Abstract
Background
Patients who experience mild ischemic stroke or transient ischemic attack (TIA) face an increased likelihood of stroke recurrence within 90 days following the initial thrombotic complication.1–4 Antiplatelet therapy plays an essential role in reducing the risk of stroke recurrence. Clopidogrel is a blocker of platelet aggregation that acts through the P2Y12-receptor pathway, the mode of action being synergistic to aspirin in aggregation assays. 5 Dual antiplatelet therapy (DAPT) with aspirin and clopidogrel leads to a synergistic effect and reduced risk of ischemic events. 6 This combination therapy has been proven to be more efficacious than aspirin monotherapy for decreasing the risk of thrombotic events in patients diagnosed with acute coronary syndromes. 7
The Clopidogrel in High-Risk Patients With Acute Nondisabling Cerebrovascular Events (CHANCE) trial showed that DAPT with aspirin and clopidogrel reduced the risk of recurrent stroke by 32% compared with aspirin monotherapy. Moreover, the risk of major bleeding remained low. 8 Recently, the Intensive Statin and Antiplatelet Therapy for Acute High-Risk Intracranial or Extracranial Atherosclerosis (INSPIRES) trial, which included over 6000 patients with mild ischemic stroke or high-risk TIA, showed that aspirin and clopidogrel therapy within 72 hours of the symptom onset reduced stroke recurrence; however, it was associated with an increased likelihood of moderate or severe bleeding. 9
We undertook a systematic review and meta-analysis of randomized controlled trials (RCTs) to assess the safety and efficacy of aspirin plus clopidogrel versus aspirin monotherapy in patients with mild ischemic stroke or high-risk TIA to provide comprehensive and robust evidence regarding the utility of both treatment modalities. This is the largest meta-analysis on this subject as we pooled the INSPIRES 2023 trial not included in prior studies.
Methods
We adhered to the guidelines established by the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) statement for conducting this systematic review and meta-analysis. 10 The protocol of review is registered with PROSPERO (ID: CRD42024499310). No ethical approval was required for the study.
Data sources and search strategy
Two investigators (A Ahsan and L Fatima) independently searched PubMed/MEDLINE, Embase, the Cochrane Library, and ClinicalTrials.gov from their inception until December 31, 2023. The researchers manually examined references of retrieved RCTs, prior meta-analyses, and reviews to ensure that all relevant articles were included. The search strategy used the following keywords and Medical Subject Headings (MeSH) terms: “aspirin”, “antiplatelet therapy”, “clopidogrel”, “ischemic stroke”, “transient ischemic attack”, and “randomized controlled trials”. The details of the search strategy are provided in Supplementary Table 1.
Eligibility criteria and outcomes
Studies were deemed eligible for inclusion in our meta-analysis if they adhered to the following criteria: (i) were RCTs comparing aspirin plus clopidogrel versus aspirin alone; (ii) included patients with a noncardioembolic mild ischemic stroke National Institutes of Health Stroke Scale (NIHSS) score ⩽ 5 or high-risk TIA; (iii) therapy was started within 3 days; (iv) evaluated at least one of the below-listed efficacy or safety outcomes.
The primary efficacy outcome was recurrent stroke (ischemic or hemorrhagic) and the primary safety outcome was moderate or severe bleeding (definitions of bleeding used in each trial are provided in Supplementary Table 2). Secondary outcomes included ischemic stroke, hemorrhagic stroke, all-cause mortality, cardiovascular (CV) mortality, myocardial infarction (MI), and major adverse cardiovascular events (MACE) defined as the composite of MI, any stroke, and CV mortality.
Study selection and data extraction
The studies obtained from the literature search were imported to EndNote X9 (Clarivate Analytics), and duplicate records were removed. Two authors (M Ahmed and A Ahsan) independently reviewed RCTs based on titles and abstracts. This was followed by a review of the full texts of the articles. A third author (L Fatima) was consulted in the event of any disagreements.
We extracted the following data from each eligible RCT: trial name, year of publication, country, total sample size, inclusion criteria, dose of aspirin and clopidogrel in the treatment arm, dose of aspirin in the comparison arm, duration of DAPT, treatment onset time, duration of follow-up, age of patients, sex, various risk factors such as hypertension, diabetes, dyslipidemia, smoking status, history of MI, and any history of previous stroke or TIA. We used a prepiloted Excel spreadsheet for data extraction.
Bias assessment of the included studies
Version 2 of the Cochrane Risk of Bias (RoB 2.0) tool for RCTs was used to assess the risk of bias. 11 Risk of bias was assessed across five domains: randomization, deviations from intended variation, missing outcome data, measurement of outcome, and selection of reported results. The trials were scored as high, with some concerns, or low risk of bias in each domain.
Statistical analysis
We used RevMan, Version 5.4 (Nordic Cochrane Center, Copenhagen, Denmark) for running statistical analysis. Risk ratios (RRs) were pooled along with their corresponding 95% CIs for all clinical endpoints. We used the DerSimonian–Laird random effects model for pooling results to account for inter-study heterogeneity. 12 The results were visualized through the generation of forest plots. Heterogeneity across the trials was assessed using the Higgins I2 test. 13 We also performed subgroup analyses based on the treatment onset time (within 24 hours vs within 72 hours) and duration of antiplatelet therapy (21 days vs 90 days). A sensitivity analysis was conducted directly based on the reported hazard ratio (HR) by the studies. In all cases, a p-value of less than 0.05 was regarded as statistically significant.
Results
Search results
An initial search of the databases yielded 2767 records. After removing 458 duplicates, 2309 unique records were subjected to title and abstract screening. This process identified 48 potentially eligible reports retrieved for full-text assessment based on the eligibility criteria. Four studies were deemed eligible and included in our meta-analysis. The details of screening and the study selection process are provided in Supplementary Figure 1.
Study and patient characteristics
All included studies were RCTs,8,9,14,15 published from 2007 to 2023, and 16,547 patients were incorporated in our pooled analysis. Aspirin plus clopidogrel was administered in 8267 patients whereas 8280 patients received aspirin alone after ischemic stroke or TIA. The median age of patients ranged from 65 to 74 years. Men constituted > 50% of the participants in each trial. All studies had a follow-up duration of 90 days. The duration of DAPT ranged from 21 to 90 days. The baseline characteristics of the included patients varied across trials: hypertension (46–69.9%), diabetes (9–28%), dyslipidemia (3.4–11.2%), current or previous smoking status (20.4–43.2%), MI (1.7–10.6%), and history of prior stroke (1.6–56.6%) or TIA (3.1–43%). The details of the baseline characteristics are provided in Table 1. The risk of bias assessment of the included trials performed using RoB 2.0 showed some concerns for one RCT but the other pooled RCTs had a low risk of bias. The details are provided in Figure 1.
Baseline characteristics of the included studies and participants.

ABCD2, age, blood pressure, clinical features, duration of TIA, diabetes mellitus; C, comparison (aspirin alone); CHANCE, Clopidogrel in High-Risk Patients With Acute Nondisabling Cerebrovascular Event 8 ; d, day; DAPT, dual antiplatelet therapy (aspirin + clopidogrel); FASTER, Fast Assessment of Stroke and Transient Ischemic Attack to Prevent Early Recurrence 14 ; INSPIRES, Intensive Statin and Antiplatelet Therapy for Acute High-Risk Intracranial or Extracranial Atherosclerosis 9 ; NIHSS, National Institutes of Health Stroke Scale; POINT, Platelet-Oriented Inhibition in New TIA and Minor Ischemic Stroke 15 ; TIA, transient ischemic attack; WHO, World Health Organization.
Mean ± SD.

Risk of bias assessment for the included trials.
Clinical outcomes
Stroke
Recurrent stroke (ischemic or hemorrhagic) was reported by all four studies on 90 days’ follow-up. Compared with aspirin monotherapy, DAPT significantly reduced the risk of recurrent stroke by 26% (RR: 0.74; 95% CI: 0.67–0.83; p < 0.00001; Figure 2A). No heterogeneity was detected among the studies (I2 = 0%). Sensitivity analysis based on the HR reported by studies showed similar results (HR: 0.73; 95% CI: 0.66–0.82; p < 0.00001; Supplementary Figure 2).
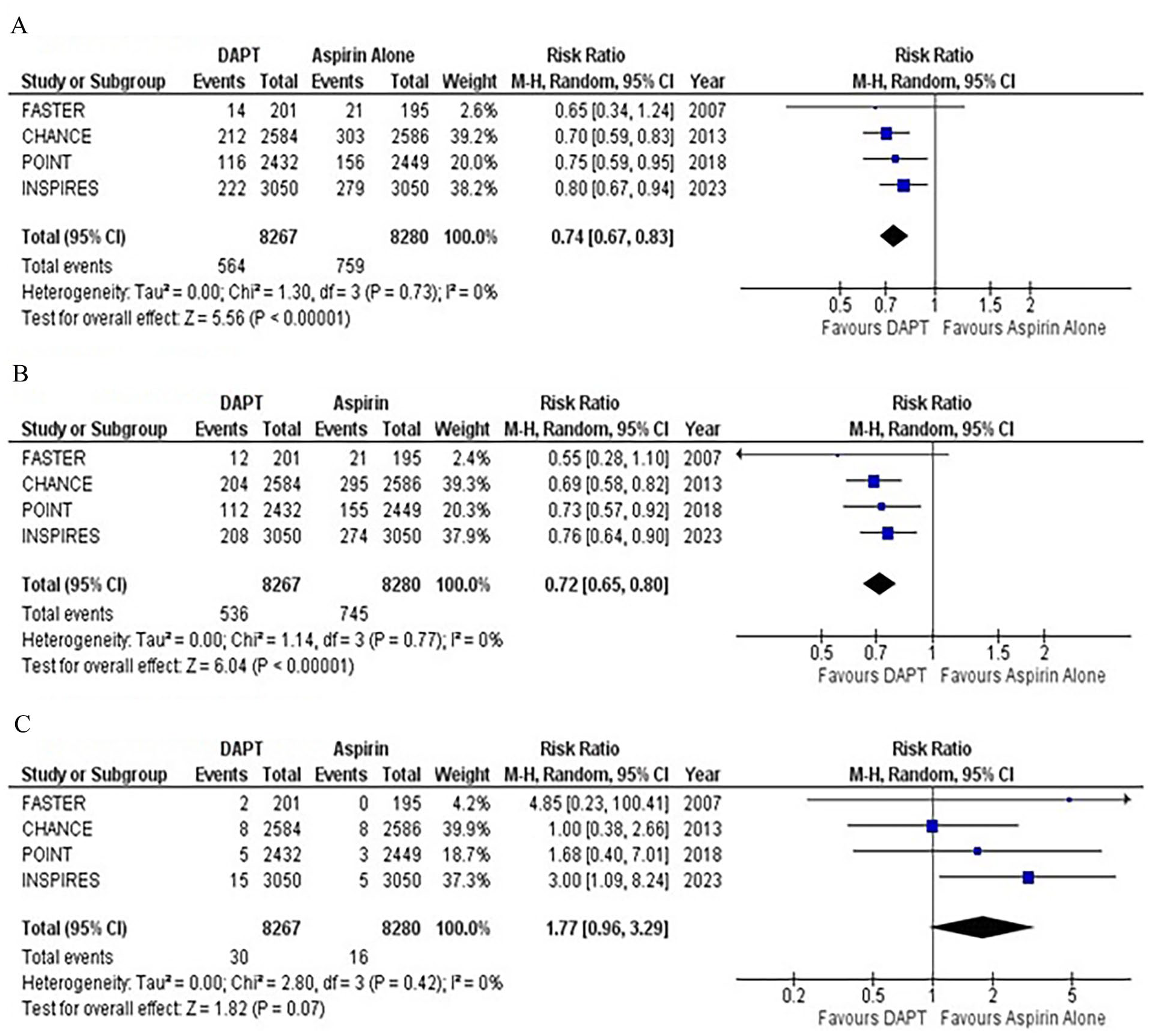
Forest plot for (
All the studies reported data for ischemic stroke. Compared with aspirin monotherapy, DAPT significantly reduced the risk of ischemic stroke by 28% (RR: 0.72; 95% CI: 0.65–0.80; p < 0.00001; Figure 2B). No heterogeneity was detected between the studies (I2 = 0%). A sensitivity analysis was conducted by pooling reported HRs, which yielded similar results (HR: 0.71; 95% CI: 0.64–0.80; p < 0.00001; I2 = 0%; Supplementary Figure 3A).
Hemorrhagic stroke was reported by four studies with nonsignificant statistical differences observed between the two groups (RR: 1.77; 95% CI: 0.96–3.29; p = 0.07; Figure 2C). No heterogeneity was detected among the studies (I2 = 0%). A sensitivity analysis was conducted by pooling reported HRs, which yielded similar results (HR: 1.71; 95% CI: 0.86–3.38; p = 0.13; I2 = 0%; Supplementary Figure 3B). Results stratified by treatment onset time showed comparable results (Supplementary Figures 4–6).
All-cause and CV mortality
All-cause mortality, reported in four studies, showed nonsignificant statistical differences between the two groups (RR: 1.25; 95% CI: 0.87–1.80; p = 0.23; Figure 3A). No heterogeneity was detected (I2 = 0%). A sensitivity analysis was conducted by pooling reported HRs, which yielded similar results (HR: 1.25; 95% CI: 0.86–1.81; p = 0.24; I2 = 0%; Supplementary Figure 7A).
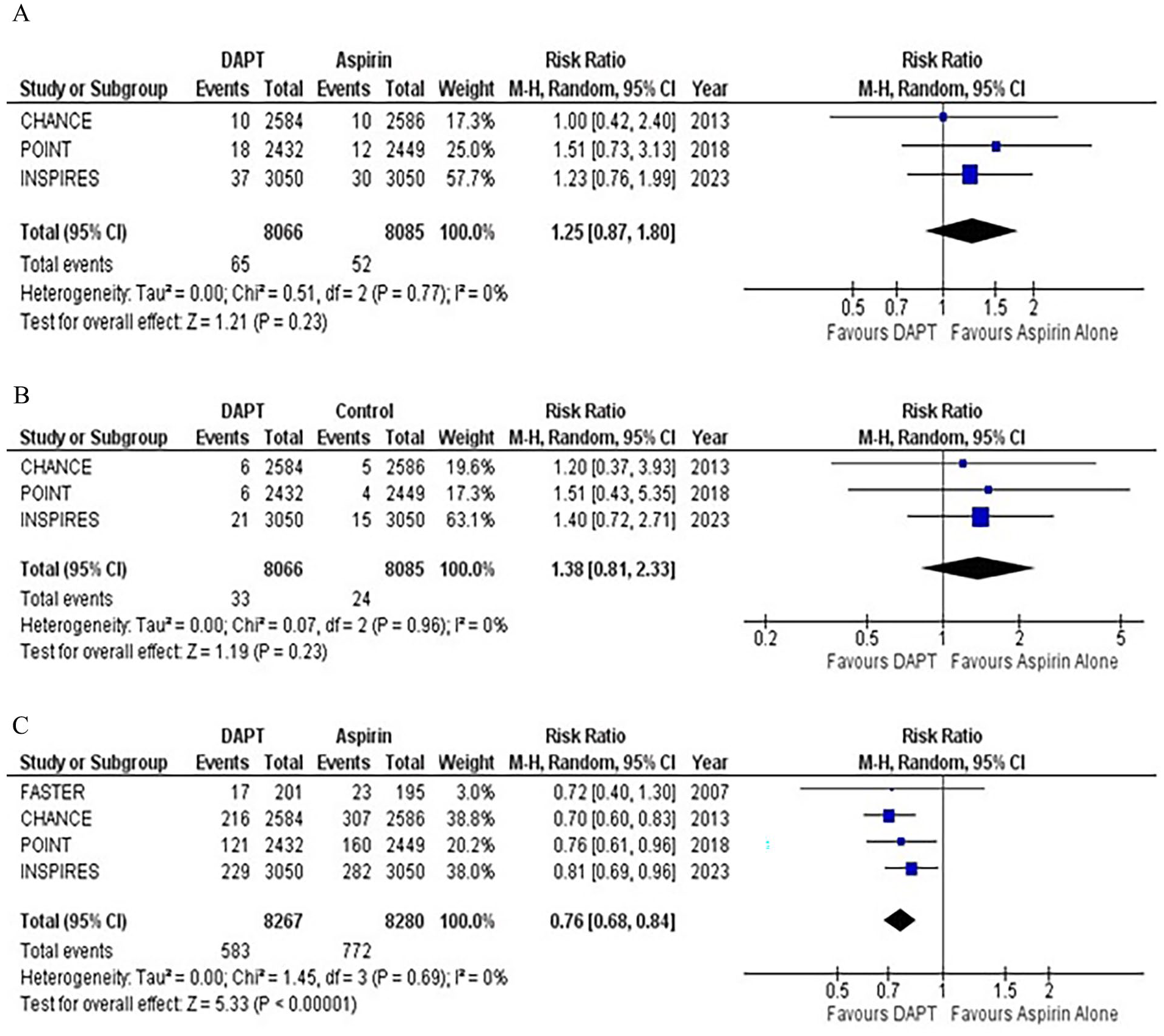
Forest plot for (
Three RCTs reported cardiovascular-related death. The risk was comparable between the two groups, with no heterogeneity reported among the outcomes (RR: 1.38; 95% CI: 0.81–2.33; p = 0.23; I2 = 0%; Figure 3B). The sensitivity analysis based on HR demonstrated similar results (HR: 1.37; 95% CI: 0.81–2.32; p = 0.24; I2 = 0%; Supplementary Figure 7B). Subgroup analysis showed that comparable results were observed in both (24 vs 72 hours) groups (Supplementary Figures 8 and 9).
MACE and MI
All studies reported the outcome of MACE. Compared with aspirin monotherapy, DAPT significantly reduced the risk of MACE by 24% (RR: 0.76; 95% CI: 0.68–0.84; p < 0.00001; Figure 3C). No heterogeneity was observed among the studies (I2 = 0%). The sensitivity analysis showed similar results (HR: 0.74; 95% CI: 0.67–0.83; p < 0.00001; I2 = 0%; Supplementary Figure 10). Subgroup analysis also showed significant reduction in MACE with both treatment onset (24 vs 72 hours) durations (Supplementary Figure 11).
MI, reported by three studies, showed no statistically significant difference between the two groups (RR: 1.63; 95% CI: 0.77–3.46; p = 0.20; Figure 4A). No heterogeneity was detected among the studies (I2 = 0%). The sensitivity analysis showed comparable results (HR: 1.62; 95% CI: 0.76–3.44; p = 0.21; I2 = 0%; Supplementary Figure 12). Similar findings were observed in subgroup analysis (Supplementary Figure 13).
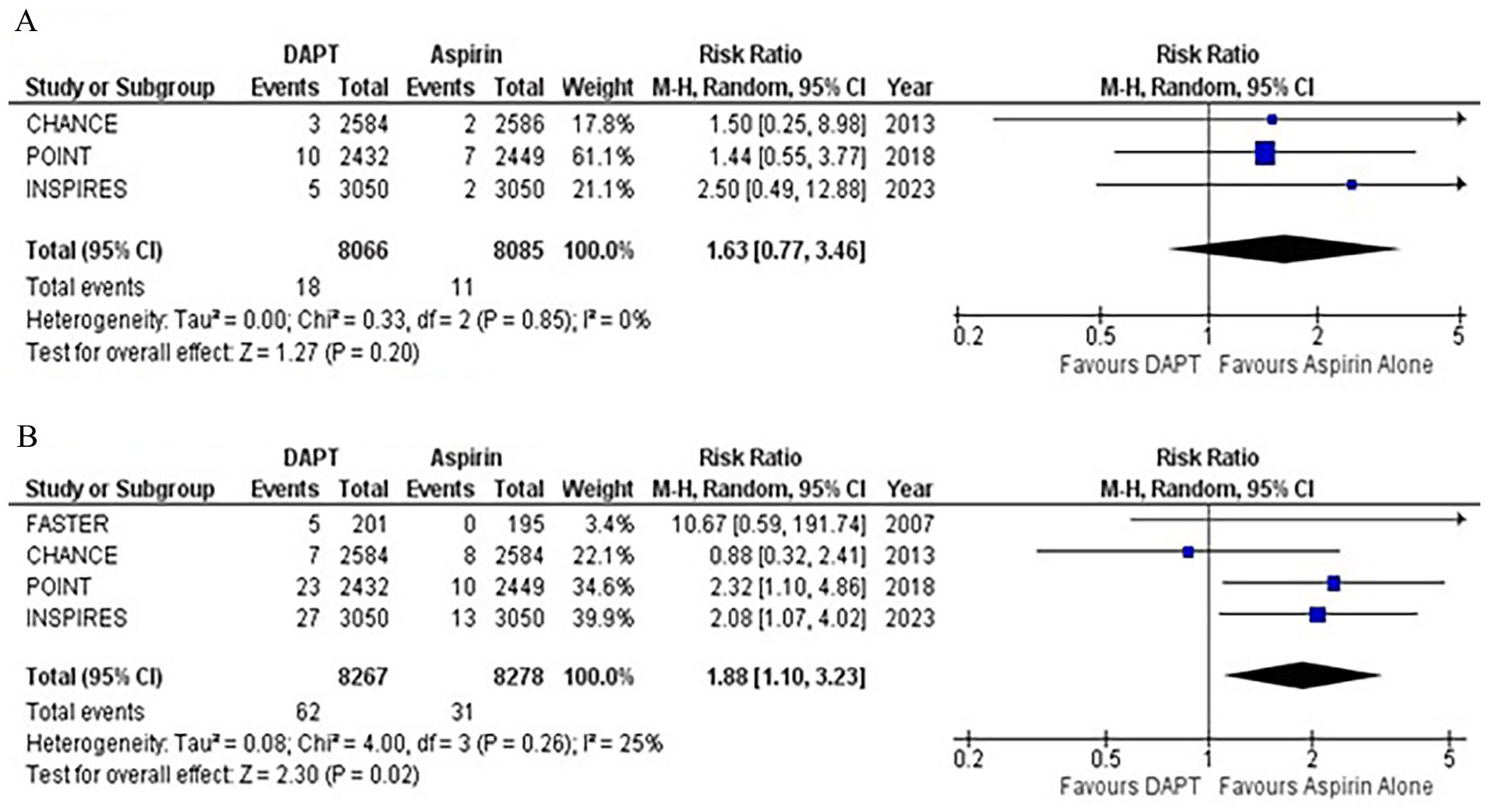
Forest plot for (
Moderate or severe bleeding
All four studies reported moderate or severe bleeding as an outcome. Compared with aspirin monotherapy, DAPT was associated with a significantly increased risk of moderate or severe bleeding (RR: 1.88; 95% CI: 1.10–3.23; p = 0.02; Figure 4B). Mild heterogeneity was observed (I2 = 25%). A sensitivity analysis was conducted by pooling reported HRs, which yielded similar results (HR: 1.98; 95% CI: 1.24–3.15; p = 0.004; I2 = 0%; Supplementary Figure 14). Point estimates on subgroup analysis showed that DAPT for 90 days was associated with a nonsignificant increase in the risk of bleeding as compared to DAPT for 21 days (Supplementary Figure 15).
Discussion
This comprehensive meta-analysis of 16,547 patients with noncardioembolic mild ischemic stroke or TIA showed that DAPT with aspirin plus clopidogrel significantly reduced the risk of recurrent stroke (ischemic or hemorrhagic), ischemic stroke, and MACE. However, it resulted in an increased risk of moderate or severe bleeding. No statistically significant changes were observed for hemorrhagic stroke, all-cause mortality, CV mortality, and MI.
The Fast Assessment of Stroke and Transient Ischemic Attack to Prevent Early Recurrence (FASTER) trial 14 was the first RCT that evaluated the efficacy of DAPT with aspirin plus clopidogrel in patients with minor ischemic stroke or TIA. This trial showed that the risk of recurrent stroke was lower in the DAPT group; however, the results failed to reach statistical significance. Moreover, the hemorrhagic complications were high with DAPT. Since then, three large RCTs have been conducted that include CHANCE, 8 the Platelet-Oriented Inhibition in New TIA and Minor Ischemic Stroke (POINT), 15 and INSPIRES trials. 9
Both CHANCE and POINT trials demonstrated that DAPT with aspirin plus clopidogrel started within 24 hours led to a significantly reduced stroke recurrence during 90 days of follow-up compared with aspirin monotherapy. In the INSPIRES trial, therapy was initiated within 72 hours after symptom onset, which resulted in a significantly reduced stroke recurrence. The results of this trial indicated that treatment started within 3 days after ischemic events was efficacious in reducing stroke recurrence, similar to the 24-hour window. Current guidelines recommend that the DAPT with aspirin and clopidogrel should be started within 24 hours after ischemic stroke or TIA. 16 This narrow time window of 24 hours has limited the application of DAPT in ischemic stroke. Our meta-analysis showed that the therapy started within a time frame of 72 hours also led to a significantly reduced risk of recurrent stroke on 90 days’ follow-up. The sensitivity analysis showed similar significant results. A secondary analysis of the POINT trial also showed that DAPT started within a period of 3 days is helpful in preventing stroke recurrence. 17
We observed that the risk of moderate or severe bleeding was significantly higher in patients who received aspirin plus clopidogrel. This increased risk was mainly driven by the POINT and INSPIRES trials. However, the risk of bleeding was low in the CHANCE trial. In the POINT trial, DAPT was continued for 90 days, which could have led to an increased risk of bleeding compared to the CHANCE trial where therapy lasted for 21 days. Moreover, the loading dose of clopidogrel used in the POINT trial was 600 mg whereas the CHANCE trial used a loading dose of 300 mg. The INSPIRES trial used a similar dose of clopidogrel as the CHANCE trial. However, the aspirin dose employed in the INSPIRES trial was slightly higher (100–300 mg loading dose followed by 100 mg daily in INSPIRES vs 75 mg in CHANCE). The difference in doses of drugs and duration of DAPT might have contributed to the high risk of bleeding observed in POINT and INSPIRES trials. DAPT for 21 days along with specified loading doses can reduce the risk of bleeding. The time-course analysis of RCTs has also shown that the therapy for 21 days is effective and safe in patients with noncardioembolic ischemic stroke or TIA.17,18 It is important to mention the use of effective medical therapy and blood pressure management, which can potentially reduce the risk of hemorrhagic complications in patients receiving DAPT.19,20 The NASCET trial 20 showed that controlling blood pressure can lower the risk of hemorrhages or subarachnoid hemorrhages. The investigators found that the incidence of hemorrhages was only 0.5% in the group receiving proper blood pressure management. Moreover, the treatment should be based on the risk profile of patients so that the patients who are more likely to experience a hemorrhagic complication are closely monitored along with a possible dose adjustment.
It is important to mention that the role of ticagrelor plus aspirin has been studied as well in minor stroke. The THALES (Acute Stroke or Transient Ischemic Attack Treated With Ticagrelor and ASA for Prevention of Stroke and Death) trial demonstrated that using ticagrelor plus aspirin in patients with minor stroke can reduce stroke recurrence. 21 However, the researchers found that DAPT with ticagrelor plus aspirin resulted in an increased risk of major bleeding. A network meta-analysis compared DAPT with aspirin plus clopidogrel and aspirin plus ticagrelor to aspirin monotherapy. 22 The investigators found that DAPT was superior to aspirin monotherapy, and two DAPT regimens had comparable efficacy for reducing stroke recurrence. The risk of major bleeding was higher in the patients who received aspirin plus ticagrelor than in the group receiving aspirin plus clopidogrel. Paciaroni et al. 23 found that clopidogrel monotherapy can significantly reduce the risk of stroke recurrence in patients with recent ischemic stroke as compared to aspirin alone. Interestingly, the risk of bleeding was also significantly lower in the clopidogrel monotherapy group. However, the study had a broader inclusion criterion and was not specifically limited to minor ischemic stroke or high-risk TIA. This shows the need for further randomized studies to assess the efficacy of various antiplatelet therapies in this population which could help mitigate hemorrhagic complications.
This is the largest meta-analysis to date that directly evaluated the safety and efficacy of aspirin plus clopidogrel in patients with mild ischemic stroke or high-risk TIA. A prior meta-analysis pooled three RCTs with a sample size of 10,447 patients. 24 We performed an extensive literature search and included the INSPIRES trial, which is currently the largest RCT that evaluated clinical outcomes after administration of aspirin plus clopidogrel versus aspirin alone in patients with ischemic stroke (NIHSS score ⩽ 5) or high-risk TIA (ABCD2 ⩾ 4). A sensitivity analysis was conducted based on HR for all clinical endpoints. Subgroup analyses were performed to assess the impact of treatment onset time and duration of antiplatelet therapy on clinical outcomes as well.
Our study has some limitations. This is a study-level analysis and individual patient data (IPD) meta-analysis will provide more insights regarding the efficacy of DAPT in minor stroke and potential effect modifiers. Secondly, the duration of DAPT and treatment onset varied across the pooled trials. It is important to mention that a large proportion of patients included in the POINT trial had a previous history of stroke or TIA, which might have influenced the results. Moreover, there was a difference in the baseline characteristics of the included studies, which could have affected the pooled estimates. The loading dose of drugs also varied across the pooled studies, which could lead to difficulty in deciding the drug dosage.
Conclusion
DAPT with aspirin and clopidogrel reduces stroke recurrence and MACE in patients presenting with ischemic stroke or high-risk TIA compared to aspirin monotherapy. DAPT is associated with an increased risk of moderate or severe bleeding when compared with aspirin monotherapy. Further evidence is still required regarding accurate drug dosage and duration of DAPT.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241265335 – Supplemental material for Efficacy and safety of aspirin plus clopidogrel versus aspirin alone in ischemic stroke or high-risk transient ischemic attack: A meta-analysis of randomized controlled trials
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241265335 for Efficacy and safety of aspirin plus clopidogrel versus aspirin alone in ischemic stroke or high-risk transient ischemic attack: A meta-analysis of randomized controlled trials by Mushood Ahmed, Areeba Ahsan, Laveeza Fatima, Jawad Basit, Abdulqadir J Nashwan, Shafaqat Ali, Mohammad Hamza, Iosif Karalis, Raheel Ahmed, Ahmad Alareed, Nkechinyere N Ijioma and M Chadi Alraies in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
