Abstract
Purpose:
To evaluate feasibility, quality of life, toxicity, and cosmetic outcome for intraoperative breast cancer brachytherapy after breast-conserving surgery using high dose rate brachytherapy.
Methods:
Fifty-two consecutive women, ⩾50 years old, diagnosed with a unifocal non-lobular breast cancer ⩽3 cm, N0, underwent breast-conserving surgery and sentinel node biopsy. Twenty-five women received intraoperative brachytherapy pre-pathology at primary surgery and the others post-pathology, during a second procedure. An applicator, connected to a high dose rate afterloader, was used. Two of the women were excluded due to metastases found per-operatively at a frozen section from the sentinel node. Quality of life was evaluated using two validated health questionnaires. Treatment toxicity was documented according to the LENT-SOMA scale by two oncologists. The cosmetic result was evaluated using the validated freely available software BCCT.core 2.0.
Results:
The clinical procedure worked out well logistically. Seven women received supplementary external radiotherapy due to insufficient margins and, in one case, poor adaptation of the breast parenchyma to the applicator. No serious adverse effects from irradiation were registered. The results from the health questionnaires showed no major differences compared with reference groups from the Swedish population. Only two women were registered as having a “poor” cosmetic result while a majority of the women had a “good” outcome.
Conclusions:
This pilot study shows that intraoperative brachytherapy is a feasible procedure and encourages further trials evaluating its role in treatment of early breast cancer.
Keywords
Introduction
Today a majority of women in Sweden diagnosed with breast cancer undergo breast-conserving surgery (BCS) 1 . The benefit of postoperative radiotherapy to the remaining breast tissue is well established in several randomized trials and in a large meta-analysis from Early Breast Cancer Trialists’ Collaborative Group (EBCTG) 2 . Conventional external radiotherapy is delivered at a total dose of 40–50 Gy over 3–5 weeks postoperatively. In order to avoid the prolonged treatment and, for some patients, excessive travel time to the hospital, some women choose a mastectomy. Moreover, screening programs and increased public awareness have led to earlier diagnosis, with many small and lymph node negative tumors diagnosed. Using existing treatment routines may result in overtreatment of some of these breast tumors, which perhaps never would have been of any clinical importance.
New technologies, such as breathing adapted treatment and proton therapy3,4, have reduced heart and lung doses. However, the risk of side effects from radiotherapy is still a reality, especially for smokers and women with cardiac risk factors 5 .
At the latest St. Gallen International Breast Cancer Conference 2017, the issue of escalating/de-escalating breast cancer treatment was highlighted 6 . The panel suggested that partial-breast irradiation (PBI) may be considered for a low-risk group of tumors defined by the American Society for Radiation Oncology ASTRO 7 and the Breast Cancer Working Group of the Groupe Européen de Curiethérapie–European Society for Therapeutic Radiology and Oncology (GEC-ESTRO) 8 , especially when endocrine therapy is prescribed. In brief, this low-risk group would include women age ⩾50 years with non-lobular T1–T2 N0 tumors, even if the selection criteria differ between different national societies 9 .
Since early local recurrences after BCS most commonly occur in the near vicinity of the primary tumor10,11, it seems logical to concentrate radiotherapy to this volume of the breast parenchyma. In the TARGIT-A trial 12 , women with early breast cancer who underwent BCS were randomized to receive conventional external beam radiotherapy (EBRT) or intraoperative radiotherapy (IORT) using the Intrabeam® system. Feasibility and safety were reported after a median follow-up of 29 months. The treatment was well tolerated 9 . The estimated 5-year risk for local recurrence for the IORT group was 3.3% and 1.3% for the EBRT group, and so the pre-defined non-inferiority margin of a 2.5% increase was not reached. IORT concurrently with BCS (pre-pathology group) showed the same results (2.1% vs 1.1%); while with delayed IORT (post-pathology group), the difference between the groups were larger (5.4% vs 1.7%). The authors concluded that IORT should be considered as an option for carefully selected patients.
PBI after BCS, as an alternative to conventional external radiotherapy, may be delivered in different forms. Pulsed dose rate (PDR) brachytherapy is already in use at the University Hospital of Örebro and a previous clinical trial shows promising outcomes after a median follow-up of 7 years 13 . The procedure consists of catheters inserted into the breast parenchyma with the guidance of an “index point” where the tumor center was located. The treatment was given by pulsed irradiation—50 Gy given in 12 pulses per day over 5 days. The 5- and 7-year rates of actuarial local control were 96% and 96%, respectively. PBI has also been delivered using a balloon device 14 . Intraoperative brachytherapy (IOBT) is in use in many countries all over the world, but so far no attempt has been made to implement this technique in Sweden. Our breast team at the University Hospital of Örebro has taken advantage of the opportunity to use a novel brachytherapy applicator connected to a high dose rate (HDR) afterloading machine to treat 50 women with early breast cancer in a pilot study. Primary endpoints were feasibility, treatment side-effects, and expenses. Secondary endpoints were patient’s satisfaction and quality of life, evaluated by two health questionnaires. We also evaluated the cosmetic outcome.
Methods and Materials
Patients
The study was performed at the University Hospital of Örebro, Sweden. Fifty-two consecutive women, ⩾50 years old, diagnosed with breast cancer who underwent BCS, were included from September 2013 to June 2015 (Fig. 2). Written informed consent was obtained. Two of the women were excluded due to metastases found per-operatively at a frozen section from the sentinel node. All of the remaining women had a mammographically unifocal breast cancer, ⩽30 mm and an axilla free from metastases. Patients with lobular cancer either on the preoperative biopsy or at final histopathological report were excluded. Patients with positive margins at the final histopathological report or extensive ductal cancer in situ received conventional EBRT as a complement to IOBT. Patients, tumors, and treatment characteristics are shown in Table 1.
Patient, tumor, and treatment characteristics.
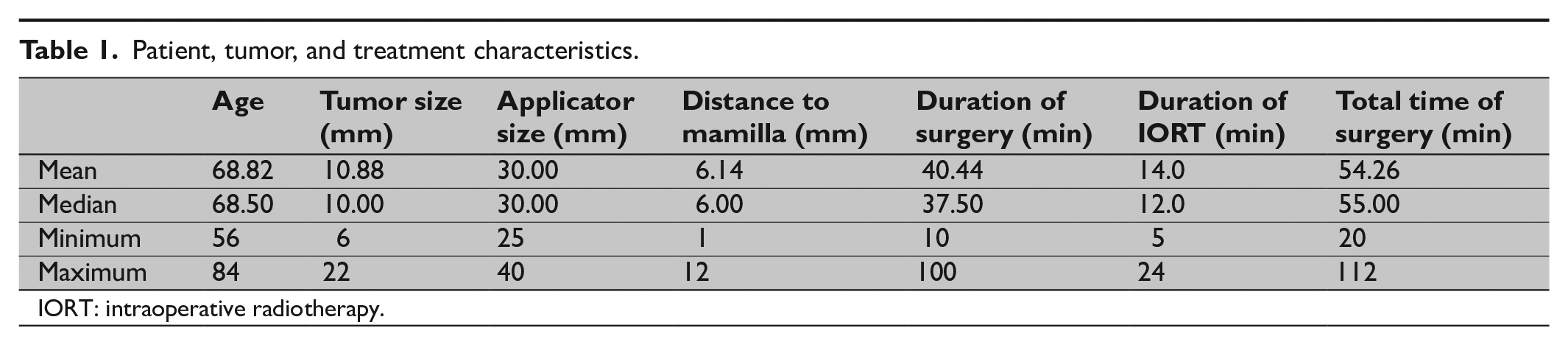
IORT: intraoperative radiotherapy.
Radiotherapy
HDR brachytherapy has for a long time been used for the treatment of prostatic and gynecological cancer15,16. The current isotope is Iridium 192, which is the most commonly used isotope for HDR brachytherapy applications. This is also the isotope used in this study. A reusable applicator of a plastic material, PEEK®, shaped according to the anisotropic radiation dose distribution was developed (Fig. 1). The applicator was attached to a pole approved to be connected to a MicroSelectron® HDR machine (Elekta AB, Stockholm, Sweden). Five sets of applicators with a diameter of 25, 30, 35, 40, and 50 mm respectively were constructed. A single dose of 20 Gy, prescribed at the applicator surface, was delivered in the wound cavity. The dose fall from the applicator surface varied due to the diameter of the applicator (Table 2). A medical physicist calculated the treatment time from a dose-plan library based on source strength and applicator dimension.

IOBT applicator.
The distance from the surface of the applicators with different diameters to the 10-Gy isodose.

The 10-Gy isodose volume outside the applicator was determined using planning CT and the treatment planning system Oncentra Brachy® (Elekta AB) (Fig. 3). Air cavities inside this shell were outlined, and their volumes were calculated as a measure of the tissue adaption to the applicator.
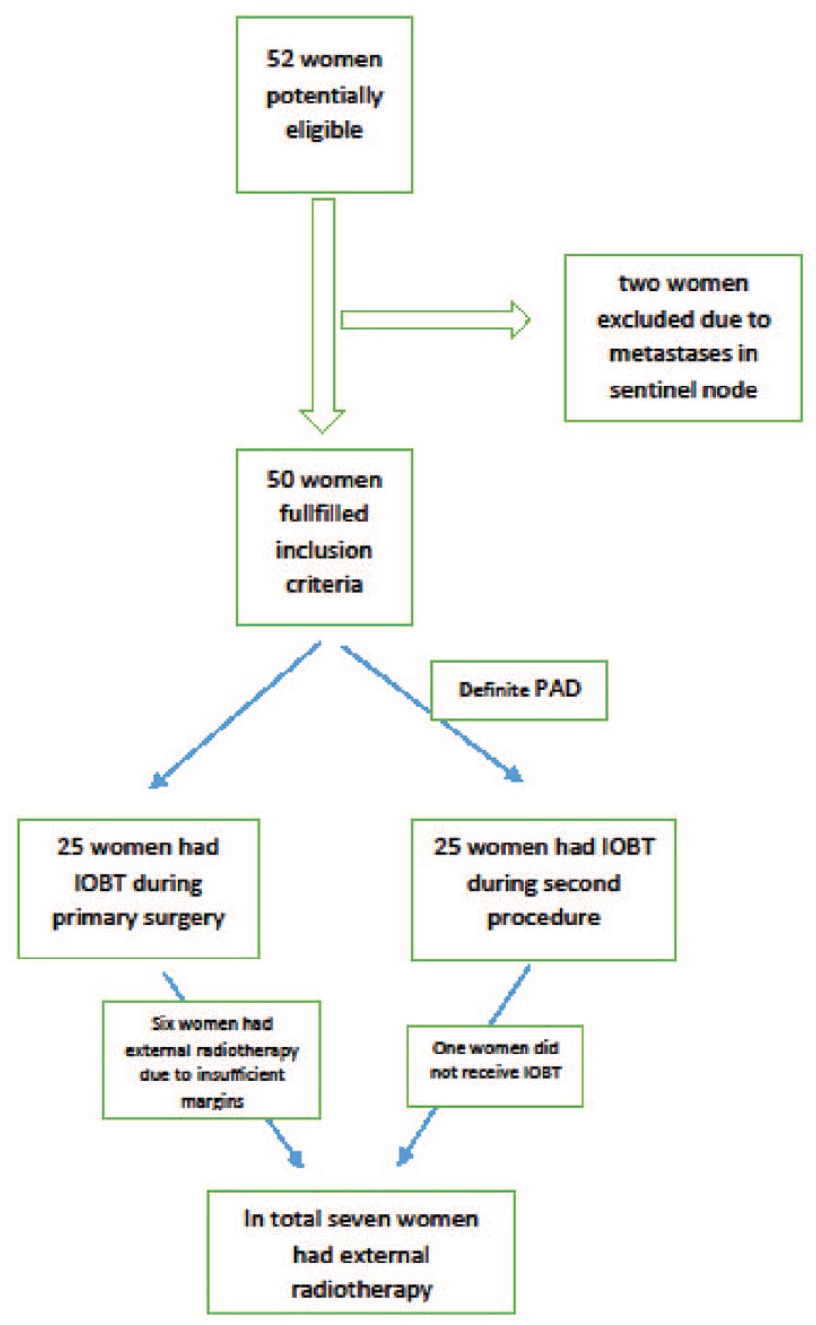
Flow chart.
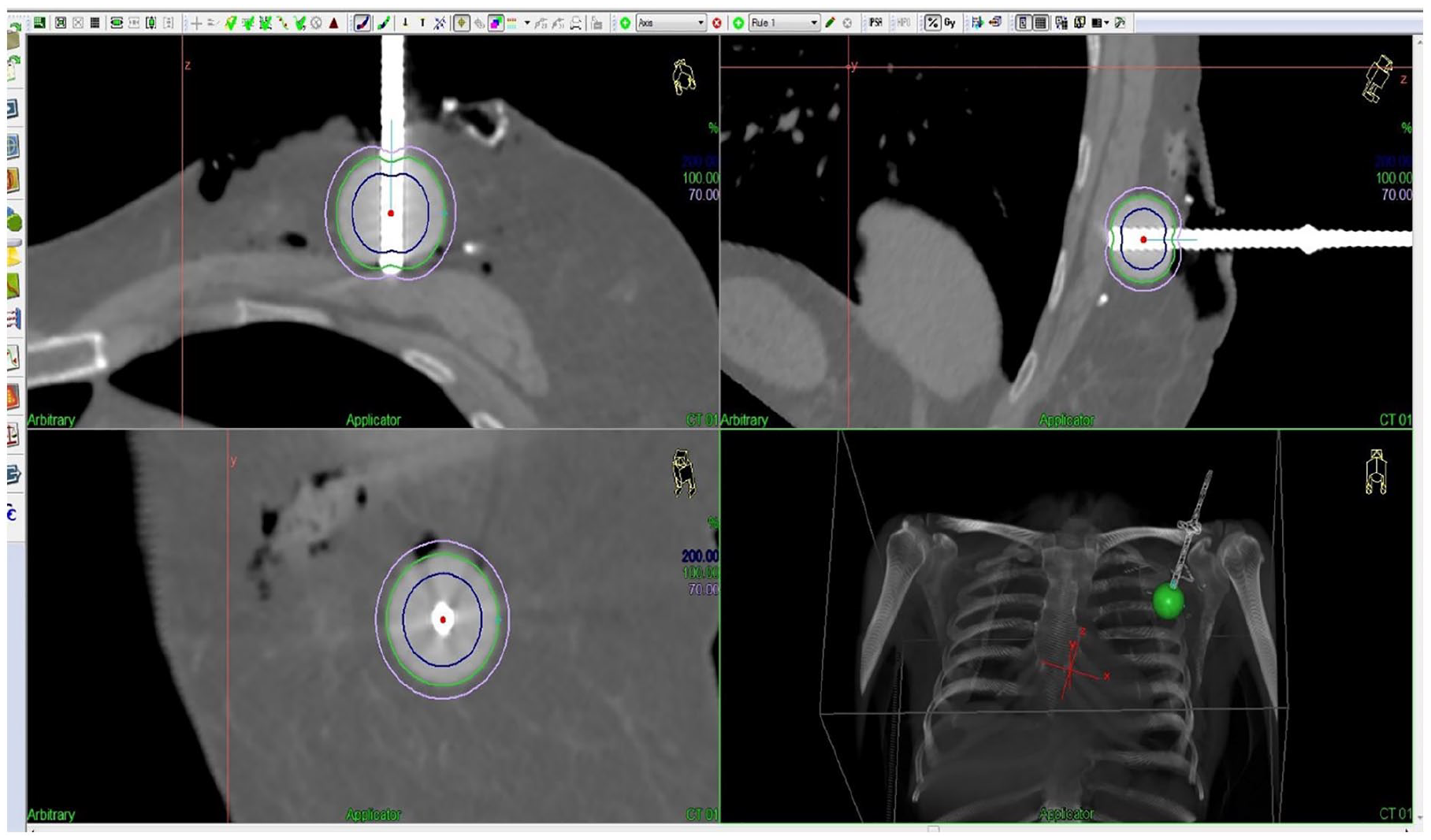
CT scanning and position of applicator.
Clinical Procedure
Twenty-five women were treated with IOBT during the primary surgery (pre-pathology group) and 25 women had IOBT during a secondary procedure, a few weeks after primary surgery when the full pathological report was known (post-pathology group). All IOBT procedures took place at the department of brachytherapy.
In the operating room, wide local excision of the primary tumor and a sentinel node biopsy (SNB) was carried out. Applicators of different sizes were tried out until the one that best fitted into the wound cavity was found. Two to four sutures were used to approximate the breast parenchyma to the applicator surface. A surgical gauze was inserted subcutaneously in order to protect the skin by creating a distance to the applicator. Local anesthesia with long duration was infiltrated around the surgical cavity. After bandaging, the patient was taken to the postoperative ward and shortly after that was transported fully awake to the department of brachytherapy. Before the start of brachytherapy, a CT scan of the thorax was performed to visualize the applicator’s adaption to the parenchyma in the surgical cavity. After the treatment, the applicator was removed, the breast parenchyma adapted, and the wound was closed. For the post-pathology group, the whole procedure took place at the department of brachytherapy. The wound was re-opened under local anesthesia, and the remaining procedure was the same as described above.
Follow-Up
The women were followed-up with a clinical control which included filling in health questionnaires (EORTC-QLQ-C30 17 and EQ-5D) (see the online supplemental material) and photographing of the breasts, at 2–4 weeks and 6 months postoperatively and then annually with the intention to have follow-up visits until 10 years postoperatively.
The study was approved by the Regional Ethical Review Board at Uppsala University, Dnr 2013/028.
Treatment Toxicity
Classification and grading of surgical and radiation side effects were documented according to the LENT (Late Effects of Normal Tissues)–SOMA (Subjective, Objective, Management and Analytical Evaluation of Injury) scale subjectively by two oncologists18,19. Symptoms were graded at a scale from 0 to 5, with the higher value the worse the outcome. Breast edema was defined as a swelling with an increased volume of the treated breast, either asymptomatic or symptomatic. Fibrosis was detected by palpation of the treated breast in comparison with the untreated side. The highest detectable grade of fibrosis in any quadrant of the breast was set as the final grade. Retraction and atrophy of the treated breast were defined as volume loss due to radiotherapy and surgery.
Quality of Life
Patient’s satisfaction and quality of life after treatment were assessed by two health questionnaires—EQ-5D-3L and the European Organization for Research and Treatment of Cancer (EORTC) score 30-item quality of life questionnaire (QLQ-C30).
EQ-5D-3L, three-level EuroQol Group’s five-dimension questionnaire, is a generic instrument for health outcome assessment 20 . It contains five dimensions: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Each dimension comprises three levels: no problems, some or moderate problems, and severe problems (see the online supplemental material). In addition, the patient is asked to indicate her health on a scale called the EQ VAS (visual analogue scale). A representative reference group for comparison was not possible to find.
EORTC-QLQ-C30 is an integrated system for assessing the health-related quality of life of cancer patients participating in clinical trials 17 . We used the latest version, version 3 21 . The EORTC-QLQ-C30 has been developed for several types of cancers, including a specific questionnaire for breast cancer (QLQ-BR23). Since this version includes several systemic therapy side effects such as hair loss, neurological symptom not applicable to our study group, we decided to use the general version consisting of 30 labels. This questionnaire evaluates five functional scales (physical, role, cognitive, emotional, and social), four symptom scales (fatigue, nausea, appetite, and pain), five single items (constipation, diarrhea, sleep, dyspnea, and financial), and a global health scale. The scoring of the EORTC QLQ-C30 was performed according to the EORTC scoring manual 22 . All scores were linearly transformed to a 0-to-100 scale. A high score for a functional scale represents a high/healthy level of functioning, a high score for the global health status/QoL represents a high quality of life, but a high score for a symptom scale/item represents a high level of symptomatology/problem.
Our results were compared to reference values for the EORTC QLQ-C30 in the Swedish population 23 .
Cosmetic Outcome
To evaluate the cosmetic outcome we used an objective assessment tool. Frontal digital photographs were taken at 2–4 weeks, at 6 months, and then annually after surgery. The photographs were analyzed by BCCT.core 2.0, a validated software which produces a composite score based on symmetry, color, and scar 24 . Forty-eight patients were assessed 1 year after surgery. The scores were categorized into Excellent (E), Good (G), Fair (F), or Poor (P).
Statistics
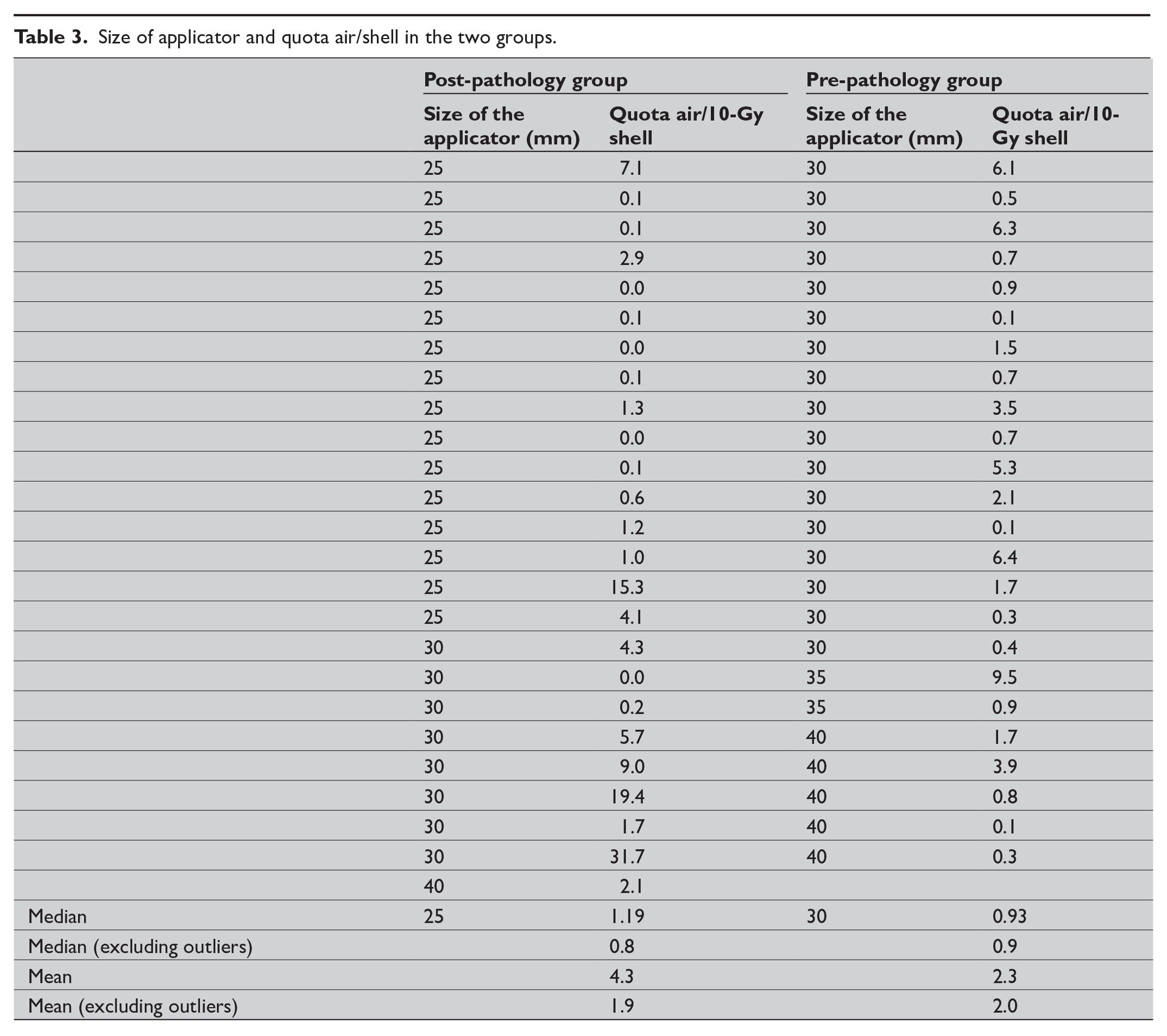
We estimated a pilot trial of 50 women to be sufficient to implement the new procedure and to evaluate potential flaws. Data from the QLQ-C30 health questionnaire were analyzed using Statistical Package for Social Science (SPSS), Version 22 (IBM, Armonk, NY). All the scores from the QLQ-C30 health questionnaire were linearly transformed into a 0–100 scale according to the manual. The data were continuous and presented in mean, range, and standard deviation. Differences in mean values for the women in the study at 1 year of follow-up were compared to the reference values of the Swedish population (see Table 3).
Size of applicator and quota air/shell in the two groups.
We used unpaired t-test to compare the continuous QLQ-C30 scores. The unpaired t-test analyses were performed with STATA release 14 (StataCorp, College station, TX). In order to evaluate the effect size, Cohen’s d was calculated for every difference in mean between the groups 25 . According to this concept, a low Cohen’s d indicates the necessity of larger sample sizes, and vice versa. “Low” are values <0.2, “moderate” are values around 0.5, and “high” are values >0.5.
Results
Feasibility
The clinical procedure worked out well logistically, both pre- and post-pathologically. Of the original 52 women, two were excluded due to metastases in the sentinel node, found per-operatively (Fig. 1). Two women who received IOBT in a second procedure felt uncomfortable while positioning the applicator. One woman reacted with hypotension and nausea after administration of local anesthesia. A few women needed an extra injection of local anesthesia when closing the wound. In the post-pathology group one woman had a CT scan where the applicator turned out to be separated from the breast parenchyma, due to a large wound cavity and difficulties to mobilize the tissue. This woman had conventional EBRT instead of IOBT.
Six women in the pre-pathology group received additional external radiotherapy due to the final histopathology report. In all six cases the in situ component presented with insufficient or indistinct margins.
Mean total surgical duration (time in operating room + IOBT time + time for wound-closure) for the pre-pathology procedure was 75 min and for the post-pathology procedure (time to re-open the wound and place the applicator + IOBT time + time for wound-closure) 38 min. Mean time in the operating room for the pre-pathology group was 62 min. Time for CT-scanning and dose planning were not included in the calculation.
The one-off cost for developing the applicators was €30,000. The cost for treating one woman with IOBT was €1950, which can be compared with a 3-week treatment of conventional external radiotherapy (15 sessions), €5330, or a 5-week treatment (25 sessions), €7640, which are the costs according to the Örebro University Hospital’s 2017 price list.
Treatment Toxicity
Few acute side effects were recorded at the initial follow-up visit. According to the LENT-SOMA scale, 11 women had no symptoms at all at 2–4 weeks and 37 women had mild (grade 1–2) side effects. Among the women who underwent complementary external radiotherapy, one was diagnosed with a radiotherapy-related breast edema 2–4 weeks after treatment. The condition was successfully treated with orally administered cortisone. Three women had a wound infection, which in two cases needed antibiotic treatment. One of these two women had her wound infection (treated with antibiotic) 3 months postoperatively.
The woman with poor adaption of breast parenchyma, who received external radiotherapy instead of IOBT, was missing in this first follow-up but not excluded. At the 6 and 12 months visit most of the grade 1–2 side effects had resolved and all the women had a satisfactory outcome (data not shown).
Quality of Life
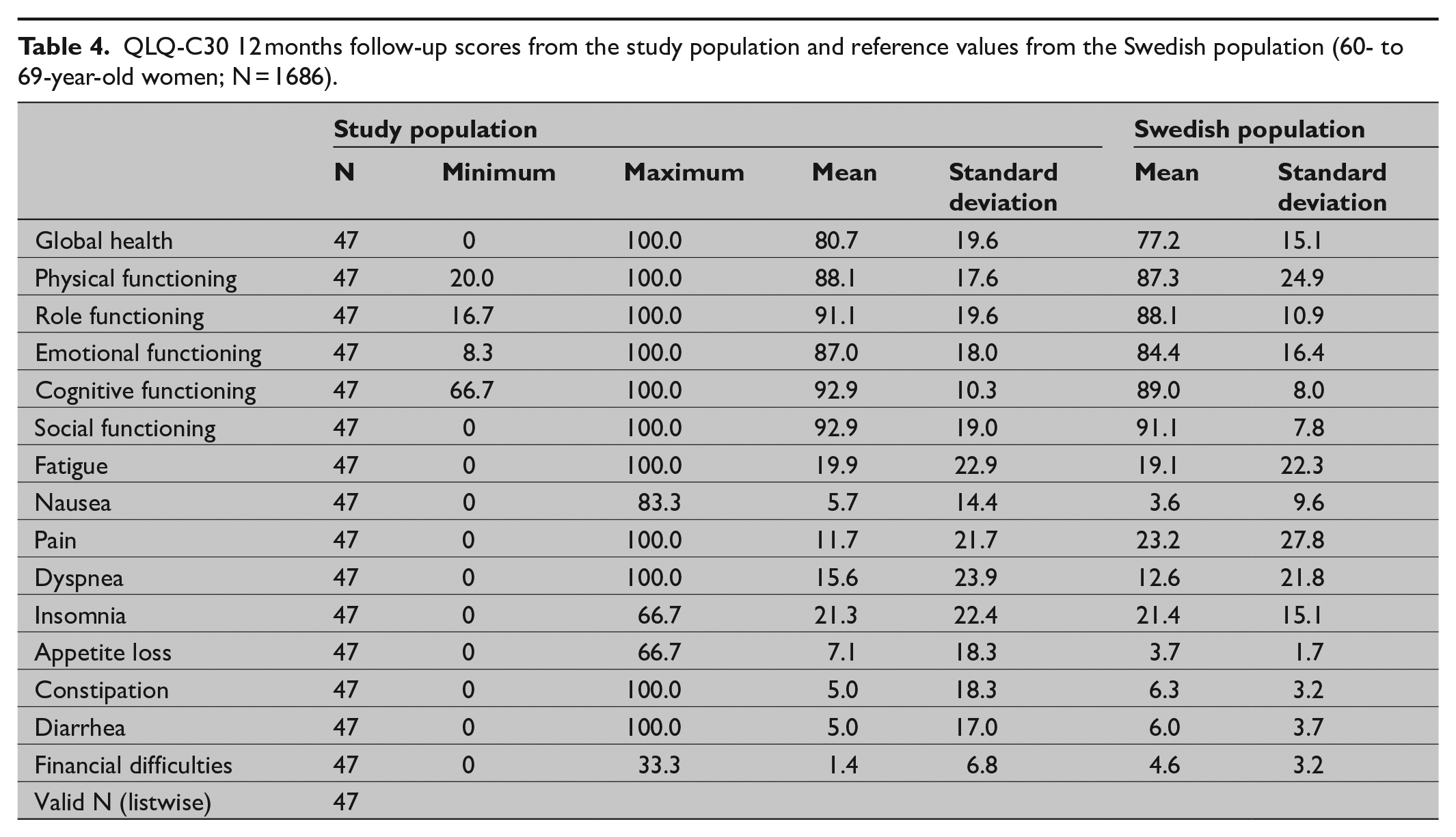
The 1-year results from the EORTC-QLQ-C30 health questionnaires are illustrated in Table 4. The numbers of missing items were low. At 1 year of follow-up, scores from three women were missing. For one additional woman three items from the scale were missing and for another woman two items. Four additional women missed one item each. Almost all missing items differed from each other; thus, biasing should not be a matter of concern. Overall, the women in the study reported a high score on the functional scales and quality of life and a low score on the symptomatic scales. The outcome was compared to a subgroup of women 60–69 years old in a random sample of adults from the Swedish population (Table 4) 23 . Concerning global health and functional scales the women in our study scored higher than the reference population, but the difference showed statistical significance only for “cognitive functioning” (data not shown). The study participants also reported a higher frequency of fatigue, insomnia, and appetite loss. The unpaired t-test showed statistically significant differences in the “appetite loss,” “pain,” and “financial difficulties” parameters. Cohen’s d was low for almost all mean differences which indicate that larger samples are needed. The EQ-5D analysis revealed overall low frequency of problems for the different dimensions, except for pain/discomfort and anxiety/depression were 14 and 15 women respectively reported moderate or severe problems (data not shown).
QLQ-C30 12 months follow-up scores from the study population and reference values from the Swedish population (60- to 69-year-old women; N = 1686).
Cosmetic Outcome
We decided to present the cosmetic results after 1 year when the wound and possible wound infections were healed. One woman was excluded due to incomplete photographing at the 1-year control. Another woman was diagnosed with subcutaneous metastases after 1 year and therefore was excluded. The woman with a large wound cavity described earlier did not receive IOBT due to poor adaption of the breast tissue to the applicator. She was still assessed in IOBT group according to the intention-to-treat concept and had a good cosmetic outcome according to the BCCT software. The pre-pathology group and the post-pathology group turned out to be evenly distributed among the five categories. The evaluation program reported “good” results in 14 women in the pre-pathology group and in 11 women in the post-pathology group. Only one woman in each group was registered as having “poor” cosmetic result.
Radiotherapy and Dose Planning
The median of the air volume proportion within the 10-Gy isodose volume were 0.9% and 1.2% for the pre- and post-pathology groups respectively. The median size of the applicator in the pre-pathology and post-pathology groups were 30 and 25 mm respectively (data not shown). The results from one woman could not be found, so the calculated number of study participants in the pre-pathology group was 24. The woman who received external RT instead of IOBT due to poor adaptation to the applicator had a proportion of 32% air within the 10-Gy tissue shell. She was still included in the calculation since she received the applicator. The median 10-Gy isodose volumes were 25 cm³ in the pre-pathology group and 15 cm³ in the post-pathology group.
Breast Cancer Recurrence
Median age of the study participants was 68.5 years and median tumor size was 10 mm (Table 1). Median size of applicator was 30 mm. The largest applicator was never used. Median duration time of IOBT was 12 min. After a median follow-up of 3.1 years, no study participant in the pre-pathology group had experienced a recurrence. One woman in the post-pathology group had an ipsilateral recurrence 1 year after primary treatment. The recurrence was located in a different quadrant than the primary tumor. She was treated with mastectomy. Another woman in this group had a contralateral cancer 3 years after IOBT. She was treated with BCS, sentinel biopsy, and IOBT for a second time. An additional woman in the post-pathology group received IOBT to the left breast after earlier had been treated for a cancer in her right breast. She was shortly afterward found to have a recurrence in her right breast with distant metastases.
Discussion
The trial shows that the procedure with BCS with pre- or post-pathology IOBT is feasible. No logistical problems were reported. No serious toxic side effects from IOBT were registered, and the grade 1–2 side effects had almost disappeared 6 months after treatment. Three postoperative infections were observed. With the reservation of low power, QoL did not differ significantly in this pilot group compared to reference groups from the Swedish population. The cosmetic outcomes were good in the vast majority of patients and evenly distributed between the pre- and post-pathology group.
There are different methods of delivering PBI, of which IOBT is one. The safety of partial-breast and reduced-dose radiotherapy is supported by a randomized, controlled, non-inferiority trial done in 30 radiotherapy centers in the United Kingdom and published recently in the Lancet 26 . Patients were randomly assigned to receive 40-Gy whole-breast radiotherapy (WBRT), 36-Gy WBRT and 40 Gy to the partial breast (reduced-dose group), or 40 Gy to the partial breast only (partial-breast group) in 15 daily treatment fractions. In terms of local recurrence non-inferiority of partial-breast and reduced-dose radiotherapy compared with the standard WBRT was found. Moreover, the GEC-ESTRO presented data from a non-inferiority, randomized trial. After a median follow-up of 6.6 years, the preset difference margin of 3% was not reached 27 .
In the TARGIT-A trial 12 , 3451 patients were enrolled at 33 centers in 11 countries and randomized to IORT with Intrabeam® or external WBRT. The 5-year risk for local recurrence overall was 3.3% (95% confidence interval (CI): 2.1–5.1) for IORT versus 1.3% (95% CI: 0.7–2.5) for external WBRT (p = 0.042).
The ELIOT trial randomized 1305 women between IOBT and WBRT 28 . In this trial ipsilateral breast tumor recurrence (IBTR) after a median follow-up of 5.8 years was 0.4% in the group receiving external radiotherapy and 4.4% in the ELIOT group. The non-inferiority margin of 4.5% was not exceeded, but still the numerical difference was 4% which prompted the authors to recommend an improved selection of patients to the ELIOT-method. Inclusion criteria in the ELIOT trial were invasive breast cancer tumor ⩽2.5 cm with no restrictions regarding axillary nodal status. Lobular tumors were also eligible.
The techniques in these two trials are fundamentally different. Whereas Intrabeam® delivers irradiation from within the undisturbed tumor bed, in the ELIOT trial, the mammary gland is mobilized, a pre-pectoral lead shield is inserted, the edges of the tumor bed are joined, and radiation is delivered. Intrabeam® uses 50-kV x-rays delivering 20 Gy to the tumor bed surface and 5–7 Gy at 1-cm depth, in 20–45 min. ELIOT uses electrons at 4–12 MeV delivering 21 Gy in 3–5 min. The TARGIT-A trial has been criticized for its short follow-up—median time 29 months, only 611 (18%) patients had a 5-year follow-up—and for misinterpretation of the non-inferiority criterion, which requires the upper CI to be less than the pre-defined non-inferiority level of 2.5% (difference between IORT group and EBRT group 1.0% unit; 95% CI: –0.68 to 2.68, pre-pathology group) 29 . Breast cancer mortality was much the same for IORT and EBRT, but significantly fewer non-breast-cancer deaths occurred in the IORT group than in the EBRT group. The authors’ explanation is fewer deaths from cardiovascular causes and other cancers in the IORT group.
The primary aim of this pilot study was to investigate feasibility and safety of a concept that was new for our breast surgical and radiotherapy team.
The Iridium 192 HDR source emitting photons with a mean energy of 374 keV giving a deeper tissue penetration around the applicator although treatment has to be given in a shielded room. This differs from Intrabeam®, which uses 50-kV photons with lower penetration but with the possibility of delivering the treatment in an unshielded operating room. Both methods have the ability to deliver high doses to the tumor bed while reducing doses to nearby critical structures which makes them suitable for the purpose of PBI. Many centers already own an HDR equipment, which should reduce the cost of initiation of the procedure. However, a shielded (operating) room is mandatory. In our trial all IOBT treatments took place in a shielded room at the department of oncology. In this way, the duration time in the operating room for the pre-pathology group was prolonged for only 2 min which made room free for the next surgical procedure without delay. The TARGIT trial reports a prolongation of surgical procedure duration time of 30 min 12 .
The TARGIT-A trial found a larger absolute risk of recurrence in the post-pathology group. The authors’ explanation is that the fresh tissue is compromised after several weeks of healing process and consequently that the treatment is not as effective as in the pre-pathology group. In our trial we objectively investigated the contact between the applicator and the target tissue through a CT scan and found a poorer adaptation between the applicator and the target tissue in the post-pathology group. To the best of our knowledge, no previous study has carried out a CT scan before start of IOBT. During the post-pathology procedure, we experienced that the breast tissue was compromised with a more solid consistency as in the TARGIT-A trial, which, together with our CT-based dose planning results, points toward performing IOBT only during primary surgery. However, waiting for definitive histopathological report could possibly limit the usefulness of the pre-pathological procedures since some patient will need the addition of complementary external XRT. In our trial there were six women (24%) in the pre-pathology group who underwent external XRT after the final histopathological report. In the TARGIT-A trial the corresponding percentage was 15%.
The frequency of additional external XRT after IORT depends on selection criteria 30 , and the inclusion criteria differs between different radiotherapy societies 31 . Exclusion criteria in our trial were lobular cancer and metastases in the axilla. In this manner, we wanted to reduce the need for additional external XRT. However, we did not succeed in our preoperative selection of patients to prevent this to happen for every fourth woman. No serious adverse toxic effects were registered which is in line with previously reported results from IORT trials32,33.
In the TARGIT-A trial, IORT was found to significantly improve quality of life 34 . In our trial evaluation of the two health questionnaires should be interpreted with caution, due to potentially low power. A few items in the QLQ-C30 health questionnaire showed statistically significant differences compared to the reference Swedish population. Results from the EQ-5D questionnaire were not compared to any reference population, but a relatively high proportion of study participants reported moderate or severe anxiety/depression and/or pain/discomfort. A drawback of this study is the lack of known state of health before treatment and cancer diagnosis. It is also fair to believe the results to be affected more by the diagnosis of cancer rather than by the IOBT procedure. Thus, giving a health questionnaire after the cancer diagnosis could also be misleading.
Since our trial was designed as a pilot study we did not include a control group. Instead, we compared the BCCT software results from the pre-pathology group with the results from the post-pathology group and found no differences. On the contrary, in the TARGIT-A trial, the cosmetic outcome for those treated with Intrabeam® was found to be superior to those patients who received conventional EBRT 35 .
One of the strengths with our trial is the CT scan images, which will possibly facilitate the identification and documentation of where the dose is delivered with respect to the excision cavity as well as the organs at risk including the skin and chest wall. It also helps the surgeon to improve the adaptation of the breast tissue around the applicator. This will increase the quality of treatment or, in cases where re-treatment is needed, to identify previously delivered dose to organs at risk.
We also believe that our careful selection of patients and tumor characteristics are necessary when offering IOBT. The TARGIT-A trial had relatively wide inclusion criteria. We included tumors with smaller sizes and excluded lobular cancer and tumors presented with metastases in the axilla. Further trials are needed to find the best suitable group of patients for this treatment.
The National Institute for Health and Care Excellence (NICE) in England concludes in their latest report that there are some patients who could particularly benefit from Intrabeam®, but the patients should be fully informed of the evidence and treatment options available. Moreover, they conclude that Intrabeam® can only be recommended if its use is accompanied by the gathering of additional information on clinical effectiveness by data collection 36 .
In conclusion, IOBT represents a promising alternative of postoperative radiotherapy for selected patients. In the absence of reliable data and longer follow-up, it should remain as a technique under investigation. Our pilot trial urges for further larger trials using this concept, which already has started at our center.
Supplemental Material
sj-pdf-1-sjs-10.1177_1457496920903975 – Supplemental material for Intraoperative high dose rate brachytherapy during breast-conserving surgery: A prospective pilot study
Supplemental material, sj-pdf-1-sjs-10.1177_1457496920903975 for Intraoperative high dose rate brachytherapy during breast-conserving surgery: A prospective pilot study by Åsa Wickberg, Göran Liljegren, Johan Ahlgren, Leif Karlsson, Anders With and Bengt Johansson in Scandinavian Journal of Surgery
Footnotes
Acknowledgements
All listed co-authors were integrally involved in the formation of this manuscript via study conception/design and/or data acquisition and analysis/interpretation. Furthermore, all authors made significant contributions to the drafting or critical revisions of the manuscript, and all authors gave final approval prior to submission for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study design was approved by the Ethics Committee at Uppsala University, Sweden (Dnr 2013/028).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was supported by grants from the Local Research Committee and the Key Foundation of Örebro University Hospital, Sweden.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
