Abstract
Background and objective:
Acute mesenteric ischemia (AMI) has a high mortality rate due to the development of bowel necrosis. Patients are often ruled outside active care if a large proportion of small bowel is necrotic. With the development of treatment for short bowel syndrome (SBS) and intestinal transplantation methods, long-term survival is possible even after extensive small bowel resections. This study aims to assess the incidence of SBS and potentially suitable candidates for intestinal transplantation among patients treated for AMI.
Methods:
This population-based retrospective study comprised patients aged less than 70 years and diagnosed with AMI between January 2006 and October 2020 in Helsinki and Uusimaa health care district, Finland.
Results:
Altogether, AMI was diagnosed in 711 patients, of whom 133 (19%) were aged below 70. An intervention was performed in 110 (83%) patients. Of these 133 patients, 16 (12%) were ruled outside active treatment due to extensive small bowel necrosis at exploratory laparotomy, of whom 6 (5%) were potentially suitable for intestinal transplantation. Two patients were considered as potential candidates for intestinal transplantation at bowel resection but died of AMI. Nine (7%) patients needed parenteral nutrition after resection, and two of them (2%) developed SBS. Only one patient needed long-term parenteral nutrition after hospital discharge. This patient remained dependent on parenteral nutrition but died before evaluation of intestinal transplantation could be carried out while the other patient was able to return to enteral nutrition.
Conclusions:
A small number of patients with AMI below 70 years of age are potentially eligible for intestinal transplantation.
Keywords
Summary of the study (for Twitter)
Patients with acute mesenteric ischemia are ruled outside active care if a large portion of the bowel is necrotic. In this study, 16 patients (12%) were ruled outside active care, of whom 6 were retrospectively deemed suitable for intestinal transplantation.
Context and relevance
Patients with acute mesenteric ischemia (AMI) are usually ruled outside active care if a large proportion of small bowel is necrotic, but with the development of treatment for short bowel syndrome (SBS) and intestinal transplantation methods, even extensive bowel resections should be considered on selective patients. In this study, analyzing 133 AMI patients below the age of 70 years, 16 (12%) patients were ruled outside active treatment due to extensive bowel necrosis, 6 (5%) of whom were retrospectively evaluated as potentially suitable for intestinal transplantation. Only two (2%) patients developed SBS after successful treatment of AMI.
Introduction
Acute mesenteric ischemia (AMI) is a surgical emergency with mortality rates ranging from 50% to 69% despite advances in diagnosis and treatment.1–3 However, mortality rates as low as 25% have been reported after the implementation of treatment pathways and enhanced use of modern technologies. 4 Long-term data have indicated that successfully treated patients have a very low risk of recurrence and often attain satisfactory recovery, although compared with the background population, mortality is still increased due to cardiovascular comorbidities.5–8
Survival from AMI is largely dependent on the extent of the necrotic bowel. Traditionally, patients are often ruled outside active care if development of short bowel syndrome (SBS) with intestinal failure (SBS-IF) and consequent dependency on parenteral nutrition (PN) seem inevitable. If ileum and colon are left intact, an adult can maintain enteral autonomy with up to a 50% small bowel resection, but dependency on long-term PN increases markedly if the length of the remaining small bowel is less than 100 cm. 9 However, quality of life (QoL) of patients dependent on long-term PN (for varying etiological reasons) is relatively good.10–12 Varying incidences of SBS after treatment for AMI have been reported, ranging from 14% to 87%.5,13–17
SBS-IF is primarily treated with PN, medication, and corrective surgery. If these prove insufficient or lead to serious complications, intestinal transplantation may be considered. With the advancement in surgical techniques, perioperative transplant management, and immune-induction therapies, intestinal transplantation has become an increasingly safe treatment option for SBS-IF,18–21 also after AMI. 22 Transplantation rates have, however, remained low. In 2020, 157 intestinal transplantations were performed in the whole world. 23 Since the beginning of the Finnish intestinal transplantation program in 2009, eight patients have received intestinal transplantation in Finland and none for AMI. 24 It is unclear whether this reflects differences in patient material or a lack of recognition of intestinal transplantation as a treatment option for patients with AMI.
This study seeks to answer the following three questions:
How many patients with AMI are deemed unfit for active treatment due to extensive necrotic bowel in the Southern Finland area?
How many of these patients would have potentially been eligible for intestinal transplantation?
How many patients develop SBS after successful treatment of AMI?
Methods
Study district
This retrospective cohort study included patients diagnosed with AMI in Helsinki and Uusimaa health care district hospitals (HUS) between January 2006 and October 2020. The district serves a population of approximately 1.6 million in a defined geographical area in Southern Finland and includes 1 tertiary and 18 secondary referral hospitals with approximately 2500 hospital beds. These government-based hospitals treat all referral patients within the area, and private referral hospitals are non-existent. The population living in the study district in 2006 was 1,463,385 (52% women, 13% aged 65 years or over, and 3% aged 80 years or over) and 1,698,974 (51% women, 18% aged 65 years or over, and 4% aged 80 years or over) in 2020. The population of Finland in 2006 was 5,276,955 (51% women, 16% aged 65 years or over, and 4% aged 80 years or over) and 5,533,793 (51% women, 23% aged 65 years or over, and 6% aged 80 years or over) in 2020. Mean population in HUS district during 2006–2020 was 1,581,701 and the mean population of Finland during the same period was 5,430,811.
Patient cohort
Patients diagnosed with AMI in the district were identified from electronic patient records by conducting a search for the International Classification of Diseases 10 (ICD-10) code K55.0 (vascular disorders of intestine) and Nomesco Classification of Surgical Procedures (NCSP) codes for procedures on mesenteric vessels (PCE17, PCF16, PCF17, PCHXX, PCJ17, PCN16, PCN17, PCP16, PCP17, PCQ16, PCQ17, and PCQ99). AMI was defined as ischemia caused by a severe decrease in blood flow to areas of the small intestine. Patients who died from AMI were identified from death certificates acquired from Statistics Finland with a search for the ICD-10 code K55.0. Patients were considered to have AMI if they were diagnosed alive in surgery or by using imaging studies or if they were diagnosed in autopsy.Exclusion criteria were ischemia caused by trauma or strangulation, permanent residence outside the HUS district, a clinical diagnosis (not confirmed in imaging, surgery, or autopsy), chronic colonic ischemia, chronic ischemia, and ischemia isolated to the colon. The institutional review board of HUS approved the study. Patient records were analyzed, and data regarding patient characteristics, treatment, and outcome were manually extracted. Last date of follow-up was defined as the date of last note in the electronic patient records (which all the hospitals in the area use). The patient records were reviewed up to 1 April 2021.
Management of AMI
In AMI patients in the HUS area, primary anastomoses are performed either in primary operation or after second-look laparotomy (if the vitality of bowel is in question or the patient is unstable). Stomas have been used rarely, mainly in patients with peritonitis and septic shock. A pathway and care bundle for AMI patients was introduced in May 2018, which has improved diagnostics and outcomes. 4 Although no changes were made to the abovementioned strategy, one of the recommendations was to keep the threshold for using damage-control strategy (with deferred anastomosis) low.
Definitions
SBS
SBS describes a malabsorption disorder caused by a loss of bowel mass due to surgical resection, congenital defects, or disease. 25
Potential recipients of intestinal transplantation
Contraindications for intestinal transplantation are similar to those for other solid organ grafts. These include systemic and untreated local infections, non-resectable malignancies, severe neurological dysfunction, major psychiatric illness, severe immunodeficiency, non-compliance, and severe cardiac or pulmonary comorbidities, or both. 26 The criteria for intestinal transplantation in our unit follow the criteria listed here. In this study, we applied a rather strict age limit to identify only patients with a high likelihood of qualifying for transplantation. Therefore, patients aged 70 years or older were not included in the analyses.
Statistics
We summarized demographics and other characteristics of the study population with descriptive statistics, including median and interquartile range (IQR) for continuous variables and frequencies and percentages for categorical variables. All statistical analyses were done using IBM SPSS Statistics version 27 (IBM Corp©, Armonk, NY, USA). As continuous variables were not normally distributed, we used Mann–Whitney U test to compare distributions of two groups. Chi-square test was used to test for independence of two categorical variables, or Fisher’s exact test when there were five or less observations per cell. The Z-test was used to compare two proportions.
Results
Patient characteristics
The initial extraction yielded 1691 patients. After applying exclusion criteria, 711 patients were diagnosed with AMI during the study period. Of these 711 patients, 133 were below the age of 70 years and formed the final study cohort (Fig. 1). Baseline characteristics of these patients are shown in Table 1. Briefly, the median age was 63 years with more women (n = 76, 57%) than men. Most had some comorbidity (n = 114, 86%), the most common of which was hypertension (n = 59, 44%), followed by diabetes (n = 39, 29%) and peripheral arterial disease (n = 39, 29%). The majority were considered functionally independent (n = 125, 94%) at baseline.
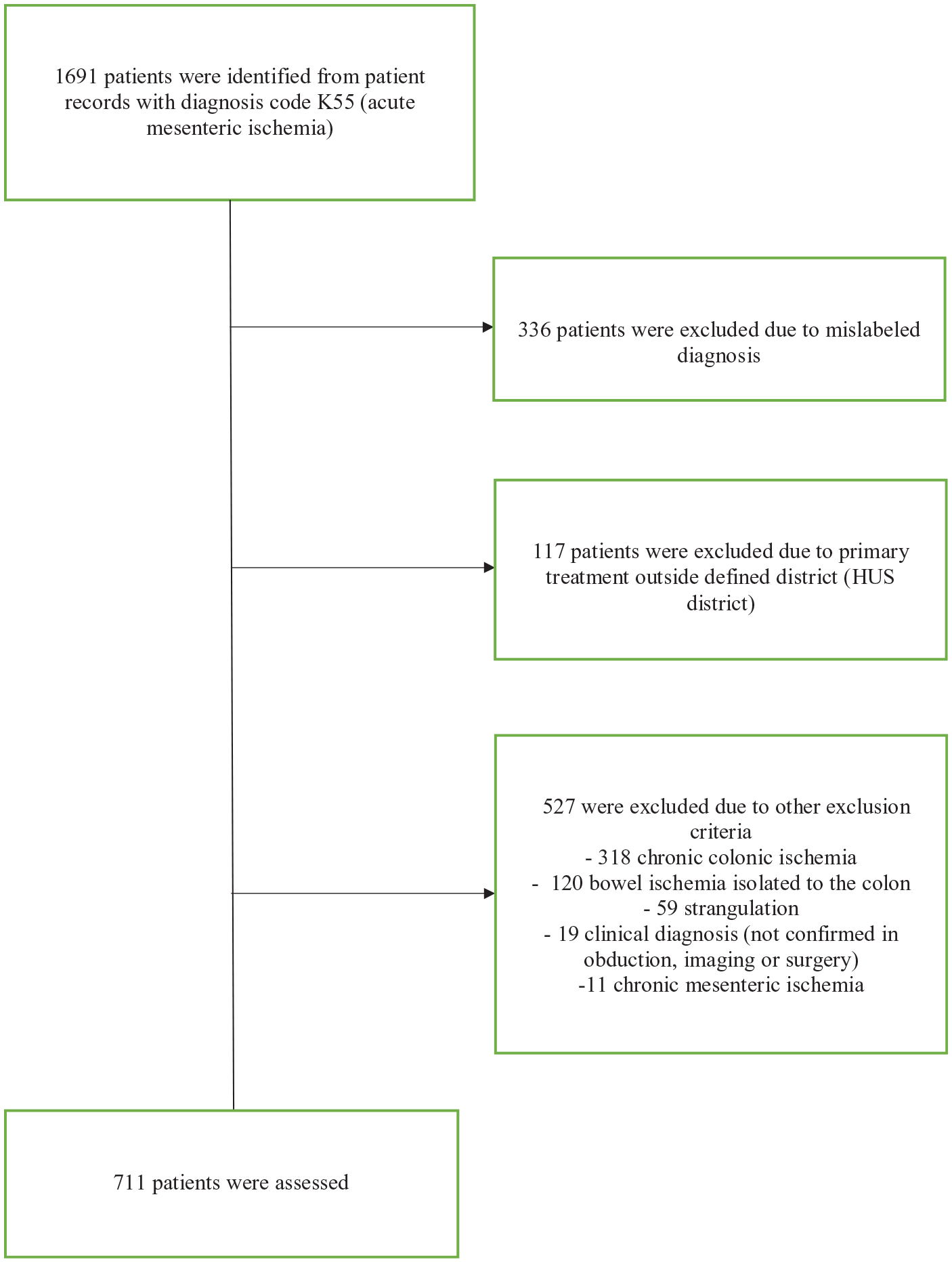
Flowchart of patient selection.
Basic demographics, comorbidities, medications, and functional status of the 133 patients below 70 years treated for acute mesenterial ischemia, n (%).
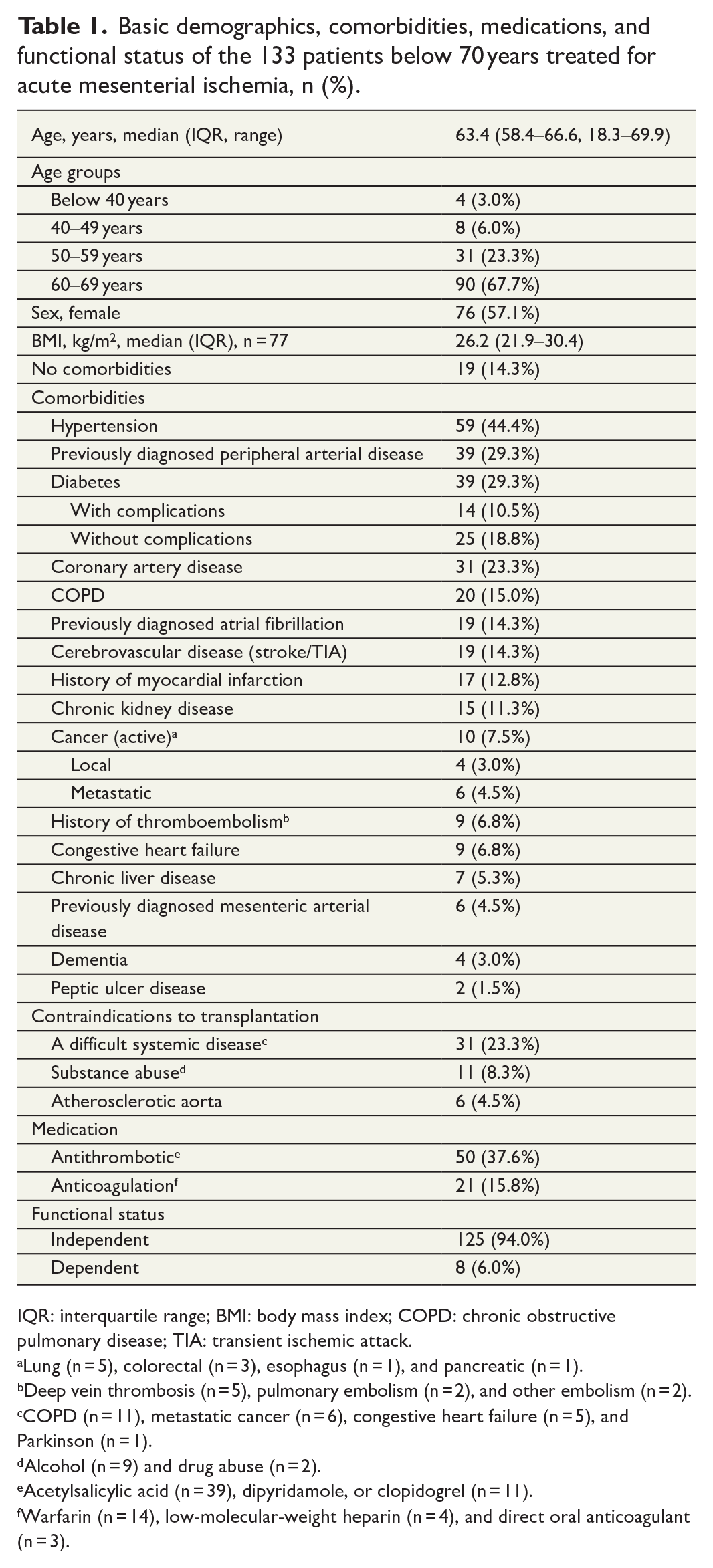
IQR: interquartile range; BMI: body mass index; COPD: chronic obstructive pulmonary disease; TIA: transient ischemic attack.
Lung (n = 5), colorectal (n = 3), esophagus (n = 1), and pancreatic (n = 1).
Deep vein thrombosis (n = 5), pulmonary embolism (n = 2), and other embolism (n = 2).
COPD (n = 11), metastatic cancer (n = 6), congestive heart failure (n = 5), and Parkinson (n = 1).
Alcohol (n = 9) and drug abuse (n = 2).
Acetylsalicylic acid (n = 39), dipyridamole, or clopidogrel (n = 11).
Warfarin (n = 14), low-molecular-weight heparin (n = 4), and direct oral anticoagulant (n = 3).
Diagnostics, treatment, and outcome
The majority (n = 119, 89%) of patients were diagnosed alive, 81 (61%) by imaging and 38 (28%) in surgery. The most common etiology was obstruction of the superior mesenteric artery (SMA) due to embolism (n = 83, 62%), followed by SMA obstruction due to thrombosis (n = 18, 14%) and non-obstructive mesenteric ischemia (NOMI) (n = 16, 12%). The rest of the patients had venous thrombosis (n = 8, 6%) or the etiology was unclear (n = 8, 6%).
Eight patients (6%) were deemed unfit for surgery, most commonly due to multiple comorbidities (n = 5). One patient died after imaging, 14 were not diagnosed alive (11%), and the rest underwent an intervention (n = 110, 83%). Sixteen (12%) patients were ruled outside active care during laparotomy due to the extensive bowel necrosis and no bowel resection was performed in these patients. The rest of the patients had bowel resections (n = 40), revascularization (n = 18), or both (n = 36).
Revascularization was performed in 57 (43%) patients, using endovascular approach in 21 (16%) and open approach in 36 (27%) patients. Laparotomy was performed in 105 (79%) patients and bowel resection was performed in 77 (58%) patients. The rate of bowel anastomosis leakage was 10% (n = 8). A stoma was performed in a total of 20 patients (15%), with one patient (1%) receiving a jejunostomy. The median length of resection was 80 cm (IQR = 30–150 cm, range = 10–250 cm). Open abdomen was applied to 36 (26%) patients. Thirty-day mortality was 42% (n = 56) and 90-day mortality was 47% (n = 62). All deaths within 90 days were caused by AMI. One-year mortality was 51% (n = 68) with two non-AMI-related deaths and 2-year mortality was 53% (n = 71) with four non-AMI-related deaths.
Parenteral nutrition
Nine (7%) patients needed PN after bowel resection, and one of them died of AMI 30 days after initial laparotomy. Seven patients were able to return to full enteral nutrition and their median time on PN was 23 (IQR = 7–84, range = 7–145) days. Only one of these patients received PN after hospital discharge but was not fully dependent on PN at this point. In these patients, the median length of the resected small bowel was 65 cm (IQR = 43–150 cm, range = 40–240 cm). In addition, a right-sided hemicolectomy was performed in three patients. One patient needed treatment for SBS after returning to enteral nutrition. The patient received nutritional counseling, was regularly monitored with blood tests, and took medication for diarrhea and vitamin supplements. The length of the resected small bowel in this patient was 240 cm, leaving an estimated 90 cm of small bowel and intact colon in continuity.
One patient remained PN-dependent and had a long-term central venous catheter inserted for PN. Intestinal transplantation was considered as a future treatment option during surgery, and complete resection of jejunum and ileum was carried out. The patient died approximately 8 months after the initial operation due to duodenal necrosis (Table 2). The patient’s suitability or need for transplantation was not further evaluated before death due to postoperative complications.
Characteristics of two patients for whom intestinal transplantation was considered as a treatment option in the acute setting.
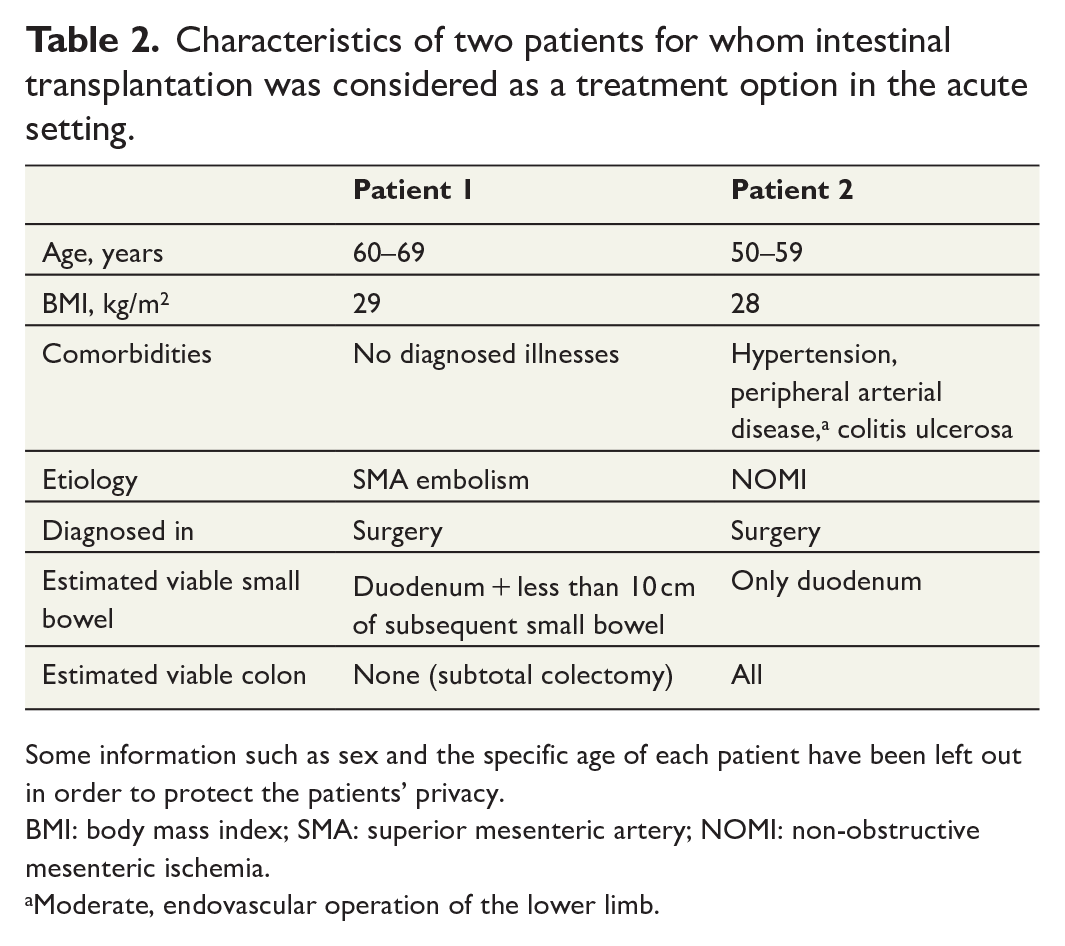
Some information such as sex and the specific age of each patient have been left out in order to protect the patients’ privacy.
BMI: body mass index; SMA: superior mesenteric artery; NOMI: non-obstructive mesenteric ischemia.
Moderate, endovascular operation of the lower limb.
Intestinal transplantation was also initially considered in another patient. The patient had all but 10 cm of her small bowel resected with possible future transplantation in mind, but the patient died 3 days after surgery (Table 2).
Therefore, the incidence of SBS was 2% in the total cohort and 3% in those who survived AMI.
Intestinal transplantation
We analyzed in detail the 16 patients who were ruled outside active care during laparotomy due to extensive bowel necrosis. Intestinal transplantation was not considered as a treatment option in any of these patients. No significant differences were found between these 16 and the rest of the patients with regard to patient demographics, comorbidities, medications, or etiology. A significantly larger proportion of these patients were diagnosed in surgery (26% vs 50%, p value = 0.047).
To assess which of these 16 patients would have been suitable candidates for intestinal transplantation, we excluded those not independent at baseline (n = 2) and those with an active malignancy (n = 2). The rest of the patients were evaluated individually, and six more patients were deemed unfit for transplantation due to severe systemic illnesses or substance abuse (Fig. 2, Table 3). In the end, six (5%) patients were retrospectively deemed potentially suitable for transplantation (Table 4). We can therefore estimate that 0.26 patients per year per million population and 1.44 patients per year in Finland have extensive small bowel necrosis due to AMI and could be potentially eligible for intestinal transplantation.
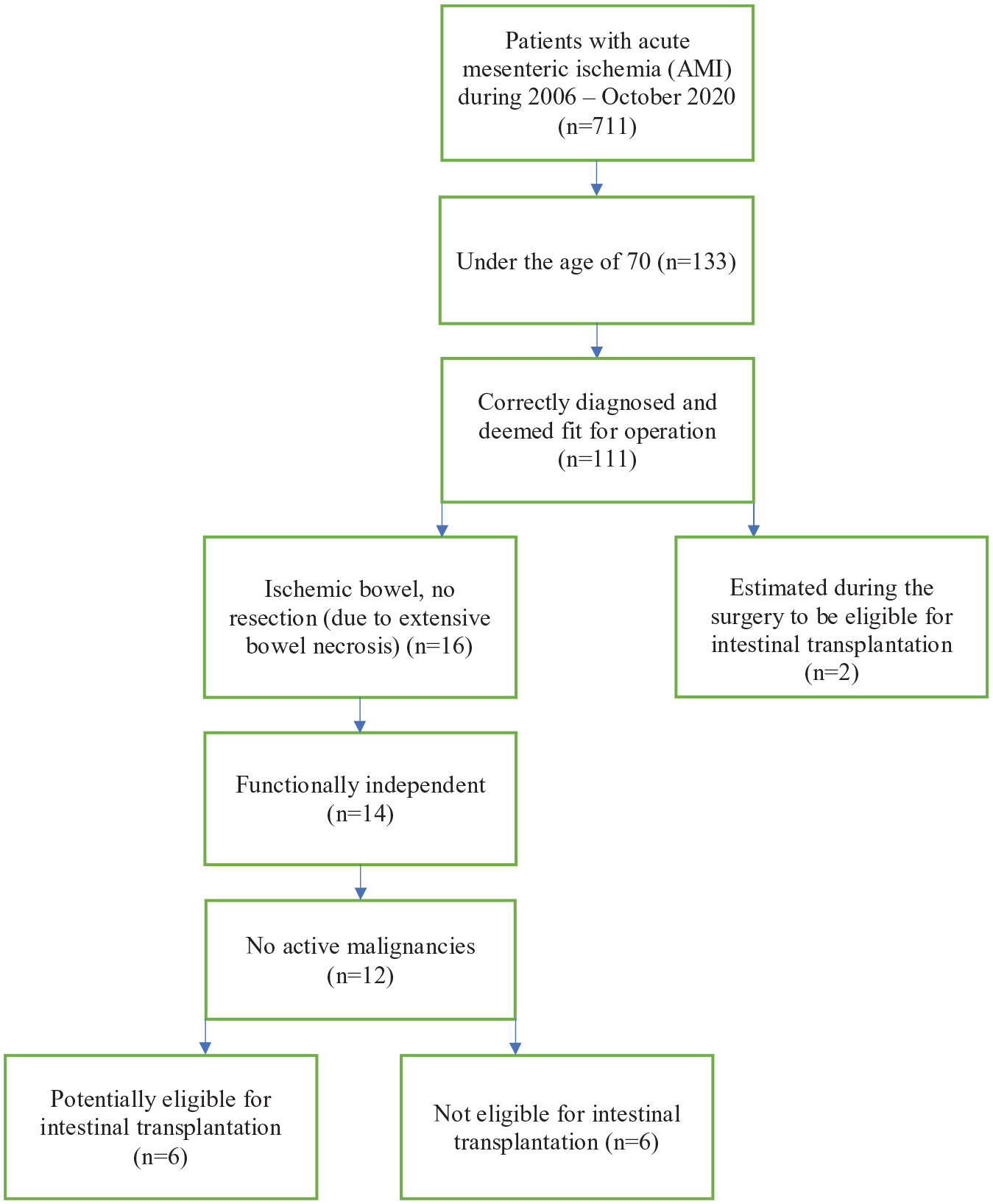
Flowchart depicting the determination of patients potentially eligible for intestinal transplantation.
Characteristics of six patients retrospectively considered not eligible for intestinal transplantation because of comorbidities.
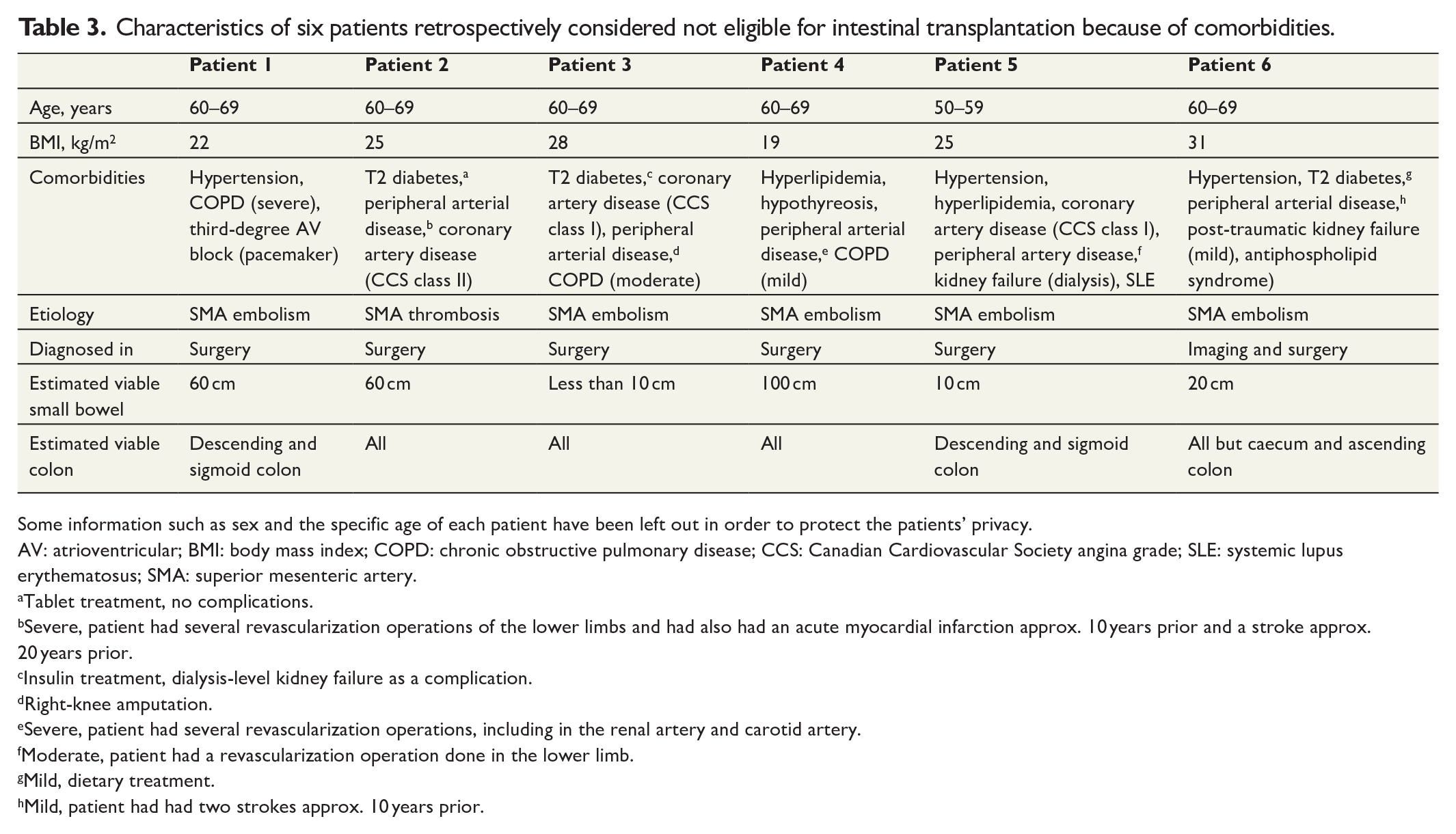
Some information such as sex and the specific age of each patient have been left out in order to protect the patients’ privacy.
AV: atrioventricular; BMI: body mass index; COPD: chronic obstructive pulmonary disease; CCS: Canadian Cardiovascular Society angina grade; SLE: systemic lupus erythematosus; SMA: superior mesenteric artery.
Tablet treatment, no complications.
Severe, patient had several revascularization operations of the lower limbs and had also had an acute myocardial infarction approx. 10 years prior and a stroke approx. 20 years prior.
Insulin treatment, dialysis-level kidney failure as a complication.
Right-knee amputation.
Severe, patient had several revascularization operations, including in the renal artery and carotid artery.
Moderate, patient had a revascularization operation done in the lower limb.
Mild, dietary treatment.
Mild, patient had had two strokes approx. 10 years prior.
Characteristics of six patients retrospectively considered potentially eligible for intestinal transplantation.
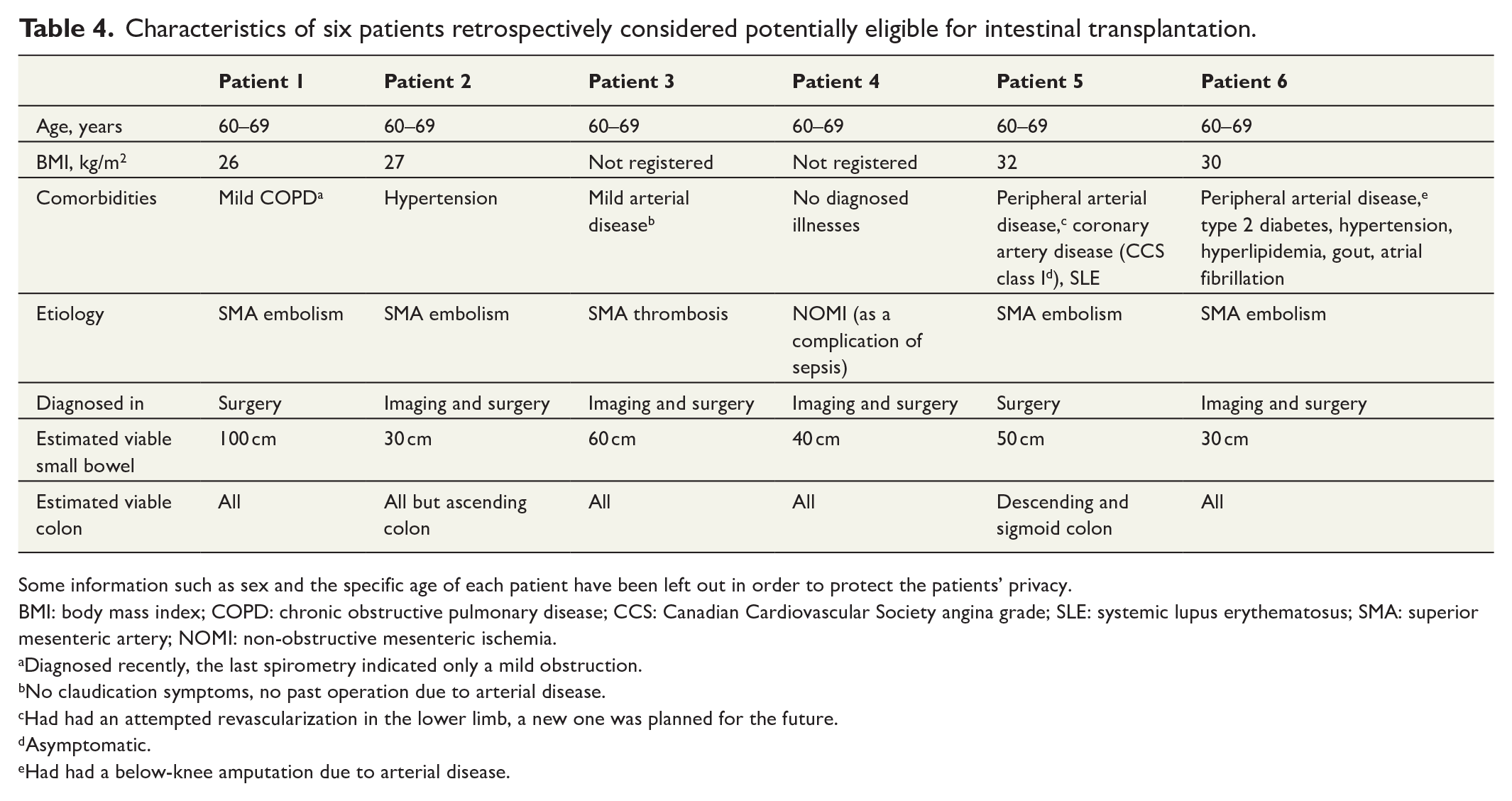
Some information such as sex and the specific age of each patient have been left out in order to protect the patients’ privacy.
BMI: body mass index; COPD: chronic obstructive pulmonary disease; CCS: Canadian Cardiovascular Society angina grade; SLE: systemic lupus erythematosus; SMA: superior mesenteric artery; NOMI: non-obstructive mesenteric ischemia.
Diagnosed recently, the last spirometry indicated only a mild obstruction.
No claudication symptoms, no past operation due to arterial disease.
Had had an attempted revascularization in the lower limb, a new one was planned for the future.
Asymptomatic.
Had had a below-knee amputation due to arterial disease.
Discussion
In this study, 16 patients aged less than 70 years were ruled outside active care due to the extensive length of the necrotic small bowel. Of these, six were retrospectively deemed potentially eligible for intestinal transplantation. Based on these numbers, we estimated that there are 1.44 patients per year in Finland who have extensive small bowel necrosis due to AMI and could potentially be eligible for intestinal transplantation should they survive AMI and develop SBS-IF. This number is an approximation as not all potential recipients would be likely to qualify for transplantation or even require it. Two patients with ultra-short small bowel after AMI were originally considered as candidates for intestinal transplantation, but neither survived beyond 8 months. One surviving patient had a clinical SBS necessitating medication and vitamin supplements.
As the two patients on whom transplantation was initially considered demonstrate even with extensive resections and adequate postoperative treatment, the patients may not survive until a further evaluation on the possibility of transplantation can be carried out. Many potential risks can materialize in this interval, such as bowel anastomosis leakages, postoperative infections, and synchronous embolization. Therefore, when considering the six patients deemed retrospectively eligible for transplantation, it is good to keep in mind that even if these patients had survived the initial operation (and been willing and qualified recipients), a positive outcome is still highly hypothetical, especially with NOMI—as was the case with one patient on whom transplantation was primarily considered and with one patient on whom it was retrospectively deemed possible—where the spread of ischemia is more unpredictable and varied.
Until recently, outcomes of intestinal transplantation have lagged behind other solid-organ transplantations. Among other things, this has been due to high rejection rates and high risks of infection due to, for example, bacterial translocation. 20 However, the safety of intestinal transplantation has been steadily increasing, mainly due to advances in immune-induction therapies.18–21
Home PN is still the primary therapeutic option for SBS-IF.27,28 Like other solid-organ transplantations, intestinal transplantation aims to improve long-term survival and QoL. As PN is a safe treatment with a high survival rate, intestinal transplantation is usually considered only if PN is no longer an option, for example, in patients suffering from severe complications. In a previous survey, a small portion of SBS-IF patients due to mesenteric ischemia (16%) were deemed as intestinal transplant candidates both with an indication and no contraindications. 29
Compared to a healthy population, patients on home PN experience lower QoL and equal or lower when compared to patients with other chronic diseases.30,31 However, poor QoL is not in itself yet an indication for intestinal transplantation due to higher survival rates in patients with home PN. Non-surgical treatment options for SBS are also improving and, especially, recently introduced glucagon-like peptide-2 (GLP-2) analogs have had promising results in the management of SBS. 32 Due to the relatively good QoL on home PN, on young AMI patients with otherwise healthy vasculature, even extensive bowel resections should be considered—and the consequent development of SBS-IF accepted—as the disease is otherwise lethal. This is in line with the 2016 European Society for Trauma and Emergency Surgery guidelines, which note that more extensive resections could be a reasonable option on younger patients. 33
The number of annual intestinal transplants has been steadily increasing until quite recently, when the total number has declined globally.34,35 This is most likely due to improved outcomes of home PN and the ability of specialized intestine failure units to better prevent and treat intestinal failure–associated liver disease, an important indication for transplantation. 34
In, for example, Italy and Nebraska, AMI is reportedly one of the most common etiologies of intestinal failure.36–38 Globally, AMI is also a common etiology among patients requiring intestinal transplantation.22,36,39 To the best of our knowledge, there are no recent long-term follow-up studies on AMI, and it is unclear how many AMI patients would develop intestinal failure and qualify for intestinal transplantation.
Intestinal transplantation is still a rare treatment in Finland. During the last 12 years, eight intestinal transplantations have been performed. Six of these have a functioning graft with a maximum 11 years of follow-up. 24 Our results suggest that intestinal transplantation may be an unrecognized option for AMI patients in Finland. Because our study was conducted in the area that also hosts the only national transplantation center, it is possible the results even overestimate the level of awareness of intestinal transplantation. Because AMI is an acute disease with a high mortality, treatment decisions are often made during on-call hours and in emergency laparotomies. This underscores the importance of specialized clinics and awareness raising.
In this study, SBS was not a common complication of AMI. We wished to investigate post-AMI patients aged 70 or below, as long-term PN and other treatment options for SBS-IF are quite demanding, and often unsuitable for older and more morbid patients. Previous studies have reported varying incidences of SBS following AMI, ranging from 14% to 87%,5,13–17 but in our cohort only two patients developed SBS. These relatively low numbers are most likely due to the age limit we applied to our cohort. This age limit also most likely explains the relatively low mortality rates and the high proportion of embolic versus atherosclerotic disease. Another possible explanation is the relatively restrictive approach to bowel resections in our study material, as a total of 16 patients were ruled outside active care due to the extent of bowel necrosis.
Differences exist between countries and centers in the strategy of bowel anastomosis following AMI. In our center, anastomoses are performed in the initial operation if the patient is stable and there is no doubt of bowel viability in accordance with ESTES guidelines. 33 As the bowel in patients with AMI is often very swollen, risks for anastomotic leaks are increased. Some centers, like the intestinal stroke center in Paris, opt for a temporary stoma for all patients undergoing resections. 40 To the best of our knowledge, there are no studies comparing these strategies on patients with AMI.
This study has several limitations. While retrospective evaluation of eligibility for intestinal transplantation is difficult, evaluation of patients’ attitudes toward treatment options is impossible. Evaluating retrospectively the degree or grade of a disease can also be challenging. To avoid overestimation of potential transplant recipients, we aimed to maintain a high limit of eligibility by, for example, setting an age limit.
In conclusion, with the development of both treatment for SBS and methods of intestinal transplantation, carefully selected AMI patients with extensive bowel necrosis should be evaluated for the possibility of intestinal transplantation before deeming them to palliative care.
Footnotes
Author contributions
V.S. came up with the idea for the paper. All authors participated in formulating the research question and methods for the study. A.L. collected the data and had primary responsibility of analyses, in collaboration with V.S., S.P., A.P., and M.T. All authors commented on the manuscript and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was received from Helsinki University Hospital Research grants, Vatsatautien tutkimussäätiö Foundation, the Finnish Medical Foundation, and Mary and Georg C. Ehrnrooth Foundation.
