prevalence of gallstone disease, 2 and older age is an independent prognostic factor for increased morbidity and mortality after emergency abdominal surgery. 19 Our results are in line with earlier studies, where postoperative morbidity and mortality after cholecystectomy varies between 24%–31% and 4%–13%, respectively.5–7 Whereas cholecystectomy does carry higher risk for mortality than conservative care, antibiotic therapy is associated with subsequent biliary events in 22% of patients. 20
Percutaneous cholecystostomy has been proposed as an alternative treatment in elderly high-risk patients. 21 However, recent studies have demonstrated that even after percutaneous cholecystostomy, new episodes of cholecystitis can occur and there is an increased reintervention and complication rate.22,23 A recent Dutch multicenter randomized study (CHOCOLATE-trial) compared undelayed cholecystectomy and percutaneous catheter gallbladder drainage for acute cholecystitis in high-risk patients. They demonstrated that emergency surgery was safe and did not increase mortality and both the 1-year complication rate and risk for recurrent biliary disease were lower after surgery (12% and 5%, respectively) compared to percutaneous drainage (65% and 53%). 22 The novel procedure of endoscopic sonography–guided gallbladder drainage may offer an alternative approach in the future. 24
Recurrent biliary events often occur in elderly patients with cholecystitis who are unfit for surgery. 25 Need for endoscopic retrograde cholangiopancreatography (ERCP) was between 6% and 10% in our patient cohort, although patients with common bile duct stones were excluded from this study. Elderly patients tend to have more biliary duct stones than younger patients and ERCP is safe and effective for patients of all ages.26,27 Cholecystectomy after ERCP is strongly recommended in young and fit patients,28–30 but in elderly or otherwise high-risk patients, this is still controversial and needs further studies.
There are limitations in this study. The first major limitation is the premature termination of the randomized trial based on the extreme difficulties in patient enrollment resulting in an underpowered feasibility pilot study. In addition to the challenges of conducting a randomized trial within an acute care setting, most patients aged over 75 years of age had severe comorbidities, such as dementia and congestive heart failure, and a higher rate of common bile duct stones, thus excluding the patients from being evaluated for trial enrollment. In addition to the small number of randomized patients, another limitation was the lack of thorough prospective assessment of all patients over 75 years of age with acute cholecystitis due to the emergency surgery setting, potentially causing selection bias. To mitigate both of these limitations, we performed a thorough retrospective study of all eligible patients in all of the four hospitals during the study period to have an overview of the whole patient cohort. In the retrospective cohort, the decision to refrain from surgery was based on surgeons’ or anesthesiologists’ decision or patients’ own will. This retrospective cohort is naturally limited by its retrospective nature. However, it is quite a large multicenter patient cohort assessing treatment outcomes in elderly patients with acute cholecystitis.
In conclusion, LC may be superior to antibiotic therapy for acute cholecystitis in the selected group of elderly patients in terms of safety and reduced rate of readmissions and reinterventions. Larger RCTs are needed to validate these pilot results in patients over 75 years of age.
Footnotes
Author contributions
All the authors contributed in writing this manuscript: study design (all authors), data collection (all authors), data analysis (A.K., M.U.), manuscript writing original version (A.K., P.S., M.U., H.P., T.R.), and final version (all authors).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical requirements
The study was conducted according to the Helsinki Declaration and approved by the Ethical committee of Kuopio University Hospital.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was obtained from all individual participants in the included the randomized trial.
