Abstract
Purpose:
Over the recent years, axillary staging of initially node-positive breast cancer patients converting to clinical node negativity after neoadjuvant chemotherapy has seen rapid changes. This narrative review aims to give a contemporary overview over published evidence and clinical practice, and thus provide some guidance to the surgical community in the process of re-evaluating and re-shaping surgical practice.
Methods:
The search strategy aimed at finding relevant studies. Only articles in English were considered.
Results:
The introduction of modern techniques offer more precise staging surgery and thus hopefully reduced arm morbidity. Clinical practice has however diverged both within countries and internationally. While some countries have adapted de-escalated axillary staging techniques such as targeted axillary dissection, targeted lymph node biopsy or sentinel lymph node biopsy, others continue to recommend a full axillary lymph node dissection. With the implementation of new techniques, many questions arise, regarding aspects of oncological safety, technical performance, budget and practicality, patient selection and indications for different levels of axillary staging procedures.
Conclusions:
There is a growing body of evidence on de-escalation of axillary surgery in the setting of cN+ → ycN0 breast cancer treated with neoadjuvant chemotherapy. However, standards differ between countries and future studies are necessary to fully assess the optimal strategy for these patients.
Keywords
Introduction
After a long historical period of unselective use of axillary lymph node dissection (ALND) as the only means of axillary staging and treatment, the need for de-escalation became increasingly obvious with the increasingly early detection of breast cancer and improved efficacy of systemic adjuvant treatment. The incentive for a search of less debilitating surgical strategies rose from the significant risk of lymphedema development after ALND, commonly combined with loss of function and sensation, pain, and a deficient range of motion. In 1977, the novel concept of the sentinel lymph node biopsy (SLNB), identifying the first lymph node receiving lymphatic drainage from the tumor area, was first described by Cabanas in patients with penile carcinoma. 1 In 1990, a blue dye-based SLNB technique for the detection of nodal melanoma metastases was presented to the Society for Surgical Oncology by Donald Morton and Alistair Cochran from the John Wayne Cancer Institute. Interestingly, the SLNB concept along with this new technique was met with much doubt, and the original manuscript rejected by several scientific journals until its publication in 1992. 2
In the following years, the SLNB was tested, implemented, and validated in prospective studies in breast cancer patients, leading to the conclusion that the sentinel lymph node (SLN) could be assumed to represent the true axillary status with an acceptable false-negative rate and could thus guide surgeons to selectively use ALND in case of a positive SLNB. The Philadelphia Consensus Conference of 2001 concluded that the SLNB was “a suitable replacement for axillary dissection as a staging and diagnostic procedure in T1 and T2 (usually 3 cm) breast cancers,” but surgeons should be practicing completion ALND until they could show a sufficient detection rate and false-negative rate (FNR) lower than 5%. 3
The increasing popularity of neoadjuvant chemotherapy (NACT) brought new challenges to axillary surgery. So far, it was agreed that an ALND was not indicated in case of an up-front negative SLNB in clinically node-negative (cN0) patients, and after the publication of the ACOSOG Z0011 trial in 2011 4 and the EORTC 10981-22023 AMAROS trial in 2014, 5 its role was questioned even in SLN-positive cases. But what if the SLN was negative after NACT, while it may have harbored a metastasis before NACT? The question of the appropriate timing of SLNB in this situation was addressed in prospective studies such as SENTINA in Germany 6 and the Swedish prospective multicenter trial.7,8 It was concluded that it was safe to schedule SLNB after instead of before NACT in cN0 patients, thus taking the degree of response to NACT into account, and accepting a pinch of uncertainty not knowing the initial pathological axillary status when deciding on surgical and adjuvant treatment strategies. It was only a matter of time until the debate, whether to see conversion through NACT as a threat for undertreatment or a bliss for the preservation of arm function, also reached the patient subgroup addressed in this review: the clinically node-positive (cN+) population receiving NACT.
Strategies to de-escalate axillary staging after NACT
Loyal to the historical tradition, the introduction of SLNB after NACT in initially cN+ patients focused mainly on FNR, used as a surrogate marker for the risk of later axillary recurrence. Disappointingly, the detection rate in this situation was significantly lower and the FNR unacceptably high, probably due to fibrotic changes in the lymphatics through NACT, altering lymphatic drainage patterns. 9 From here, the development took to two potential solutions: First, to find ways to decrease FNR and second, to question whether the FNR was as important for oncological outcomes as expected. Alluding to the fact that the true metastastic node (and thus probably the original SLN) can be missed by the SLNB procedure after NACT, strategies were sought to guarantee that this initial metastastic node was securely retrieved at post-NACT surgery. A logical conclusion was to mark this specific node up-front for its easier identification after NACT. In the NSABP Z1071 trial, a metal clip was initially placed in the metastastic node of a retrospectively studied subgroup which decreased the FNR to 1.4% when the clipped node was removed at SLNB. 10 Since then, the term “targeted axillary dissection” (TAD) refers to the marking of an initial axillary metastasis before NACT and its subsequent location and removal in combination with an SLNB procedure after NACT. Without the SLNB, the procedure is simply called “targeted lymph node biopsy” (TLNB).
Other strategies to decrease the FNR in SLNB were subsequently identified in several studies: The use of dual instead of single tracer, the use of immunohistochemistry to detect even small metastastic deposits such as isolated tumor cells, and an increased number of removed SLNs. It should, however, be pointed out that the accuracy of SLNB is compromised when picking random lymph nodes in the vicinity of the SLN to increase nodal yield; on the contrary, palpation of the axilla and removal of nodes suspicious to the touch are included in the SLNB concept.
The second potential solution to the FNR problem was the hypothesis that FNR may not directly translate into axillary recurrence rates. Already in up-front SLNB in cN0 patients, long-term axillary recurrence rates are surprisingly low considering an occult FNR of 7%–10% in patients where ALND is routinely omitted after a negative SLNB. 11 Similarly, the low axillary recurrence rates after SLNB as single axillary staging procedure in cN+ patients converting to ycN0 reported, for example, from the IEO in Milan give food for thought, and remind us that we might not need to be focusing solely on low FNRs if their clinical importance is not confirmed in oncological follow-up studies. 12 Here, one should eagerly expect further long-term results from larger studies such as the prospective EUBREAST 3/AXSANA 13 and Dutch MINIMAX studies. 14
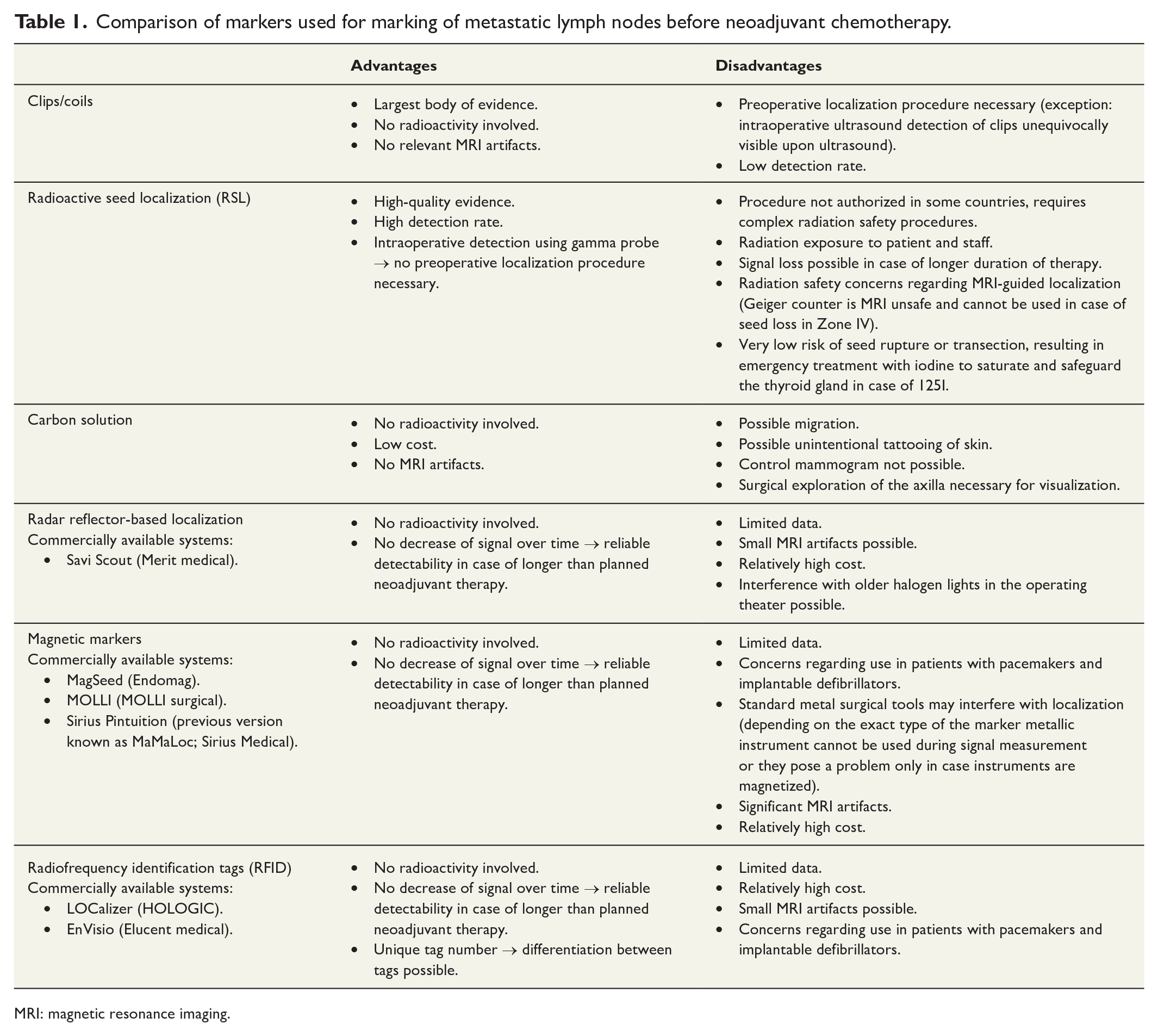
How to mark metastastic lymph nodes?
Several different markers of metastastic nodes prior to NACT have been developed, which can be basically divided into markers identifiable by imaging (usually intra- or preoperative ultrasound, the latter necessitating a second localization procedure), visual inspection during surgery or by different types of probe-detectable signals (Table 1). Irrespective of the type of marker, it is usually placed into the lymph node metastasis under ultrasound guidance.
Comparison of markers used for marking of metastatic lymph nodes before neoadjuvant chemotherapy.
MRI: magnetic resonance imaging.
The majority of studies on TAD and TLNB evaluated the use of clips/coils.10,13,15–17 Since these markers cannot be detected using special probes, the detection rate depends on their visibility upon imaging, particularly using ultrasound. Once the clip/coil has been identified, it is usually localized in a second marking procedure using either a wire or a probe-guided detection marker, or—in case of excellent ultrasound visibility—it may be localized directly in the surgical theater using intraoperative ultrasound, such as practiced in the ILINA trial. 18 The prospective SenTa registry, the largest available dataset on clip-based TAD, reported an FNR of 4.3% and a detection rate of 78% among 423 patients in whom TAD was attempted. In further 34 patients, the TLN was not detectable by preoperative ultrasound and TAD thus not attempted. Therefore, the real detection rate may be as low as 72%. 15 Indeed, a recent survey found that only 65% of respondents using clips or coils describe the detection rates as very good or good. 19 While there is insufficient evidence to recommend a specific type or shape of clip or coil, it may be wise to use larger clips or those with a 3D shape that might enhance sonographic visibility (Fig. 1).20,21
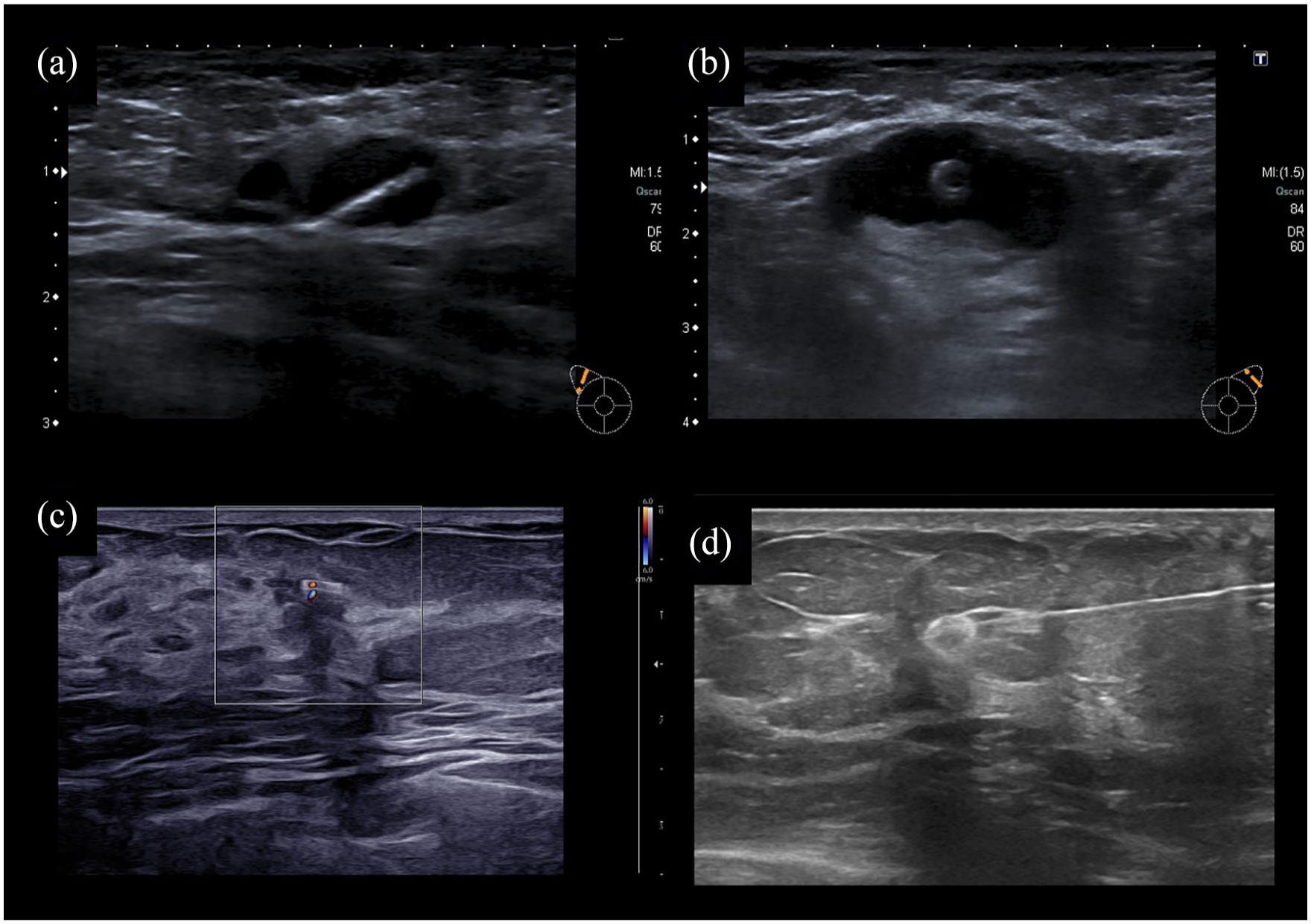
Examples of different clip types used for lymph node marking. (A, B) Ultrasound image of clip of various shapes directly after placement into the sonographically visible hypoechogenic lymph node metastasis. (C) In case of some clips the so-called “twinkle artifact” can be observed. 22 This artifact can be helpful in case of complete remission on imaging. (D) Wire localization of the clip before targeted axillary dissection.
Another marker for TAD/TLNB is carbon solution, which implies an ultrasound-guided injection of a highly purified carbon suspension into the cortex of the biopsy-proven metastastic lymph node and the adjacent soft tissue. The largest study on the carbon-based TAD was the German-Swedish prospective TATTOO trial. 23 In the first publication, the trial reported a high detection rate (94%) and an FNR of 9%. Recently, updated results from the extended TATOO trial focusing only on cases with a completion ALND were presented, which included an additional 39 patients enrolled in Sweden where recruitment was extended beyond October 2019. 24 Four out of 65 cN+→ ycN0 patients had a negative TAD but metastases on ALND, corresponding to an FNR of 6.2%. Interestingly, all false-negative procedures were performed in the first 2 years of the trial, suggesting that a learning curve must be taken into consideration when implementing surgical de-escalation in the clinical routine.
Among markers enabling probe-guided detection, the use of radioactive seeds was examined in the prospective single-arm RISAS trial, including 227 patients from the Netherlands who had a TAD and completion ALND following NACT. 25 TAD could be successfully performed in 98% of patients with an axillary pCR rate of 35%. The TLN was identical with the SLN in 71% of patients. There were five false-negative cases, corresponding to an FNR of 3.47% (95% confidence interval (CI): 1.38–7.16). The trial was concluded to be negative, however, because the upper range of the 95% confidence interval of the FNR crossed the pre-specified non-inferiority boundary of 6.24%. Interestingly, the four false-negative cases all occurred among the first 10 patients. 26
Other markers for probe-guided techniques used in clinical routine are magnetic seeds, radar reflectors and radiofrequency markers (Fig. 2). These rely on tagging the TLN with a marker that can be detected during surgery using a special probe, either sterile for single use or covered in a sterile sheath, thus eliminating the need of a second preoperative localization procedure. Examples of commercially available markers can be found in Table 1. Current evidence on the use of these techniques before NACT is limited. While many centers use such markers to preoperatively localize clip-marked nodes after NACT,27,28 fewer studies report on their pre-NACT placement into the TLN despite the obvious advantage of sparing the patient another invasive procedure.29–31 Most markers were initially approved for short-time use (e.g. 30 days) and only later, the approval was extended to implantation for either an unlimited duration or 180 days. One common limitation of these techniques is the potential signal loss in case of deeply situated lesions. However, with pressure applied to the tissue, the distance between the marker and the probe can be reduced and successful marker retrieval achieved in most cases.

Examples of markers allowing probe-guided detection (note that the depicted size does not correctly compare the different markers shown; for exact dimension see main body of the manuscript). (A) MagSeed (5 mm × 1 mm). (B) Sirius Pintuition (5 mm × 1.65 mm). (C) Savi Scout (12 mm × 1.6 mm). (D) LOCalizer (11 mm × 2 mm).
Two magnetism-based detection techniques are widely used in Europe: MagSeed and Sirius Pintuition. MagSeed is a 5 x 1 mm metallic seed made of surgical stainless steel, deployed by an 18-gauge needle (Fig. 2A). It is currently approved in the EU for long-term implantation in any soft tissue. The seed is temporarily magnetized only in the presence of the Sentimag probe. However, non-metallic instruments are recommended during the procedure, because ferromagnetic devices interfere with seed localization. The Sentimag probe may be used both for the localization of the MagSeed-marker target node and the SLN, if a lymphatic tracer such as Magtrace is used. Sirius Pintuition is a permanent magnetic seed (5 mm × 1.65 mm; 14-gauge needle) that is approved in the EU for placement for up to 180 days in any soft tissue (Fig. 2B). The probe used for detection has an additional tool to show not only the distance to the seed, but the angle as well. The probe is compatible with standard metal instruments, as long as they are not magnetized, since this may lead to interference and/or marker dislocation. Thus, it might be prudent to have one set of non-metallic instruments available. The main disadvantage of magnetic markers is the creation of 5–6 cm artifacts surrounding the marker when using magnetic resonance imaging (MRI). If tumor response is to be assessed by MRI, magnetic markers should not be placed in the vicinity of the tumor area before NACT. 21
Savi Scout detection is based on radar reflector technology. The marker is longer (12 mm × 1.6 mm, 16-gauge needle) and consists of two antennae (Fig. 2C). It is currently approved in the EU for marking (>30 days) a biopsy or soft tissue site intended for surgical removal. Minor artifacts may occur on MRI. 32 Since the technology is based on infrared light, the acoustic and visual signal appears only once the probe touches the skin or tissue and signal loss has been described in case of a large hematoma. 33 The use of Savi Scout for lymph node marking was reported by small studies.34–36
The LOCalizer system uses radiofrequency identification (RFID) tags (Fig. 2D) that are placed into the TLN or a non-palpable breast lesion. These have a unique number that is displayed on the screen when the probe detects a tag. RFIDs may cause MRI artifacts which appear to be smaller than for magnetic markers but larger than those produced by radar reflectors. 32 The system is approved in the EU for long-term placement in the breast but not in the axilla; some studies however report on the use in the axilla as well.37,38
How many lymph nodes should be marked?
In case of two or more suspicious lymph nodes, the question arises whether more than one lymph node should be marked. Currently, there is not enough evidence to answer this question and in previous studies, usually only one lymph node was marked. Hypothetically, if only one lymph node is marked and later removed, no information on the response of further nodal metastases will be acquired. This may be problematic since a heterogeneous response to NACT is common especially in hormone receptor-positive HER2-negative subtypes. 39 Furthermore, a large number of nodal metastases, especially when matted together, may significantly change local functional anatomy, since lymphatic and blood vessels may get obstructed.
Data from smaller studies indeed suggest that the FNR can be reduced by the marking and removal of more than one lymph node. Lim et al. 40 evaluated this approach in 14 patients with one to three abnormal lymph nodes scheduled for NACT. First, the most suspicious node was biopsied and, if proven metastatic, designated as the first target node and marked at a second setting. During this second setting, further sonographically abnormal lymph nodes underwent fine-needle aspiration and marking. Patients with more than one metastatic lymph node had each node marked with a different type of marker to aid individual lymph node identification (UltraCor Twirl, HydroMARK, and UltraClip dual trigger). All target lymph nodes were removed at surgery, but no SLNB, so no TAD was performed. All patients received a completion ALND. The first clipped node predicted the true axillary status with an FNR of 7.1%. When the second clipped node was also considered, the FNR fell to 0%.
Marking multiple nodal metastases obviously results in the removal of a higher number of lymph nodes. Natsiopoulos et al. 41 used carbon solution for the marking of every biopsy-proven or strongly suspicious node: Two nodes were marked in 27% of patients, and three or more in 36%. As expected, the median number of nodes excised during TAD was 4 (range 2–10) which is higher than in comparable studies. 10 How this relatively high number of removed lymph nodes may affect long-term health-related quality of life (QoL) and arm morbidity remains to be clarified. In a large population-based study including 1338 elderly patients with breast cancer, the most important factor associated with lymphedema was the number of removed lymph nodes. 42 The excision of 1–5 nodes increased the risk of lymphedema by the factor 2.11 compared with no nodes removed, whereas removal of 6–10, 11–15 and >15 nodes increased the risk by factor 4.68, 5.61 and 10.50, respectively. 43 QoL and arm morbidity are primary endpoints of the AXSANA study and will be repeatedly evaluated during follow-up. 13
Another aspect that needs to be considered is the cost of multiple markers. Unsurprisingly, the number of marked nodes is generally higher in studies using the relatively cheap carbon solution. In contrast, probe-guided detection markers cost between 200 and 400 EUR, depending on the marker and clinical setting. Furthermore, reimbursement regulations differ between countries, which may affect clinical routines regarding the number of lymph nodes marked.
How to identify potential candidates for de-escalated axillary staging?
So far, national and international guidelines recommend de-escalation of axillary surgery after NACT only in patients converting to a clinically negative lymph node status (cN+→ ycN0).21,43 However, standards regarding assessment of axillary response differ between countries and hospitals. In some, nodal status is assessed using clinical examination only, while in others full imaging workup including ultrasound, MRI or even PET/CT is performed. Hypothetically, patients risk under- or overtreatment with potentially serious consequences if the accuracy of preoperative assessment is low. 44 To date, no TAD studies have reported any oncological follow-up. Therefore, careful patient selection is necessary to safely implement de-escalated axillary surgery into the clinical routine. High-quality standardized examination after NACT is crucial for the selection of patients in whom full ALND may safely be omitted.
A recent systematic review showed that the sensitivity of ultrasound for the prediction of residual nodal metastases is higher than that of clinical examination, MRI or PET. 44 In the large German SENTINA trial, the accuracy of ultrasound was 55% and that of clinical examination 48%. 6 Among 592 initially node-positive patients converting to ycN0 based on clinical examination and ultrasound, 48% had one or more metastatic lymph nodes upon histopathological evaluation, while 23% of patients with suspicious nodes upon ultrasound after NACT had no residual axillary disease. Similar results were reported by others.45–47 Several studies evaluated additional strategies to improve the accuracy of imaging. Caudle et al. 48 performed fine needle aspiration of clipped nodes in 50 patients after NACT. However, the FNR of fine needle aspiration was higher and the sensitivity lower than that of ultrasound alone, so this approach cannot be recommended.
Interestingly, several studies reported an association between diagnostic accuracy of ultrasound after NACT and tumor subtype, with the highest positive predictive value in luminal and highest negative predictive value in triple-negative tumors.44,49,50
Another important debate regards the oncological safety of TAD in patients presenting with higher nodal burden. On one hand, patients with a higher number of suspicious nodes prior to NACT (e.g. ⩾3 or ⩾4) were included into most of previous studies investigating the technique. On the other hand, however, none of them reported on the FNR in this specific subgroup.10,13,15 The response to NACT is not uniform for all metastatic nodes, 39 and in theory, the higher the number of metastatic lymph nodes at the time of diagnosis, the higher the chances of leaving residual metastases behind with a de-escalated axillary procedure. In addition, as stated above, a higher nodal burden may result in the obstruction of lymphatics and blood vessels. As a consequence, an SLN after NACT may not represent the true “first” node. For these reasons, the German AGO Breast Committee updated recommendations for TAD in March 2022 (Fig. 3): TAD and ALND are viewed as equivalent techniques in patients with up to three suspicious nodes at time of diagnosis and good response to NACT (ycN0). In cases with four or more suspicious lymph nodes, however, ALND is the preferred method and TAD should be approached with caution.

Current recommendations of the AGO Breast Committee regarding targeted axillary dissection.
What to do in case of a “lost marker”?
One of the most frustrating experiences in theater is the “lost marker” situation in TAD. While detection rates using probe-guided detection techniques and carbon solution are very high (⩾93% in most studies), this situation may occur more frequently when nodal metastases are marked using a clip or coil.15,23,24,51 In the SENTA trial, the detection rate was as low as 72%. 15 Furthermore, a correctly placed marker may be dislocated through downstaging of the metastatic lymph node associated with morphological changes such as nodal shrinkage. 44
In this context, two scenarios need to be considered. In the first one, the placement of a wire or a secondary localization marker into the initially clipped node after NACT is impossible because the clip is not visible upon imaging. If the clip cannot be identified by ultrasound, additional imaging such as mammography or computed tomography may be helpful.44,52 However, this is time-consuming and may result in delays. In this case, it is recommended to perform SLNB and examine the SLN using specimen radiography (Fig. 4). In 48%–75% of patients, the SLN is also the TLN. If the clip cannot be identified, completion ALND should be considered.20,21
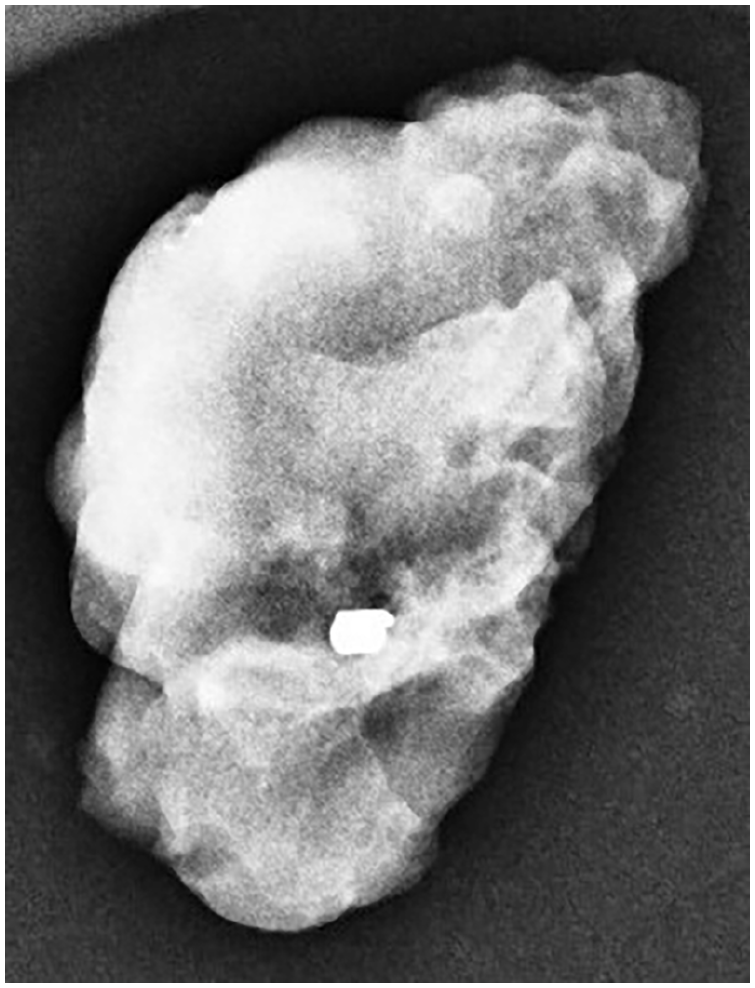
Specimen radiography of a target lymph node confirming the removal of the metallic clip used for node marking before neoadjuvant chemotherapy.
In the second scenario, the TLN is sent for radiography but the clip is not found in the specimen. If the clip is also absent from the SLN specimen, a completion ALND is recommended. Interestingly, Hartmann et al. 17 reported an intraoperative detection rate of the clipped TLN of 67%. All ten patients in whom clip removal could not be confirmed on specimen radiography had postoperative imaging with mammography and computed tomography. The lost clip was still visible in two of these patients, suggesting that it may have been accidentally removed from the surgical site by swabs or instruments.
Most guidelines do not include specific recommendations on the technique of TAD and the management of “lost markers.” The AGO Breast Committee guidelines recommend completion ALND in case a TAD cannot be successfully performed. If the patient received ALND and the clip is still in the patient’s body, no further intervention is recommended. An exception should be made for patients with radioactive seeds since these must be removed due to radiation protection laws.
Footnotes
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jana de Boniface declares that there is no conflict of interest. M.B.-P. received honoraria for lectures and participation in advisory boards from Roche, Novartis, Pfizer, pfm, Eli Lilly, Onkowissen, Seagen, AstraZeneca, Eisai, AstraZeneca, Amgen, Samsung, Canon, MSD, GSK, Daiichi Sankyo, Gilead, Sirius Pintuition, Pierre Fabre, ExactSciences, and study support from: EndoMag, Mammotome, MeritMedical, Gilead, Hologic.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: J.d.B. is supported by a Young Clinical Investigator Award by the Swedish Cancer Society.
