Abstract
Background and objective:
The option to treat early colon cancer (CC) with local resection, as well as trials investigating neoadjuvant treatment, has increased the importance of identifying early-stage disease in the workup. Most CC patients are T- and N-staged preoperatively with CT, although its reliability in staging early CC remains elusive. The aim of this study was to investigate CT-staging accuracy in early CC by evaluating pT and pN stages in patients staged as cT1-2, and cT and cN stages in patients with pT1 tumors.
Methods:
Retrospective population-based cohort study on data from the nationwide Swedish colorectal cancer registry on all CC patients staged as cT1-2 and all patients with pT1 undergoing surgical resection 2009–2018. CT-acquired T- and N-stages were compared with final histopathology. Factors potentially influencing accuracy were analyzed with uni- and multivariate logistic regression.
Results:
Computed tomography (CT) staged 4849 patients as cT1-2, whereas 2445 (50%) were pT3 and 453 (9%) pT4. Positive predictive value of the cT1-2 stage was 40%. Of 1401 pT1 patients, 624 (45%) were staged as cT1-2, 139 (10%) as cT3, 15 (1%) as cT4 and 623 (44%) as cTx. In all, 1474 (30%) of the cT1-2 patients were pN+, whereas CT staged 1062 (72%) as cN0. A total of 771 patients were staged as cN+, whereas 403 (52%) were pN0. Overall accuracy in determining N+ was 67%, with 26% sensitivity and 88% specificity. Positive and negative predictive values in determining N+ were 48% and 73%, respectively.
Conclusions:
This nationwide population-based study shows that CT-staging carries a substantial risk of understaging locally advanced tumors as cT1-2 and pT1 tumors as cTx, in addition to poor N-staging. Thus, CT obtained T- and N-staging should not be used for deciding treatment strategies in early CC.
Keywords
Introduction
Preoperative staging of patients with colon cancer (CC) is fundamental when deciding on treatment strategies. Major surgical resection has historically been the mainstay treatment for all stages of CC 1 although developments in endoscopic techniques enabling local resection of early T1 CC, as well as neoadjuvant treatment (within trials), have widened the therapeutic spectrum. To correctly identify patients with early CC, eligible for local resection, and to differentiate them from patients with more advanced stages where neoadjuvant treatment might be beneficial, has consequently become increasingly important. Local resection is feasible in T1 CC without suspicion of lymph node metastases (LNM), and risk factors of LNM in the resected specimen ultimately define the need for subsequent surgery. Local resection carries many benefits in terms of decreased morbidity, mortality, and cost compared with surgery.2,3 Despite this, previous studies have shown that 84%–92%4–6 of T1 CCs are N0 and hence potentially curable by local resection, yet the majority of patients undergo surgery. 1
Computed tomography (CT) is widely used in the workup of CC mainly because of its use in detecting distant metastasis, as well as being universally available and easily reproducible. 7 However, T- and N-staging of CC performed with CT is more challenging, and previous studies have shown substantial variations in both T- and N-staging accuracy.8–11 Thus, the majority of studies investigating CT-acquired T- and N-staging accuracy in CC are of limited size and have had an emphasis on radiological methodology, raising concerns on generalizability to a clinical setting. Furthermore, it is well known that CT cannot reliably differentiate T1 from T2 and the two early stages are, therefore, often combined as cT1-2. Distinguishing early-stage tumors confined to the bowel wall (T1-2/UICC stage I) from tumors invading beyond the muscularis propria (T3-4/UICC stage II) in addition to nodal status is, therefore, the goal of staging in early CC. Apart from a recent large population-based study from Denmark showing an accuracy of 76% when distinguishing localized limited disease (T1-2/UICC stage I) from more advanced stages, 12 valid population-based data on CT TN staging of early CC are lacking.
Based on the considerations above, the aim of this nationwide retrospective cohort study was to investigate CT staging of early CC by evaluating pT and pN stages in patients staged clinically as cT1-2 as well as examining the cT and cN staging diversity of pT1 lesions. Secondarily, we examined factors potentially influencing staging accuracy with uni- and multivariate regression analysis.
Methods
Patients
The Swedish Colorectal Cancer Registry (SCRCR) is a nationwide registry with prospectively collected data on preoperative staging, operative surgical details, histopathology, oncologic treatment, and follow-up (5 years). The SCRCR has been shown to have high validity and completeness compared with the mandatory Swedish Cancer Registry, covering 99% (median) of all CCs during the study period. 13
All patients undergoing surgical resection for CC between 2009 and 2018 clinically staged as cT1-2 as well as all pT1 patients were identified in the SCRCR. Patients undergoing endoscopic resection as final treatment were not included. Exclusion criteria were neoadjuvant treatment, emergency resection, unspecified pathological stage, time from diagnosis to surgery exceeding 1 year, and missing data on T-stage, N-stage, resection method, or cancer location.
Staging
Routine workup for CC comprises colonoscopy and CT of the thorax and abdomen for local staging and distant metastasis screening. Patients are staged according to the UICC TNM classification, version 8. 14 All CT images are reviewed twice, and the second read is performed by an experienced gastrointestinal radiologist prior to a multidisciplinary team conference. Stages reported in the registry and herein are based on the final CT-based stage, decided on at the multidisciplinary team conference. Nodal status is reported as N0, N1, or N2 in the SCRCR, herein recategorized as positive (N+) or negative (N0) nodal status. Information on the specific methodology of CT scans is not registered in the SCRCR and, therefore, not included as a parameter in the present study.
Clinical T- and N-stages, determined with CT, were compared with the histopathological T- and N-stages. CT T-stages 1 and 2 are combined in the SCRCR as cT1-2 (considered accurate when pT1 or pT2 was found in the specimen). Cases clinically staged as cT1-2 were analyzed to determine the accuracy of T- and N-staging in this group.
Factors including age, sex, year of surgery, and tumor location were tested for potential impact on T- and N-staging accuracy in the cT1-2 group. Time from diagnosis to surgery refers to the date the tumor was discovered, since date of CT scan is not recorded in the SCRCR, to the date of surgery. Tumor location was presented as right colon, comprising cecum, ascending colon, hepatic flexure and transverse colon and left colon, comprising splenic flexure, descending colon, and sigmoid colon.
Statistics
T-stage accuracy was expressed as positive predictive value (PPV), and N-stage accuracy was expressed as sensitivity, specificity, PPV, negative predictive value (NPV), positive likelihood ratio, and negative likelihood ratio. All pT1 cases were analyzed separately to determine T- and N-staging accuracy in this group. Uni- and multivariate logistic regression were used to investigate the relationship between age, sex, year of surgery, and tumor location with staging accuracy in the cT1-2 group. Python (with add-on packages Pandas and NumPy) and R (R core team 2020, R foundation for statistical computing, Vienna, Austria) programming languages were used for analyses. The p values <0.05 were considered significant. Data are presented as median and range.
Ethics
The study was approved by the regional ethical review board, Lund University (2017/546) and performed in accordance with the declaration of Helsinki. All patient data retrieved from the SCRCR were coded for anonymity.
Results
Baseline characteristics cT1-2 group
In total, 5694 patients with surgically resected CC staged as cT1-2 were identified in the SCRCR, whereas 845 were excluded (Fig. 1). The remaining 4849 patients with a median age of 74 and consisting of 50% women constitute the cT1-2 group in the study population (Table 1). Median time from diagnosis to surgery was 35 days, and 4607 (95%) patients underwent surgery within 90 days.

Inclusion of cT1-2 cases.
Characteristics of CT cT1-2 group.
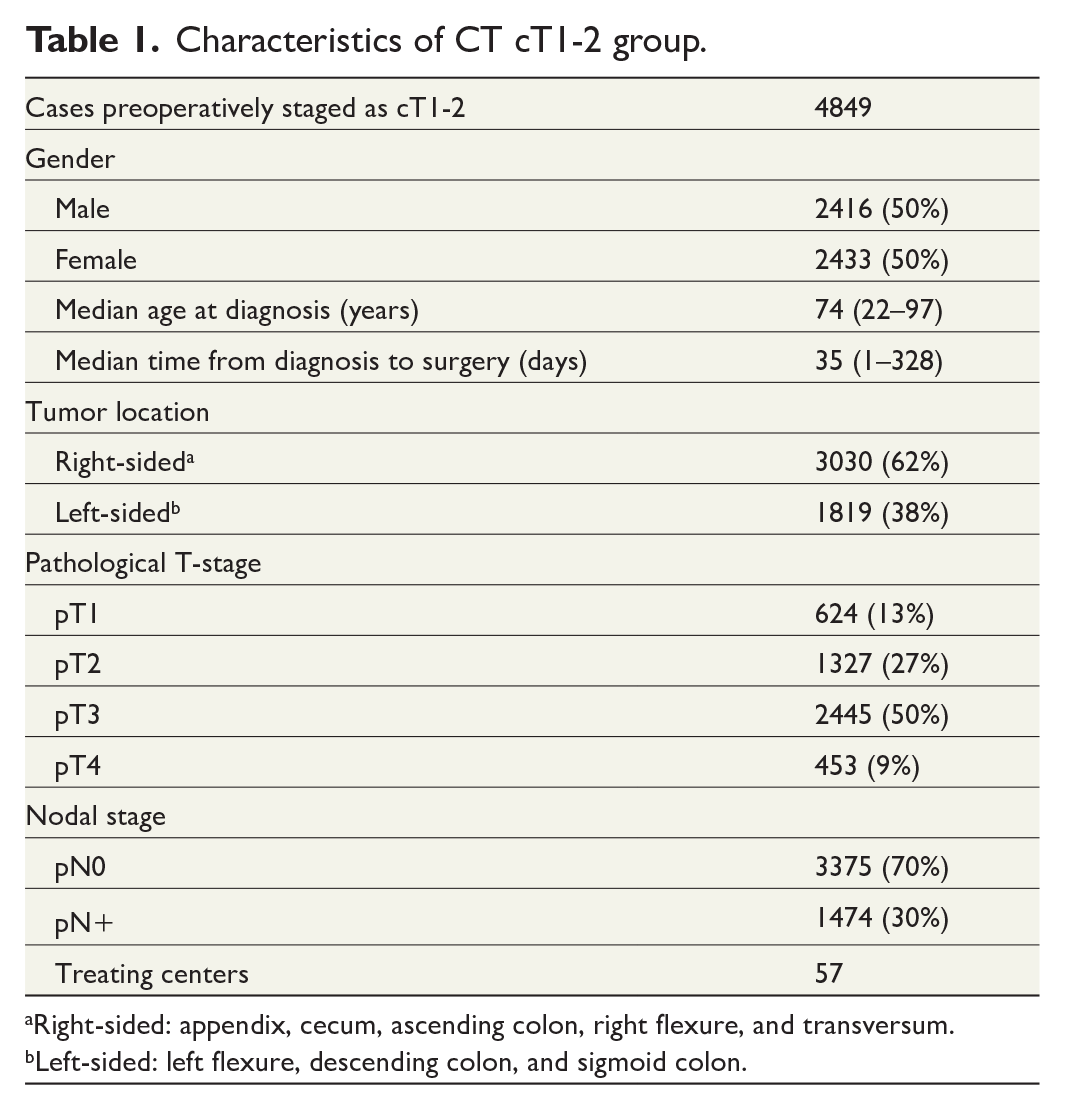
Right-sided: appendix, cecum, ascending colon, right flexure, and transversum.
Left-sided: left flexure, descending colon, and sigmoid colon.
T-stage accuracy in cT1-2 group
A total of 4849 lesions were staged as cT1-2, whereas 1951 (40%) were correctly staged, comprising 624 (13%) pT1 and 1327 (27%) pT2 tumors. Thus, the accuracy, expressed as PPV, for the CT-acquired cT1-2 stage was 40%. The majority of cases staged as cT1-2 by CT were understaged and histopathology revealed 2445 (50%) pT3 and 453 (9%) pT4 tumors.
Uni- and multivariate analysis identified tumor location (left-sided tumors being more accurately staged) as a significant factor influencing T-stage accuracy (Table 2). Lower age was also identified as a significant factor in increasing T-stage accuracy in uni- but not multivariate analysis (Table 2).
Factors potentially influencing accuracy in tumor staging of early CC (cT1-T2).
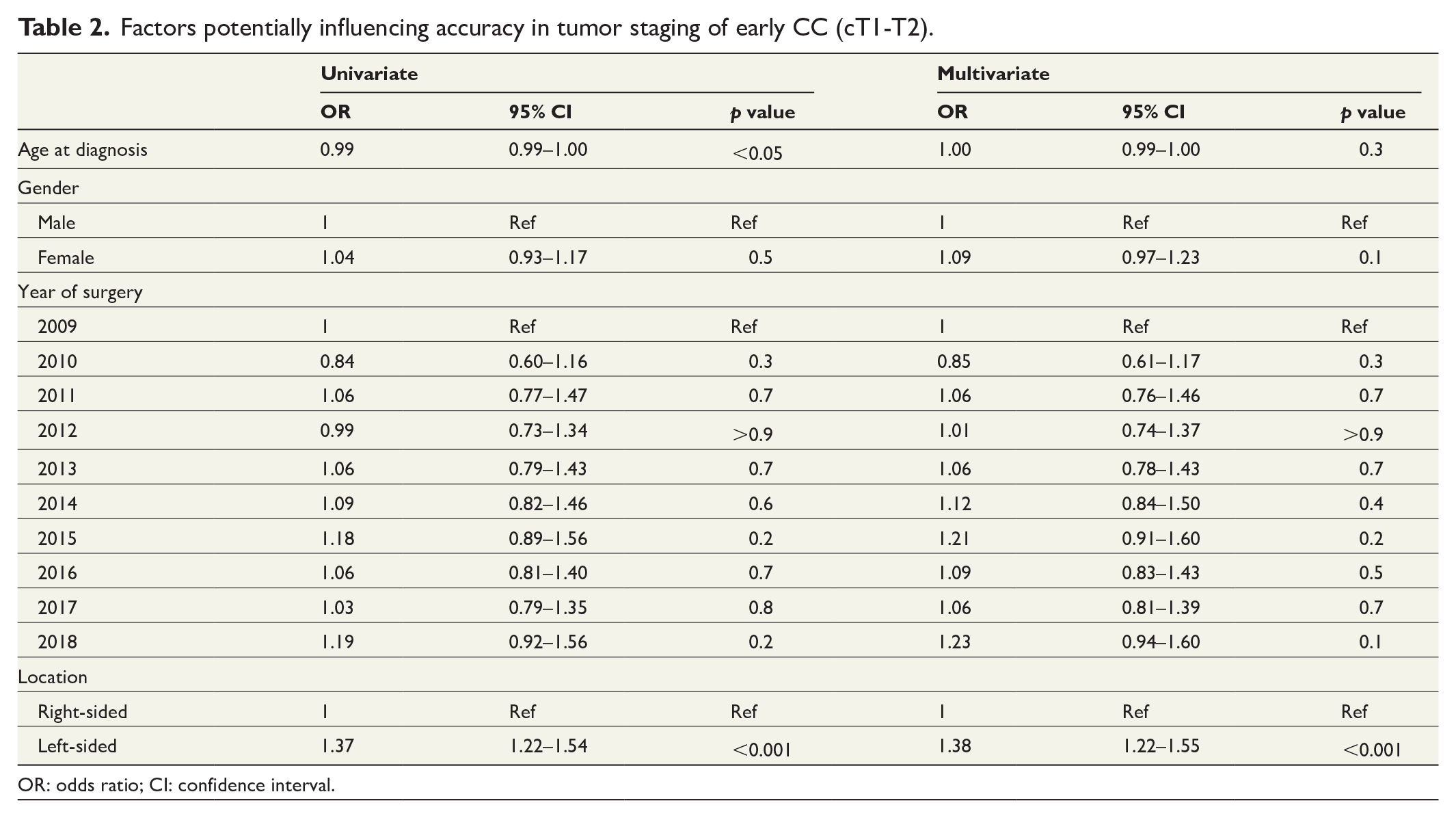
OR: odds ratio; CI: confidence interval.
N-stage accuracy in cT1-2 group
Overall, 1474 patients (30%) in the cT1-2 group were pN+. CT staged the majority, 1062 (72%) of these erroneously as cN0 and 44 (3%) were staged as cNx (Table 3). On the contrary, CT staged 771 patients as cN+, whereof more than half, 403 (52%), were in fact pN0 and thus overstaged.
Accuracy of CT in staging nodal metastases in CT cT1-2 group.
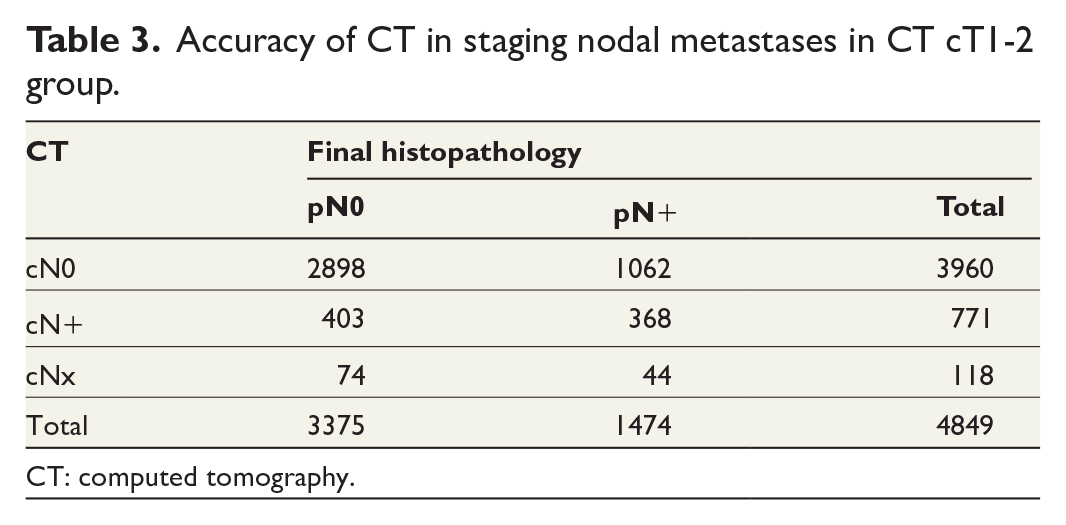
CT: computed tomography.
The overall accuracy in determining pN+ status was 67%, with a sensitivity and specificity of 26% and 88%, respectively. PPV, NPV, positive likelihood ratio, and negative likelihood ratio were 48%, 73%, 2.1, and 0.85, respectively.
Uni- and multivariate analysis identified higher ageas a significant factor associated with better nodal staging accuracy. Right-sided tumor location was also a significant factor increasing nodal staging accuracy in uni- but not multivariate analysis (Table 4).
Factors potentially influencing accuracy in nodal staging of early CC (cT1-T2).
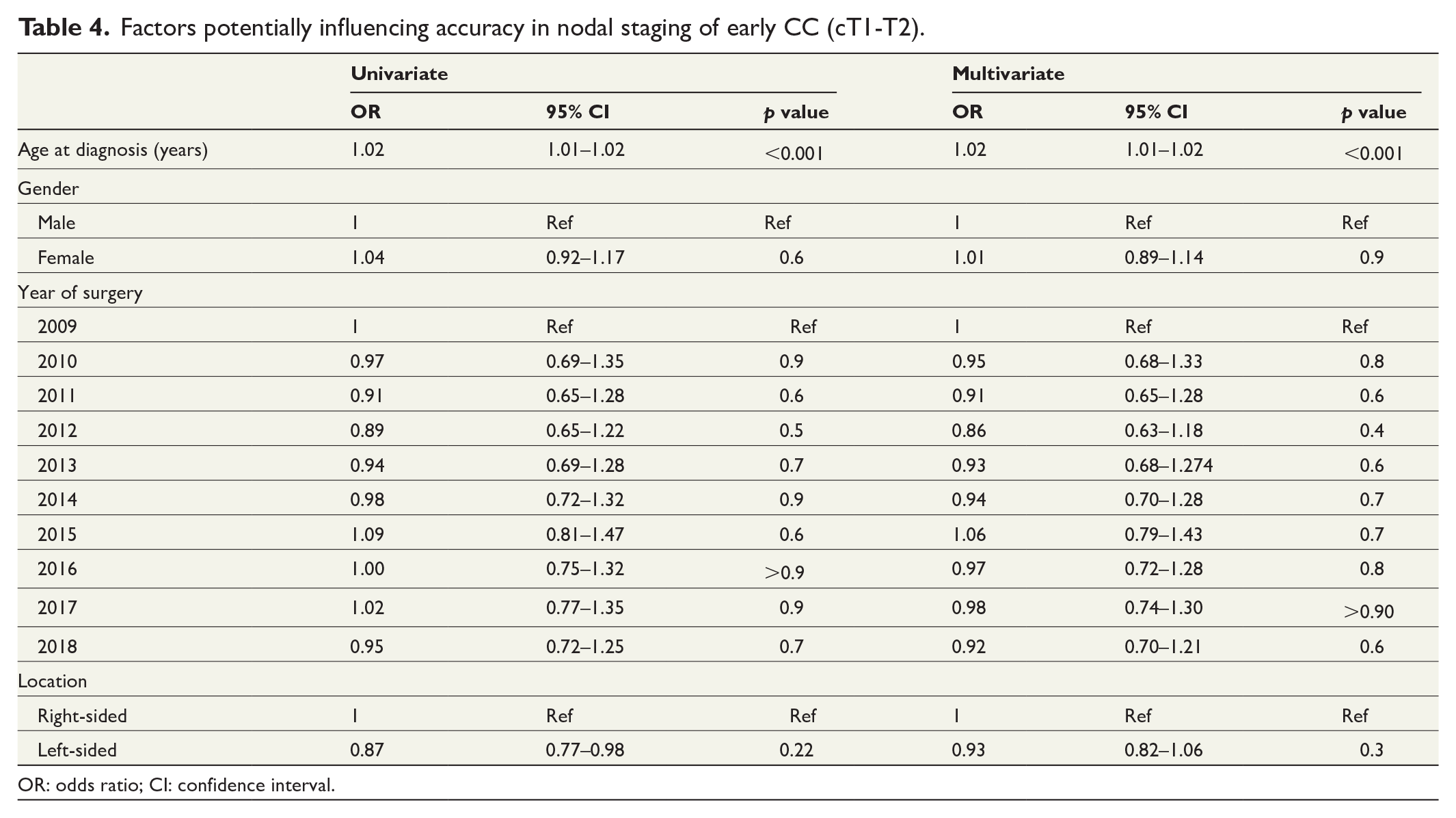
OR: odds ratio; CI: confidence interval.
Baseline characteristics pT1 group
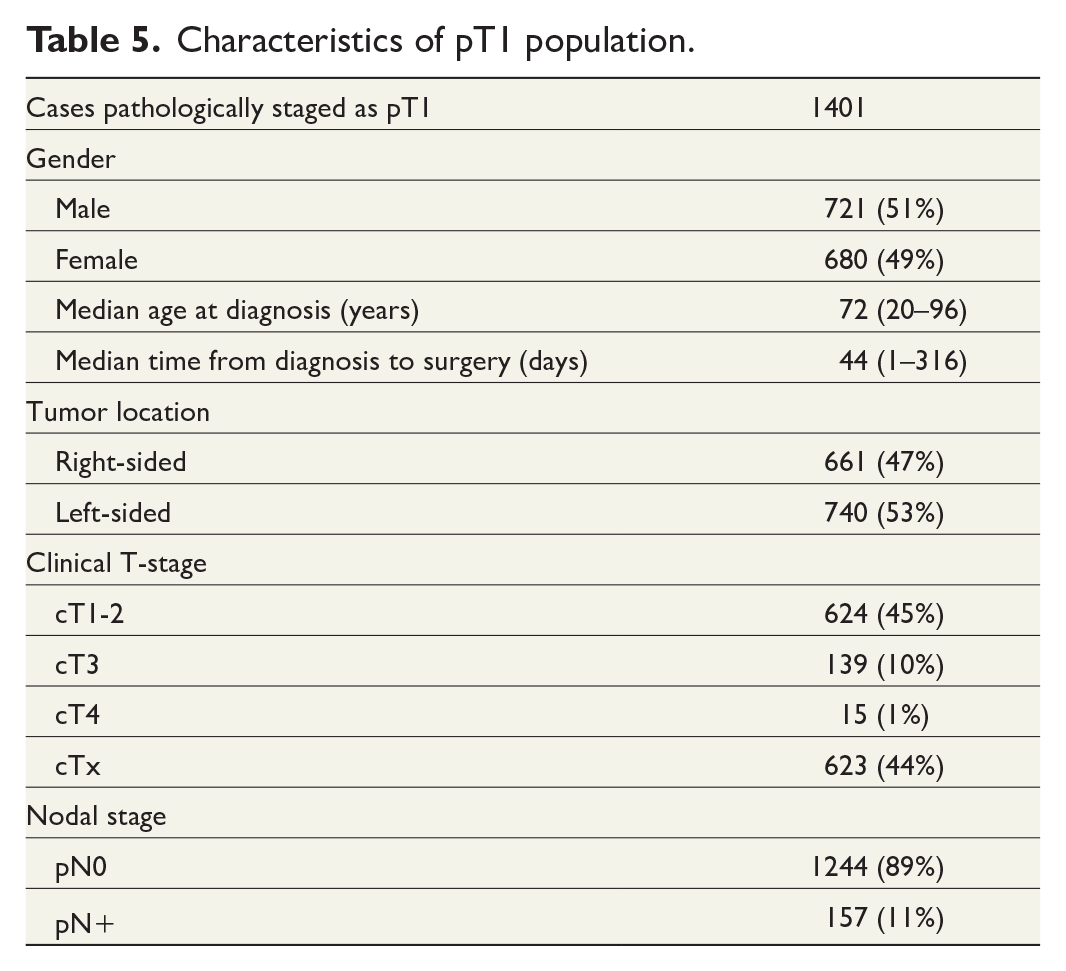
A total of 2056 patients with surgically resected pT1 CC (regardless of clinical stage) were identified in the SCRCR. After excluding 655 patients, 1401 remained for analysis and constitute the pT1 group in the study population (Fig. 2). The median age in the pT1 group was 72, and 680 patients were (49%) women (Table 5).
Inclusion of pT1 cases.
Characteristics of pT1 population.
T-stage accuracy in pT1 group
CT staged 624 (45%) of the 1401 patients with pT1 CC correctly as cT1-2, while 154 (11%) were overstaged as cT3 or cT4 and 623 (44%) were undeterminably staged as cTx (Table 5).
N-stage accuracy in pT1 group
In total, 157 (11%) of patients with pT1 CC had LNM on pathological examination and the majority of these, 105 (67%) were staged as cN0, while 23 (15%) were staged as cNx. Thus, 29 (18%) of the 157 pN+ patients were correctly staged as cN+ by CT. Concomitantly, CT staged 109 patients as cN+, whereas the majority 80 (73%) were pN0 (Table 6). Sensitivity and specificity were 22% and 93%, respectively. PPV, NPV, positive likelihood ratio, and negative likelihood ratio were 27%, 91%, 2.97, and 0.91, respectively.
Accuracy of CT in staging nodal metastases in pT1.
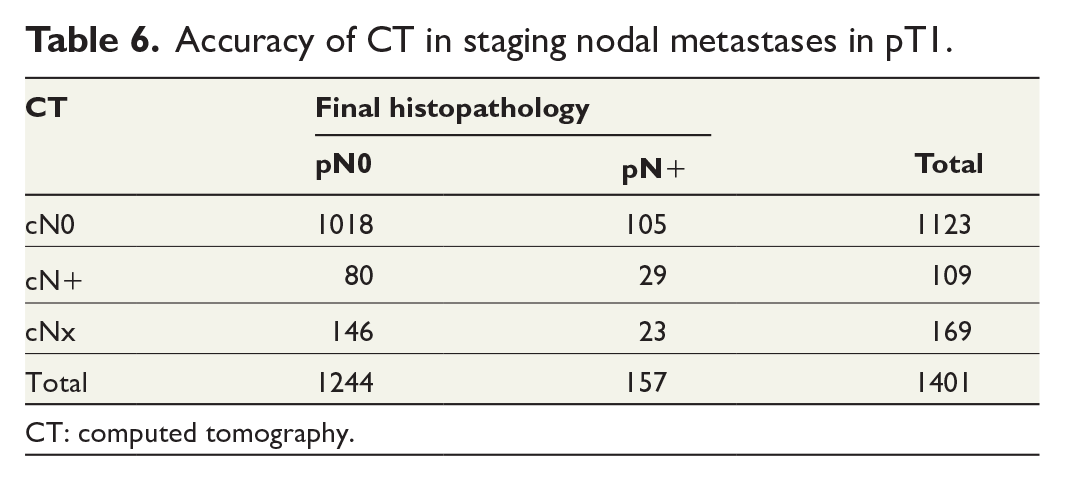
CT: computed tomography.
Combined T- and N-staging in early CC
A total of 3960 patients were staged by CT as cT1-2 cN0. Histopathology revealed that 2480 (63%) of these patients were understaged and either pN+ or pT3-4. In total, 1480 (37%) of the 3960 cT1-2 cN0 patients were correctly staged, comprising 512 (35%) pT1 pN0% and 968 (65%) pT2 pN0. In all, 1244 of the 1401 pT1 cases were pN0 and, 512 (41%) of these were correctly staged preoperatively as cT1-2 cN0.
Discussion
In this large nation-wide cohort study, we investigated T- and N-staging accuracy in early CC performed with CT from a clinical perspective. We found that CT-based T-staging of early CC was gravely inaccurate with a substantial risk of understaging pT3 and pT4 tumors as cT1-2 as well as over- and undeterminably staging pT1 tumors. In addition, CT-based N-staging was also highly inadequate with most patients staged as cN+ in fact being pN0 while the majority of patients with pN+ were staged as cN0.
Eligibility for local resection is ultimately decided by endoscopic findings such as morphology, size, and surface pattern, although pre-therapeutic CT plays an important role in staging potential candidates for local resection. It is well established that CT cannot differentiate T1 from T2, and the two stages are, therefore, often combined as cT1-2. Herein, we found a PPV of 40% for the CT-acquired cT1-2 stage. Thus, the majority of patients staged as cT1-2 were understaged and in fact pT3 or pT4. Our findings are within the lower range of previous studies, reporting PPVs of 33%–95% and overall T-stage accuracies of 58%–94%. However, these studies are in general of limited size15–18 and many focus on radiologic methodology.19,20 It is also important to note that most previous studies investigate CT accuracy in all T-stages and staging of early CC might be more challenging. Dehal et al. 21 found that early CCs were more inaccurately staged compared with more advanced stages with 46% of cT1 and 23% of cT2 tumors being understaged. In addition, a recent Danish population-based study by Olsen et al. 12 found that 56% of patients staged as cT1-2 were pT3 or pT4, a finding which is almost identical to our results. Nevertheless, investigation of CT obtained cT1-2 stage does not account for possible misclassification of pT1 tumors, and we, therefore, included analyses of CT-staging on all patients with pT1 CC. Interestingly, we found that merely 45% of pT1 tumors were correctly staged as cT1-2, whereas 10% were overstaged as cT3 and 44% were undeterminably staged as cTx.
Furthermore, we found that more than half of cases staged as cN+ were pN0. Conversely, most patients (72%) with N+ disease were erroneously staged by CT as cN0. Our findings are within the wide span of previous studies, reporting N-stage accuracies ranging from 44% to 90%.9,17,21–23 Notably, the large variation in CT-acquired N-staging found in the literature is possibly explained by a lack of standardization of CT protocols. Nevertheless, our findings support previous conclusions in the literature regarding CT N-staging as unreliable, one recent study even describing it as a “flip of the coin.” 24
When combining the results of T and N staging in early CC, we found that a minority of patients (41%) with pT1N0 disease, potentially curable by local resection, were correctly staged as cT1-2N0 preoperatively. The main reason for the low staging accuracy of pT1N0 tumors was the large proportion of cTx staged pT1 tumors, which does not disqualify patients from local resection. However, 79% of patients staged as cT1-2 cN0 were understaged and in fact pT3-4 or N+. Hence, it is primarily the risk of understaging locally advanced tumors and N+ cases that severely hampers the clinical use of CT when allocating patients to local resection. On that note, numerous studies are currently investigating the role of preoperative chemotherapy in more advanced stages of CC, which relies on correct preoperative CT staging.25–28 Although we have not investigated CT-acquired cT3 and cT4 stages, our study demonstrates the inability of CT in discriminating early CC from more advanced stages as well as N0 from N+. Thus, our findings raise concerns about treatment algorithms based on CT-acquired clinical stage, with the risk of both over- and undertreating patients.
Interestingly, left-sided tumor location was associated with increased T-stage accuracy in uni- and multivariate regression analyses. This might be explained by morphological differences in left- versus right-sided CC with sessile and pedunculated tumors being more frequent in left-sided CC29,30 and possibly easier to detect on CT. In addition, low age was associated with increased T-stage accuracy whereas high age was associated with increased N-stage accuracy in uni- and multivariate regression analyses. We have no rational explanation for these findings, and we have not been able to find any support or explanation in the literature. Nevertheless, it is important to note that ORs for both tumor location and age were very close to one and the clinical importance of these findings is thus questionable.
Our study is strengthened by the large population-based sample size and prospectively collected data, potentially mirroring the clinical aspects of CT obtained CC staging better than in a controlled setting. The study contains certain limitations comprising lack of information on CT protocols and equipment as well as not including benign cases, potentially overstaged by CT as CC, disenabling calculations of cT1-2 sensitivity and specificity. Furthermore, we did not include patients with T1 CC treated with endoscopic resection, which is another limitation. The reason for this was that endoscopic cases lack information on pN, and the aim was to analyze both T- and N-staging performed with CT. In addition, body mass index (BMI), which potentially could influence CT accuracy, was not included as a factor in the regression analyses since the coverage for BMI was too low, constituting another limitation.
In conclusion, our results show that CT staging of early CC is inadequate with a substantial risk of understaging locally advanced tumors as cT1-2 and pT1 CC as cTx, in addition to unreliable nodal staging. Based on our findings, CT-based staging of early CC should be interpreted with great caution and not used to decide treatment strategies.
Footnotes
Correction (January 2023):
This article has been updated with minor textual changes since it was published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Crafoord Foundation (20210916). Lions Research Fund Skåne (2021-11)
