Abstract
Background:
Soft tissue defects or periprosthetic infections after total knee arthroplasty (TKA) are severe complications that may lead to loss of the arthroplasty or the limb. Reconstructions with medial gastrocnemius flaps (MGF) are occasionally used to provide soft tissue coverage around the knee.
Aims:
The study aimed to establish the rate of implant survivorship after MGF reconstruction for soft tissue coverage in the treatment of exposed or infected TKA and to establish functional outcome.
Methods:
A retrospective analysis was performed on all patients who received soft tissue coverage with an MGF of an exposed or infected TKA between 2000 and 2017 at the Department of Hand and Plastic Surgery at Umeå University Hospital. The outcomes were implant survivorship and patient-reported outcome measures (PROMs) using the five-level EQ-5D version and The Knee Injury and Osteoarthritis Outcome Score.
Results:
Forty-seven patients (mean age = 67 years, 30 women) were included. The mean time between flap coverage and follow-up was 6.7 (±3.4) years. Implant survivorship was observed in 28 of 47 (59.6%) patients at follow-up. Flap failure was rare, with only 3 of 47 (6.4%) cases. Of the 20 patients who answered the PROMs, 10 of 20 experienced moderate to severe pain or discomfort.
Conclusions:
Due to unfavorable underlying conditions, MGF reconstruction after TKA is often associated with a compromised functional outcome. Because donor site morbidity is limited and flap failure is unusual, the procedure can be considered prophylactically in a small subset of patients with risk factors to prevent soft tissue defects and periprosthetic joint infection.
Context and relevance
Soft tissue defects after total knee arthroplasty are serious complications with the risk of periprosthetic infection, potentially leading to loss of the joint. Reconstruction with medial gastrocnemius flaps is occasionally used to provide soft tissue coverage. There are no current guidelines for performing a flap reconstruction other than in the presence of superimposed soft tissue. We demonstrated implant survivorship in 28 of 47 patients. However, of the 20 patients who responded to the patient-reported outcome measurements, 10 of 20 experienced moderate to severe pain or discomfort. Because donor site morbidity is limited and flap failure is uncommon, the procedure might be considered prophylactically in a small subset of patients with risk factors.
Introduction
Periprosthetic joint infection (PJI) and early wound complications necessitating surgical treatment after total knee arthroplasty (TKA) are challenging complications with a reported incidence of approximately 1.5% and 0.33%, respectively.1,2 For TKA with early surgical treatment of wound complications, the 2-year cumulative probability of major subsequent surgery (component resection, muscle flap coverage or amputation) is 5.3% and PJI 6.0%. 2 In contrast, for knees without early surgical intervention for the treatment of wound complications, the 2-year cumulative probabilities are 0.6% and 0.8%, respectively, 2 emphasizing the importance of obtaining primary wound healing after TKA.
TKA is vulnerable to soft tissue defects due to the superficial location of the arthroplasty, the need for early active motion, and the frequent occurrence of previous surgical incisions. 3 Soft tissue defects can have adverse effects, with postoperative wounds progressing to substantial skin necrosis and subsequent exposure to the arthroplasty that could lead to prosthesis revision, arthrodesis, or amputation of the limb. An exposed or infected TKA can be covered with a medial gastrocnemius flap (MGF), mediating wound closure without excessive tension and restoration of well-vascularized soft tissue, facilitating improved delivery of oxygen, systemic antibiotics, and immune modulators to the infected joint.3–5 The MGF is an alternative therapeutic option because of its surgical accessibility, substantial size, and mobility. 6 Moreover, the functional loss and comorbidity associated with the flap are generally limited owing to compensation provided by the remaining soleus and hemi-gastrocnemius muscle. 6
Although MGF is a reliable option for reconstructing an exposed or infected TKA, some patients experience persistent or recurrent infections, reflecting case complexity and not necessarily an issue with the flap.
This study aimed to establish the rate of implant survivorship after MGF reconstruction for soft tissue coverage in the treatment of exposed or infected TKA and establish the outcome of patients with implant survivorship using patient-reported outcome measures (PROMs).
Methods
Settings and location
A retrospective analysis was performed on all patients who received soft tissue coverage with an MGF of an exposed or infected TKA between 2000 and 2017 at the Department of Plastic Surgery at Umeå University Hospital. This hospital is a third-level university hospital with a catchment area of emergency care for about 160,000 inhabitants and a referral center for plastic and orthopedic surgery for approximately 900,000 inhabitants.
Patients and data collection
Some 49 patients were identified through patient medical records. Follow-up time is defined as the time from coverage with an MGF until death or the end of follow-up (March 2021).
Surgical procedure
The revision TKA was performed by a consultant orthopedic surgeon subspecialized in reconstructive surgery, and soft tissue reconstruction was conducted by a consultant plastic surgeon.
The DAIR (debridement, antibiotics, irrigation and implant retention) procedure was used for early (<6 weeks from primary TKA) PJI. The method includes removal of skin margins, excision of any sinuses, synovectomy, and exchange of removable implants (i.e. polyethylene insert). Patients undergoing two-stage exchange arthroplasty for PJI were first treated with implant resection, antibiotic-loaded cement spacer placement, and organism-specific intravenous antibiotic therapy along with wound management. The second stage involved delayed reimplantation and flap coverage. Microbiological testing was performed (conventional cultures/sonication of resected implants).
The medial gastrocnemius muscle was released from its distal attachment and brought anteriorly through a subcutaneous tunnel. The flap was sutured to surrounding soft tissue and covered with a split-thickness skin graft harvested from the contralateral anterolateral thigh. After MGF reconstruction, the knee was immobilized and kept in extension for 5–7 days to ensure skin graft survival. The flap was protected with an ointment compress, dry dressing, and a bolster dressing. The knee was then mobilized according to a special schedule depending on the maturation of the skin graft: 5-min motion three times a day for the first day, increasing with 5 min of movement every day until free motion after 1 week. The culture was performed during flap placement as sonication of the extracted prosthesis and tissue culturing, with an antibiotic-free interval before placement of the flap if clinically possible. However, in some cases, when flap placement was performed shortly after another surgical procedure, there was no antibiotic-free interval, resulting in negative cultures. The type and length of the postoperative antibiotic treatment depended on several factors, including the microbiological culture and the clinical and laboratory response.
Outcome measurements
The primary outcome measure was implant failure, defined as amputation, arthrodesis, persistent joint infection, or remaining spacer. The date for implant failure for patients with persistent joint infection was defined as the date for the last surgical procedure (DAIR in one case and secondary arthroplasty in the other case) and the date for implant failure for patients with remaining spacer was defined as the date when the decision not to perform a secondary arthroplasty was taken, where the decision not to perform additional surgery was based on the presence of extensive comorbidity. A secondary arthroplasty is defined as implantation of a knee prosthesis as part of a two-stage procedure, with extraction of the primary arthroplasty and implantation of an antibiotic-loaded cement spacer as the first procedure.
Patients with implant survivorship who were alive at the end of follow-up were asked to answer two PROMs: the five-level EQ-5D version (EQ-5D-5L) and the Knee Injury and Osteoarthritis Outcome Score (KOOS). The patients were contacted by mail, and all provided informed written consent.
The EQ-5D-5L is a validated non-disease-specific instrument to measure five dimensions of generic health-related quality of life (QOL): mobility, self-care, usual activity, pain/discomfort, and anxiety/depression.7,8 All items are divided into five response levels: 1, no; 2, mild; 3, moderate; 4, severe; and 5, extreme problems/unable. The EQ-VAS answers how good or bad an individual’s health is today on a scale from 0 to 100. The EQ-5D index is on a scale from −0.224 to 1, where 1 represents perfect health, 0 represents death, and values below zero represent “worse than death.” 9 The KOOS questionnaire is a validated instrument to measure patients’ opinions about their knees and associated problems. 10 The questionnaire contains five domains: (1) pain frequency and severity during functional activities, (2) symptoms such as the severity of knee stiffness and the presence of swelling and range of motion restriction, (3) difficulty experienced during activities of daily living (ADL), (4) difficulty experienced with sport and recreational activities, and (5) knee-related QOL. All items are rated on a 5-point Likert-type scale (0–4), after which the scores are transformed into a 0–100 scale, with lower scores indicating more extreme and troublesome symptoms.
Ethics
The study was performed according to the principles of the Declaration of Helsinki and the ethical guidelines of the Swedish research council. Ethical approval was obtained from the Regional Ethical Review Board in Umeå (Dnr 2018/289-31).
Statistics
Patient characteristics and frequencies of events were summarized using descriptive statistics. The limited sample, not reaching at least 10 events per variable required to obtain an acceptable regression coefficient, did not allow us to perform any regression analysis. 11 Implant survivorship was illustrated by a Kaplan-Meier survival curve. Potential risk factors for implant failure were analyzed with independent sample t test for continuous variables and Pearson chi-square test for categorical variables. The EQ-VAS was compared with a population reference data for Sweden 12 using one sample t test. Test results with p values of less than 0.05 were considered statistically significant. The statistical analyses were performed using SPSS (IBM SPSS Statistics for Macintosh, Version 22.0, Armonk, NY: IBM Corp. USA).
Results
Study population
A total of 49 patients received soft tissue coverage with an MGF of an exposed or infected TKA during the study period. Of these 49 patients, 2 were excluded (1 due to lack of information in the medical records of performed surgeries and 1 due to perioperative death). The mean age for MGF reconstruction was 67.3 (±11.16) years, and the mean time between flap coverage and follow-up was 6.7 (±3.4) years. Twelve patients were dead at follow-up, with the 1-year mortality following flap surgery being 2 of 47 (4.3%). Of 47 flaps, 33 (70.2%) were performed at extraction of the arthroplasty and implantation of an antibiotic loaded cement spacer, 11 (23.4%) at DAIR, and 3 (6.4%) at superficial debridement. The decision on when to place the flap during the treatment process was based on when the soft tissues were deemed insufficient and not on the belief that placement at one stage was more advantageous. Flap placement was performed after one revision in 22 (46.8%) cases, after two in 16 (34.0%) cases, and after three in 9 (19.1%) cases. Thus, flap placement after one revision means flap placement at the time for extraction of the arthroplasty and implantation of an antibiotic loaded cement spacer, at DAIR, or at superficial debridement, while flap placement after two or more revision means that one respectively two or more revisions have been performed prior to the flap surgery. Demographics and treatment modalities are presented in Table 1.
Clinical characteristics and surgical history.
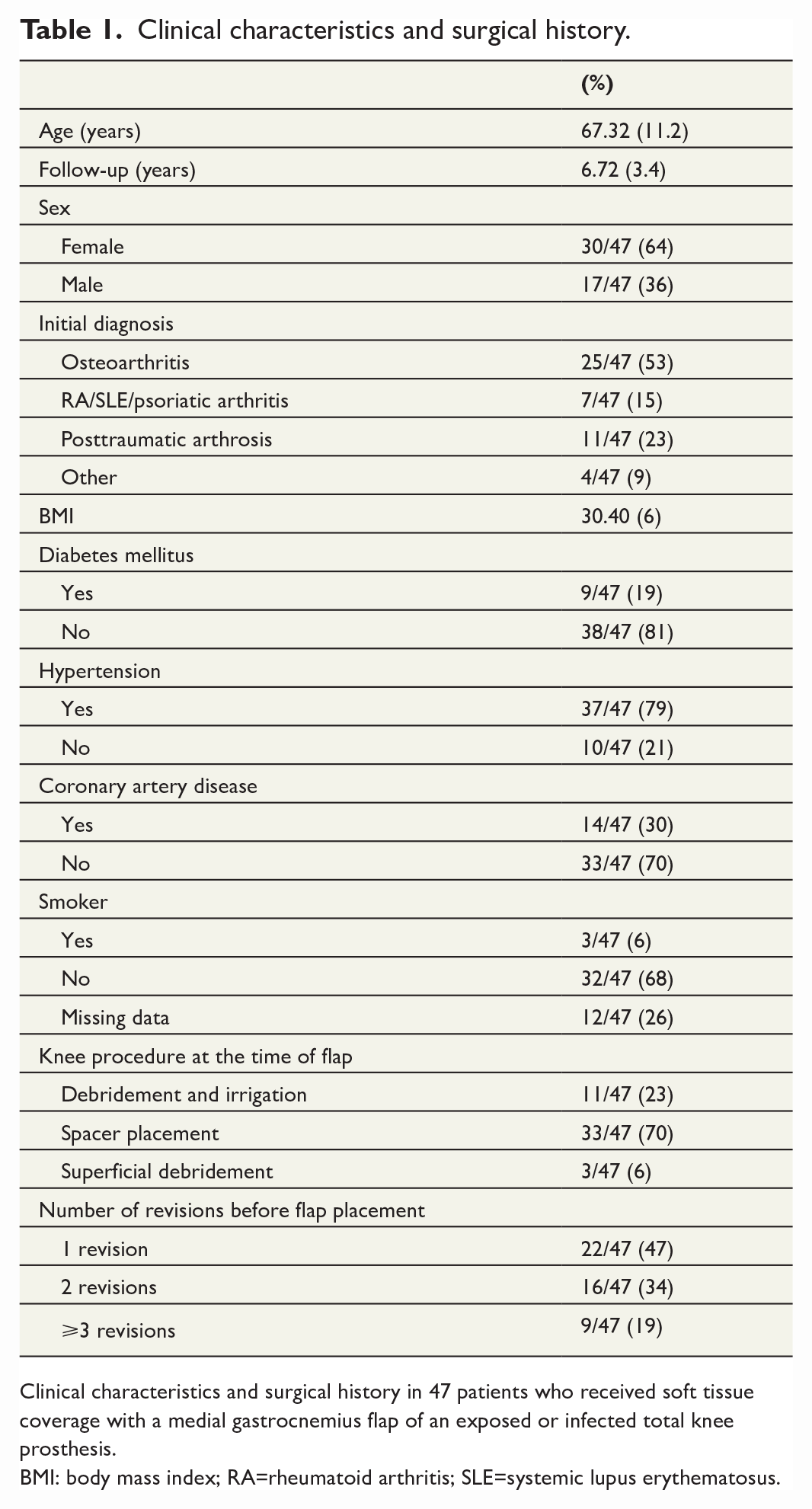
Clinical characteristics and surgical history in 47 patients who received soft tissue coverage with a medial gastrocnemius flap of an exposed or infected total knee prosthesis.
BMI: body mass index; RA=rheumatoid arthritis; SLE=systemic lupus erythematosus.
Implant survival
Implant survivorship was observed in 28 of 47 (59.6%) patients at follow-up: 9 (19.1%) had their primary TKA remaining and 19 (40.4%) had a secondary arthroplasty. Implant failure was observed in 19 of 47 (40.4%) patients at follow-up: 11 (23.4%) underwent amputation, 3 (6.4%) had an arthrodesis, 2 (4.3%) had a persistent or recurrent infection, and 3 (6.4) had a remaining spacer. Implant survivorship is illustrated by a Kaplan-Meier survival curve in Fig. 1. Flap failure was rare, with only 3 of 47 (6.4%) cases. Out of the three cases with flap failure, two patients underwent amputation and one received a latissimus dorsi flap and had the primary TKA remaining at follow-up.
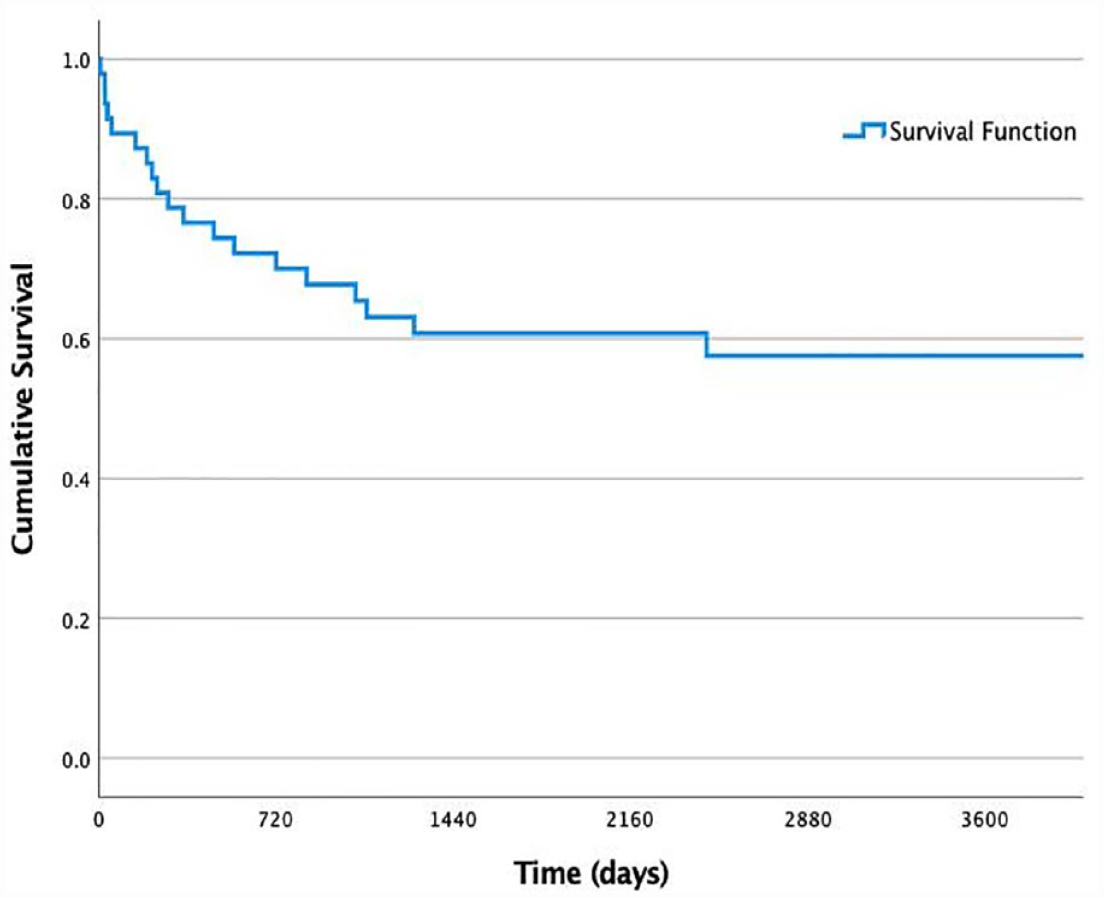
Kaplan-Meier survival curve for illustration of implant survival (in days) after flap surgery.
Factors associated with implant failure
Several potential risk factors for implant failure were identified from the literature and presented as patients with implant survivorship (n = 28) and patients with implant failure (n = 19) (Table 2). The number of patients in each group can differ due to missing data on the specific factor of interest. Since the treatment differed between the patients, regarding both the number of revisions before the flap placement and the length of the prior antibiotic treatment, with some patients not having an antibiotic-free interval before the placement of the flap, the microbiological data are presented as presence or absence of polyflora in the peroperative culture and not analyzed more in detail. Of the analyzed risk factors, only body mass index (BMI) was significantly associated with implant failure (32.1 and 27.9 in implant survival vs implant failure respectively (p = 0.020); however, the analysis was unadjusted.
Factors associated with implant failure.
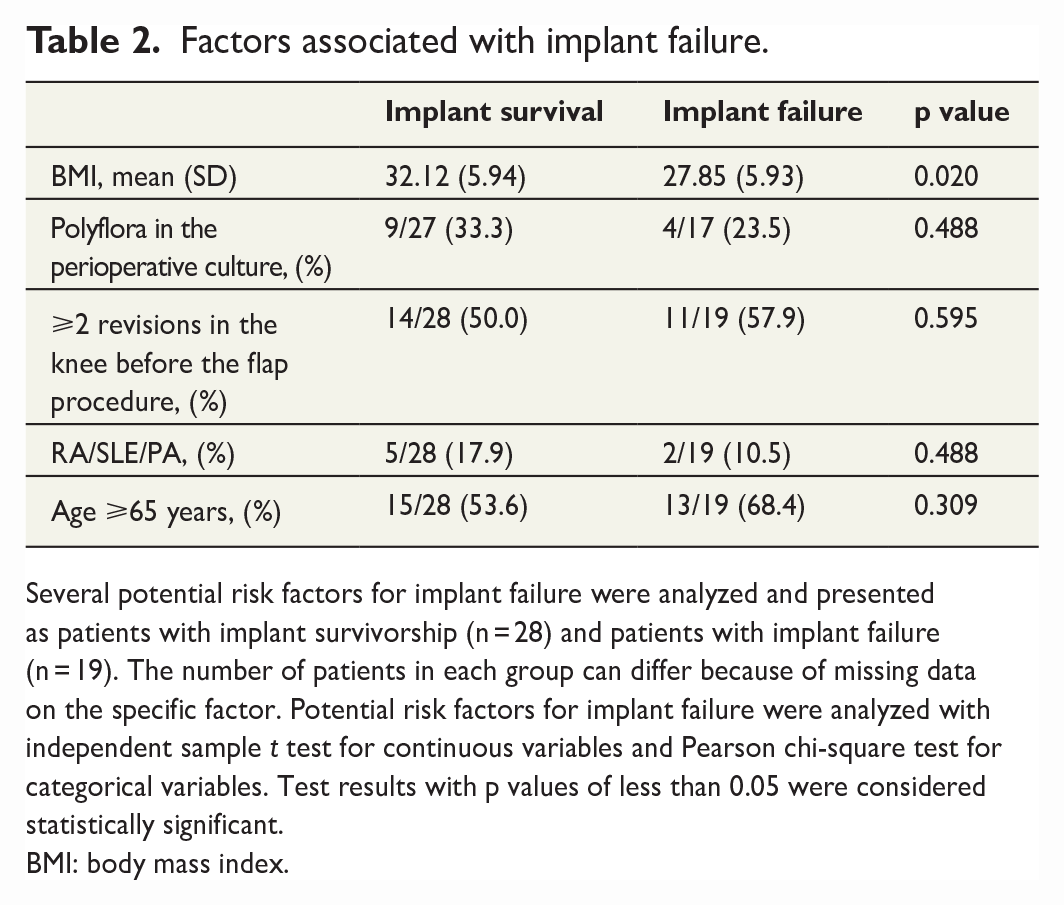
Several potential risk factors for implant failure were analyzed and presented as patients with implant survivorship (n = 28) and patients with implant failure (n = 19). The number of patients in each group can differ because of missing data on the specific factor. Potential risk factors for implant failure were analyzed with independent sample t test for continuous variables and Pearson chi-square test for categorical variables. Test results with p values of less than 0.05 were considered statistically significant.
BMI: body mass index.
Patient-reported outcome measures
PROMs (EQ-5D and KOOS) were used to describe the functional outcome of patients with implant survivorship. Of 26 alive patients with implant survivorship, 20 answered, yielding a response rate of 76.9%.
For EQ-5D mobility, 12 of 20 patients experienced no or mild problems walking, while 7 of 20 experienced moderate to severe problems and 1 could not walk. For EQ-5D self-care, most patients (15 of 20) experienced no or mild problems with washing or dressing and 5 of 20 experienced moderate to severe problems. For EQ-5D usual activities (e.g. housework and family activities), 12 of 20 patients experienced no or mild problems, 6 of 20 experienced moderate to severe problems, and 2 of 20 could not perform the activities. Regarding pain and discomfort, 10 of 20 experienced no to mild problems and 10 of 20 experienced moderate to severe problems. The data are summarized in Table 3. When answering “how good or bad the health is today,” the mean value of the EQ-VAS was 62.63 (± 26.00), which was significantly lower than the population reference data for Sweden (62.63 vs 76.6; p = 0.031). The mean EQ-5D index value was 0.64 (± 0.28), with only 1 of 20 (5) scoring less than zero.
EQ-5D-5L health profile.

The mean value of KOOS-pain was 65.45 (± 29.14); the mean value of KOOS-sport was 23.75 (± 25.69); the mean value of KOOS-ADL was 57.20 (± 28.71); the mean value of KOOS-QOL was 42.00 (± 24.66); and the mean value of KOOS-symptom was 63.05 (± 24.07). Distribution of KOOS is presented in a histogram in Supplementary Fig. 1.
Discussion
Reconstruction with MGF as part of treating an exposed or infected TKA is often associated with a high number of implant failures and a compromised functional outcome, inferring the case complexity of the patient population.
Because the donor site morbidity is limited and flap failure is uncommon, the procedure might be considered preventive in a small subset of patients with high risk to avoid subsequent soft tissue defects and risk of PJI.
After MGF coverage of an exposed or infected TKA, we demonstrated implant survivorship in 59.6% of our patient cohort. Of the remaining patients, 23% had undergone an above-knee amputation, 6% had an arthrodesis performed, 4% had a persistent or recurrent infection, and 6% had a remaining spacer. Flap failure was rare, observed in only three patients. The reported implant survivorship varies between 48% and 92%,4,13,14 with most studies based of small heterogeneous cohorts. Corten et al., 14 analyzing 24 patients with an infected TKA reconstructed with an MGF, showed that 92% of the knees were successfully reconstructed. In contrast, in a study with 31 patients, Tetreault et al. 15 showed a higher failure rate, with 52% of patients having a persistent or recurrent infection and a 48% prosthesis retention rate at a 4-year follow-up. Houdek et al. 16 included 83 patients and found a 68% revision-free survival rate and a 79% amputation-free survival rate at 10 years of follow-up. Kwiecien et al. 17 reported that a functional joint was preserved in 31 (54%) of 58 patients. Recurrent periprosthetic infection and amputation were common (58% and 25%, respectively). Moreover, the implant survival rates decreased with time, with the overall 2, 5, 10, and 15 years after MGF coverage being 84%, 77%, 68%, and 68%, respectively. 16 The high conversion rate to arthrodesis or amputation most likely relates to the complexity of populations eligible for MGF. Although the retention rate varies among different studies, flap-related complications are rare.3,13
No current guidelines exist on indications to perform an MGF reconstruction other than in the presence of superimposed soft tissue. 17 However, we propose that a flap reconstruction should not only be considered when the soft tissue is deemed insufficient, but also in the case of PJI without an exposed TKA, as restoration of well-vascularized soft tissue facilitates delivery of oxygen, systemic antibiotics and immune modulators to the infected joint, potentially improving the outcome. In the present study flap reconstruction was performed most often with the insertion of a spacer (70%), followed by a DAIR (23%). Successful results have been reported with MGF reconstruction as part of the first stage (infection extraction of the arthroplasty and implantation of an antibiotic spacer) 14 or as part of the second stage (reimplantation).14,18 Studies performed on larger samples suggest delaying flap coverage until after infection treatment may be advisable if the soft tissue is amenable to closure at resection or spacer exchange without excessive tension. 15 However, it is plausible that patients who underwent flap coverage at the time of spacer placement had more severe soft tissue defects, contributing to poorer outcomes. An advantage of the latter approach is that a re-elevation of the flap during subsequent implant revision can be avoided. On the other hand, the performance of flap coverage at the time of explanation allows unrestricted rehabilitation following later second-stage reimplantation. Comparing patients with pre-existing soft tissue defects who require flap reconstruction with patients with no pre-existing soft tissue defects but with extensive debridement during revision TKA requiring immediate proactive flap coverage, patients with pre-existing soft tissue defects have a higher rate of implant reinfection (58% versus 27%) and amputations (25% versus 0%), with a functional joint preserved in 54% and 80% of the cases, respectively. 16 These results underscore the need to consider early MGF reconstruction to treat an infected TKA.19,20
Several risk factors have been suggested to indicate poor outcome. However, due to the rarity and complexity of these situations, most studies are of heterogeneous patient populations with small cohorts, insufficient for stratified analysis. We showed that BMI was significantly associated with implant failure; however, the analysis was unadjusted. Tobacco use, diabetes mellitus, peripheral vascular disease, and growth of multiple organisms have been proposed to be associated with poor outcome and could be included in the optimization before undertaking the surgical procedure.15,16,21 In a large cohort study, 16 the risk of implant failure was increased by morbid obesity, five and more prior surgical procedures on the knee and the risk of amputation was increased in patients aged ⩾65 years, and a wound size >50 cm2.
Despite the limited number of patients with PROMs, we found that half of the patients experienced moderate to severe pain or discomfort. The mean EQ-VAS was 62.6 compared with 58.3 in patients on the waiting list for knee arthroplasty and 69.8 in a matched control group. 22 Our findings are in concordance with several studies14–16 showing that even patients with successful soft tissue reconstruction experience limitations in physical activities. Three studies with patients undergoing TKA with soft tissue defects reconstructed with MGF reported an overall poor functional outcome.14–16,23
Our study has limitations inherent to the retrospective study design, and further limitations are the heterogeneous patient selection and inadequate sample size to detect factors significantly associated with implant failure in adjusted analysis. Notwithstanding these limitations, this study adds data on complex revision TKA with compromised soft tissues and presents MGF as a valid option.
Conclusion
Reconstruction with MGF as part of the treatment of an exposed or infected TKA is often associated with a high number of implant failures and a compromised functional outcome, reflecting the case complexity of the patient population. Because donor site morbidity is limited and flap failure occurs rarely, the procedure can be considered preventive treatment in a small subset of patients with high risk to avoid subsequent soft tissue defects and risk of PJI.
Supplemental Material
sj-tiff-1-sjs-10.1177_14574969231175562 – Supplemental material for Outcome following soft tissue coverage with a medial gastrocnemius flap of an exposed or infected total knee arthroplasty
Supplemental material, sj-tiff-1-sjs-10.1177_14574969231175562 for Outcome following soft tissue coverage with a medial gastrocnemius flap of an exposed or infected total knee arthroplasty by Rebecca Wiberg, Camilla Mukka, Olof Backman, Göran Stålhult, David Edmundsson and Sebastian Mukka in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
The authors thank all participating patients for contributing to the present study.
Author contributions
RW and SM designed the study. RW drafted the initial manuscript and performed the statistical analysis. SM performed survival analysis. All authors analyzed and interpreted the data and revised and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SM declares lecture fees from DePuy Johnson & Johnson, but not related to the present study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
