Abstract
Background and objective:
The reported rate of surgical site infections (SSIs) in breast cancer surgery varies widely in previous literature. The use of antibiotic prophylaxis is controversial but recommended by several guidelines. The aim of this study was to evaluate the efficacy of routine antibiotic prophylaxis in patients with breast cancer undergoing mastectomy.
Methods:
In this retrospective single-institution study, we reviewed 1413 consecutive female breast cancer patients who underwent mastectomy and/or axillary lymph node surgery between years 2012 and 2019. Prophylactic antibiotics for all patients undergoing mastectomy was introduced in our hospital in 2016 and before that the prophylaxis was prescribed individually on surgeons’ preference. All patient records for 30 postoperative days were evaluated in detail and all SSIs were recorded. The rate of SSIs was compared between patients who received antibiotic prophylaxis and those who did not. A multivariate logistic regression model was used to define the odds ratio (OR) for the efficacy of antibiotic prophylaxis.
Results:
A total of 335 patients underwent mastectomy without antibiotic prophylaxis and 1078 with prophylaxis. The rate of SSIs was 6.9% in patients who received prophylaxis and 6.3% in patients without prophylaxis (p = 0.70). The rate of SSIs was similar before and after the introduction of regular antibiotic prophylaxis and there was no difference in any of the patient subgroups investigated. In multivariable logistic regression analysis, the OR for antibiotic prophylaxis was 1.04 (95% CI: 0.62–1.73, p = 0.88).
Conclusions:
Routine use of antibiotic prophylaxis did not reduce the rate of SSIs in mastectomy. Unselective antibiotic prophylaxis for all patients does not seem mandatory in mastectomy.
Context and Relevance
The reported rate of surgical site infections in breast cancer surgery varies widely in literature. Most previous trials have considerable heterogeneity in patients and procedures, and many trials have been conducted in the 1990s, after which there has been many kinds of advancement regarding the breast cancer surgery. The evidence of the efficacy of antibiotic prophylaxis in clean breast surgery is controversial.
The present study of 1413 patients shows no difference in the rate of surgical site infections between mastectomy patients receiving and not receiving antibiotic prophylaxis in any of the investigated patient groups. We conclude that antibiotic prophylaxis does not seem to reduce the rate of SSIs in mastectomy, and it may not be warranted in the procedure.
Introduction
Approximately one of eight women in Western countries will develop breast cancer (BC) during their lifetime. 1 Despite BC surgery has evolved rapidly during past decades, and most patients are nowadays treated with breast conserving surgery (BCS), mastectomy is still frequently needed in patients with large tumor-to-breast ratio or contraindications to adjuvant radiation therapy (RT). Although BC surgery is considered clean surgery, surgical site infection (SSI) is the most common complication of mastectomy. 2 In all BC surgery, the rate of SSI is 1%–15% and in mastectomy even higher, 3%–41%.2–6 Thus, the risk of SSI seems to be higher than in most other clean surgical operations (1.5%–3.4%).7,8
Complications after BC surgery lead to increased morbidity, delayed adjuvant treatment, and increased cost of care.9,10 It has been shown that delays in adjuvant therapy may result in inferior oncological outcomes in patients with BC. 11 Therefore, all measures to decrease the number of complications are needed. Surprisingly, there is a limited amount of published data regarding the incidence of postoperative complications in BC surgery. 2
Surgical antibiotic prophylaxis (SAP) is widely used to reduce intraoperative wound contamination and to minimize the risk of SSI. The efficacy of SAP is proved in many procedures involving contamination, but in most clean surgery, the indication is not clear. 12 In contrast, unnecessary use of SAP exposes the patients to the risks of antibiotic side effects, such as allergic reactions and Clostridium difficile infection, and it also contributes to the emergence of antimicrobial resistance. 13 Thus, SAP should only be used when it is shown to be effective. It has been shown that in some clean procedures, SAP is more frequently used than it is indicated.14,15 Several randomized controlled trials (RCT) have been conducted to investigate the efficacy of SAP in BC surgery, but the trials have been highly heterogeneous and produced conflicting results, as some trials have found no efficacy16,17 and some have proposed a remarkable decrease in the rate of SSI.18,19
In our hospital, SAP for all patients undergoing mastectomy was introduced in April 2016, following several international guidelines on antibiotic prophylaxis. 3 Prior to April 2016, SAP was used according to surgeon’s preference based on the patient’s estimated individual risk for SSI.
The purpose of this study was to evaluate the efficacy of routine SAP in mastectomy by comparing the rate of SSI before and after introducing regular SAP and comparing the patients receiving and not receiving the SAP.
Methods
In total, 1423 consecutive female BC patients undergoing mastectomy without immediate breast reconstruction, in the Department of Plastic and General Surgery, Turku University Hospital, Turku, Finland, between 1 June 2012 and 31 December 2019 were reviewed in this retrospective study. Patient information was acquired from the Auria Clinical Informatics Register and electronic patient records. For 30 postoperative days, the number of SSIs, any unplanned returns to care (RTC), and rehospitalizations for SSI were recorded.
All patient records were acquired for 30 postoperative days, including the information of any laboratory tests related to SSI (C-reactive protein (CRP), leukocyte count, bacterial culture samples) and information from the Hospital District Follow-up Register for Infection and Antibiotic usage (SAI Register). The information of any antibiotic prescriptions was acquired from the national Prescription Center. The information acquired from all different sources was scrutinized to ensure they all produced similar results and that the data were accurate and comprehensive.
No patient group was excluded from the study, except those with missing data. Information of previous ipsilateral BC and RT was recorded. If the patient had a previous BCS and the mastectomy was performed due to oncological reasons (positive or close resection margins), it was recorded. Patients having neoadjuvant chemotherapy were ensured to have a normal neutrophil count before the operation.
All SSI diagnoses were re-evaluated on the basis of the Centers for Disease Control and Prevention (CDC) and National Healthcare Safety Network (NHSN) Criteria.20,21 The diagnosis was considered SSI, when any of the following criteria was met: (1) purulent drainage from the incision or puncture; (2) organisms isolated from aseptically obtained culture of fluid or tissue; (3) deliberate opening of the incision by a surgeon in patients having either tenderness, localized swelling, redness, or warmth; or (4) diagnosis of SSI by surgeon or attending physician.
The research protocol of the study was approved by the Hospital District of Southern Finland (T218/2019).
Surgical procedure
SAP was prescribed for all patients undergoing mastectomy after April 2016. Before 2016, SAP was given according to surgeons’ preference based on the patient’s estimated infection risk. When the prophylactic antibiotic was used, a single dose was administrated during the induction of anesthesia, within 60 min before surgical incision. If there were no contraindication, cefuroxime 1.5 g was given intravenously. For cefuroxime, intraoperative redosing is recommended when the duration of the operation exceeds 4 h, but none of the operations surpassed this limit. In case of contraindications for cefuroxime, the secondary choice for SAP was clindamycin 600 mg intravenously.
Most patients were operated using an ultrasonic SonoSurg® (Olympus Medical Instruments, Tokyo, Japan) instrument. The length of operation was recorded from the start of the incision to the completion of the skin closure. Sentinel node biopsy (SNB) was performed, if no axillary lymph node metastases were detected clinically or in preoperative ultrasound examination. Axillary lymph node dissection (ALND) was performed, if nodal metastases were detected preoperatively or intraoperatively. If mastectomy was a reoperation after BCS due to insufficient margins, axillary surgery had been performed with the initial operation in most patients.
The surgeon was considered to be experienced when one had performed more than 50 mastectomies overall.
Intracutaneous sutures were used in wound closure. A single drain was applied (two for bilateral mastectomies). The removal of the drain was instructed on the sixth postoperative day, or earlier if the discharge from the drain was less than 80 mL/day. The postoperative control was instructed 2–3 weeks after the operation.
Treatment of SSIs
The SSIs were usually treated with intravenous antibiotics combined to repeated punctures of the purulent drainage. Incisions were avoided. Patients requiring hospitalization were discharged when the fever had subsided, the leukocyte count was normalized, and the CRP value was fallen below level of 100 mg/L. Patients with superficial infections and low CRP and leukocyte counts were treated as outpatients, but in such cases, the antibiotic treatment was usually initiated in the emergency department with a single dose of intravenous cefuroxime, and most often the antibiotic treatment was continued with cefalexin 500 mg three times a day for a week.
Statistical analysis
The data were analyzed using JMP 15 Pro (SAS Institute Cary, North Carolina, USA) analysis software. The incidence of SSI and other adverse events was compared between patients receiving (SAP) and not receiving (no-SAP) antibiotic prophylaxis. A case–control comparison was made between no-SAP patients and a matched control group. To form the control group, a Visual Basic tool was programmed to pick the most matching patients from the SAP group for each patient in no-SAP group.
The comparisons were performed with the chi-square test for categorical variables and Student’s t-test for continuous variables with normal distribution and the Mann–Whitney U-test for variables not normally distributed. A two-tailed test with the significance level of 5% was considered significant. In subgroup analysis, the odds ratio (OR) for SSI was determined in relation to SAP versus no-SAP for each subgroup separately.
The variables having a p-value less than 0.20 in univariable analysis were included in a multivariable logistic regression analysis. In analysis regime, the least meaningful variable was removed, and the analysis was repeated until only variables with statistical significance (p < 0.05) remained. The analysis was repeated with SAP factor included to ensure that similar results were achieved. As a result, the OR for SSI in SAP versus no-SAP groups was defined.
Results
An inquiry to Auria Clinical Informatics Register yielded information of 1423 patients. However, 10 patients were missing the information of SAP, leaving 1413 patients eligible for the analysis. Mastectomies were performed by 26 surgeons. In total, 706 patients underwent mastectomy prior to the implementation of the regular SAP, and 707 patients after that (Table 1). Before the regular SAP was introduced, 47% (330/706) patients received SAP while 53% (376/706) did not (Table 2). In case–control comparison, a highly similar patient group “Matched SAP” was acquired (Table 2).
Demographics of the patients operated before and after the introduction of the regular SAP and for all patients receiving SAP and not receiving SAP, respectively.
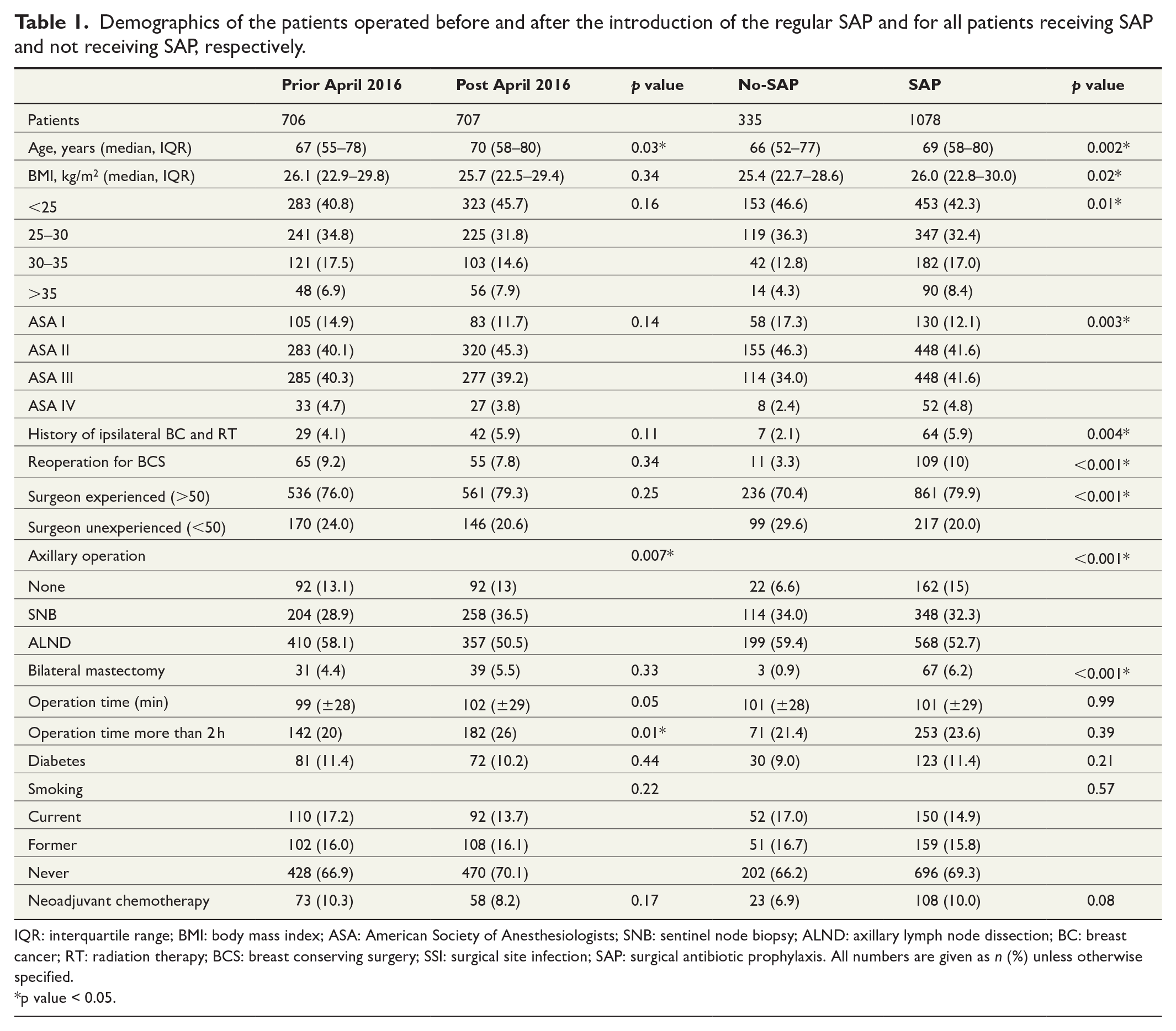
IQR: interquartile range; BMI: body mass index; ASA: American Society of Anesthesiologists; SNB: sentinel node biopsy; ALND: axillary lymph node dissection; BC: breast cancer; RT: radiation therapy; BCS: breast conserving surgery; SSI: surgical site infection; SAP: surgical antibiotic prophylaxis. All numbers are given as n (%) unless otherwise specified.
p value < 0.05.
Patients categorized according to the SAP versus no-SAP prior to April 2016 and matched control patients in SAP group (p-value in comparison to no-SAP group).
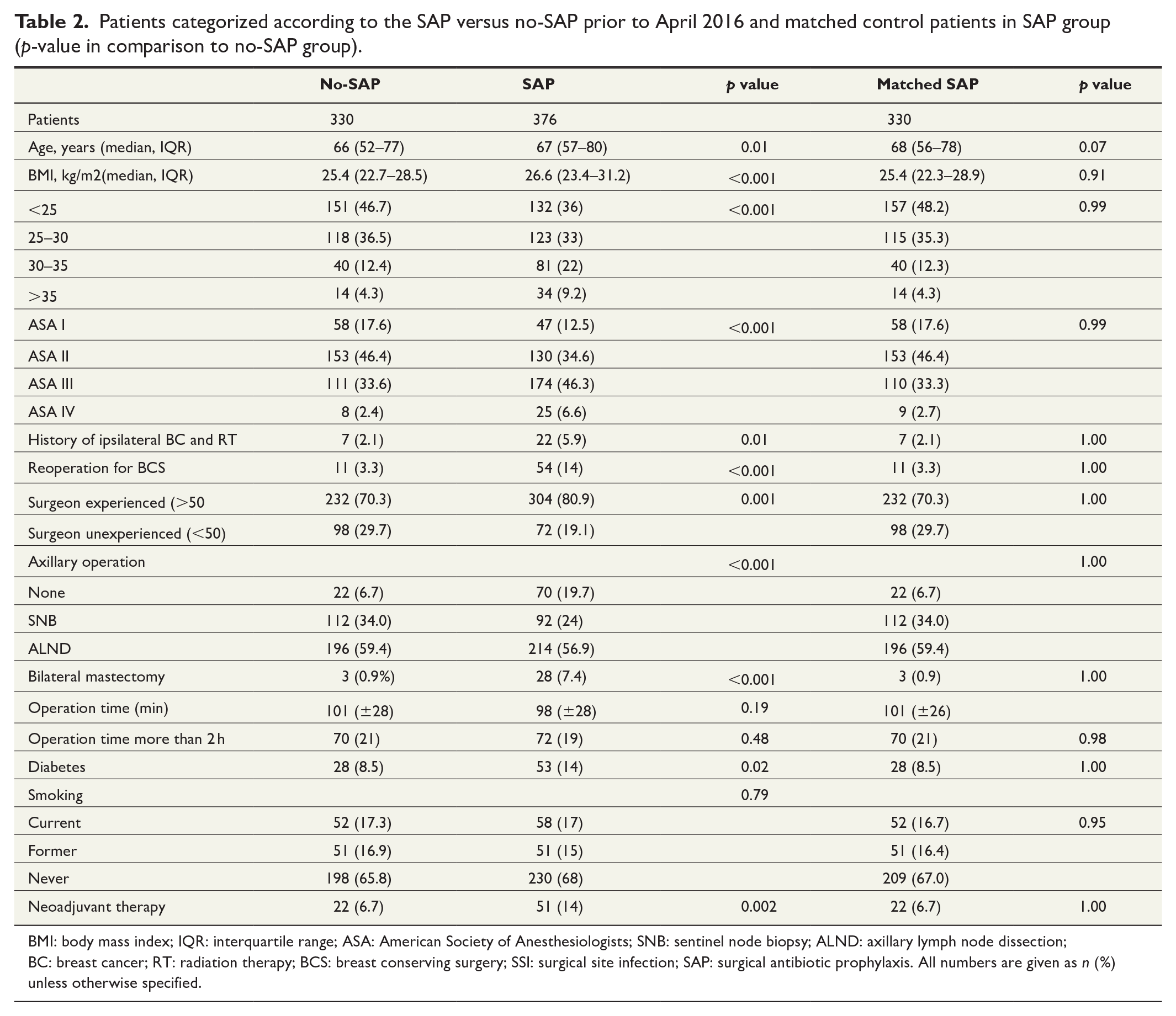
BMI: body mass index; IQR: interquartile range; ASA: American Society of Anesthesiologists; SNB: sentinel node biopsy; ALND: axillary lymph node dissection; BC: breast cancer; RT: radiation therapy; BCS: breast conserving surgery; SSI: surgical site infection; SAP: surgical antibiotic prophylaxis. All numbers are given as n (%) unless otherwise specified.
After the introduction of regular SAP, five patients were operated without SAP. When the SAP was prescribed, cefuroxime 1.5 g intravenously was chosen in 927 patients (86%). In case of contraindication for cefuroxime, intravenous clindamycin was used in 134 patients (12%). Two patients had levofloxacin, three patients had penicillin, and one patient had cefalexin. In 11 cases, the SAP was given, but the administered antibiotic was not recorded. In patients receiving clindamycin, the rate of SSI (7.5%, 10/134) was similar to the patients receiving cefuroxime (6.7%, 62/927, p = 0.74).
Patients operated before introducing the regular SAP were slightly younger than the patients operated after it. The number of operations lasting for more than 2 h was higher in the patients operated after April 2016 due to the longer examination time of frozen section study.
The patients having SSI and not having SSI are compared in Table 3. The patients having SSI were detected to be more often obese, having more often history of BC treatment including RT and that the operation time was longer in average. This is associated with more patients having SSI in ALND group, although it was not shown to be a risk factor for SSI in comparison of axillary procedures. When the operating time within a specific axillary procedure group was investigated, the operating time was similar in patients with and without SSI.
Patients having SSI and not having SSI, respectively. All numbers are given as n (%) unless otherwise specified.
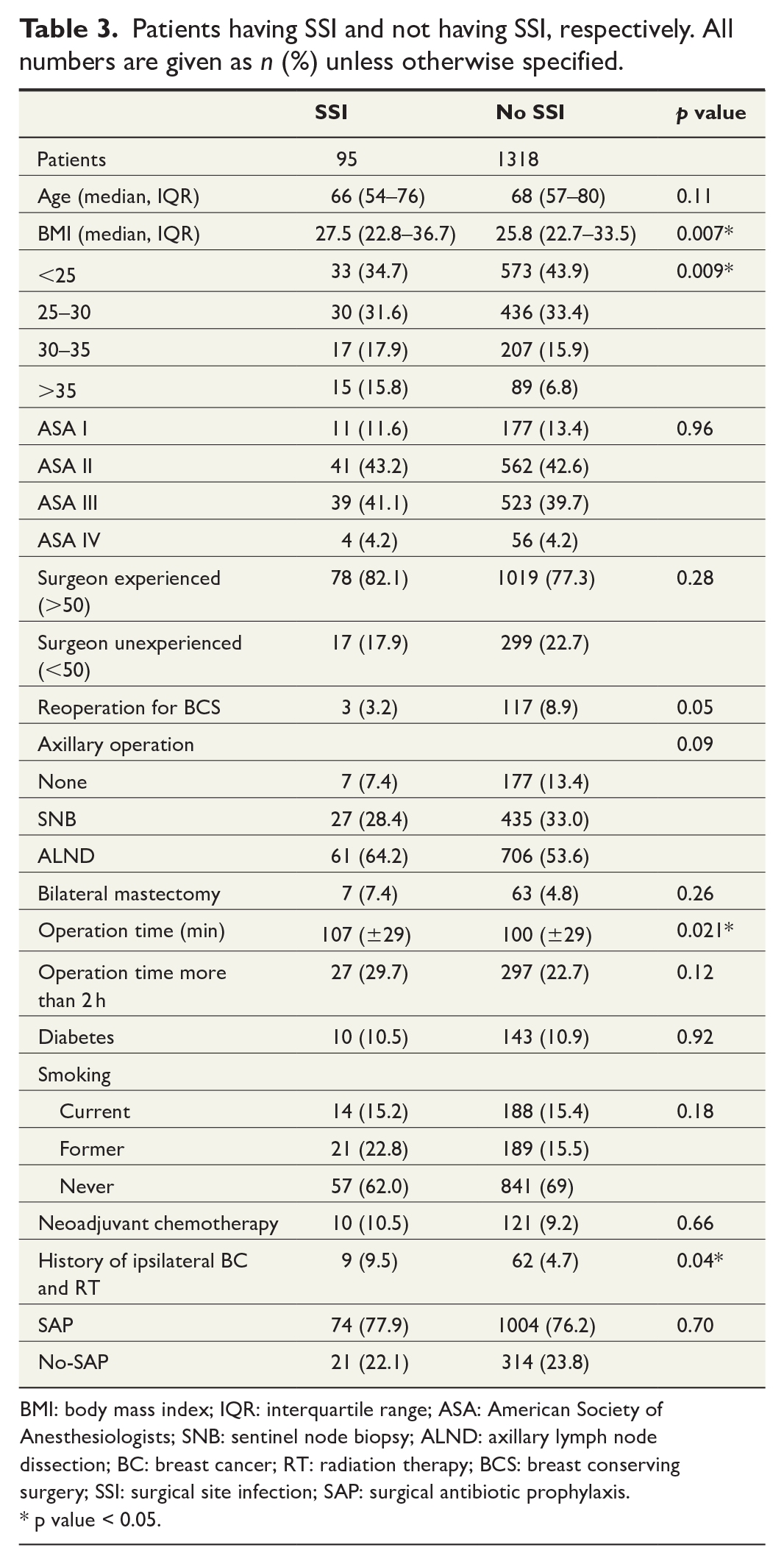
BMI: body mass index; IQR: interquartile range; ASA: American Society of Anesthesiologists; SNB: sentinel node biopsy; ALND: axillary lymph node dissection; BC: breast cancer; RT: radiation therapy; BCS: breast conserving surgery; SSI: surgical site infection; SAP: surgical antibiotic prophylaxis.
p value < 0.05.
A subgroup analysis was performed to the factors usually suggested to be risk factors for SSI. The OR for SSI with or without SAP in each subgroup was determined, but none of the values was of a statistical significance (Table 4).
Subgroup analysis of the patients having SSI.
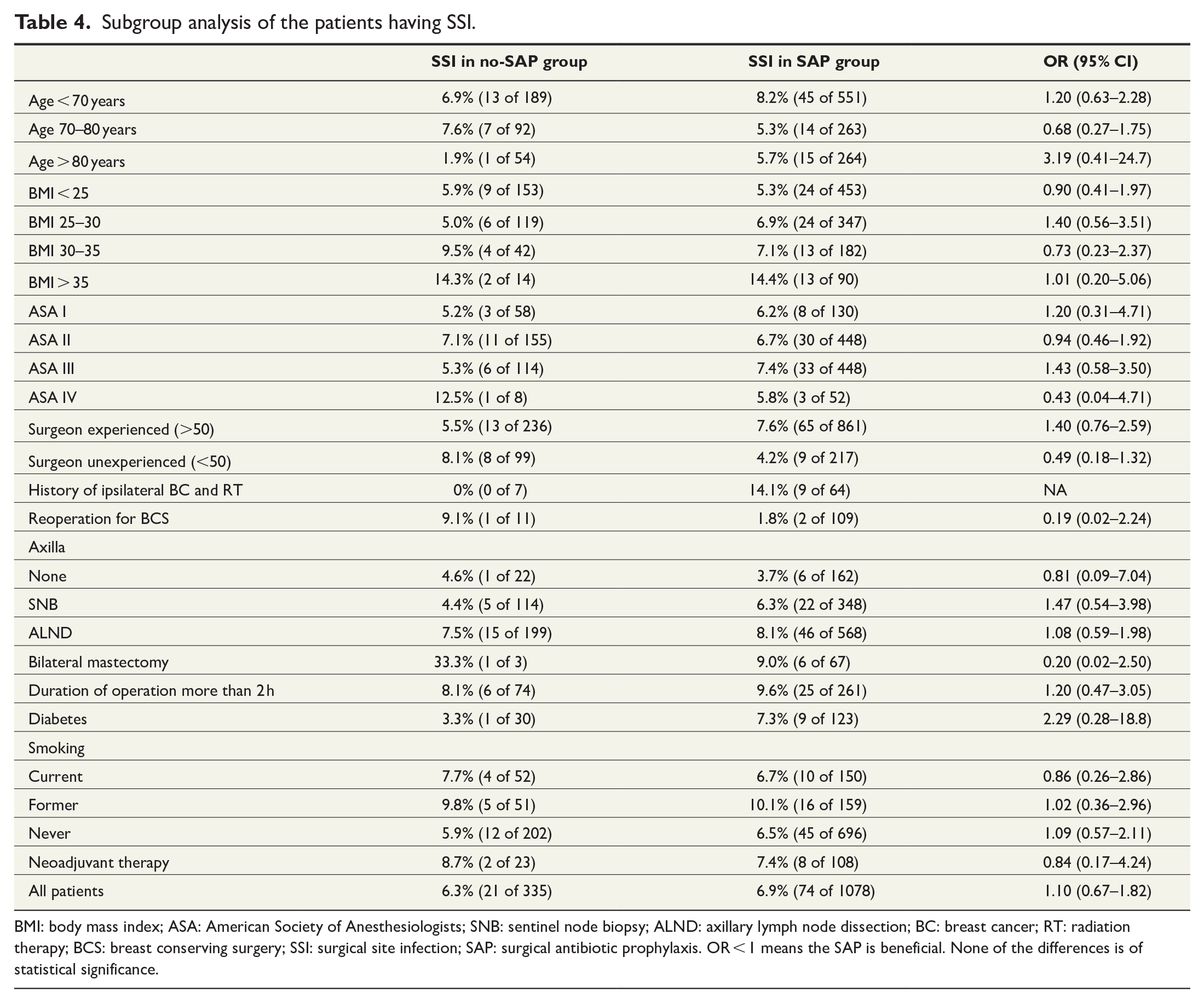
BMI: body mass index; ASA: American Society of Anesthesiologists; SNB: sentinel node biopsy; ALND: axillary lymph node dissection; BC: breast cancer; RT: radiation therapy; BCS: breast conserving surgery; SSI: surgical site infection; SAP: surgical antibiotic prophylaxis. OR < 1 means the SAP is beneficial. None of the differences is of statistical significance.
The rate of SSIs and related adverse events are presented in Table 5. There is no difference between patients receiving versus not receiving SAP in any comparison in terms of SSI, rehospitalization or any RTC.
All unplanned RTC, SSIs, and rehospitalizations are listed in relation to SAP.
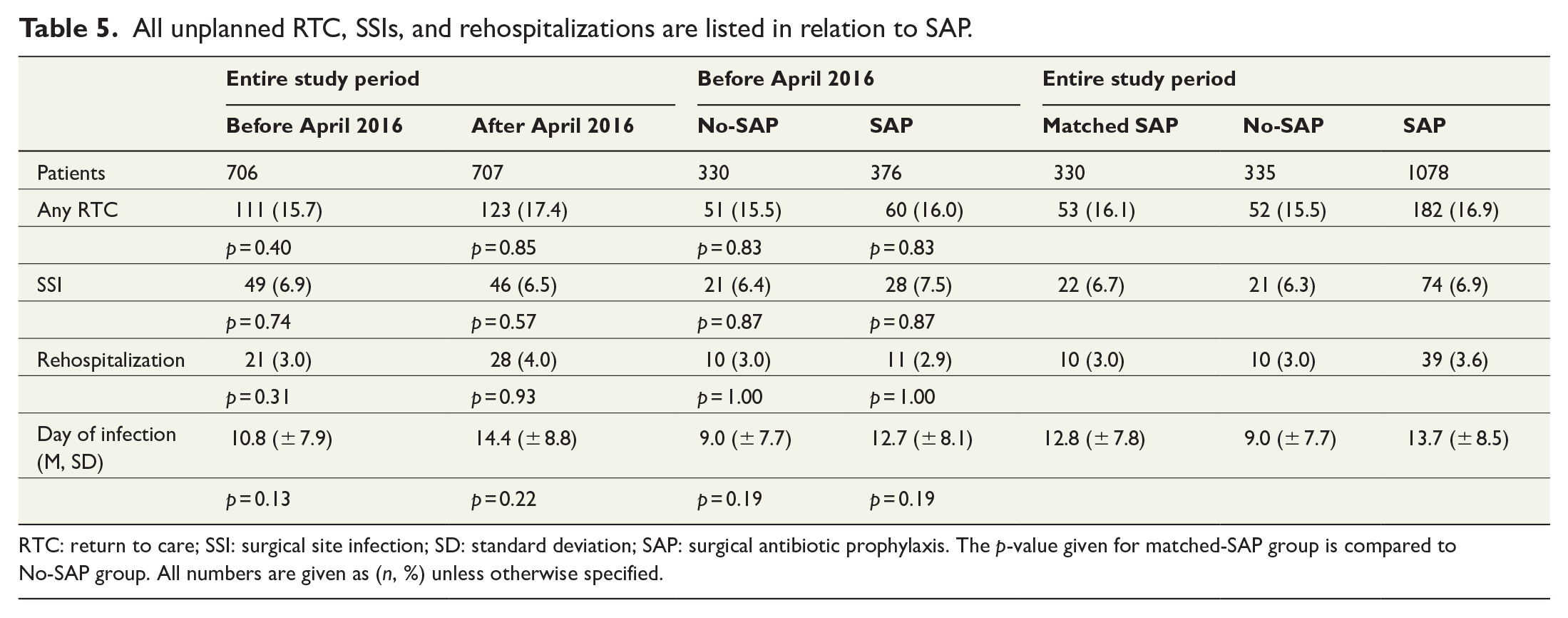
RTC: return to care; SSI: surgical site infection; SD: standard deviation; SAP: surgical antibiotic prophylaxis. The p-value given for matched-SAP group is compared to No-SAP group. All numbers are given as (n, %) unless otherwise specified.
A multivariable logistic regression analysis was performed to evaluate the efficacy of SAP. The OR for SAP versus no-SAP was 1.04 (95% CI: 0.62–1.73, p = 0.88). The risk factors for SSI with statistical significance were high BMI (p = 0.016), previous BCS, and RT (p = 0.018) and the axillary procedure performed (p = 0.023).
Discussion
The rate of SSI was found to be almost identical in all patients receiving SAP (6.9%) compared to the patients not receiving SAP (6.3%, p = 0.70), and in case–control comparison (6.4% versus 6.7%, p = 0.87). There was no change in the rate of the SSI after the regular SAP was introduced (6.5%) compared to the time before the introduction (6.9%, p = 0.74). To date, this study is the largest study to investigate in detail the patients undergoing mastectomy and considering SAP effect on SSI.
The factors associated with the risk of SSI were longer operation time, high BMI, and previous BCS with RT. All these have been suggested to be risk factors for SSI in previous literature. 22 Multiple other factors, such as older age, smoking, diabetes, preceding BCS, preceding chemotherapy, and lack of surgeon experience, have been proposed to be risk factors for SSI. 22 In this study, none of these factors were associated with the elevated risk of SSI. The patients having operation time more 2 h has previously been suggested to be a risk factor for SSI, 23 but this does not get support from this study. The operating time in average was longer in patients suffering SSI, but this was shown to be a consequence from longer operating time associated with ALND, which showed to be a risk factor for SSI in multivariable analysis. When axillary procedures were evaluated separately, operating time had no association with SSI.
The risk of SSI was detected to be highly elevated in obese patients with BMI over 35 kg/m2 (14.4%, 15/104), even though the SAP was commonly used in these patients. As obese patients frequently have a high breast volume, BCS should be a viable option in most patients in this group. Therefore, BCS should probably be recommended as a primary choice in these patients to minimize the risk of delays in adjuvant therapy.
The mean time for the occurrence of the SSI was 13 days in the SAP group and 9 days in the no-SAP group. These findings are in concordance with the previous literature, showing an onset of SSI to be 9.6–11 days in no-SAP group and 11–17 days in SAP group,17,24,25 but in this study, the difference was non-significant.
In previous literature, the reported rate of SSI after mastectomy has varied widely. There have been several RCTs considering SAP in mastectomy with highly varying results.4,16–18,22–26 Most of these trials include several types of breast surgery, not just mastectomy. In the trials giving numbers for mastectomy separately, the rate of SSI is usually 6%–19%, but rates up to 41% (16/39) have been presented. 4 However, many of these studies include a rather small number of patients. Many have considerable heterogeneity in patients and procedures and many date back to the 1990s, after which there has been many kinds of advancement regarding the BC surgery. As Amland et al. 23 state, at the time of the trial, there was no proper definition for SSI and in part, the wide variation of results probably relates to the application of different definitions for SSI.
A Cochrane review considering 10 RCTs with 2823 patients concluded that there is no clear consensus on behalf of SAP, but that the SAP probably is effective and should be recommended in BC surgery. 3 According to this review, the risk ratio (RR) for SSI after SAP is 0.67 (95% CI: 0.53–0.85), but only one of the included RCTs yielded results of statistical significance. The overall risk of SSI for all BC surgery in the review was 10.5% without prophylaxis and 7.1% with prophylaxis, exceeding the rate of what we have shown in this study and what is generally considered appropriate for clean surgical operations.7,8 These high numbers are regardless of the fact that the Cochrane review includes BCSs, which is considered to have a substantially lower risk of SSI compared to mastectomy.4,25
In addition, there have been large retrospective studies with a very low rate of SSI. Yang et al. evaluated 458 mastectomy patients and found an SSI rate of only 1.1% in SAP group and 1.2% in control group. 27 In studies based on National Surgical Quality Improvement Program (NSQIP), the rate of SSI after mastectomy is reported to be 2.3%–4.34%.2,28,29 Palubicka et al. have investigated 865 patients and report an SSI rate of 5.3%. 30
The rate of SSI seems to be highly variable, and even if the definition of SSI was varied, this is not probably the only difference between the trials. The practice around mastectomy has evolved rapidly over last decades, and it may be that all trials from the 1980s and 1990s do not correspond with the circumstances of the present day. In many hospitals, open biopsy is no more used, new energy instruments have substituted scalpels and electrocautery, intracutaneous sutures have substituted skin clips, and hospitalization time has diminished, all changes that may have effect on the rate of SSI. Therefore, it is not justified to expect that the research conducted at present day would produce similar results than the trials in the 1990s. In addition, differences in local treatment protocols and especially in basic infection-control strategies may have an effect on the results. 31 In the previous literature on the subject, there are vast differences in hospitalization time (from median 1 19 –14 days 27 ) and patient demographics (mean BMI of 30 kg/m2) 19 to median weight of the patients (57.5 kg), 27 operation time (50 17 –163 min 19 ), and the duration when the drains are removed (from 5.7 26 –14 days6,19). There is variation in the manner of skin closure, as for example, Gulluoglu et al. used skin clips on all patients. Gulluoglu et al. also had the most SSIs (65%) on a first week after the operation, 18 whereas most other studies have had the most SSI on Days 10–17.17,24,25 Many of these factors obviously could have an impact on the rate of SSI. As the results from the previous heterogeneous trials are varying, it is rather difficult to make straight comparisons between the present study and the previous ones.
Limitations of this study
The main limitation with this study was its retrospective nature. However, we do not expect this to cause significant bias to the results. The SSI after mastectomy most frequently occurs 10–14 days after the operation, and we do not think the awareness whether patients have received SAP should have had an effect to the treatment or making SSI diagnosis that late after the operation.
The electric prescription was used in private healthcare a year later (during 2014) than in the public healthcare, and therefore it is presumable that some antibiotic prescriptions prior to that time are not found in our data. Nonetheless, the data used in this study were acquired from multiple parallel sources, and the conformity of the information was ensured. Furthermore, in our country, we have a publicly funded healthcare system, and the BC treatment, especially hospitalization, is practically exclusively offered by public healthcare. Considering this, we suppose that there should not be a significant lack of information.
This study was conducted in a Western country university hospital, and thus the results cannot be generalized to apply in all different circumstances. We are waiting to see whether future research, such as recently published protocol of a prospective multicenter trial in Pakistan, 32 would produce comparable results.
Future research
We propose further research to evaluate the reasons why the rate of SSI in the literature is so varying. We suggest that BCS and mastectomy should be investigated separately, as these procedures probably present a different rate of complications. Well-conducted RTCs with homogeneous population are needed to clarify the subject. Prospective trials concerning different instruments and skin closing materials, such as antibacterial triclosan-coated sutures, could potentially yield results that may help in preventing SSIs.
Conclusion
Prophylactic antibiotics do not seem to be necessary for patients undergoing mastectomy. We suggest larger prospective study to confirm the finding. The true incidence of SSI and affecting factors remain ambiguous, and more evidence of the effectiveness of antibiotic prophylaxis in mastectomy, and other breast surgery, is still needed.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data sharing
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions. This research study was conducted retrospectively from data obtained for clinical purposes. The research protocol of the study was approved by the Hospital District of Southern Finland (T218/2019). No ethical approval was required for this retrospective study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by a grant from Vappu Uuspää Foundation. The funding source had no role in the design, conduct, analysis, or reporting of the study.
