Abstract

Background
With an aging population, acute cholecystitis (AC) presents increasing challenges, particularly due to comorbidities and the use of anticoagulants that may limit surgical treatment options.1,2 The Swedish Registry of Gallstone Surgery (GallRiks) does not capture data on patients managed conservatively or with delayed surgery, creating a gap in understanding treatment strategies for AC. 3 The aim of this study was to evaluate patient-related factors associated with non-surgical management of AC in Dalarna County, Sweden, including age, sex, American Society of Anesthesiologists (ASA) classification, and the use of anticoagulants. Another aim was to assess the risk of readmission following non-surgical treatment.
Methods
This retrospective observational study follows the STROBE guidelines for cohort studies. 4 Patients admitted to Falun and Mora Hospital in 2022 were screened for eligibility. Inclusion criteria required a diagnosis of AC based on International Classification of Diseases: 10th Revision (ICD-10) codes (K80.0, K80.1, K80.4, K81.0, K81.1, K81.8, and K81.9). Exclusion criteria included acalculous cholecystitis, biliary pancreatitis, gallbladder malignancies, cancer of unknown origin, prior cholecystectomy, or the absence of a Swedish Social Security number.
Patients were categorized into three groups:
Immediate surgery group (ISG): Underwent surgery during the index admission.
Delayed surgery group (DSG): Underwent elective or acute surgery within 1 year after index admission.
Non-surgery group (NSG): No surgery within 1 year after index admission.
Anticoagulant treatment was defined as regular use of warfarin, heparin, or direct oral anticoagulants, including apixaban, rivaroxaban, or edoxaban. The ASA classification served as a surrogate measure of overall comorbidity.
Statistical analyses were conducted using SPSS (IBM SPSS Statistics for Windows, Version 25.0; IBM Corp., Armonk, NY, 2017), with the ISG serving as the reference group. Nominal variables were analyzed using the chi-square test, and ordinal or continuous variables using the Mann–Whitney U test, with statistical significance defined as p < 0.05.
Results
Of 452 patients assessed, 316 met the inclusion criteria: 228 (72.4 %) underwent immediate surgery (ISG), 35 (11.1 %) delayed surgery (DSG), and 53 (16.8 %) received non-surgical management (NSG). NSG patients were older (mean 79 vs 59 years in ISG, p < 0.001) and predominantly male (69.8 %, p = 0.002). ASA 2 was the most common classification overall (50.9 %), but ASA 3 was more frequent among NSG patients (71.7%) compared to ISG (22.4 %, p < 0.001). Anticoagulant use was more prevalent in DSG, 9 of 35 (25.7 %) and NSG 14 of 53 (26.4 %) compared to ISG, 15 of 228 (6.6 %) (p < 0.001) (Table 1).
Demographic and clinical characteristics of patients with acute cholecystitis.
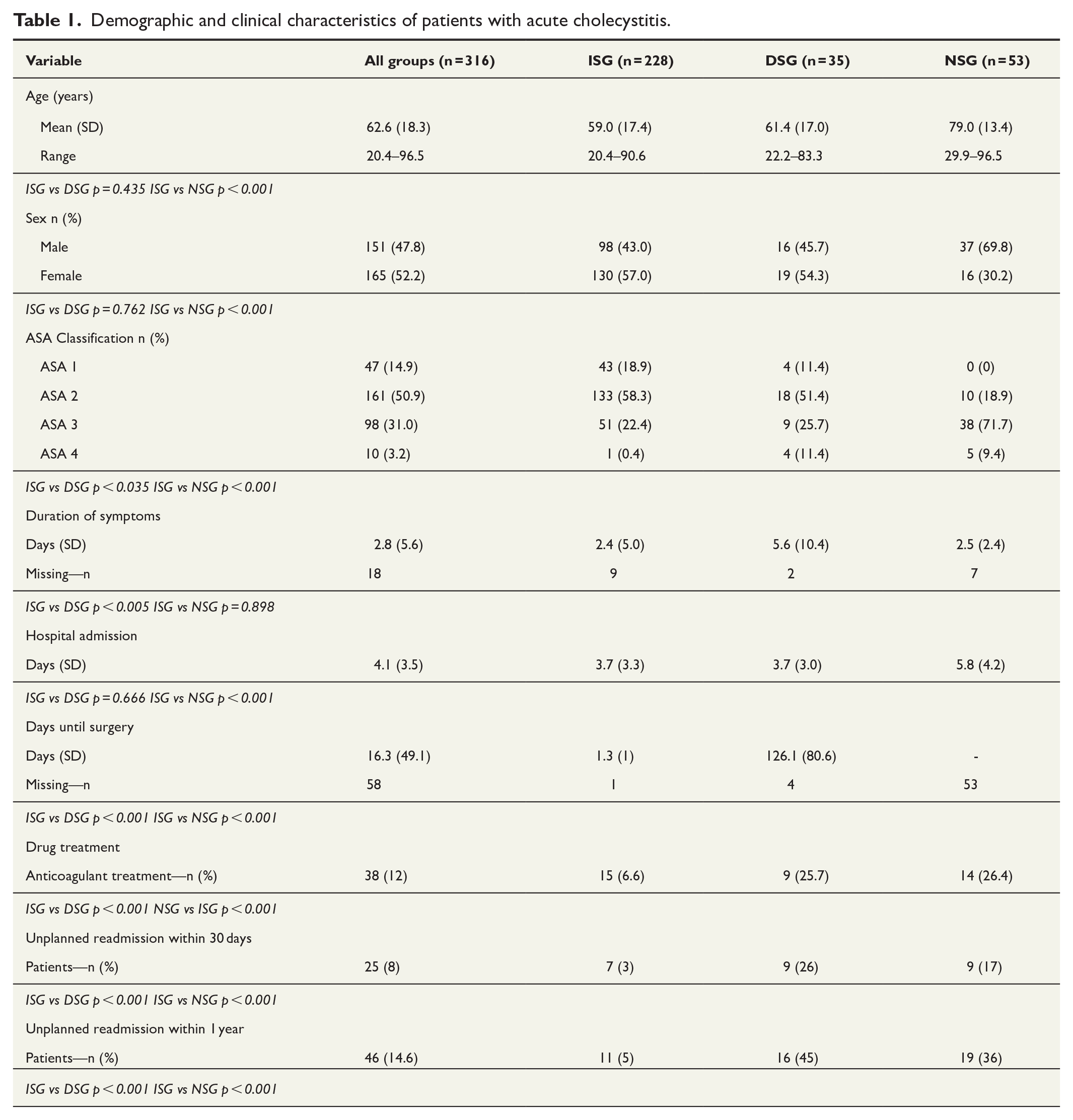
Patients in the ISG had the lowest risk of unplanned hospital readmission at 1 year (11 of 228 (5%)) within 1 year compared to DSG and NSG (16 of 35 (45%) and 19 of 53 (36%), respectively, ISG vs DSG p < 0.001 ISG vs NSG p < 0.001). Among DSG patients, 6 of 35 (17 %) were readmitted for a new episode of AC. The mean time from symptom onset to admission was 2.4 days in ISG versus 5.8 days in DSG (p < 0.005) (Table 1).
Discussion
Among 316 patients, 53 (16.8 %) received non-surgical treatment. These patients were typically older, male, had a higher ASA classification and were more frequently treated with anticoagulants. They also had higher rates of unplanned readmission at both 30 days and 1 year.
Our findings align with Feuerwerker et al, 5 who reported that AC patients on antithrombotic therapy were older and less likely to undergo early surgery. As antithrombotic therapy becomes more common, 6 non-surgical and delayed surgical approaches remain necessary alternatives when surgical risk is high.
While this study offers valuable insights into AC management patterns in Sweden, its generalizability is limited by its single-region design. The small size of the delayed and non-surgical groups also restricts the statistical power for subgroup analyses. Selection bias may have occurred due to the retrospective nature of data collection. When selecting patients admitted to hospital, those managed in elderly care settings or not admitted to hospital, may have been missed. Furthermore, anticoagulant use could confound age-related treatment decisions. Other factors, such as surgeons’ preference, patient preferences, or local access to operating theaters, may have influenced treatment decisions but could not be assessed in this retrospective study.
In summary, patient-related factors such as advanced age, higher ASA classification, and anticoagulant therapy are associated with non-surgical management of AC. As Sweden’s population continues to age, further research is needed to optimize treatment strategies and minimize readmission in this frail patient group.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.B. is an unpaid member of the leadership board of GallRiks and vice president of the Swedish Association for Emergency Surgery and Traumatology. The authors declare no other conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CKF—Center for clinical research in Falun provided the funding of admission for ethical review at the Swedish Ethical Review Authority (grant no. 91091, Region Dalarna).
Consent Statement
Ethical approval from the Swedish Ethical Review Authority (DNR: 2024-03365-01) was granted. Informed consent was waived due to the study’s retrospective design. Pseudonymised data and GDPR protocol was used. Declaration of Helsinki principles was applied.
Clinical Trial Statement
The study was prospectively registered on Researchweb.org (Nr 282534). However, due to its retrospective design, which relied on existing medical records, it was not registered in a clinical trial database such as ClinicalTrials.gov.
