Abstract
Background & objective:
Arterial vascular trauma bears a great risk of poor functional outcome or limb loss. The aim of this study was to analyze amputation rates of patients after vascular trauma and to perform a predictor analysis for the risk of major amputation.
Methods:
In a single-center retrospective analysis of 119 extremities treated for arterial vascular injury between 1990 and 2018 amputation rates and factors associated with limb loss were assessed. All patients were treated for traumatic vascular injuries; iatrogenic injuries were not included in the study.
Results:
During the study period, a total of 119 legs in 118 patients were treated after arterial vascular trauma. The in-hospital major amputation rate was 16.8% and the mortality rate was 2.5%. In the predictors analysis, vascular re-operation (amputation rate 53.8% vs 12%, odds ratio = 8.56), a Rutherford category ⩾IIb (25.4% vs 4.2%, odds ratio = 6.43), work-related or traffic accidents (28.2% vs 0%, odds ratio = 4.86), concomitant soft tissue or bone injuries (26.7% vs 7.3%, odds ratio = 4.64), polytrauma (33.3% vs 12%, odds ratio = 3.68), and blunt trauma (18.9% vs 0% for penetrating trauma, odds ratio = 1.64) were found to be associated with amputation.
Conclusions:
Lower extremity arterial vascular trauma was associated with a significant major amputation rate. Several predictors for limb loss were identified, which could aid in identifying patients at risk and adapting their treatment.
Introduction
Arterial vascular trauma (AVT) of the lower extremities constitutes a potential life-threatening situation in affected patients with possible permanent functional impairment or limb loss. In some patients, the combination of ischemia with concomitant soft tissue, bone, and/or venous injuries deteriorate the chance of limb salvage (LS).
Timing of the revascularization has substantial impact on outcome with declining rates of LS after 1 h of ischemia time.1 –3 In patients with severe concomitant injuries requiring surgery, a triage of the injuries has to be done, and sometimes, the decision between limb and life has to be made in favor of the patients’ life. 4 The usage of vascular shunts for bridging the time until revascularization has been reported, but routine application has not yet gained acceptance.1,5 The standard approach for treatment of AVT is open surgical repair; however, endovascular treatment is becoming increasingly popular. 6 In a retrospective analysis of the National Trauma Database in patients with AVT, endovascular treatment seemed to be a viable treatment option. However, treatment of patients with complex injuries may be challenging and open repair or a combination of endovascular and open surgery might be favored. 7
Severe trauma can also impede the postoperative management after revascularization. Infection of the soft tissue or the vascular reconstruction due to contamination, limitations of anticoagulation owing to concomitant injuries or devastating accompanying injuries themselves may result in major amputation.8,9
Material and methods
The aim of this study was to analyze the outcome of patients undergoing revascularization after AVT in terms of mortality and major amputation rates. Furthermore, a predictor’s analysis for the risk of amputation after AVT was performed. Retrospective evaluation of all patients treated for lower extremity AVT between January 1990 and December 2018 at the Department of Vascular Surgery, University Hospital of Innsbruck, a mature level I trauma center, was performed. Patients with iatrogenic vascular injury were not included in the study.
By scanning the clinical information system (Cerner KIS®, Berlin, Germany), data about the patients and the management were obtained. Data of patients primarily treated at other trauma centers were extracted from discharge and transfer letters. Patients with traumatic amputation of the extremity were not included in the study. The mangled extremity severity score (MESS) was calculated retrospectively quantifying the severity of the AVT. 10 The severity of the ischemia was categorized according to the Rutherford classification of ischemia. 11
Primary endpoint was amputation and mortality rates. Secondary endpoints were trauma-related data (trauma mechanism, type, and localization), perioperative characteristics (revascularization technique, fasciotomy details, and technique of skin closure), and the duration of hospitalization. After separation in an LS and amputation group, a predictor ranking for the risk of amputation was performed.
The study was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice guidelines. It was approved by the Medical Ethics Committees Innsbruck (1079/2020).
Statistics
All data were stored in Microsoft Excel (Microsoft; Redmond, USA). Statistical analyses were performed with SPSS 26.0 for Windows (SPSS, Armonk, NY, USA), GraphPad Prism 8.4.3 (GraphPad Software LLC, La Jolla, USA), and WEKA 3.8.4 (University of Waikato, Waikato, NZ). 12
Patients were divided into two groups: those with major amputation as the outcome (AP) and those with LS. A Mann–Whitney test was used for continuous data of both groups due to a non-Gaussian distribution; Fisher’s exact test was used for categorical variables. Invalid or missing data were not utilized for further analysis; no imputation technique was employed.
To evaluate the influence of various predictors on the occurrence of amputation, a Bayes Network with a 10-fold cross-validation was employed to generate a predictor ranking via a gain-ratio feature evaluation. Model results included correct classification rate (CCR), mean absolute error (MAE), relative absolute error (RAE), and weighted average for the true positive rate (TPavg), false positive rate (FPavg), precision (Pavg), and odds ratios (ORs) calculated from contingency tables. In case of continuous or ordinal variables, the ideal cut-off was determined using receiver-operating characteristics (ROC) curves and Youden’s J to convert into binary variables. Predictor properties reported include gain ratio, amputation rate, and OR including 95% confidence intervals (CIs). Statistical significance was considered for p-values < 0.05.
Results
Between January 1990 and December 2018, a total of 119 lower extremities were treated after AVT. The demographic data and details on the vascular trauma are given in Table 1. Patients were predominantly male (96 of 119 cases, 81%) and the mean age was 37.5 (±17.5, range 12–84) years. The majority of patients were involved in traffic accidents (n = 46, 39%) followed by sports/leisure accidents (n = 36, 30%), mainly skiing casualties. In 31% (n = 37), the type of accident leading to AVT could not be determined retrospectively. The main mechanism of injury was found to be blunt trauma in 106 cases (89%). All patients underwent open surgical reconstruction for reperfusion. On surgical exploration, transection and traumatic disruption (n = 57, 48%) were the most common types of arterial injury (Table 1). The popliteal artery (n = 56, 47%) followed by the superficial femoral artery (n = 27, 23%) were the most common sites of arterial trauma, whereas in 28 cases (24%), more than one vessel of the lower extremity was affected. Reverse interposition of a venous graft (n = 74, 62%) was the most common choice of repair. Due to long-segment vessel injury, bypass surgery was performed in 22 cases (19%). A temporary intravascular shunt for bridging ischemia was used in three patients only. Concomitant osseous injuries (n = 99, 83%) requiring surgery were found in 94 cases (79%). Concomitant venous injuries (n = 34, 29%) were predominantly reconstructed (n = 21, 18%); ligation of the vein due to extensive bleeding or disruption was performed 12 times (10%). Decompressive fasciotomy was performed in 88 (74%) extremities. Details on the fasciotomy procedures are given in Table 2. A split skin graft for closure of the fasciotomy wound was used in 12 (10%) of the patients; myocutaneous flap plastic was necessary three times (3%). Fourteen (12%) patients of other nationalities were transferred to their home countries before final wound closure after a mean hospital stay of 33.6 ± 28.5 days. Wound closure was performed 15.4 ± 14.3 days after revascularization. Major amputation was necessary in 20 (17%) patients. Below-knee amputation was performed in 12 patients, and the remaining 8 patients underwent above-knee amputation. No patient suffered limb loss after a sport or leisure accident-related trauma. In-hospital mortality was 2.5% (n = 3); on average, the patients died 29 ± 21.6 days after revascularization. Reason for death was sepsis due to extensive traumatic soft tissue damage after successful revascularization of a fully disrupted superficial femoral artery in one patient. The other two underwent revascularization of the common/superficial femoral artery for disruption; however, due to severe concomitant visceral trauma with spleen rupture and colon perforation, multi organ failure was the cause of death in both.
Details on the injury.
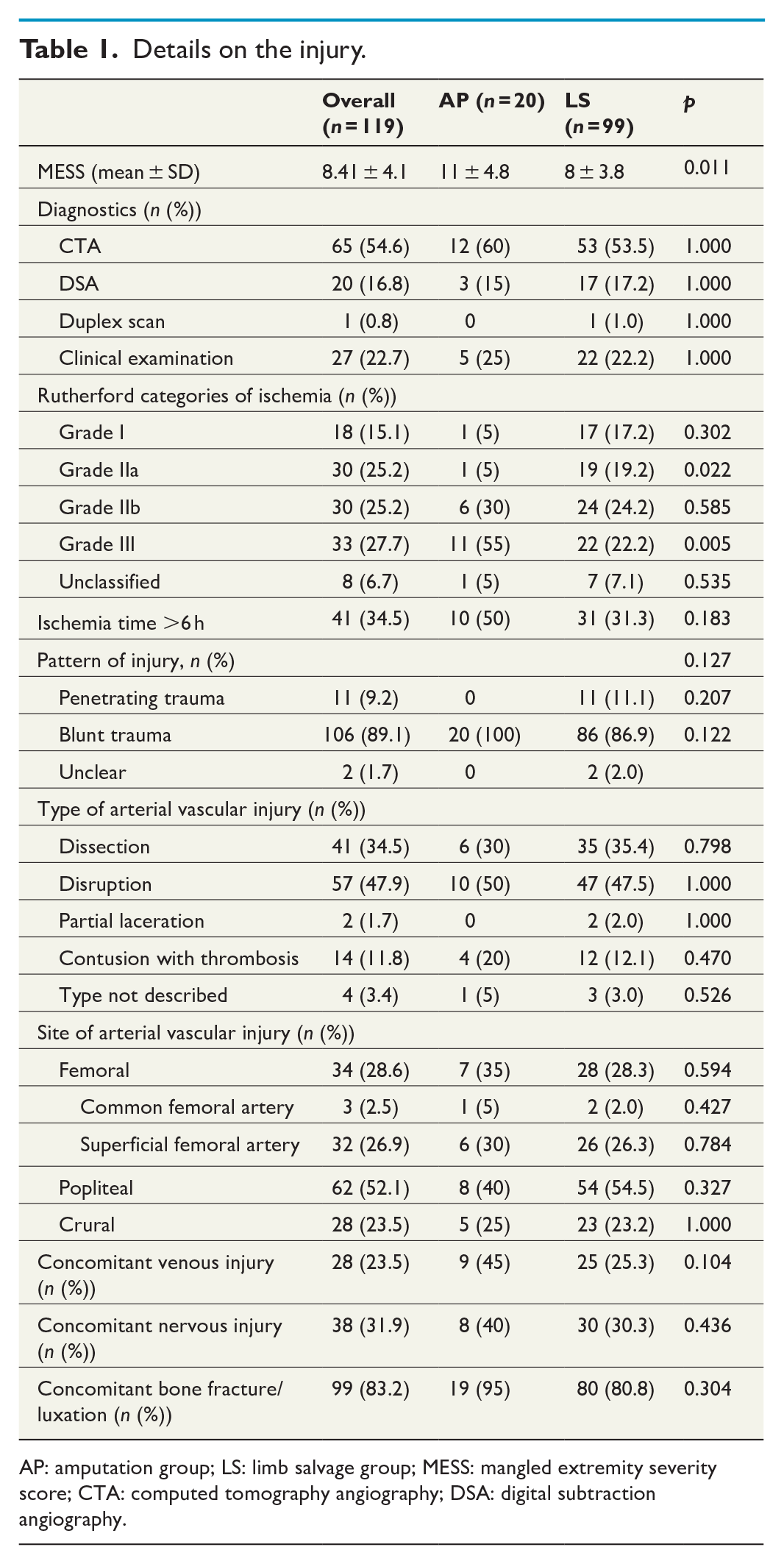
AP: amputation group; LS: limb salvage group; MESS: mangled extremity severity score; CTA: computed tomography angiography; DSA: digital subtraction angiography.
Details on the vascular repair and the postoperative course.
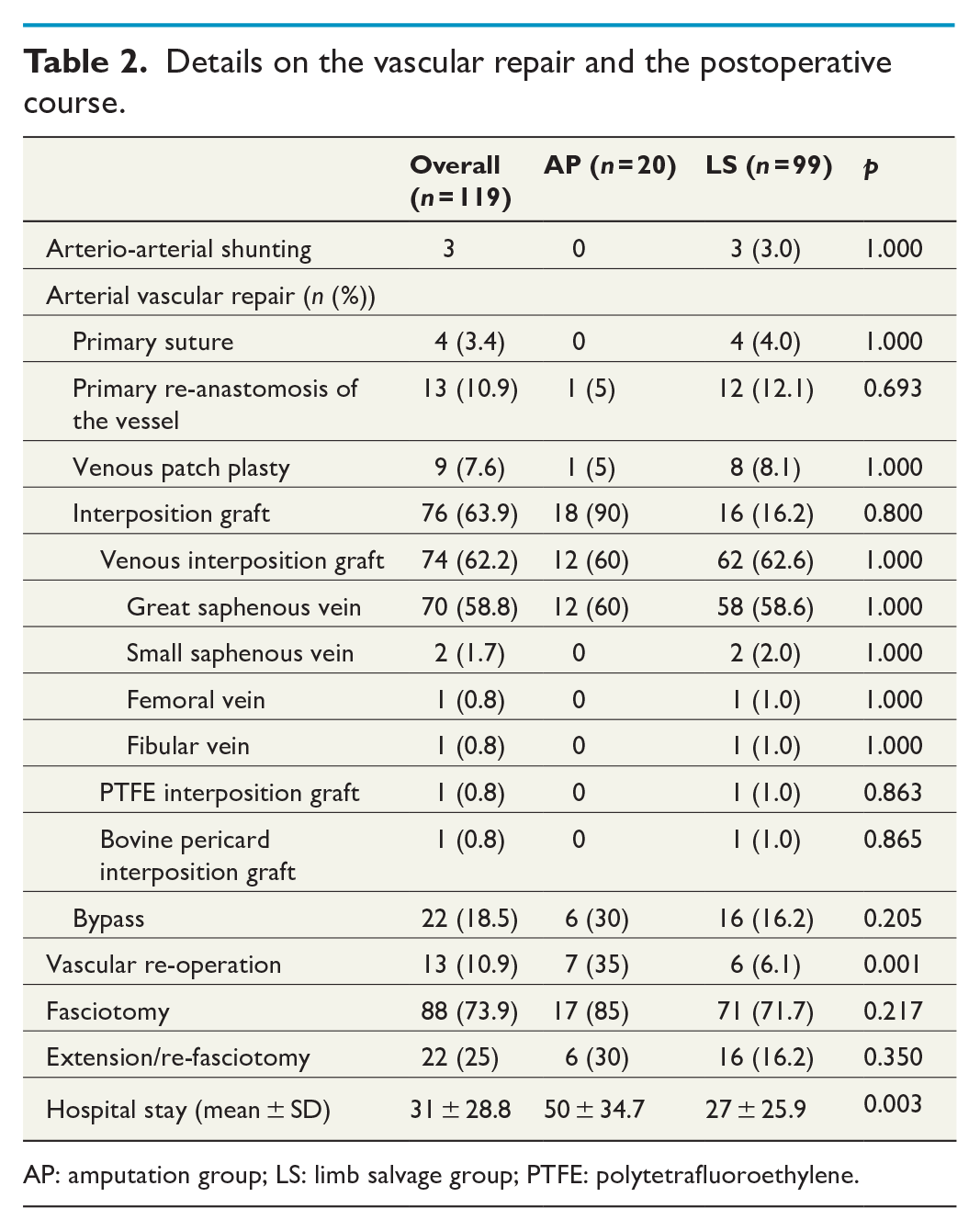
AP: amputation group; LS: limb salvage group; PTFE: polytetrafluoroethylene.
For a further subgroup analysis, the patient cohort was divided in a major amputation group (AP, n = 20) and an LS group (n = 99). There was no significant difference in regard to age (38 ± 18.1 vs 35 ± 14.3 years, p = 0.588). Women were statistically as likely to undergo amputation as men (91 vs 81%, p = 0.357). The mean length of hospital stay in the AP group was significantly longer (53.1 ± 34.1 days) compared to the LS group (27.4 ± 26.0 days; p = 0.0003).
A significant difference between the two groups was found in regard to trauma type with traffic (70%) and work-related accidents (30%) dominating in AP cases. Vascular trauma mechanism did not differ significantly between the groups with similar distributions of blunt (89 vs 100%), penetrating (10% vs 0%), and unspecified trauma (1.0 vs 0.0%) (Fig. 1B, p = 0.127). The rates of dissections (36 vs 30%), disruptions (48 vs 50%), and occlusions (11 vs 15%) were also comparable (Fig. 1C, p = 0.732). No significant difference in distribution of affected vessels was found (Fig. 1D, p = 0.442).
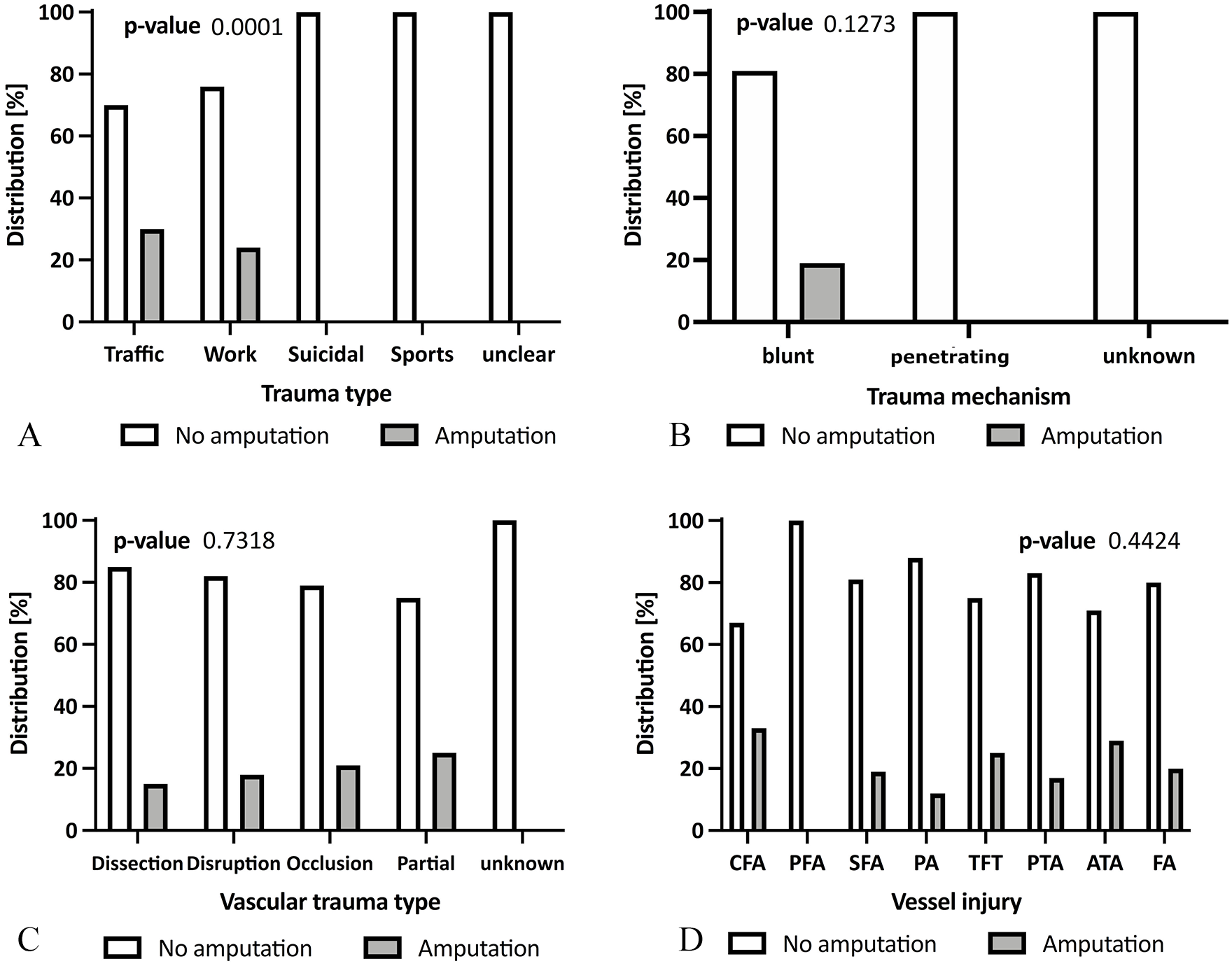
Comparison of limb salvage and amputation cases. In (A), different trauma types responsible for the injury were outlined. (B) Shows the predominance of blunt trauma in the patient cohort. In (C), the distribution of the different vascular trauma types causing the ischemia is outlined. (D) shows that the popliteal artery (AP) was the most affected vessel followed by the superficial femoral artery (SFA), posterior tibial artery (PTA), anterior tibial artery (ATA), fibular artery (FA), tibiofibular truncus (TFT), common femoral artery (CFA), and profound femoral artery (PFA).
In contrast, the Rutherford categories of ischemia at presentation were significantly different between both groups. While most patients in the LS group presented with Rutherford grade IIa (31.5%), the majority in the AP group were categorized as Rutherford IIb (32%) or III (58%) (p = 0.001). The mean MESS score was similar in the LS and the AP groups with 8.3 ± 4.1 versus 8.9 ± 3.9 points (p = 0.419).
Even though there was a higher rate of fasciotomies in the AP (85%) compared to the LS (72%) group, the difference was not significant (p = 0.217). Delayed fasciotomy (>12 h after AVT) because of deferred revascularization or belated diagnosis of compartment syndrome (CS) was found in 21% of the overall cases and was more common for LS (24%) than AP (7.1%) (p = 0.164). Patients in the AP group presented with a median ischemia time of 7.75 (6.25–22.50) h, whereas patients of the LS group underwent revascularization after a median time of 6 (5.25–8.00) h (p = 0.091).
Predictors for lower extremity amputation
The Bayes network analysis correctly classified 97 out of 119 cases (CCR = 81.5%). An overview of the ranked predictors with a gain ratio above 0.01 is given in Table 3.
Predictor ranking for lower extremity amputation.
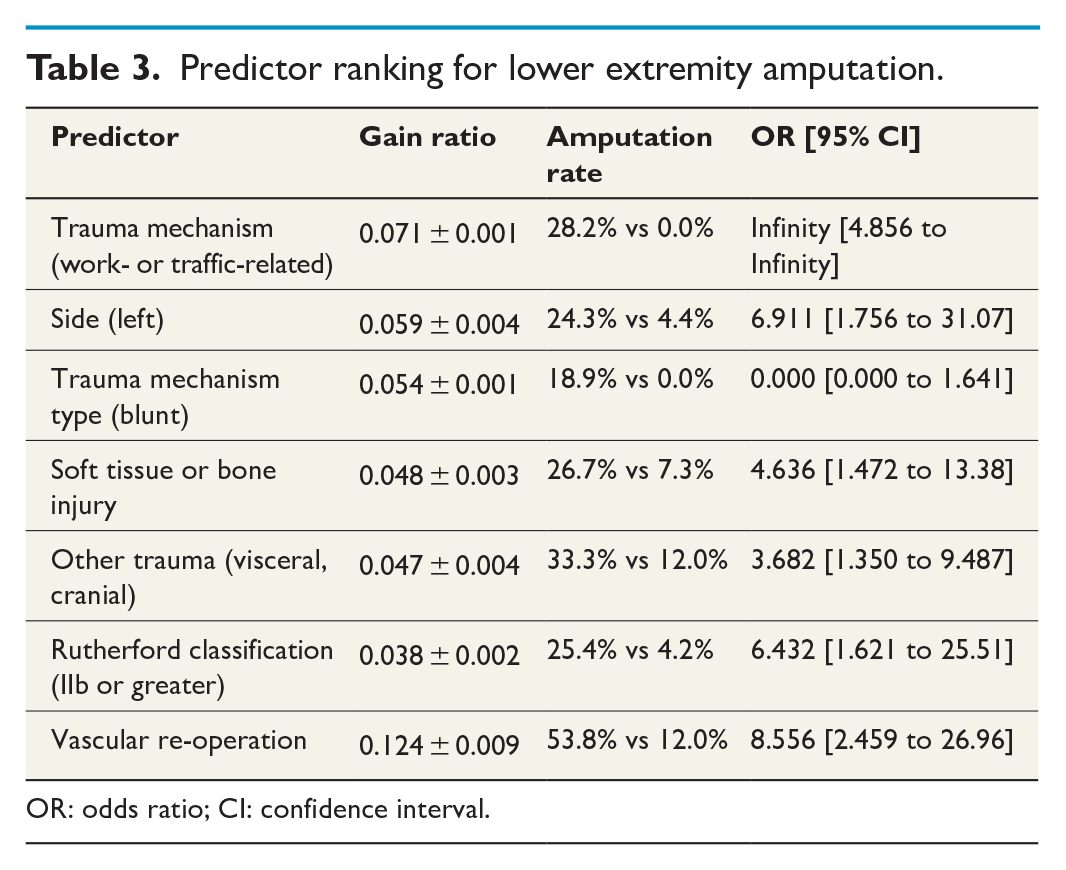
OR: odds ratio; CI: confidence interval.
The single strongest predictor was vascular re-operation (gain ratio 0.124 ± 0.0009; p = 0.001) with a significantly higher amputation rate of 53.8 versus 12.0% and an OR of 8.56. Various factors concerning trauma type and extent ranked also high: trauma type, that is, work- or traffic-related injury (gain ratio 0.071 ± 0.001; p < 0.0001), blunt trauma mechanism (0.054 ± 0.0001; p = 0.207), soft tissue or osseous injury (0.048 ± 0.003; p = 0.007) as well as trauma to other regions of the body (e.g. cranial and visceral) (0.047 ± 0.004; p = 0.017) all conferred significant predictive value. Curiously, injury side (left extremity) was also among the strongest predictors (0.054 ± 0.001, p = 0.005).
The Rutherford classification demonstrated the highest group separation at classification level IIb or higher (gain ratio 0.038 ± 0.002; p = 0.002) with an amputation rate of 5.6% when classified as Rutherford I and 3.3% for Rutherford IIa, yet 20.0% at Rutherford IIb and 33.3% at Rutherford III. The MESS score, however, did not confer predictive power with a gain ratio of 0.001 ± 0.009.
Discussion
In this study, the outcome of patients after AVT of the lower extremity was retrospectively analyzed over a time period of 28 years. A hospital mortality rate of 2.5% and an amputation rate of 17% can be reported. Predictors for major amputation were vascular re-operation, trauma mechanism and trauma type, concomitant injuries, and the severity of ischemia at presentation.
Compared to the literature, the results are comparable despite differences in the mechanism of injury.2,4,8 Many studies are reports of national trauma databases or military studies with penetrating injuries, mainly gunshot wounds, causing AVT. In our patient cohort, most AVTs were caused by blunt injuries as a consequence of car or sports accidents. Blunt trauma was found to be a significant predictor of amputation with an amputation rate of 19%. No patient with a penetrating trauma was affected by limb loss. Despite these results, our overall amputation rates are not significantly higher compared to the meta-analysis of Perkins et al. 4 as well the trauma database analysis of Kauvar et al. 8 Even though having more penetrating traumas in their patient cohort, blunt trauma was found to be a risk factor for amputation in both studies.
The strongest predictor for amputation in our analysis was vascular re-operation with a fourfold increased risk of limb loss. Ten out of the thirteen patients underwent re-operation due to thrombosis of the vascular reconstruction. In 50% of the patients with occlusion of the reconstruction, no technical or morphological problem causing the thrombosis could be identified. After thrombectomy, all reconstructions remained patent. Dysfunction of the coagulation system due to extensive trauma or after massive blood transfusion, irreversible trauma to the arteries, or insufficient heparinization are possible causes of the thrombosis. Sufficient anticoagulation following revascularization after AVT can be a challenging issue due to concomitant injuries, especially visceral or cerebral trauma, resulting in the need of reduction or even denial of anticoagulation to reduce the risk of potential life-threatening bleeding complications. In two of the ten patients, thrombosis of the vein bypass was suspected due to compression of the graft by CS. After fasciotomy and thrombectomy, the bypasses remained patent. In these cases, decompression of CS was not only crucial for the protection of the muscle and nerve tissue but also for the preservation of graft patency. The remaining three patients underwent surgery due to technical problems of the reconstruction (kinking of the anastomosis, too small diameter of the vein and remaining intimal lesion distal to the anastomosis).
Vascular re-operation without thrombosis of the reconstruction was required because of severe septic bleeding in three cases. In several guidelines, antibiotic therapy covering gram-positive and gram-negative bacteria is recommended in patients with type III Gustilo–Anderson fractures and extensive soft tissue injury.13 –15 All patients of our cohort with the above-mentioned fractures were treated with broad spectrum antibiotics; however, septic bleeding occurred.
This study indicates that concomitant injuries to AVT—either to soft tissue, bones, or other regions of the body—bear a great risk of limb loss. In patients with severe concomitant injuries, a threefold increased amputation rate was seen in our patient cohort. In such patients, a triage of the treatment of different injuries has to be made, and sometimes, the decision life before limb has to be made. 4 Extensive trauma cases are more prone to complications like infections or development of a CS with increased risk of permanent functional impairment or even limb loss.1,9,16 In our predictors analysis, the type of trauma—precisely work-related and traffic accidents—was risk factors for amputation. The majority of patients suffering work-related accidents were forestry or construction workers with high velocity or impact injuries. Traffic accidents belong to the same trauma category. High impact traumas cause severe injuries, in many cases polytrauma with the above-mentioned problems and consequences.
In patients with concomitant bone injuries to AVT, the decision whether to perform osteosynthesis or revascularization first has to be made. There is general consensus about the threshold of 6 h of ischemia time until revascularization; however, the benefit of revascularization as fast as possible has already been shown in the literature.1,3,4,9 As a consequence, interdisciplinary consensus between vascular and orthopedic surgeons to decide whether to perform revascularization or osteosynthesis first has to be achieved. Temporary intravascular shunting during osteosynthesis, as reported previously and performed in three of our patients, would be an easily applicable tool to facilitate both simultaneously. 1 Barros D’Sa et al. 17 showed a significant reduction of amputation rates after introduction of arterial and venous shunting in trauma patients in Belfast (pre-shunting 32.4% vs 8.8% post-shunting, p = 0.009). However, routine use of temporary shunting, especially in peripheral hospitals without vascular surgical expertise could be challenging. Incorrect insertion, dislocation or migration and thrombosis are well-known complications regarding vascular shunting. Therefore, special training in the treatment of vascular injuries (e.g. in military settings) or vascular surgical expertise is necessary for the correct handling of shunts and possible complications.
The decision and timing to perform the revascularization should be determined by the severity of ischemia of the extremity. In this study, a clear difference in the amputation rates was seen between the Rutherford categories, an objective indicator for the severity of ischemia. 11 The cut-off was found to be at Rutherford category IIb with a sixfold increase of amputation rates compared to patients at category I or IIa. So, at the clinical evaluation, sensory loss and motor impairment are crucial markers for the risk of amputation.
Limitations of the study
This is a retrospective study with all the biases of retrospective data collection including sample and recall bias. The calculation of the MESS score is not routinely done at our department, so the calculation was performed retrospectively. This could be a reason for the lacking predictive power of the MESS score in the Bayes network analysis.
Conclusion
Lower extremity AVT was associated with a significant major amputation rate. Several predictors for limb loss were identified, namely, vascular re-operation, a Rutherford category ⩾IIb, work-related or traffic accidents, concomitant soft tissue or bone injuries, polytrauma, and blunt trauma. This could aid in identifying patients at risk of major amputation and in adapting their treatment.
Footnotes
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by M.K., F.E., and L.G. The first draft of the manuscript was written by M.K., and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
