Abstract
Background:
The prevalence of abdominal aortic aneurysms is higher in population with other vascular comorbidities, especially among men. Utility of screening among patients with cerebrovascular disease is unclear.
Objective:
To determine the prevalence of abdominal aortic aneurysm in male patients with diagnosed cerebrovascular disease manifested by transient ischemic attack or stroke.
Methods:
Between May 2013 and May 2014, all consecutive male patients undergoing carotid ultrasound in single tertiary center with a catchment area of 179,000 inhabitants were evaluated for ultrasound screening of abdominal aortic aneurysm. Abdominal aortic aneurysm was defined as maximum diameter of infrarenal aorta 30 mm or more.
Results:
Of 105 (n = 105) consecutively evaluated male patients, only 69% (n = 72) were eligible for the study and underwent aortic screening. Reason for ineligibility was most often poor general medical condition (n = 29). Mean age of screened patients was 66 years (SD 9.8 years). Half of the screened patients suffered stroke (n = 36). The incidence of abdominal aortic aneurysm was 5.6% (n = 4). All found abdominal aortic aneurysms were small and did not require immediate surgical intervention. During a follow-up period of over 4 years, none of the aneurysms exhibited tendency for growth.
Conclusions:
The male population with cerebrovascular disease is comorbid and frail. Only, moderate prevalence of abdominal aortic aneurysms can be found in this subpopulation.
Introduction
Abdominal aortic aneurysm (AAA) is a vascular disease characterized by degeneration and pathological dilatation of abdominal aorta. Diameter of ⩾30 mm is considered as the threshold diameter for diagnosis 1 .
The prevalence of AAA is 3.3% among men aged between 65 and 74 years 2 . Age and male gender and smoking are the predominant risk factors for AAA. Other significant predictors for AAA are the positive family history for AAA, hypertension, and other vascular diseases 3 . However, recent studies implicate remarkable decline in the prevalence of AAA in general population4 –6.
AAA is asymptomatic in almost all clinical cases, and thus, the diagnosis is usually coincidental upon radiologic imaging of the abdomen for other indications. The natural history of the disease is characterized by gradual growth in caliber, but the individual growth rate varies greatly from stable to rapid expansion. Aneurysm size is the predominant and best-known predictor for rupture abdominal aortic aneurysm (RAAA). RAAA demands emergent surgical intervention for salvage, but the course of events after rupture is often grave—the overall mortality to RAAA is up to 80% despite the surgical treatment3, 6 –8. It has been estimated that 1%–2% of deaths of men are caused by RAAA9, 10. Nevertheless, if AAA is treated electively prior to rupture, the operative mortality is less than 5%11,12 and life expectancy mimics the healthy population13 –17. There is a well-established consensus that threshold diameter for treatment should be 50 mm for women and 55 mm for men, and when rapid AAA growth is observed (⩾1 cm/year), fast track referral to a vascular surgeon with additional imaging should be considered.
AAA is usually diagnosed with abdominal ultrasound (US) examination. It has been shown that AAA-related mortality declines with screening programs among men in the general population, but there are controversies over the cost-effectiveness of large screening programs4, 18 –20. At the time of recruitment for this study, a universal consensus for screening for AAA was still lacking, thus making the idea of selective screening for AAA an intriguing idea.
Coexistence of other vascular diseases with AAA has been well-established, and increased prevalence of AAA in population with peripheral artery disease (PAD) and coronary artery disease (CAD) has been proven in multiple studies21 –25. Cerebrovascular disease (CVD) is considered to share the common etiology and pathogenesis with other major vascular diseases26 –28. Even so, recent data on coexistence of AAA with CVD remain scarce.
The early warning sign manifestation of CVD is transient ischemic attack (TIA), where ischemic insult to brain tissue is often still reversible and with treatable cause without permanent loss of function. If symptoms persist, the condition is defined as stroke. The most common treatable cause for TIA and stroke is carotid artery stenosis. Thus, the routine clinical work-up after TIA and stroke includes screening for carotid artery stenosis, which is done with either computer tomography angiography (CTA) or duplex–Doppler ultrasound (ddUS). This makes the idea of annexing abdominal aorta screening for AAA with ddUS of carotid arteries intriguing. Only one previous dedicated screening study has studied the prevalence of AAA among patients with stroke of TIA. Among all patients, the prevalence of AAA was found to be 5.6%, but in post hoc analyses among male patients over 59 years of age, the prevalence was very high reaching up to 11% 26 .
The objective of this study was to replicate and confirm previous findings and study the applicability of screening for non-diagnosed AAAs in men with TIA or stroke undergoing carotid artery US.
Material and Methods
Study Population
In this prospective screening study, consecutively 105 patients were evaluated for eligibility (between May 2013 and May 2014) and 72 patients were recruited and screened during this 1-year period. We included male patients who presented neurological symptoms caused by TIA (or subsequently developing stroke) and thus were referred to ddUS imaging of the carotid arteries. The exclusion criteria were already diagnosed or treated AAA, being unable to provide informed consent and obvious contraindication for elective treatment of AAA (advanced dementia, frailty, or delirious due to previous illness). Study patients were recruited by attending neurologists and screened by the research group physicians upon admission to neurologic ward or emergency department surveillance ward.
The study was conducted in the North Karelia Central Hospital in Joensuu, Finland, with a primary catchment population of 179,000. The study was approved by the ethical committee of the University of Eastern Finland (5/13). The study is registered in the ClinicalTrials.Gov with number NCT01850732. Every participating patient provided a written informed consent prior to screening.
US Screening
The screening for AAA was done using a convex probe on portable US device (Philips HD11 XE, 2009). Largest caliber of infrarenal aorta was registered in sagittal and coronal plane. The dimension of the aorta was measured from outer-to-outer artery wall with threshold diameter of 30 mm to diagnose AAA. Prior to the study, the physicians conducting the screening were validated for competence by consultant radiologist. Radiologist was also available for second opinion in cases of poor visibility to abdominal aorta.
Demographic data of the patients were recorded. If patient was diagnosed with AAA, he was informed and referred to a vascular surgeon.
Statistical Methods
Demographic and descriptive data of the study population are presented using frequencies and percentages for categorical variables. Mean values and standard deviations (SDs) are reported in continuous variables.
Results
Of the 105 patients evaluated for this study, only 72 patients were eligible and finally enrolled. That yields total of 33 drop-outs, which indicates screening coverage of 69% in the initially targeted population. Two of the excluded patients were due to refusal to participate in the study. Two patients were excluded due to previously diagnosed AAA. The rest of 29 patients (29% of the initial target population) were excluded due to severe acute or chronic illness (being unable to provide informed consent and obvious contraindication for elective treatment of AAA).
General characteristics and demographic data of the screened population are presented in Table 1. The prevalence of AAA among the screened population was 5.6% (95% confidence interval (CI) 2.2–13.4; n = 4). All found aneurysms were small, with maximal diameter varying between 30 and 32 mm, and those patients were referred to vascular surgeon for follow-up. 75% (n = 3) of the patients with AAA found in screening were active smokers. None of the AAAs were palpable in clinical examination and were thus discovered exclusively due to the screening.
Demographics of 72 men with TIA symptoms.
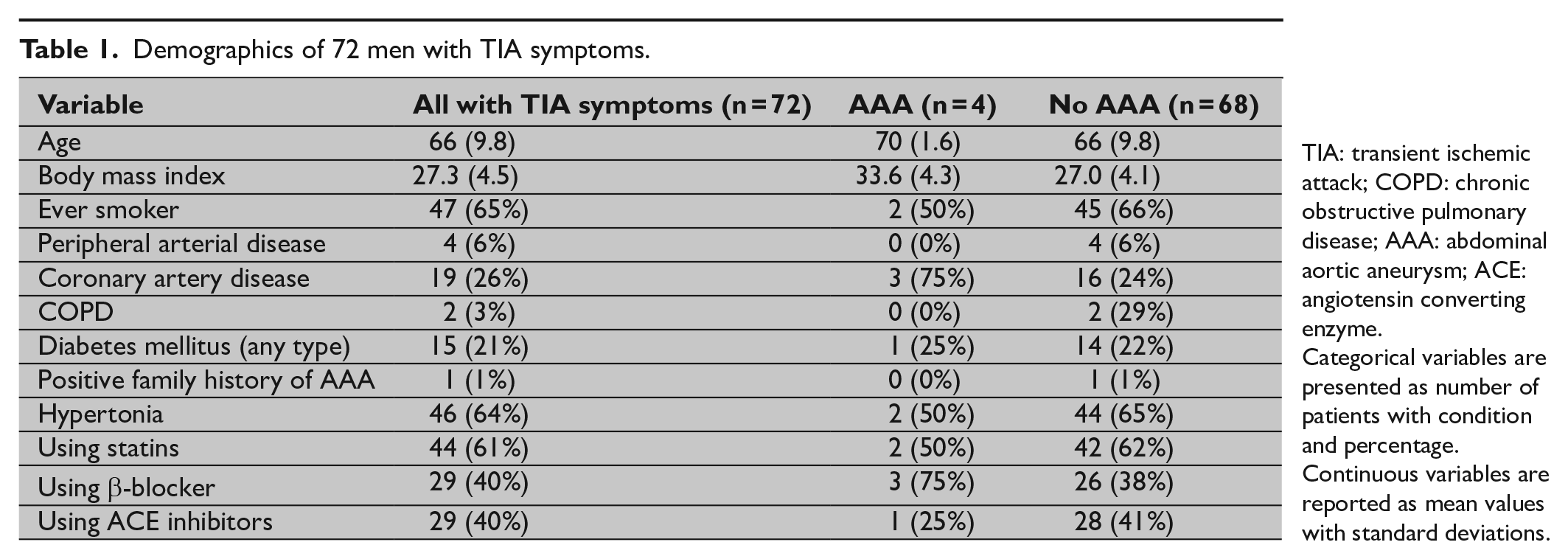
TIA: transient ischemic attack; COPD: chronic obstructive pulmonary disease; AAA: abdominal aortic aneurysm; ACE: angiotensin converting enzyme.
Categorical variables are presented as number of patients with condition and percentage. Continuous variables are reported as mean values with standard deviations.
In 36 (50%) of the originally 72 TIA patients, the symptoms persisted over 24 h and the eventual diagnosis was stroke. Vast majority of the patients (n = 64, 89%) had also computer tomography of the brain to evaluate ischemic changes. The etiology of TIA proved to be cardiogenic in 8% (n = 6) and carotid stenosis in 92% (n = 66) of the patients. None of the patients with cardiogenic etiology had AAA; thus, the prevalence of AAA was 6.0% (4/66) in patients with carotid stenosis.
25% (n = 18) of the patients had at least one revascularization in history, and most of them being cardiac (percutaneous coronary intervention (n = 9), coronary bypass (n = 7), carotid endarterectomy (n = 1), and lower extremity bypass (n = 1)). Only one AAA was found in this subgroup from a patient with universal atherosclerosis and revascularization for both PAD and CAD in history. He was the only smoker among revascularized patients.
Discussion
According to this prospective study on screening of AAA in patients with acute TIA symptoms, the incidence of AAA may be slightly increased in male patients with TIA or stroke, especially when the indication is carotid stenosis. Significant proportion (almost one-third) of the initially evaluated target population was too sick to be eligible for screening (let alone for the treatment of AAA); hence, they were excluded from the study population. For this reason, increasing the sample size by extending the recruitment period was not justified.
Previously, one similar screening study has reported a prevalence of 5.8% among all patients with TIA or stroke 26 . In post hoc analyses, the same study also found that when limiting the analysis to male patients above the age of 59 years would yield a prevalence of 11% for asymptomatic AAA 26 . Our results do not support this conclusion, although we acknowledge that our sample size for patients eligible for screening is small and thus limits the interpretation of the results. Another study conducted among a men and women with TIA, stroke, or internal carotid artery stenosis revealed a prevalence of 5.6% for AAA 27 . Furthermore, it is worth noting that this screening study concentrated only on the clinically significant, previously undiagnosed AAAs. Thus, this article does not reflect the true overall prevalence of AAA in population with CVD.
Overall, there is a vast body of literature of AAAs in conjunction with other vascular diseases3, 6, 28, 29, and compared to this, the recent data on coexistence of AAA with carotid artery stenosis, stroke, or TIA are scarce. Majority of the previous publication on this subject do not report drop-out percentages, and these problems have been discussed earlier 30 . However, when the findings of this study are compared to the recent data of Swedish and English national gender- and age-based screening programs4, 5, the prevalence of newly diagnosed AAAs in our study population is remarkably high.
All found AAAs in this study were small and did not show a significant tendency for growth during a follow-up period of 4 years. Male patients hospitalized for TIA or subsequent stroke are a highly comorbid population. Due to the small overall sample size, the estimate of the prevalence of previously undiagnosed AAAs based on this study population is questionable, and no conclusions can be made to justify routine screening of this kind of subpopulation. Most likely, if targeted screening strategy would prove itself useful, it would be based on different primary inclusion criteria than TIA symptoms upon arrival.
Conclusion
The male population with TIA symptoms is rich in comorbidities, and only 71% of this population would be candidates for targeted AAA screening. Only a limited number of previously undiagnosed small AAAs were found in this population.
Footnotes
Acknowledgements
The authors thank Tapio Hakala, M.D., Ph.D., consultant cardiothoracic surgeon in the North Karelia Central Hospital and the founding member of this study group, who passed away before writing the manuscript. The authors also thank Matti Turtiainen, M.D., consultant radiologist in the North Karelia Central Hospital, who provided assistance and second opinion on cases with poor visibility to aorta.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the EVO funding (Erityis Valtion Osuus, i.e. State Subsidy Fund) of the University of Eastern Finland (research grant) and the EVO funding of the Kuopio University Hospital.
