Abstract
Background and Aims:
Approximately 75% of patients admitted with small bowel obstruction have intra-abdominal adhesions as their cause (adhesive small bowel obstruction). Up to 70% of adhesive small bowel obstruction cases, in the absence of strangulation and bowel ischemia, can be successfully treated with conservative management. However, emerging evidence shows that surgery performed early during the first episode of adhesive small bowel obstruction is highly effective. The objective of this narrative review is to summarize the current evidence on adhesive small bowel obstruction management strategies.
Materials and Methods:
A review of the literature published over the last 20 years was performed to assess Who, hoW, Why, When, What, and Where diagnose and operate on patients with adhesive small bowel obstruction.
Results:
Adequate patient selection through physical examination and computed tomography is the key factor of the entire management strategy, as failure to detect patients with strangulated adhesive small bowel obstruction and bowel ischemia is associated with significant morbidity and mortality. The indication for surgical exploration is usually defined as a failure to pass contrast into the ascending colon within 8–24 h. However, operative management with early adhesiolysis, defined as operative intervention on either the calendar day of admission or the calendar day after admission, has recently shown to be associated with an overall long-term survival benefit compared to conservative management. Regarding the surgical technique, laparoscopy should be used only in selected patients with an anticipated single obstructing band, and there should be a low threshold for conversion to an open procedure in cases of high risk of bowel injuries.
Conclusion:
Although most adhesive small bowel obstruction patients without suspicion of bowel strangulation or gangrene are currently managed nonoperatively, the long-term outcomes following this approach need to be analyzed in a more exhaustive way, as surgery performed early during the first episode of adhesive small bowel obstruction has shown to be highly effective, with a lower rate of recurrence.
Keywords
Introduction
Small bowel obstruction is a common cause for surgical admission and accounts for about 20% of all surgical emergency room visits for abdominal pain in Western countries (1). The management of adhesive small bowel obstruction (ASBO) is based on the three fundamental steps: clinical evaluation, biological tests, and CT imaging. As ASBO is commonly caused by previous abdominal surgeries, surgical treatment may seem like an ambiguity. On one hand, successful conservative treatment may leave adhesions and so, induce future episodes of bowel obstruction. On the other hand, in the collective imagination, surgery may be the source of new adhesions and, when an extensive resection is required, lead to significant consequences such as short bowel syndrome.
International guidelines for ASBO have long advocated a trial of conservative management (2–5), as up to 80% of ASBO cases, in the absence of clinical and/or radiological suspicion of strangulation and bowel ischemia (noncomplicated ASBO), can be successfully treated with conservative management (6–8). However, emerging evidence shows that surgery performed early on either the day of or day after admission, during the first episode of ASBO, is highly effective, with an increase of quality adjusted life years (8–10). Therefore, although most ASBO patients without suspicion of bowel strangulation or gangrene are currently managed nonoperatively, the long-term outcomes following this approach need to be analyzed in an exhaustive way in order to assess which patients could benefit from early surgery and why, and which others should be managed conservatively due to the high risk of adverse outcomes.
Nonadhesive etiologies of small bowel obstruction include incarcerated abdominal wall hernias, malignant or benign obstructing tumors, Crohn’s disease, volvulus, post-radiation strictures, and other infrequent causes such as bezoars (8). A recent systematic review and meta-analysis of six studies, analyzing the safety of foregoing surgery in patients without abdominal or pelvic surgery history presenting with small bowel obstruction, found a high prevalence of malignant etiologies (7.7%–13.4%), most of which were not suspected before surgery. In almost half of these patients, a trial of nonoperative management failed and patients required operative interventions (11). Moreover, a recent Cochrane review investigating the role of water-soluble contrast media (WSCM) in inoperable small bowel malignant obstructions reported limited evidence to determine the therapeutic effects and safety of conservative management with WSCM (12). Therefore, patients presenting with small bowel obstruction with no history of previous abdominal or pelvic surgery should be excluded from the treatment pathways discussed in this clinical practice review. The objective of this article is to summarize the current evidence on ASBO management strategies with a special focus on patient-centered outcomes.
Who are the ASBO patients?
Approximately 75% of small bowel obstructions are due to intra-abdominal adhesions, usually as a consequence of previous surgery (13,14). ASBO occurs in up to 9% of patients following abdominal operations (15,16), with up to a third of patients with ASBO receiving operative intervention (17,18). Known risk factors for ASBO include multiple abdominal procedures, as well as surgery involving the pelvis and rectum (19).
The risk of ASBO is the highest following colorectal and oncologic gynecological surgery (16). 10% of patients develop at least one episode of ASBO within 3 years after colectomy (20). Also the 5-year incidence rate of ASBO varies by procedure, with a maximal risk after partial colectomy and minimal risk after appendectomy (21). Behman et al. (22) found that, among 22,197 patients admitted to hospital for the first time with a diagnosis of ASBO in Ontario, Canada, between 2005 and 2011, mean age was 64.5 years and 52.2% were women.
Laparoscopy is associated with a significant reduction in the rate of adhesions across multiple abdominal procedures compared to open surgery, with the period of greatest risk for ASBO being the first 2 years after surgery (21). In the systematic review of cohort studies by Ten Broek et al. (16), the incidence of reoperation for ASBO was 1.4% after laparoscopic and 3.8% after open surgery.
A recent population-based analysis found that between 2005 and 2014, there has been a decrease in the population-based rate of hospital admissions for a first episode of ASBO in Canada, which may reflect an increase in the use of minimally invasive techniques such as laparoscopic and robotic surgery or, alternatively, an increase in the use of adhesion barriers (17). In parallel, a significant trend toward increased operative intervention, with surgery occurring earlier on the day or day following admission, has been reported. These changes might be explained by strong evidence from several recent studies showing that delayed operation (>72 h) in ASBO patients is associated with an increase in morbidity and mortality rates (23,24).
How to diagnose an ASBO?
Clinical assessment, physical examination findings, and laboratory tests alone are insufficient to make an accurate diagnosis of ASBO, in particular regarding the presence of bowel strangulation. Plain abdominal X-ray, ultrasonography, computed tomography (CT), and magnetic resonance imaging (MRI) are currently utilized to confirm the suspected clinical diagnosis of ASBO.
Due to easier availability and lower costs, X-ray is typically the initial imaging, although it has a poor sensitivity (46%–69%) and specificity (57%–67%) (25). Ultrasonography also represents a user friendly and inexpensive diagnostic tool that could be performed at bedside. A recent meta-analysis including 11 studies reported a high sensitivity (92%) and specificity (97%) of ultrasonography for small bowel obstruction (26). However, the most relevant limitation of ultrasonography in the diagnosis of ASBO is related to the scarce accuracy in the detection of bowel ischemia, which is the main indication for emergency surgery in these patients (27).
Nowadays, there is a wide consensus regarding the use of CT in the evaluation of patients with ASBO (2). Increased utilization of CT imaging in the diagnostic pathway of ASBO patients has allowed a better identification of high-grade obstructions and those that are unlikely to resolve nonoperatively (28). Furthermore, CT scan also has approximately 90% accuracy in predicting strangulation and the need for urgent surgical operation (29).
The recent systematic review and meta-analysis by Li et al. (28) showed that the pooled sensitivity and specificity of CT for ASBO were 91% and 89%, respectively, with an accuracy for etiology of adhesions, hernia, and tumor of 95%, 70% and 82%, respectively, and sensitivity and specificity of 92% and 77% for detecting the transition point. CT findings—including decreased enhanced bowel wall, wall thickness, mesenteric congestion, and mesenteric and/or peritoneal free fluid—have been reported as related to bowel ischemia (30,31). Within this context, hypoenhanced bowel wall (specificity 95%) is highly predictive of ischemia, while the absence of mesenteric fluid (sensitivity 89%) represents a reliable finding to exclude strangulation (29). Three CT findings were significantly associated with strangulation in the multivariate analysis included in a recently published study by Millet et al.: reduced bowel wall enhancement (odds ratio = 7.8), diffuse mesenteric haziness (odds ratio = 6.1), and a closed-loop mechanism (odds ratio = 6.5). The model combining these three features had an area under the curve of 0.91 and a high negative predictive value (97%). Among the strangulated cases, reduced bowel wall enhancement (odds ratio = 3.9) and mesenteric fluid (odds ratio = 3.6) were confirmed to be predictive of resection. Peritoneal fluid showed a sensitivity of 89%, which was higher compared to that of mesenteric fluid (73%), and hypoenhanced bowel wall, free peritoneal gas, and pneumatosis or venous gas were variables associated with high specificity (96%–99%) to bowel strangulation (32).
Another prospective observational study identified one clinical symptom (no flatus) and two CT findings (free fluid and high-grade obstruction) as significant predictors for early surgical exploration in ASBO patients (33). Failure to detect patients with strangulated ASBO and bowel ischemia is still associated with significant delay in surgical intervention and subsequent high mortality and complication rates (23). For all these reasons, adequate patient’s selection through physical examination and CT is the key factor of the whole management strategy.
Why to Operate on ASBO Patients?
For all patients admitted to hospital with ASBO, managed operatively and nonoperatively, the 1-year mortality rate is estimated to be 3%–23% (23,34–36). The majority of deaths occurred within 90 days of admission (62.5%) in the population-based study by Behman et al. (34).
Potential explanations for the high mortality rate in patients with ASBO include prolonged malnutrition, and the risk of ischemic bowel with perforation, sepsis, and bacterial translocation. Furthermore, delays in diagnosis or timing of surgical exploration may increase the morbidity and mortality risk. Indeed, trials of nonoperative management for uncomplicated ASBO exceeding 3 days are associated with increased morbidity and postoperative length of hospitalization (24,37).
The current standard for the management of ASBO patients without signs of bowel ischemia and/or peritonitis, which advocates a trial of conservative management and suggests surgical intervention only in the absence of clinical resolution, does not consider the long-term outcomes of nonoperative management. It also does not recognize the evidence that surgery mitigates the probability of future recurrences of ASBO (2,38). With each further episode of ASBO, the risk of subsequent additional episodes increases and the time between episodes decreases (39,40).
In the longitudinal, propensity-matched, retrospective cohort study published by Behman et al., which enrolled 27,904 subjects admitted with their first episode of ASBO, patients who underwent operative management for ASBO had a lower risk of recurrence (13% vs 21%, hazard ratio (HR) 0.62) compared to patients treated conservatively. The 5-year probability of experiencing another recurrence increased with each episode until surgery, at which point the risk of subsequent recurrence decreased by 50%. Behman also demonstrated that the chance of being treated surgically decreases with each episode (from 22% for patients admitted for their index episode of ASBO to approximately 16% of patients admitted for their second episode of ASBO, to 11.8% for patients admitted for their third episode), thus highlighting the importance of a careful evaluation for the risk/benefit of surgery during the first episode of ASBO. Although, in this study, authors accounted for measured confounders in the propensity-score matching, an important unmeasured factor was patients’ surgical history. Indeed, patients with complex abdominal surgical histories may have been less likely to receive operative treatment, while also being more likely to experience recurrences of ASBO (41).
Medvecz et al. found that, among the patients managed nonoperatively during initial admission, at least one recurrence developed in 25.6% compared with 19.0% of patients initially managed surgically. At each subsequent recurrence, patients initially managed nonoperatively tended to have a higher proportion of subsequent recurrences, and for those patients with multiple recurrences, each additional nonoperative recurrence increased the risk of future episodes (42).
The risk of recurrence and readmission may result in increased long-term risks as compared with surgical management. A population-based study reported in-hospital and 1-year mortality rates of 7% and 23%, respectively, for ASBO patients (36), and several other studies have demonstrated that nonoperative management is associated with an increased risk of long-term adverse outcomes (24,41,43). Reasons for which, fit-for-surgery patients may benefit from an early surgical intervention during the first episode of ASBO. The advantages of surgical treatment of ASBO, however, should be balanced with the risks associated with surgery, mostly related to patient’s comorbidity and frailty status.
The systematic review and meta-analysis by Hajibandeh et al. showed that, although considering the baseline suspicion of strangulation as a major confounding factor, operative management of ASBO was associated with a lower risk of recurrence, but higher risk of mortality and complications. When the baseline suspicion of strangulation was higher in the operative group, the risk of mortality and complications were higher and the length of hospital stay was longer in the operative group, but the risk of recurrence was lower. Conversely, when the baseline suspicion of strangulation was low in both groups, there was no difference in any of the adverse outcomes, except recurrence, which was lower in the operative group (44).
In the study by Behman et al., the logrank test comparing operative and nonoperative management for the first episode of ASBO suggested a statistically significant survival benefit associated with surgical management, mediated through prevention of recurrences. The 5-year cumulative incidence of recurrence was significantly lower among patients managed operatively than among patients managed nonoperatively (9).
The study by Wessels et al. identified that, following the readmission for ASBO in patients who were initially treated nonoperatively, the decision for operative management was the greatest for younger patients who tended not to have comorbidities. This, if on one hand could be explained by the potential higher severity of ASBO in patients who suffered from more severe symptoms and thus necessitated operative management, on the other hand confirm that younger patients with lower comorbidity indexes might benefit from surgery during the first admission for ASBO (18). Conversely, older patients with significant cardiac or pulmonary history and suspected dense adhesions basing on clinical history and CT findings can benefit from conservative treatment accepting a high risk of recurrence, provided there are no signs of bowel strangulation and/or ischemia.
When to Operate on ASBO Patients?
Fig. 1 illustrates our suggested decision-making flowchart in patients with ASBO.
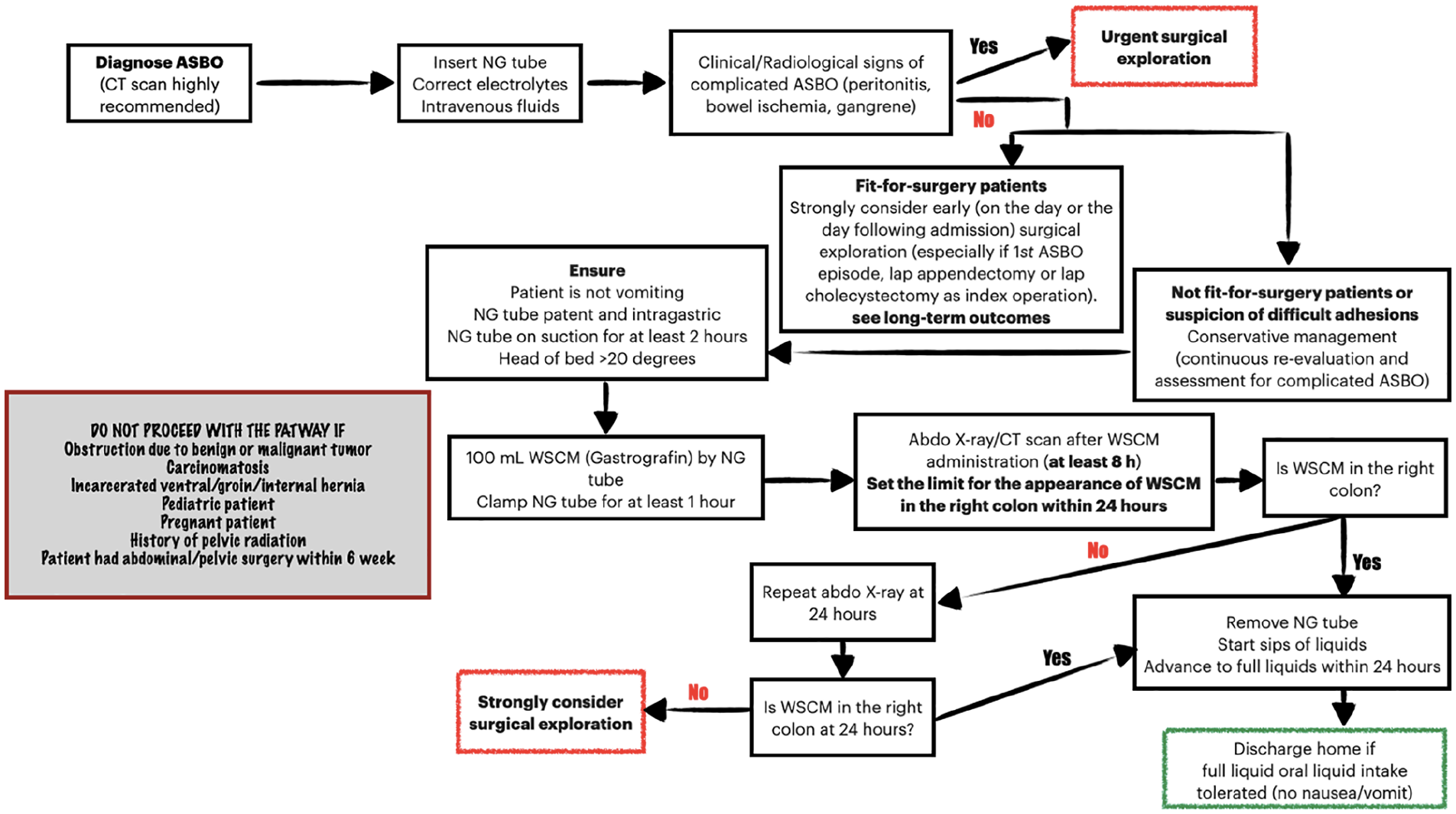
Decision-making flowchart in patients with ASBO.
Emergent surgical exploration remains mandatory for ASBO patients in the presence of signs of peritonitis, that might reveal strangulation or bowel ischemia (2). Patients with signs of strangulation or peritonitis (fever, tachycardia and leukocytosis, metabolic acidosis, and continuous abdominal pain) are absolutely not candidates for conservative treatment. Conversely, up to 70% of non-complicated ASBO can be resolved by conservative treatment with gastrointestinal (GI) decompression with nasogastric or nasointestinal tubes (no significant difference currently exists between nasogastric and nasointestinal tubes in terms of ASBO resolution), intravenous fluid, analgesics, correction of electrolyte imbalance, and a regular clinical and imaging reassessment (45).
Our decision-making protocol proposes an early (within 24 h from admission) surgical intervention in ASBO patients without signs of strangulation and/or bowel ischemia, if they are deemed fit-for-surgery, and especially if the clinical history (first episode of ASBO, appendectomy or cholecystectomy as index surgical operation) and CT findings suggest the absence of difficult adhesions.
In our experience, patients managed with early adhesiolysis are younger and less comorbid than those who undergo nonoperative treatment or nonearly operation. This is in line with the results published by Behman et al. (9).
Conversely, our protocol for not fit-for-surgery patients in the absence of signs of strangulation and/or bowel ischemia suggests the conservative management with WSCM administration, continuous clinical re-evaluation, and abdominal X-ray or CT scan at definite time points: 8 h, and, if the WSCM has not reached the right colon at 8 h, at 24 h as the very latest point in time. The combination of enteral decompression, nil per os status, and bowel rest represented the standard of nonoperative management until the mid-2000s, when the incorporation of a small bowel follow-through, serial abdominal X-rays to follow the progress of WSCM reached great success (46).
Early identification and prompt operative intervention in patients expected to fail nonoperative management following WSCM administration are likely the reason for the reported shorter length of hospital stay after a WSCM trial. The real value of WSCM is related to its diagnostic attributes in demonstrating patients who are unlikely to resolve with a conservative treatment, therefore leading to a more expedite decision-making regarding surgical intervention.
The second potential benefit of WSCM is therapeutic, as the contrast’s high osmolarity might facilitate decrease in edema of the small bowel and help relieve intraluminal pressure. The high osmolarity of WSCM can promote shifting of intestinal wall edema at the obstruction point. This might increase the pressure gradient across the obstruction as well as bowel motility to help the obstruction resolve (47).
Recent studies have identified a substantial reduction in need for operation with the incorporation of a WSCM trial into conservative treatment strategies compared to patients who did not receive contrast (20.8% vs 49.0%) (48). However, these results were not confirmed neither by a 2007 Cochrane systematic review (49) nor by subsequent meta-analyses, which demonstrated that WSCM does not reduce the need for operative management in ASBO nor impact mortality rates (50).
The recent World Society of Emergency Surgery guidelines, published in 2018, advocate WSCM imaging on admission and re-evaluation at the 24–36 h in patients without overt signs of bowel ischemia or peritonitis (2). However, a point of debate is the fixed time points of clinical observation, as many large retrospective population-based trials suggest a strong association delay to surgery and an increased rate of complications and death (23,36).
Protocols and time intervals for X-rays following WSCM administration vary in the literature, but most studies suggest allowing anywhere from 8 to 36 h for the contrast to reach the cecum, after which the obstruction is very unlikely to resolve without a surgical operation. Ceresoli et al., with their systematic review and meta-analysis, compared accuracy in predicting ASBO resolution at three discrete time intervals post WSCM administration: 2–6 h, 8–12 h, and 24–36 h. The reported sensitivity and specificity were 82% and 95% in the 2–6 h group, 97% and 89% in the 8–12 h group, and 99% and 93% in the 24–36 h group, respectively (47).
Several studies showed that the timing between hospital admission and surgical management of ASBO has significantly shortened during the last few decades. Due to this change in management strategies, in-hospital mortality rate decreased significantly by 5.6% (51). These data have been confirmed by the recent systematic review and meta-analysis by Thornblade et al. (52), who found that rates of bowel resection were lower in patients submitted to early surgery (mean 1%), with a median time cut-off for defining early surgery of 1.5 days, than those who underwent delayed surgery (mean 8%), with a significant difference between the two groups also in terms of mortality (<1% in the early group vs 5% in the delayed group) as well as costs, which were lower among patients undergoing early surgery.
What should be done for a Successful Adhesiolysis in ASBO Patients?
As reported in most recent World Society of Emergency Surgery guidelines of ASBO, open surgery is still the technique of choice for strangulating ASBO or in case of ischemic bowel loops (2). The laparoscopic approach was historically contraindicated because of the reduced intra-abdominal working space, and increased risk of both trocar injury and bowel perforation in presence of distended bowel loops. With advances in laparoscopic surgical training and technology innovations, however, laparoscopic adhesiolysis has shown the potential to be an advantageous alternative to open surgery in the management of ASBO.
Recent systematic reviews and meta-analyses reported reduced risk of mortality, morbidity (with particular regard to the incidence of pneumonia and wound infection), and reoperation rates related to laparoscopic adhesiolysis. Further potential benefits of laparoscopy in ASBO patients include faster recovery, less pain, and fewer recurrence of adhesions (53–55). According to the higher quality evidence published to date, laparoscopy in the treatment of ASBO should be used only in selected patients with an anticipated single obstructing band at preoperative CT scan, and there should be a low threshold for conversion to an open procedure in cases of high risk of bowel injuries, such as those in which diffuse adhesions are encountered.
Farinella et al. (56) reported that predictors for a successful laparoscopic adhesiolysis include ⩽2 laparotomies in patient’s clinical history, appendectomy as index operation, no previous median laparotomy incision, and a single adhesive band. The latter point is of paramount relevance as even CT scan has shown to not be 100% sensitive in the identification of patients with a single adhesive band. In fact, in the study by Sallinen et al. (57), despite rigorous selection of patients based on physical findings and CT imaging, a third of patients in both study groups had more adhesions than the expected single band.
Currently, only 50%–60% of surgeons would consider using laparoscopy for ASBO according to surveys from the United Kingdom and the United States (58) and, in Italy, the proportion of surgical units approaching a small bowel obstruction by laparoscopy in more than 50% of cases is limited to 13.4% (59). The lack of widespread adoption of laparoscopy for ASBO could be due to the fact that laparoscopic adhesiolysis can be a technically demanding procedure, and it has been associated with a higher risk of iatrogenic bowel injury in the past (60,61).
Careful preoperative selection of patients who might be best suited for a laparoscopic approach to ASBO is the key. The other key factor to guarantee the patient with best outcomes possible is related to the level of laparoscopic expertise of the surgeon, who must be able to carry out a safe step-by-step operation. Only hemodynamically stable patients without diffuse peritonitis and/or septic shock without any suspicion of bowel perforation should be considered for a laparoscopic adhesiolysis. Furthermore, patients having CT scan findings consistent with a clear transition point and, therefore, suspected to have a single completely obstructing adhesive band with completely collapsed distal small bowel loops are the ideal ones for the laparoscopic approach (2,4).
Meta-analyses published before 2016, in fact, demonstrated a trend toward higher incidence of intraoperative bowel injury in patients undergoing laparoscopic adhesiolysis (55). The results of the most recent meta-analysis published to date, which included data from 18 case-controlled studies on 38,927 patients, conversely, demonstrated that the effect size in favor of open procedures actually disappeared, leading to an insignificant result with an overall cumulative relative risk (RR) of 2.61 when laparoscopic adhesiolysis was compared with open adhesiolysis, and even to an opposite trend toward a slightly higher risk of intraoperative bowel injury during open adhesiolysis (19.7% vs 12.9%) (53).
However, in all the studies addressing the role of laparoscopy in ASBO patients, a significantly higher proportion of the American Society of Anesthesiologist (ASA) 1 and 2 patients in the laparoscopic group and a significantly higher proportion of patients with ASA 3 and 4 in the open adhesiolysis group were reported. Selection bias can be suspected also based on the threefold higher incidence of bowel resection in the open adhesiolysis groups, as reported by Quah et al. (53), that would indicate that patients who underwent open adhesiolysis had a higher incidence of strangulation, perforation, and/or a higher rate of more complex adhesions. It seems clear that surgeons preferentially select patients who had a lower likelihood of a more complex ASBO with strangulation, bowel gangrene, or multiple adhesions to have a laparoscopic approach (62).
Results from the first, and thus far the only, published prospective randomized trial comparing laparoscopic versus open adhesiolysis for ASBO have recently been published. Patients with ASBO that had not resolved with conservative management were randomized to have either open or laparoscopic adhesiolysis. Only patients with high likelihood of a single adhesive band on CT were included in the trial, thus reducing the inhomogeneity of the population under study. Study results showed that postoperative length of hospital stay for open surgery patients was on average 1.3 days longer than that in the laparoscopic group (p = 0.013), with an equivalent rate of postoperative complications within 30 days and iatrogenic bowel injury occurring in 22%–24% of the patients in each group. Laparoscopic adhesiolysis provided quicker recovery compared to open adhesiolysis, and has been demonstrated to allow quicker return of bowel function, all without increasing complications. Moreover, pain on visual analog scale (VAS) was lower in the laparoscopic group than in the open surgery group on postoperative days 3 and 4 (57).
Absolute contraindications to laparoscopic adhesiolysis are represented by hemodynamic instability and preoperative shock, diffuse peritonitis and/or evidence of severe intra-abdominal sepsis, high suspicion of perforated bowel, and high probability of diffuse and dense matted adhesions (patients with multiple previous laparotomies). Further relative contraindications to laparoscopic adhesiolysis (but not to diagnostic laparoscopy) are represented by suspicion of bowel strangulation and/or bowel ischemia without gangrene or perforation, localized clinical peritonitis, CT findings of free abdominal fluid, CT or X-ray findings of small bowel distension with a size >4 cm, and CT additional findings such as small bowel feces sign, small bowel wall thickening, and mesenteric edema or vascular engorgement. All these findings, although usually represent relative contraindication to operative laparoscopy, are not exclusion criteria from diagnostic laparoscopy, although the risk of conversion in presence of these signs is high (Table 1) (52,60).
Patients’ selection criteria for laparoscopic adhesiolysis in patients with ASBO.

CT: computed tomography; ASBO: adhesive small bowel obstruction; WSCM: water-soluble contrast media; NG: nasogastric.
The conversion rate from laparoscopic to open surgery in ASBO varies widely from the 40% rate reported in systematic reviews and meta-analyses of nonrandomized studies (53,54) to the 25% rate reported in the LASSO randomized controlled trial (57). The most frequent reason for conversion to open surgery is the presence of dense adhesions, followed by the inability to assess the transition point and the viability of the bowel, bowel injuries, bowel perforation, bleeding, inability to achieve a reliable intra-abdominal view, and the need of bowel resection due to gangrene presence.
From a technical perspective, routine manual decompression of bowel loops during open adhesiolysis is unnecessary. The randomized clinical trial by Ezer et al. (63) showed that the resumption of a regular diet, incidence of respiratory complications, bacterial translocation, wound infection, and postoperative hospital stay were not significantly different in patients for whom the intestinal content of the dilated small bowel was caressed to the stomach and aspirated via a nasogastric tube and those for whom the intestinal content was left uncaressed. Moreover, adhesiolysis should be limited to only the obstructing band or to the adhesions that need to be divided to reach the transition point. Further extensive unnecessary adhesiolysis may be potentially harmful and should be therefore avoided (60).
The intraoperative use of adhesion barriers in prevention of future adhesions after surgery is still a matter of debate. It has been shown that oxidized regenerated cellulose and hyaluronate carboxy methylcellulose reduce adhesion formation (64). Further studies have shown that the use of icodextrin 4% solution in ASBO reduces intra-abdominal adhesions and the risk of re-obstruction (65).
Where to Operate on ASBO Patients?
Operative management for ASBO within 24 h of admission is currently not uniformly adopted, as the proportion of patients managed with early surgery varies from a low of 0% to a high of 33%. Early surgical management is prevalent across large academic hospitals and among hospitals with higher volumes of admissions for ASBO. Moreover, hospitals with a higher proportion of ASBO patients treated with an early surgical operation seem to have a lower likelihood of serious complications, bowel resection, and death (9).
The study by Behman et al. (17) showed that being treated at a hospital with a greater number of beds was one of the additional factors that were significantly associated with greater odds of early operative management in patients with ASBO, with all the related benefits, as described above. This finding may be a reflection of the resources available to larger urban hospitals, with particular regard to the ability to perform after-hours procedures, including the laparoscopic ones. Since surgery, and even more so early surgery, is one of the factors related to better clinical outcomes in patients with ASBO, it is evident that patients referred to tertiary or, in general, high-volume centers have more chance to be treated according to the principles of best evidence practice.
Focus on the Surgical Technique: Laparoscopic Adhesiolysis for ASBO patients
When the laparoscopic approach is attempted for adhesiolysis in ASBO patients, extra care should be taken in placing the first trocar and establishing the pneumoperitoneum. The first trocar should be placed 10 cm away from the previous scar. Usually, the left upper quadrant is chosen as the safest place to put the first trocar if the patient has a midline incision. The Hasson technique is preferred by most authors because it is generally a safe method for accessing the peritoneal cavity, especially in dealing with particularly dilated bowel loops. Alternatively, a blunt dilating tip optical trocar under direct vision at the level of the Palmer’s point may be employed. Closed techniques to establish the pneumoperitoneum or blind insertion of the first port in close proximity to previous scars should be discouraged.
Once the first trocar is placed, the objective is to reach an adequate intra-abdominal visualization and working space to permit the insertion of the remaining trocars. Once the pneumoperitoneum has been gradually established, the surgeon must assess if there is adequate room for good intra-abdominal vision and further safe trocar placement. If not, a timely conversion is recommended, as the lack of a proper vision inside the abdomen mandates withdrawing from any attempt to handle the bowel loops using laparoscopic instruments.
At least three trocars are used, but the number of trocars finally depends on the intra-abdominal situation. Good instrument triangulation should be planned on the basis of the planned site of dissection, with additional trocars placed as needed. Depending on the available laparoscopes, 5 mm trocars or 11 mm trocar for the camera and usually two 5 mm trocars for the laparoscopic instruments are used.
Adhesions to the abdominal wall should be taken down first with laparoscopic cold scissors, identifying the white lane where the parietal peritoneum meets the adhesion and dissecting them in a bloodless plane. Blunt and sharp dissection is preferable to use of electrocautery, due to the risk of transmitting the heat to adjacent bowel loops with subsequent thermal injury, which may cause early perforations or evolve to delayed perforations only several days afterward.
Further recommendations are not to immediately search for the transition point and for the strangulating band by manipulating the distended and frail bowel loops, but rather to start with the identification of the cecum and the ileocecal valve, then starting to explore from the collapsed distal ileal loops in a distal-to-proximal fashion. Only collapsed bowel loops should be manipulated in order to avoid injuries. The distal collapsed loops can be run until reaching a collapsed bowel loop which is fixed and stuck. This is usually the location of the transition point, at which a single obstructing band can be seen. The adhesion should be underpassed with the aid of blunt maneuvers (using a suction device or spreading the two branches of an atraumatic grasper) in order to visualize and isolate the band and at the same time to obtain enough space from the adjacent bowel loops. Once a window is obtained, the band can be carefully cut using cold scissors.
High-energy devices may be safely used if adequate working space inside the abdominal cavity exists and if it is certain that no bowel is hidden in or besides the adhesions. Due to the lack of tactile feeling through the adhesions, laparoscopic adhesiolysis should be safely carried out according to the principle that “if you can see through it, you can cut it.”
If the anatomy is still unclear despite meticulous dissection, changing the position or the angle of the camera and/or placing additional trocars as needed may yield better visualization and working triangulation. Although adding additional 5 mm trocars may facilitate the procedure with less morbidity than converting to an open midline incision would, the principle of erring on the side of safety by maintaining a low threshold for conversion should guide any further surgical action.
Upon completion of the adhesiolysis, it is advisable to run the small bowel loops twice to ensure that there are no missed serosal injuries or full thickness enterotomies. Careful observation for any serosal tears over the loops which were manipulated may allow laparoscopic repair, thus preventing postoperative peritonitis.
Any injuries that are identified intraoperatively should be repaired laparoscopically in a single layer if the surgeon feels confident with this task. Otherwise, if the surgeon does not feel comfortable repairing bowel laparoscopically, two different strategies may be adopted: the injured bowel can be grabbed with a laparoscopic locking bowel grasper so that it can be brought out through a mini-laparotomy, typically made by extending one of the trocar-site incisions, and repaired extracorporeally. Alternatively, a full conversion to laparotomy may be done, especially if doubts about further missed bowel injuries exist.
Conclusion
The management of ASBO continues to evolve (Table 2). Although most ASBO patients without suspicion of bowel strangulation or gangrene are currently managed nonoperatively, the long-term outcomes following this approach need to be analyzed in a more exhaustive way, as surgery performed early during the first episode of ASBO has shown to be highly effective, with a lower rate of recurrence. Laparoscopic adhesiolysis has shown the potential to be an advantageous alternative to open surgery in the management of ASBO in selected patients with an anticipated single obstructing band, especially in high-volume hospitals for ASBO admissions, where patients treated with an early surgical operation have a lower likelihood of postoperative complications, bowel resection, and death.
Five key points on management strategies in patients with ASBO.

ASBO: adhesive small bowel obstruction; CT: computed tomography.
Footnotes
Acknowledgements
The authors thank the members of the World Society of Emergency Surgery working group on adhesive small bowel obstruction for the crucial advancements in the treatment strategies of this disease.
Author Contributions
M.P. contributed to the study conception and design, literature search, acquisition and interpretation of evidence; drafting and critically revising the article for important intellectual content; and final approval of the version to be published. M.K. contributed to the interpretation of evidence; drafting and critically revising the article for important intellectual content; and final approval of the version to be published. S.D.S. contributed to the study conception and design, literature search, acquisition and interpretation of evidence; drafting and critically revising the article for important intellectual content; editing and revising the English for the final version to be published; and final approval of the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Educational videos on how to do it:
