Abstract
Background:
Patients presenting with synchronous colorectal liver metastases are increasingly being considered for a curative treatment, and the liver-first approach is gaining popularity in this context. However, little is known about the completion rates of the liver-first approach and its effects on survival.
Methods:
A systematic review and meta-analysis of liver-first strategy for colorectal liver metastasis. The primary outcome was an assessment of the completion rates of the liver-first approach. Secondary outcomes included overall survival, causes of non-completion, and clinicopathologic data.
Results:
Seventeen articles were amenable for inclusion and the total study population was 1041. The median completion rate for the total population was 80% (range 20–100). The median overall survival for the completion and non-completion groups was 45 (range 12–69) months and 13 (range 10.5–25) months, respectively. Metadata showed a significant survival benefit for the completion group, with a univariate hazard ratio of 12.0 (95% confidence interval, range 5.7–24.4). The major cause of non-completion (76%) was liver disease progression before resection of the primary tumor. Pearson tests showed significant negative correlation between median number of lesions and median size of the largest metastasis and completion rate.
Conclusions:
The liver-first approach offers a complete resection to most patients enrolled, with an overall survival benefit when completion can be assured. One-fifth fails to return to intended oncologic therapy and the major cause is interim metastatic progression, most often in the liver. Risk of non-completion is related to a higher number of lesions and large metastases. The majority of studies stem from primary rectal cancers, which may influence on the return to intended oncologic therapy as well.
Keywords
Introduction
Colorectal cancer remains the second leading cause of cancer death worldwide, and 15% present with synchronous liver metastases.1,2 The only potentially curative treatment is a resection of the complete cancer burden. 3
In the early 2000s, the overall 5-year survival for surgically treated stage IV colorectal cancer with isolated liver metastases was around 26%. 4 During the last decade, larger studies (>500 patients) have reported a 5-year overall survival over 50%.5,6 The dramatic increase in overall survival is likely an effect of novel chemotherapeutic agents and extended criteria for resectable colorectal liver metastases. 7
Historically, management has tended to involve any one of three main strategies; either resecting the primary tumor first and the metastases later (if still resectable without progression); a synchronous resection of the entire tumor burden (this for a select few patients with smaller tumor burden only); and the liver-first approach (with aim to control distant metastasis before operating the primary).
The liver-first approach was originally described with the aim to control aggressive metastatic disease in the liver and has traditionally been preceded by chemotherapy. 8 Since the original description in 2006, the liver-first approach has had a tremendous breakthrough in some parts of the world. In Sweden, for example, 59% of rectal cancers and 40% of the total cohort of colorectal cancers with resectable synchronous colorectal liver metastases during the period of 2008–2015 were treated by means of the liver-first approach. 6 In a French multicentre study covering a similar time-period, the reported use of the liver-first approach was 13%, increasing from 7% in the early 2000s. 9 In the United Kingdom, the proportion of patients undergoing a liver-first approach increased from 10% in 2010 to 15% in 2015. 10 Obviously, there are international discrepancies to the pace in which the method is utilized, but there is clearly a progressive trend. In some centers, tailored variants of the liver-first approach have started to emerge to suit specific circumstances. Some of the most popular are the up-front hepatectomy (precluding induction chemotherapy in up-front resectable metastases) and the interval-resection approach for locally advanced rectal cancers.11,12 However, to this date, there are no randomized studies on the subject. In fact, modern knowledge about the liver-first approach is solely based on retrospective data.
Previous systematic reviews have found no significant difference in 5-year overall survival between the different surgical treatment approaches for colorectal cancer with synchronous liver metastases.13–16 Furthermore, data from Sweden indicate no significant differences in the proportion of T4 and node-positive primary tumors between the different strategies. This, together with the fact that liver-first patients more often have large or bilobar lesions, suggests that the nature of the liver disease alone probably is the primary decisive factor for choosing either strategy.5,6 Nevertheless, with reference to the available literature at hand, the liver-first approach is widely accepted to be a feasible and even preferred strategy when the metastatic burden in the liver needs to be addressed quickly, so as to not lose the patients because of imminent irresectability. On the contrary, about one-third of patients enrolled for the liver-first approach do not proceed beyond liver surgery, mainly due to liver disease progression.6,13,14 Very little is known about the failing to return to intended oncologic therapy (RIOT) in a liver-first setting.
Objectives
There are several retrospective studies of the pros and cons of the liver-first approach versus primary-first and synchronous strategies. There are also several systematic reviews comparing the different treatment approaches in propensity score-matched settings.13–16 However, little is known about the completion rates of the liver-first approach and its true effect on survival. This study was performed to evaluate the completion rates of the liver-first approach and its effect on survival.
Methods
A systematic review and meta-analysis was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and Meta-Analysis of Observational Studies in Epidemiology (MOOSE) checklist.17,18
Search strategy and key words
An electronic search in the PubMed database was performed by the first author to identify all applicable studies. Search terms were: “colorectal cancer,” “colorectal liver metastasis,” “synchronous liver metastasis,” “stage IV synchronous liver-only metastasis,” “liver-first,” “reversed,” “colon/colorectal/rectal,” “synchronous,” and “metastases,” in combinations with the Boolean terms AND/OR. The inclusion dates for the search was set from 31 January 2009 to 31 December 2019. The search was repeated to capture further published studies until 29 February 2020.
Inclusion and exclusion criteria for studies
Eligibility criteria were (1) date of publication not older than 2009; and (2) data accessible in absolute numbers on how many patients were intended for the liver-first approach, and how many actually completed the whole treatment paradigm.
Exclusion criteria were (1) case reports and case series with less than 10 were excluded; (2) articles presented in languages other than English; and (3) if more than one article presented the same set of data, only the most recent study was included.
Data collection
For all included studies, the collection of data was separated into the following nine categories:
Completion (how many of the patients undergoing liver surgery eventually completed the whole treatment program).
Non-completion (what were the main causes of program failure and at what stage).
Demographic data (age, gender, American Society of Anesthesiologists (ASA) class).
Preoperative chemotherapy (how many got it, number of cycles, response).
Primary tumor characteristics (location, T- and N-stages, symptomatic or not).
Liver disease characteristics and extrahepatic status (tumor distribution (bilobar or not), number and largest size of metastases, presence of extrahepatic metastases)
Genetic status (presence of K-RAS/B-RAF or other mutations, either in primary or liver metastases).
Liver surgery details (extent of liver surgery was defined as major (>2 segments) or not, resection margins (R0 or not), and complications according to the Clavien–Dindo classification of surgical complications). 19
Survival (overall survival data, preferably separate data for completion and non-completion groups).
Outcomes assessed
The primary outcome was to assess the completion rate of the liver-first approach.
Secondary outcomes were causes of non-completion and a comparison of the overall survival between completion and non-completion groups.
Intention-to-treat was defined as the total group enrolled for a specific treatment, including those who failed.
Completion was defined as completing both liver and primary tumor resection.
Completion rate was defined as the percentage of the intention-to-treat that successfully underwent “completion.”
Statistical analyses
The meta-analysis was conducted using the software Review Manager (RevMan, version 5.3: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). Remaining analyses were conducted using the IBM SPSS software for Mac, version 22.
A pooled meta-analysis was performed for studies where survival data (of patients who could complete the whole liver-first approach versus patients who did not) could be assessed from the articles. Univariate hazard ratio (HR) with 95% confidence interval (CI) was estimated from raw and non-raw survival data comparing the program completion and non-completion groups from 5-year time-to-event data assessed in included articles. The survival data from each article were pooled in random effect meta-analysis.
Bivariate (Pearson) correlation analyses were performed with selected variables to evaluate any possible influence on feasibility.
Results
The initial search yielded 129 results. Titles implicating irrelevant subjects were not further assessed. Abstracts with relevant titles were screened and 33 items were extracted to examine eligibility. Also, references identified in the original search were cross-checked for additional eligible articles. An additional seven articles were extracted in this manner. In total, 40 articles were read in full text. A four-phase flowchart is depicted in Fig. 1.
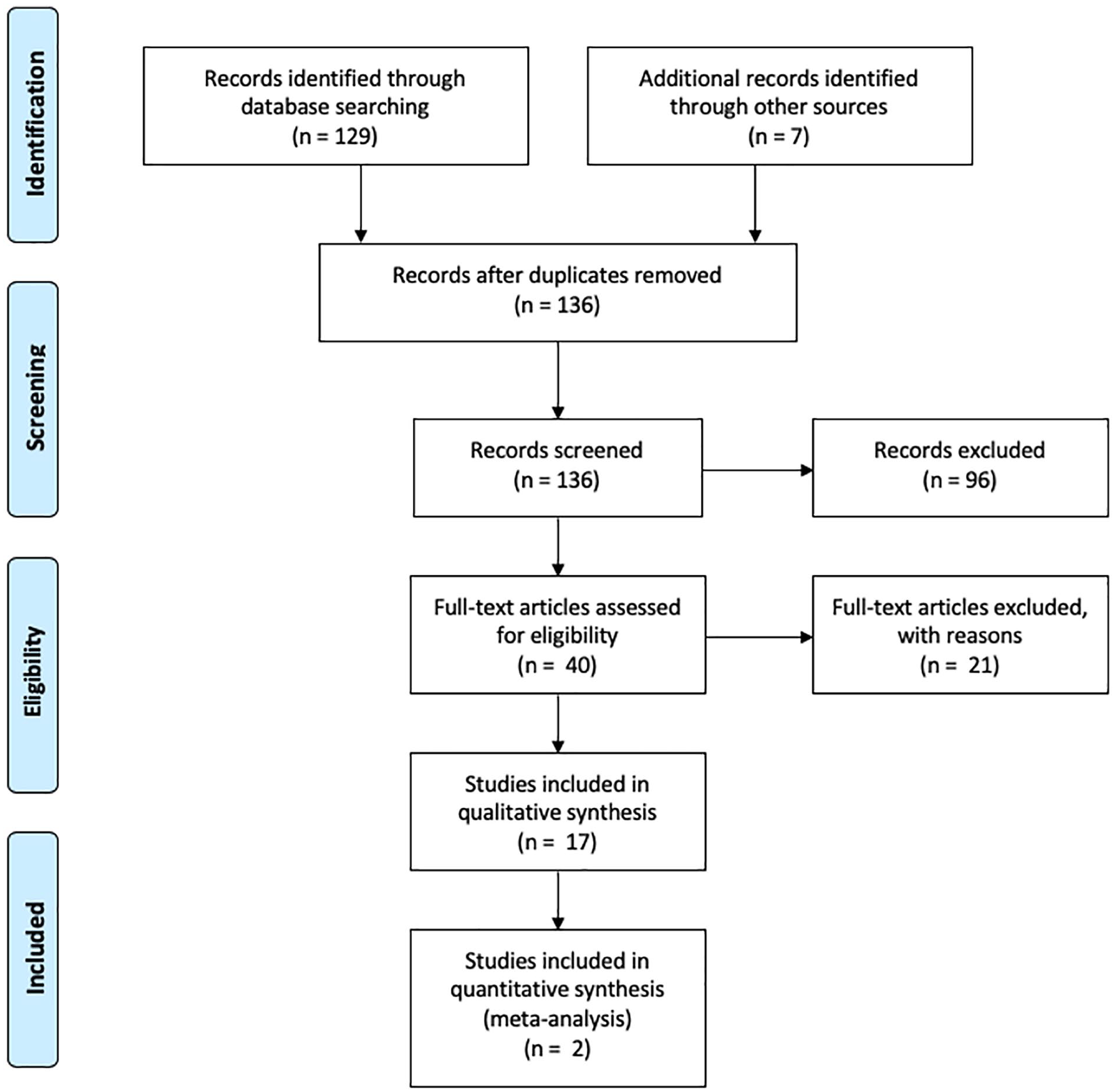
Search strategy in accordance with PRISMA 2009 flow diagram.
A total of 17 studies were eligible for the systematic review, with a total of 1041 patients.6,12,20–34
There were 10 studies presenting mixed cohorts with both rectal and colon cancer patients, and only 1 presented separate data for these subgroups. 6 One study presented patients with colon primaries only. 22 The remaining studies included only rectal cancers.
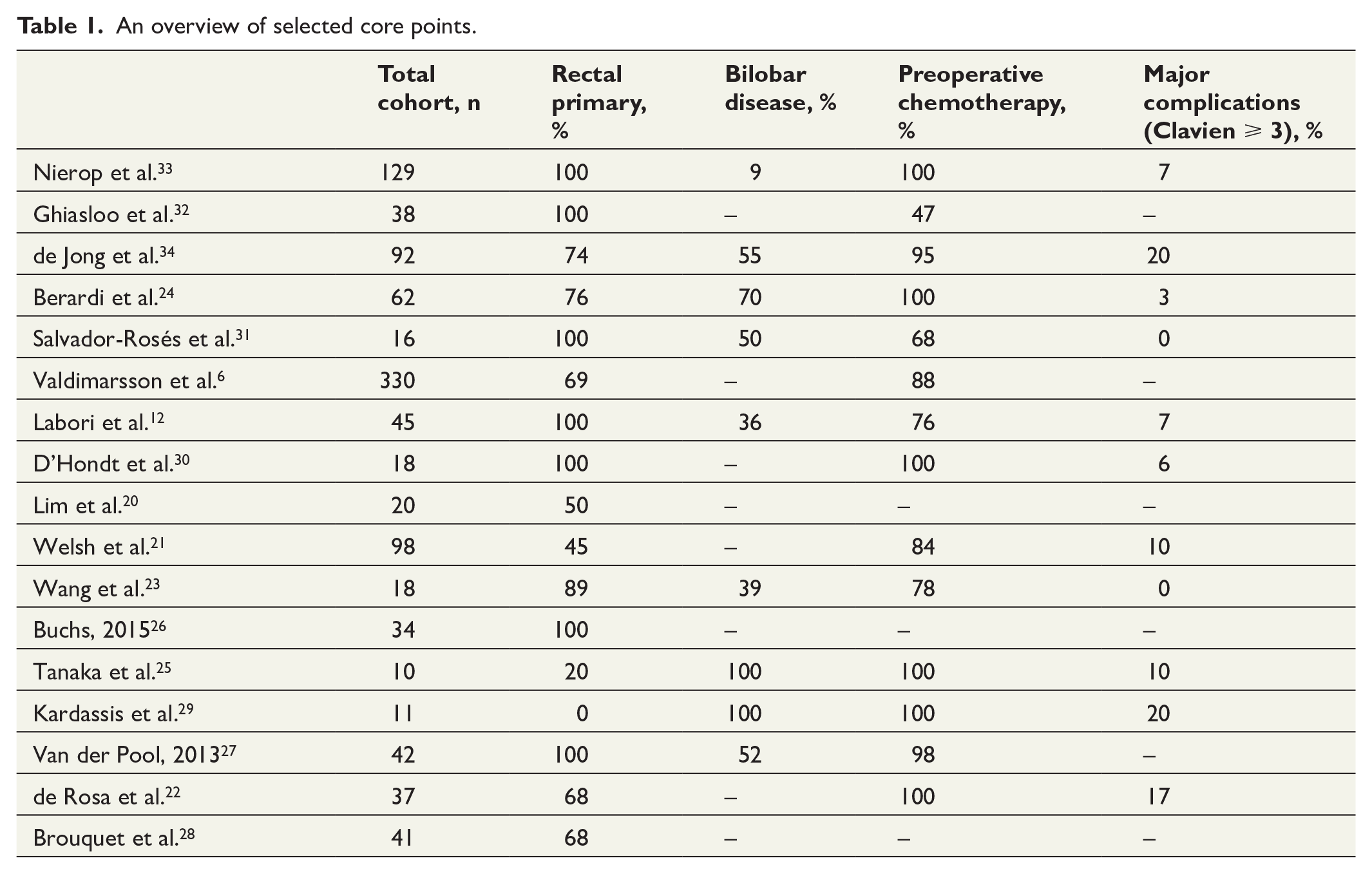
Thirteen studies presented data on all subjects intended for a liver-first approach, whereas the rest presented data only on those that completed all sequences. No study presented complete data separately for completion and non-completion groups. Core descriptive data of the included studies are presented in Table 1.
An overview of selected core points.
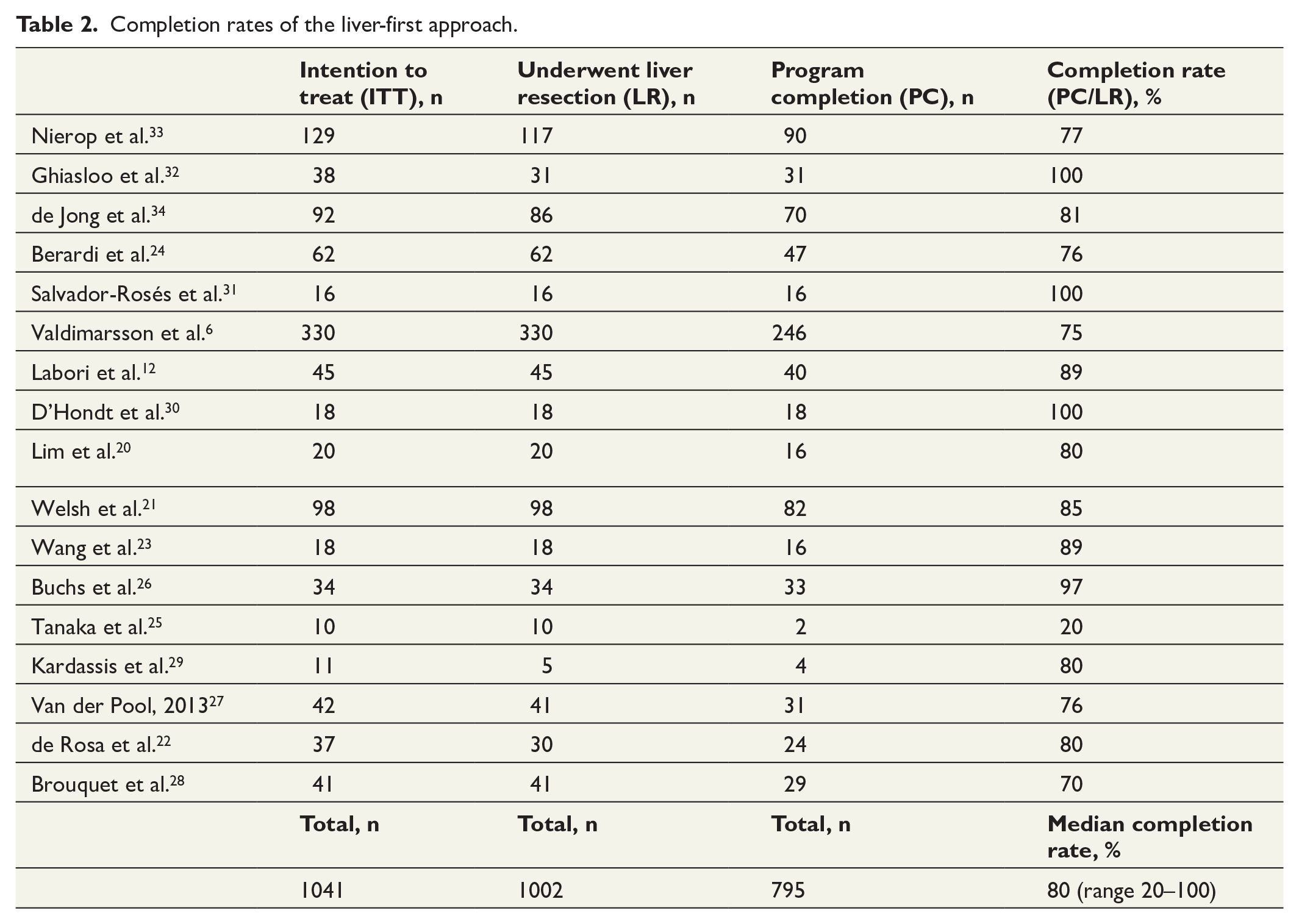
Completion rate of the liver-first approach
A total of 1041 patients were intended for a liver-first approach. Eventually 1002 patients underwent liver surgery, and 795 (79%) of these completed all sequences (Table 2). The median completion rate was 80% (range 20–100).
Completion rates of the liver-first approach.
Out of the total of 1041 patients, 39 patients either progressed during neoadjuvant chemotherapy or were subjected to open/close only due to unexpected progression. Consequently, a total of 1002 patients underwent at least one round of liver resection, and 207 of these were not able to complete the program. Four studies did not present any data on the cause of non-completion. In the remaining 123 failing patients, the major cause of non-completion was interim metastatic progression (76%), mainly in the liver. Other causes of non-completion were interval cancer-related death, insufficient future liver remnant for a second liver resection, progression of the primary tumor, and postoperative death due to liver surgery. Only one patient was lost to follow-up. A summary of causes for non-completion can be seen in Fig. 2.
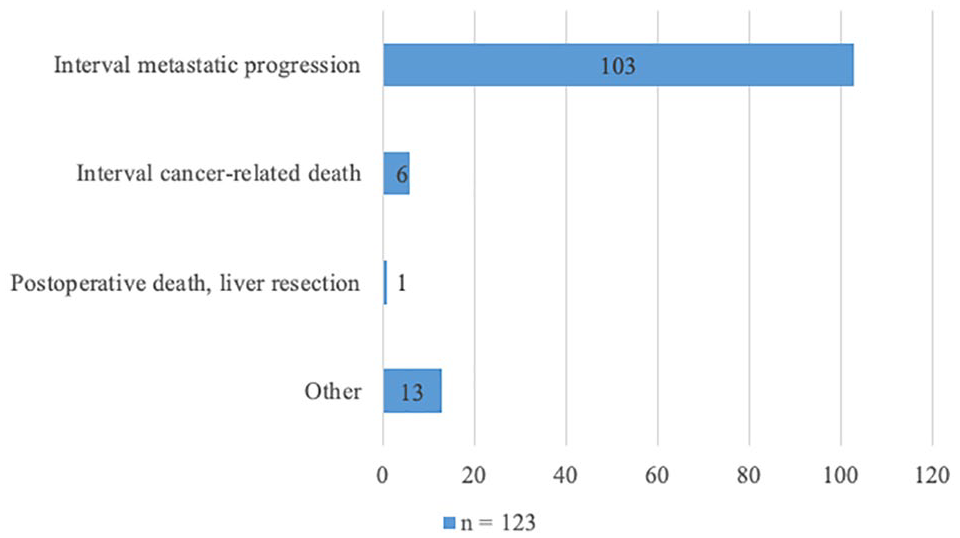
Major causes for non-completion of the liver-first approach.
Demographic data
A median of 69% (range 35–87) of patients intended for a liver-first approach were of male gender, and the overall median of the median age in the individual studies was 62 years (range 60–66). A median of 27% (range 13–88) of patients had an ASA score of 3 or more.
Preoperative chemotherapy
From 14 studies with available total cohort data—a median of 96.5% (range 47–100) received preoperative chemotherapy. Only four studies reported the number of cycles given, and five studies reported the radiologic response according to the RECIST (Response Evaluation Criteria in Solid Tumors) criteria. 35
Primary tumor characteristics
A total of 16 studies presented data on whether the primary tumor was a rectal or colon cancer. Out of 1007 patients, 755 (75%) had a rectal cancer primary. Only four studies presented data on whether the primary tumor was symptomatic or not. Clinical T4 primary tumor stage and/or node-positivity for the total cohorts was only presented in three studies.
Liver disease characteristics and extrahepatic status
A bilobar distribution was reported in a median of 52% (range 9%–100%) of patients. The overall median of the median size of the largest liver lesion and the median number of lesions in the individual studies was 2.85 cm (range 2.4–5.3) and 3.5 (range 2–5), respectively. By a Pearson correlation test, both the median number of lesions and the median size of the largest metastasis showed a significant negative correlation with completion rate (Table 3).
Bivariate association analyses between liver disease, surgical variables, and completion.
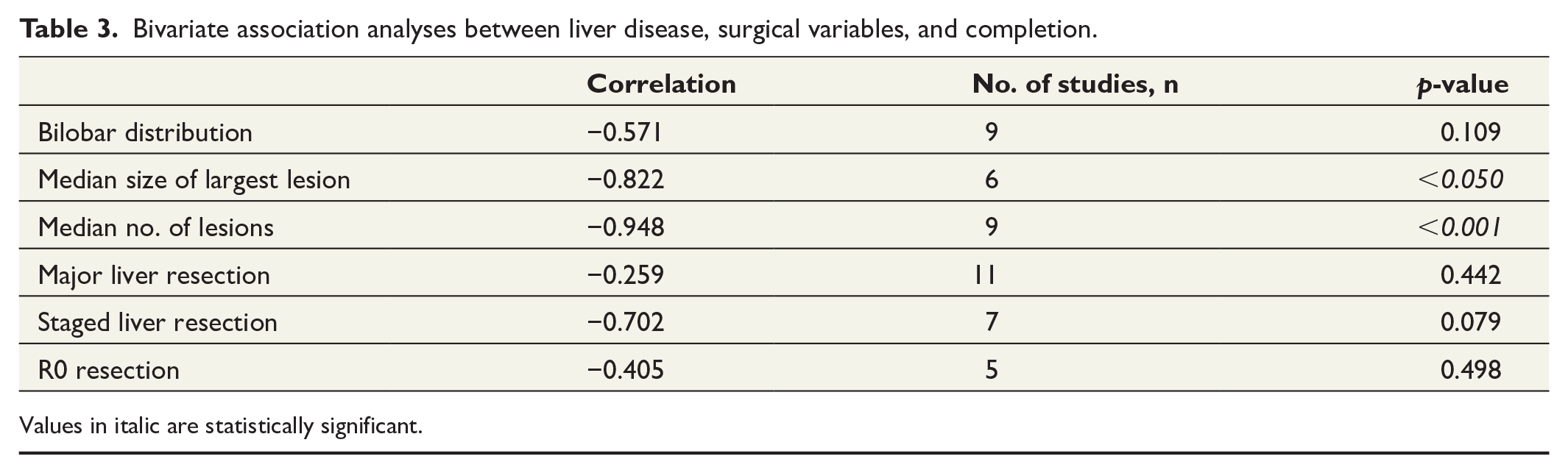
Values in italic are statistically significant.
Genetic status
Only two studies presented any data on genetic mutations. de Jong et al. 34 reported K-RAS mutations in almost 40% of their liver-first patients. Salvador-Rosés et al. 31 reported mutated type K-RAS in 22% of their “interval-approach” patients, compared to 35% in the rectum-first cohort.
Liver surgery details
Major hepatic resections were performed in a median of 50% (range 18%–80%) of patients, and 12% (range 0%–60%) underwent a staged liver resection. R0 resections were reported in a median of 57% (range 50%–92%) specimens. By a Pearson correlation test, there were no significant correlations between major hepatic resections, staged liver resections or R0 resections and completion rate (Table 3). Ten articles reported on major complications of the total cohorts according to Clavien–Dindo, or in such a manner that a Clavien–Dindo score could be calculated. The median rate of complications with a score ⩾3 was 9% (range 0–20).
Survival data
The median overall survival for the completion and non-completion groups was 45 months (range 12–69) and 13 months (range 10.5–25), respectively. A meta-analysis (Fig. 3) showed a significant survival benefit for the completion group, with a univariate HR of 12.0 (95% CI, range 5.66–24.41).

Forest plot depicting an overall survival comparison between completion and non-completion groups. CI: confidence interval.
Discussion
This systematic review shows that about one in every five patients undergoing at least a first round of liver resection in a liver-first approach do not RIOT, most commonly caused by progression of metastases in the liver. A higher number of lesions and larger metastases seem to impose a particularly high risk of not completing the treatment plan. In the available studies, about 50% of patients had a bilobar distribution and underwent a major liver resection. In this study, we observed a wide range of completion rates (20%–100%), a solid example of the selection and referral bias that includes this topic. Notably, the included studies are heterogeneous in their inclusion of synchronous colorectal liver metastases, with variation in referrals for primary resection, use of chemotherapy for down-staging or down-sizing, and with variable use and indications for treatment targeted to the primary tumor. Of note, three-quarters of the studies involved patients with a rectal cancer as primary tumor. Also, the figures probably also represent the poorly understood biological heterogeneity of this disease. 36
The meta-analysis showed a significant survival benefit for patients completing the treatment plan versus those who did not. While the results of our meta-analysis might not be surprising, there are no previous statistical measures of the actual survival benefit—a relevant factor in the risk stratification when selecting stage IV colorectal cancer patients for curative treatment.
In the 1990s and early 2000s, candidates for liver metastasectomy in this context consisted mostly of patients with unilobar disease and few metastases. 4 Today, the concept of resectability in stage IV colorectal cancer has been substantially widened. This is mainly due to novel chemotherapeutic agents as well as a general shift of focus from the metastatic burden to the remaining functioning liver remnant. The liver-first approach is an embodiment of these advancements and offers a powerful onco-surgical tool for biologically favorable patients with a significant metastatic burden in the liver and little or no symptoms from their primary. In our review, negative resection margins (R0) were only achieved in 50% of cases, suggesting that curative treatment was attempted in a significant fraction of inappropriate patients, probably with an unfavorable tumor biology. However, in some European countries, there might still be a considerable under-treatment of colorectal liver metastases.10,37
Critics of the liver-first approach have emphasized a concern that postponing resection of the primary tumor might bring about life-threatening complications, such as intestinal obstruction. In addition, some have argued that resecting the primary tumor could have survival benefits even if the liver metastases are not addressed surgically. 37 According to a multidisciplinary international consensus from 2015, a “primary-first” strategy should be considered if severe intestinal symptoms are present or imminent. 38 However, in this review, we observed that several studies included locally advanced rectal cancers and symptomatic colonic tumors, which (if needed) were successfully treated with stenting or deviating stomas parallel to a liver-first approach. Albeit risky, this could be a feasible strategy when addressing patients with both a symptomatic primary (emergency obstruction) and a wide distribution of liver metastases, which could become non-resectable if not removed promptly. However, the report of these cases may also reflect a strategy where the patient is initially deemed irresectable and non-operable, and stent or deviation strategy is done to start chemotherapy. Some of these patients are then “converted” from an assumed palliative state, to potentially resectable. This may also occur in patients who are already under treatment with a liver-first approach and develop obstructive symptoms.
Another example of non-adherence to international guidelines found in this review, was the relatively high prevalence of “up-front” hepatic resections in individual studies.31,32 Even though some centers opt for an up-front treatment in resectable colorectal liver metastases today, the initial rationale behind the chemotherapy-induced approach described by Mentha et al., was not only to down-stage or down-size but also—due to the sequential and extended nature of the treatment—to gain an early systemic disease control and hypothetically eliminate occult metastatic disease. On the contrary, treating resectable patients with preoperative chemotherapy poses a risk of “disappearing metastases,” potentially causing a significant therapeutic dilemma. 39 The rationale of precluding the induction treatment has often been a fear of causing parenchymal toxic effects, which could have perioperative and postoperative implications.40–43
Previous reports have emphasized the need for randomized prospective trials to determine the best order of treatment for stage IV colorectal cancer with colorectal liver metastases. In contrast, the clinical and biological heterogeneity of this disease indicates that there is no one best treatment for all. The question is not whether the liver-first approach is superior or non-inferior to other treatment regimens in general, but rather for which patients it is the best option. This review shows that 20% of liver-first patients do not complete intended surgery beyond liver resection, a potentially unnecessary major procedure. The initial spark for this study was an idea to characterize these non-completers, in an effort to allow for better patient stratification in the future. However, apart from identifying some cautious indicators of poor prognosis, the general lack of descriptive data on the non-completion groups precluded any solid conclusions. It remains unknown what characterizes these 20% patients that do not proceed to complete curative resection. An early identification of this subgroup could allow for an early palliative plan instead of being subjected to a potentially unnecessary major surgical procedure.
It is possible that in an era with state-of-the-art tissue ablation instruments, and potent hemostatic agents readily available, the technical challenges of performing complicated liver resections are no longer the biggest issue. Therefore, speaking in terms of resectability is perhaps no longer equivalent with curability. How can we decide an evidence-based distinction for what is curable rather than resectable? Studies investigating genetic aberrations have demonstrated that the majority of changes in colorectal primary tumors (such as RAS-mutations) were maintained in the colorectal liver metastases. 36 Probably, in the future, there might be a place for preoperative biopsies of liver metastases to assess tumor biology as a part of a prospective modern risk score for a better patient stratification and utilization of resources.44,45
Conclusion
In accordance with previous articles, this systematic review concludes that liver-first is an appropriate treatment option for selected patients with synchronous colorectal liver metastases. It offers potential complete resection for a majority of patients enrolled. Furthermore, this review found that overall survival is significantly better for those that complete their liver-first plan versus those who do not.
On the contrary, 20% patients do not complete their designated plan and are transferred to palliative care, most often due to liver disease progression in the post-hepatectomy period. To avoid performing unnecessary liver surgery in the future, further studies are needed to more appropriately identify subgroups for such an advanced and costly treatment. New and improved risk scores and guidelines are needed, perhaps with more focus on tumor biology aspects.
Footnotes
Author contributions
Adam Zeyara and Roland Andersson proposed the research idea. All authors contributed to design, protocol, and planning of the study. Adam Zeyara wrote the protocol, conducted the literature search, and wrote the first manuscript draft. Data collection and analyses were performed by Adam Zeyara. William Torén assisted in performing the meta-analysis. All authors contributed to several rounds of revisions. All authors read and approved the final manuscript for submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Institutional funding only.
Data transparency
All data are available upon request.
