Abstract
Background and Aims:
Pancreatic neuroendocrine tumors (PNET) arise from uncontrolled proliferation of neuroendocrine cell and further genetic alterations that may induce hormone secretion such as glucagon/insulin/gastrin/VIP. Their incidence is rapidelly growing, especially because of the frequent incidental diagnosis of small asymptomatic non-functionnal neuroendocrine tumors with the widespread use of cross-sectional imaging. The vast majority of pancreatic neuroendocrine tumors are sporadic but up to 5%–10% of them arise from genetic syndromes, the main one being Multiple Endocrine Neopalsm type 1 (MEN1). Appropriate management of patients with PNET is a complex challenge for surgeons, and require extensive medical collaboration. This review aims to summarize major and recent updates regarding the medico-surgical management of PNETs.
Material and Methods:
Review of pertinent English language literature.
Results:
This article provides a concise summary of the clinical presentation, diagnosis, surgical management, alternative treatments and follow up of PNETs.
Conclusion:
PNET are a rare, heterogeneous group of neoplasms with a generally favorable prognosis at least compared to pancreatic adenocarcinoma. Surgical resection is the cornerstone of their management, particularly for localized disease, and should always be discussed in multidisciplinary tumor board.
Keywords
Introduction
Appropriate management of patients with pancreatic neuroendocrine tumors (PNETs) is a complex challenge for surgeons. PNETs incidence has significantly increased in the past decades, in part related to the widespread use of cross-sectional imaging and is currently representing approximately 3%–5% of all diagnosed pancreatic tumors (1). The very high heterogeneity of PNETs makes standardization of therapeutic strategies and optimal management difficult. Surgical options have evolved ranging from extended oncological resection to parenchyma sparing resection and even now a “wait-and-see” policy. This review aims to summarize the major update regarding the medico-surgical management of PNETs.
Epidemiology and Physiopathology
PNETs arise from uncontrolled proliferation of neuroendocrine cell and further genetic alterations that may induce hormones secretion such as glucagon/insulin/gastrin/VIP. Epidemiologic studies have shown a rising annual incidence of 0.8 per 100.000 persons for PNETs, at least in part related to the increased detection of “incidentalomas” (2). Some risk factors of PNETs occurrence have been identified such as family history of cancer, diabetes, tobacco and alcohol consumption, but they are associated with a very low relative risk (3).
The vast majority of PNETs is sporadic but up to 5%–10% of them arise from four genetic syndromes: multiple endocrine neoplasia type 1 (MEN-1); Von Hippel–Lindau syndrome (VHL); neurofibromatosis type I (NF1), and more rarely tuberous sclerosis complex (TSC). MEN-1 is the most common genetic syndrome associated with PNETs. About 70%–80% of patients with MEN-I will develop PNETs (4, 5). Non-functional (NF)-PNETs account for 60%–85% of PNETs (6).
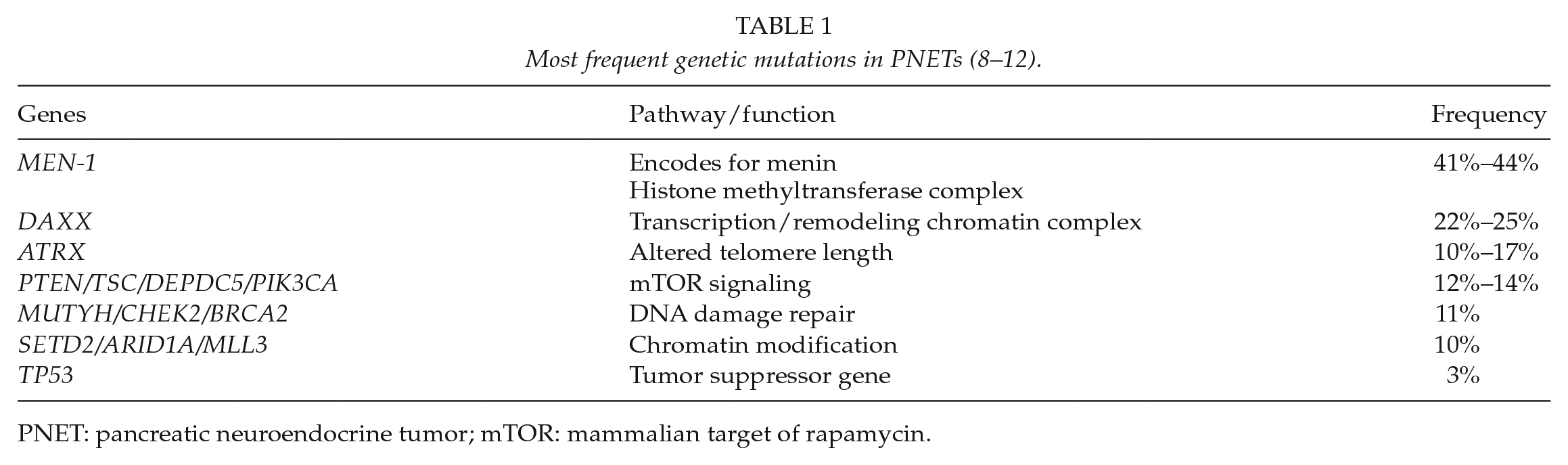
PNETs include a wide range of neoplasms from indolent well-differentiated tumor to poorly differentiated carcinomas underlying biological and molecular heterogeneity (7). High-throughput analyses have improved our understanding of the disease and pathogenesis of PNETs. Recent whole-exome sequencing of PNETs have reported the most common genes altered in sporadic PNETs implicated in chromatin remodeling (8) (Table 1). Somatic mutations of MEN-1 encoding for the menin were found in 44% of the patients highlighting its major implication in pathogenesis not only in the setting of MEN syndrome but also in sporadic PNETs. Genetic alterations in the transcription/chromatin complex: death-domain-associated protein (DAXX)/alpha-thalassemia retardation syndrome X-linked (ATRX) that promotes activation of the alternative lengthening of telomeres (ALT) was identified in 45% of sporadic PNETs. Loss of DAXX/ATRX protein seems to be related to chromosome instability, tumor size, Ki-67, and metastasis disease (9) and defines a subset of patients with more aggressive tumors and poor oncological outcomes (10). Actionable mutation in phosphatidylinositol-3-kinase (PIK3)/Akt/mammalian target of rapamycin (mTOR) pathways genes (14%) (8, 11) has been described and may predict response to therapies targeting this pathway, with encouraging results of Everolimus (mTOR inhibitor) in PNETs (7, 12, 13). Finally, studies have shown that loss of Rb expression and KRAS-mutation were commonly detected in neuroendocrine carcinomas (NECs) and could predict response to platinum-based chemotherapy (14).
PNET: pancreatic neuroendocrine tumor; mTOR: mammalian target of rapamycin.
Diagnostic
Clinical and biological diagnostic
Diagnosis of PNETs is usually delayed as most of them are small and indolent neoplasms. PNETs account for up to 50% of incidentalomas in some surgical series (15). Approximately half of PNETs are diagnosed with metastatic disease (16).
NF-PNETs
The vast majority of PNETs (80%–90%) are NF. Usually, unspecific symptoms such as abdominal pains or bowel disorder lead to morphological exploration and “incidental diagnosis.” Depending on the location of the neoplasms, they may also be revealed by jaundice (17%–50%), acute pancreatitis, anorexia or nausea (45%), weight loss (20%–35%), bowel obstruction or less frequent symptoms such as intra-abdominal hemorrhage or palpable mass related to locally advanced tumors (17).
Functional PNETs
Clinical syndrome of F-PNETs is related to specific hormone hypersecretion. The most frequent secreted peptides are in decreasing order insulin, glucagon, gastrin, VIP, and somatostatin. Insulinomas account for 30%–40% of F-PNETs and are mainly sporadic neoplasms. Gastrinomas are the most frequent F-PNETs in MEN-1 syndrome (54%) (18) (Table 2).
F-PNET: functioning pancreatic neuroendocrine tumor.
Plasma chromogranin A (CgA) is widely used as a circulating marker for PNETs. About 80% of PNETs have an increased CgA level. It is correlated to the tumor burden and liver metastasis, especially in well-differentiated tumor (24). Many conditions unrelated to PNETs may induce CgA increased, including all causes of hypergastrinemia (Helicobacter pylori infection, Biermer’s disease, atrophic gastritis, drugs, etc.) as summarized in Table 3. High level of CgA should be considered only with a normal level of gastrin in plasma. It might also be useful as a biological marker of relapse and response to treatment and has a prognostic value. Neuron-specific enolase has a poor sensibility (40%) and specificity (70%) for the diagnosis of PNETs (25) and has limited interest. Similarly, pancreatic polypeptide (PP) is elevated in less than 50% of the patients with PNETs and is not considered as a specific biomarker. Specific hormones assay (insulin, gastrin, glucagon, VIP, etc.) should not be systematically performed but only according to the clinical symptoms. Other biomarkers based on gene transcripts are under investigation such as the NETest, encompassing the detection of 51 circulating transcripts, but are not currently used or recommended for clinical practice (26).
Non-neuroendocrine conditions with elevated chromogranin A.
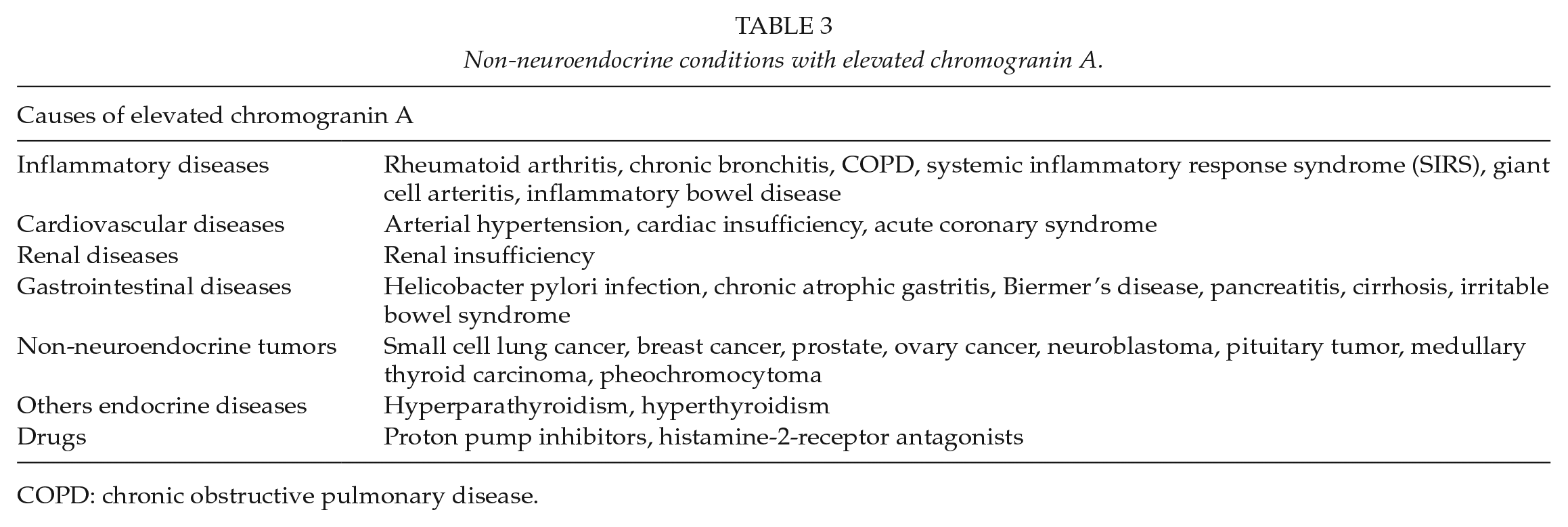
COPD: chronic obstructive pulmonary disease.
Pathological diagnostic and classifications
The pathological diagnosis of PNETs is confirmed when at least two markers among CgA, Synaptophysin or CD56 is expressed in immunohistochemistry. The histoprognostic classification of PNETs is a proliferative activity-based system defined by the Ki67 index and the mitotic count (number of mitoses per 10 high-power fields).
The revised 2017 World Health Organization (WHO) classification introduced the following modifications (Table 4):
It raised the cut-off Ki-67 for G2 tumors to 3%.
The Grade-3 PNETs were divided into two subsets of tumors according to the degree of differentiation: well-differentiated neuroendocrine tumor of high-grade NET-G3 and poorly differentiated NEC NEC-G3. NET-G3 represents up to 20% of G3 tumors with a Ki-67 index ranging from 20% to 50% but should be managed as G2 tumor.
The term mixed adenoneuroendocrine carcinoma (MANEC) was substituted by mixed neuroendocrine–nonneuroendocrine neoplasm (MiNEM).
2017 WHO histoprognostic classification of pancreatic neuroendocrine neoplasms.
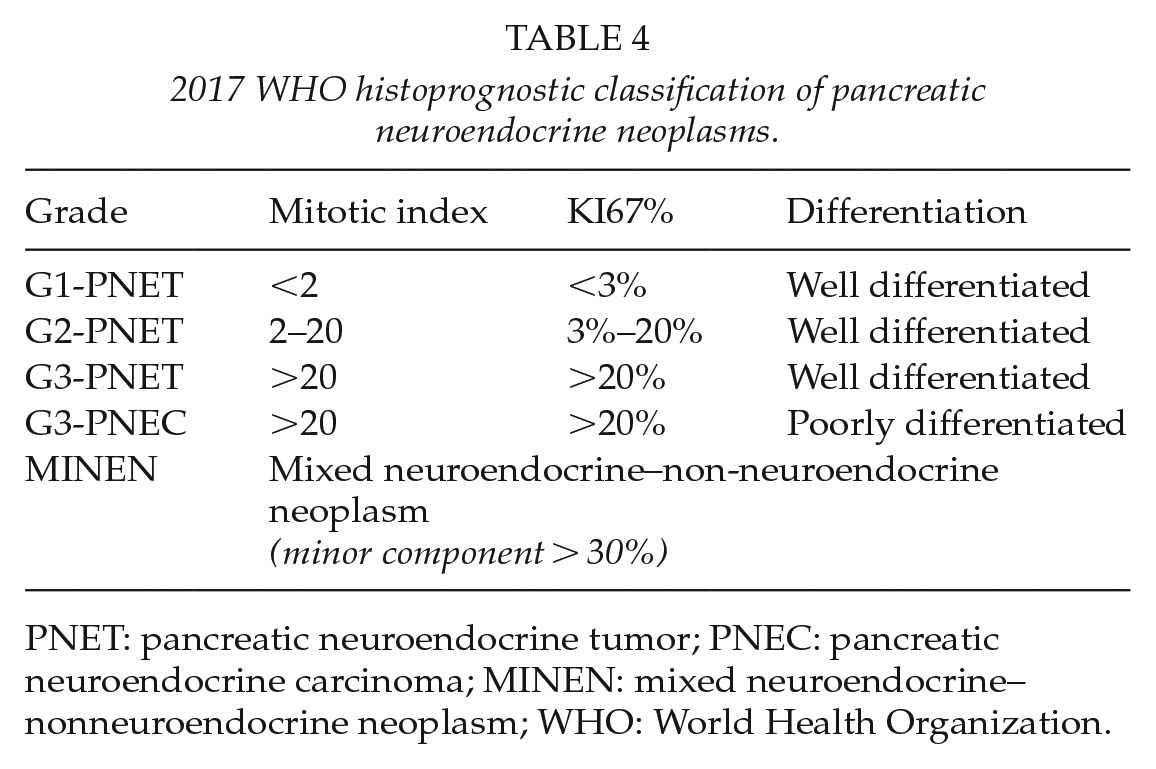
PNET: pancreatic neuroendocrine tumor; PNEC: pancreatic neuroendocrine carcinoma; MINEN: mixed neuroendocrine–nonneuroendocrine neoplasm; WHO: World Health Organization.
Classification of PNETs according to the tumor–node–metastasis (TNM) staging is also recommended to define prognostic subset of tumor. The 8th AJCC edition (2017) introduced new criteria and modifications on T-stage and M-status, asserted by the European Neuroendocrine Tumor Society (ENTS; Table 5). It is important to note that poorly differentiated NECs are not covered by this staging system at this location; the pancreas exocrine staging system should be used instead. Studies have shown that this new classification is more accurate than the previous 7th AJCC edition to define the prognosis of patients with PNETs. Due to their rarity and challenging diagnosis, cases without evident diagnosis should be referred to an expert pathologist.
AJCC 8th TNM staging classification.
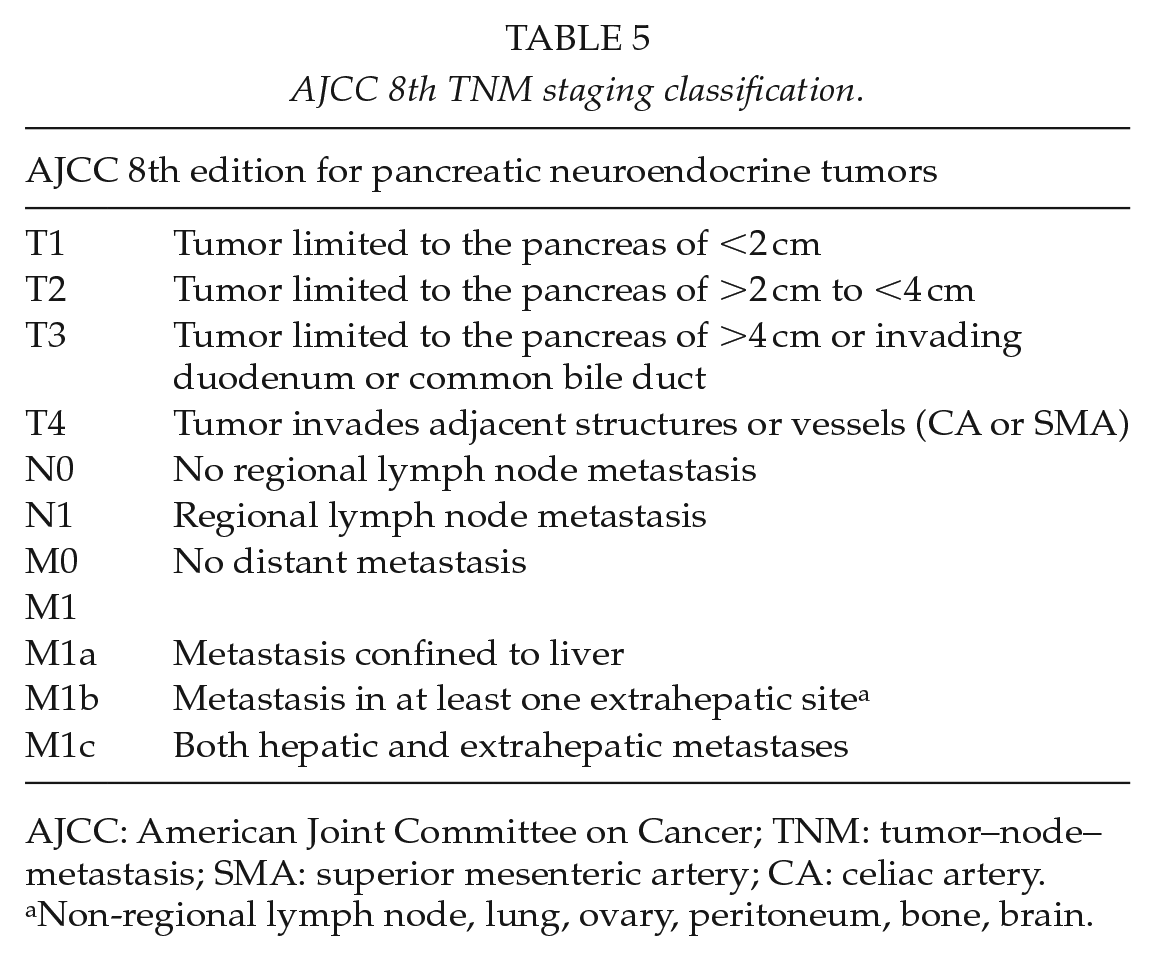
AJCC: American Joint Committee on Cancer; TNM: tumor–node–metastasis; SMA: superior mesenteric artery; CA: celiac artery.
Non-regional lymph node, lung, ovary, peritoneum, bone, brain.
Morphological diagnostic
Standard morphological examinations
Multiple detector computed tomography (MDCT) scan is the first-line imaging modality for the detection of PNETs. The imaging protocol for PNETs includes a delayed arterial (30 s) and portal venous (60–90 s) phases to increase the rate of detection of such tumors. PNETs are usually hypervascular tumors that tend to be easier identified from the surrounding pancreatic parenchyma in the delayed arterial phase (30 s) enhancement. A “washout” is observed in the portal venous phase. F-NETs are usually detected in an earlier stage with small size and a lower contrast from the pancreas. Calcifications can be depicted in the unenhanced phase (5%). Cystic component is commonly described in PNETs associated with MEN-1 (10%). The sensitivity and specificity of MDCT are 82% and 96%, respectively (27). MDCT also allows detection of local extension, vascular involvement, and distant metastases. Some studies have shown that liver metastasis of PNETs has a specific vascular pattern (hyper/iso-attenuation in portal phase) (28). However, magnetic resonance imaging (MRI) with gadolinium-enhanced sequences and diffusion-weighted sequences is the more accurate modality for the detection of liver metastases as well as small tumors (29).
Endoscopic ultrasonography (EUS) with contrast-enhanced has a higher sensitivity to detect small tumors (<20 mm) and regional lymph node (LN) metastasis (30). It may be of interest for vascular invasion and to assess the distance from the main pancreatic duct when enucleation is considered and allows biopsy in case of uncertain diagnosis.
Nuclear medicine imaging
Functional imaging used the expression of somatostatin receptor (SR) that can be uptake by radiometabolic somatostatin analog (SA). The expression of SR has been reported in about 70% of PNETs, especially in well-differentiated tumors. This functional imaging enables to evaluate tumor stage, distant metastasis and assess the expression of SR to select patients for peptide receptor radiometabolic treatment (PRRT).
The 111In-pentetreotide somatostatin receptor scintigraphy (SRS) also called Octreoscan has a sensibility and specificity of 90% and 80%, respectively, for the diagnosis of well-differentiated PNETs larger than 1 cm. However, SRS has several convenient such as a poor spatial resolution and a limited interest in NF-small NET of <1 cm. Thus, studies have reported that MRI with gadolinium-enhanced sequences and diffusion-weight sequences was more accurate than SRS for distant metastasis, especially for small liver metastasis (31).
Positron emission tomography (PET)/CT associated with 68Gallium-labeled somatostatin analog (68GA-SA-PET/CT) has shown improved sensitivity, up to 97% for the detection of gastro-pancreatic NETs. 68GA-SA-PET/CT has also demonstrated higher sensitivity than Octreoscan to detect LN metastases and distant metastases (liver, peritoneum, and bone) (32). Three SAs are currently available for 68Gallium-PET/TDM: 68GA-DOTATOC; 68GA-DOTATOTE, 68GA-DOTANOC. These three tracers have a higher affinity for SR than Octreoscan but no difference has been reported among them for the detection of PNETs (27). Fluorodopa (F-DOPA) PET/CT has shown a more limited interest for PNETs due to the physiologic uptake of the pancreatic gland (33) and is in clinical practice more used for the management of small bowel NETs.
For poorly differentiated NETs with low expression of SR, NEC or well-differentiated PNETs with Ki67 of >10%, fluorodeoxyglucose (FDG)-PET/TDM is more suitable as studies have reported its better sensitivity in this setting (34). It can also be used as a prognostic tool.
Surgical Treatment
Surgical management of PNETs has substantially evolved in the last decades. A better understanding of the natural history and biological behavior of these tumors currently supports conservative’s strategies for selected patients. Radical oncological surgery is not mandatory for all PNETs anymore and may be avoided for indolent, small tumors. Because of the complexity of the disease, and the necessity of a multidisciplinary management all patients with PNETs should be discussed within a dedicated tumor board to define the best suitable strategy for each tumor.
Which surgery?
Standard pancreatectomy versus parenchymal sparing surgery
When surgery is considered for PNETs, two surgical strategies can be discussed: standard pancreatic resection with standard lymphadenectomy versus parenchyma sparing surgery (PSS) possibly in addition to an LN picking.
For standard pancreatic resection, that is, distal pancreatectomy and pancreaticoduodenectomy, they should be performed for PNETs at risk of nodal involvement. Regional lymphadenectomy is mandatory as we know that node involvement is a strong prognostic factor of survival (35). For left-sided tumors, depending on the location of the tumor from the splenic vessels and the hilum of the spleen, spleen preserving-distal pancreatectomy can be considered for small presumably benign PNETs (36).
PSS including enucleation and central pancreatectomy has been investigated as an alternative to standard resection for small- and low-grade neoplasms (37, 38). When limited resection is considered, LN picking can be performed to assess node invasion. Enucleation may be performed for a small low-grade or benign PNETs located further than 2–3 mm from the main pancreatic duct. This should be preoperatively assessed by echoendoscopy, MRI, and/or intraoperatively by echography. Central pancreatectomy, for low-grade tumors located in the pancreatic neck and the first part of the body, is rarely performed. It is associated with a very significant risk of pancreatic fistula, because of two pancreatic stumps, and consequently, significant postoperative morbidity and non-null mortality. Consequently, this procedure, for supposed non-malignant lesions, should be performed with caution in selected patients, young enough to benefit in the long term of the excellent preservation of pancreatic endocrine and exocrine function, and fit enough to sustain the significant postoperative morbidity. Regarding postoperative pancreatic function, PSSs are only associated with 5% of postoperative exocrine and endocrine insufficiency, and excellent overall and recurrence-free 5-year survival of >95% in selected PNETs.
Laparoscopy versus laparotomy
Open approach for pancreatectomy remains the standard of care, and to date, there is no clear consensus regarding indications for a minimally invasive approach in pancreatic surgery.
Regarding pancreaticoduodenectomy, laparoscopic approach did not show any advantage over open pancreaticoduodenectomy (39). In addition, a recent Dutch multicentric randomized controlled study comparing laparoscopic and open pancreaticoduodenectomy has been prematurely stopped because of increased mortality in the laparoscopic arm (40). This is different for pancreatectomy without reconstruction, that is, distal pancreatectomy. Several studies and meta-analysis have reported that laparoscopic distal pancreatectomy was a safe and effective procedure with reduced complication rate and shorter hospital length stay (39, 40). However, very few of these studies specifically deal with PNETs.
In the area of robotic surgery, few teams have reported their preliminary experience of robot-assisted pancreatectomy (78, 79) with encouraging results. Further studies are now required to better assess the role of the robotic approach in pancreatic surgery. This is likely to be very little compared to the classical laparoscopic approach for distal pancreatectomy but might improve the postoperative outcome of pancreaticoduodenectomy.
For which tumors?
The likely benefit of surgery should be balanced with the postoperative morbidity, mortality, and impaired functional results of pancreatectomy (Fig. 1).
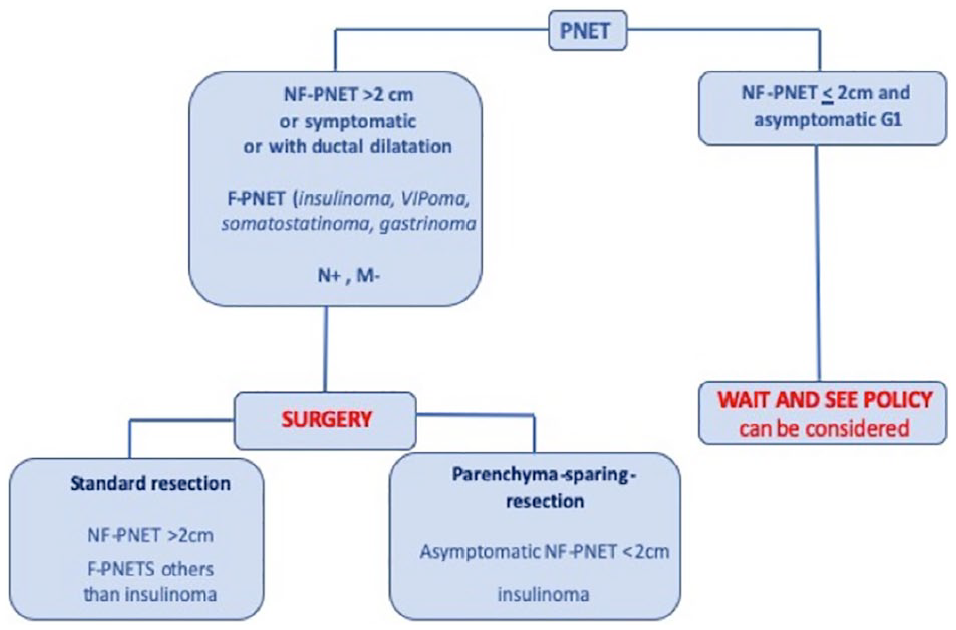
Simplified algorithm of PNET management.
Some PNETs must be operated without controversial matters, that is, symptomatic NF-PNETs, NF-PNETs greater than 2 or 3 cm, NF-PNETs with atypical imaging features especially main pancreatic duct dilatation and of course all functional PNETs including insulinomas, sporadic gastrinomas, VIPomas, somatostatinomas, and so on.
A better understanding of the natural history of small sporadic NF-PNETs recently led to reconsider surgical indications (17, 42, 43). Non-surgical management can be considered for asymptomatic sporadic NF-PNETs of <2 cm, so-called “incidentalomas,” MEN-1 NF-PNETs or MEN-1 gastrinomas of <2 cm (44, 45). However, this surveillance strategy can be an alternative to surgery only with an active serial imaging follow-up. This also requires a well-established PNETs diagnosis on somatostatin–receptor imaging and/or ideally with EUS-fine-needle aspiration (FNA). It is important to note that most of the series that have reported this non-surgical strategy are retrospectives studies. The major convenient of this strategy is to undertreat about 10% of patients, with a possible nodal metastatic involvement (46). The rate of secondary resection ranges from 10% to 25% in the literature and development of metastases occurs in less than 10% of the patients (18, 44). On the contrary, most of the patients with G3 poorly differentiated NECs, extra-liver metastasis or unresectable liver metastasis, with not possible of complete resection, will have no benefit from surgery.
For which results?
Oncological outcomes after surgery for PNETs are widely heterogeneous depending on various factors such as tumors’ grade, size, and stages. Recent meta-analyses reported a 5-year disease-specific survival greater than 90% in resected patients without synchronous liver metastasis (41). Data from the Surveillance, Epidemiology, and End Results (SEER) have reported median overall survival ranging from 12 years for G1-PNETs to less than a year for G3 tumors (1). If overall survival is most of the time excellent, tumor recurrence is a frequent event. After R0-resection, cumulative incidence of recurrence can reach 27% within 3 years and 40% within 5 years (47). Recently, Zaidi et al. (48) have validated a recurrence risk score (RRS) to guide tailored surveillance strategies. This score has included the following four well-known independent prognostic factors: symptomatic tumor, size of >2 cm, Ki67% of <3%/3%–20%/>20%, positive LN. Patients were scored on a scale from 0 to 10 points. This recurrence risk score (RRS) seemed stratified three groups of patients with a low (0–2), moderate (3–5), and high (6–10) rate of recurrence with a 2-year relapse of 2%, 14%, and 33%, respectively.
Regarding postoperative pancreatic function, after standard resection, exocrine and endocrine pancreatic insufficiency range from 9% to 30% and 5% to 25%, respectively. It decreases below 5% in the case of atypical sparing pancreatic resection (49). These functional results are nevertheless balanced by an increased rate of postoperative morbidity, especially pancreatic fistula with a rate of 45% after enucleation in prospective studies (41).
Alternative Treatment of PNETs
Radical resection of PNETs has been advocated as the only curative option but is associated with significant morbidity. Thus, the risk–benefit balance of the surgery has to be carefully weighed. In this setting, local ablation of tumors, as with radiofrequency ablation (RFA), might be considered. RFA is a thermal technique using a high-frequency alternating current through a needle electrode that is inserted in the core of the tumor. It produces necrosis and possibly systemic antitumor immune response and tumor regression (50, 51). RFA can be delivered by diverse approaches depending on the tumor location: percutaneously, intraoperatively, and by endoscopy ultrasound (EUS) approaches. Oleinikov et al. (52) reported that the majority of patients (85.7%) with insulinoma would prefer RFA of their tumor instead of surgery.
The success of this technique for liver tumors encouraged its use for pancreatic neoplasms. But in contrast to the liver parenchyma, thermal injuries may have more serious consequence in the pancreas, which is surrounded by close critical anatomic structures such as the duodenum, stomach, transverse colon, common bile duct, portal vein, and superior mesenteric vessels (53). Date et al. (53) suggested that a temperature of about 90 °C is sufficient for a successful procedure. For pancreatic tumors, a feasibility study was performed in pig models and confirmed the feasibility and safety of performing EUS-guided RFA (54, 55). This technique may be offered as an alternative, for selected patients who are of advanced age, those with a poor general condition with an increased risk of postoperative complications.
In the literature, 62 patients with PNETs were treated by RFA, and the majority of the authors used the EUS approach (56–65). This minimally invasive technique offers the best combination of precise localization coupled with real-time visualization. The majority of the tumor was localized in the head (45%) or the body (45%) of the pancreas. The mean diameter of the PNETs was 2.1 cm. Complete response after RFA was defined as normalization of previously elevated serum hormone levels if the patients had a functional PNET, or as the absence of enhancing tissue at the tumor site on contrast-enhanced imaging studies: reduction in the vascularity or appearance of central necrosis. The rate of clinical success was 83.3% to 100% (56–65). Pancreatitis was the most frequent adverse event and it was sometimes related to thermal injury of the main pancreatic duct after RFA. This complication was observed in 30% of the cases for Rossi et al. (56) caused mild to severe pancreatitis that was associated with pancreatic fluid collection, which required percutaneous drainage and temporary placement of a main pancreatic duct prosthesis. These complications were caused by the absence of an internal cooling mechanism used for percutaneous or intraoperative approaches. But, recently two studies confirmed that the risk of pancreatitis after EUS-RFA was also present, and it was 7% and 11% for Barthet et al. (60) and Oleinikov et al. (52), respectively. No signs of thermal injury of major vessels or digestive organs were described in patients treated by EUS-RFA.
PNETs can be safely and successfully treated with percutaneous, EUS, or intraoperative RFA but only for selected patients. There is growing interest in pancreatic RFA, but we would have to reckon some limitations such as the lack of standardization, insufficient to cover the tumors’ volume, the need for dose–effect dosimetry studies, and none randomized studies in the literature.
Follow-Up of Patient with PNET
The routine follow-up of patients with PNETs must include clinical examination, biochemical markers, and conventional imaging. This follow-up must be adapted to the aggressivity of the tumor assessed by various factors such as tumor grade, stage, quality of the initial surgical treatment, and patient’s health status. A main objective of the follow-up is to be able to propose an effective and curative treatment in the case of recurrence. There is nevertheless a wide variation in the frequency of follow-up according to international guidelines (66). We can define several situations (Table 6):
1. For French guidelines, it is necessary to define two different groups of tumors (67):
For patient with G1 or G2-PNETs: conventional abdominal imaging (CT scan and/or MRI) associated with clinical exam need to be repeated within 3–6 months after the surgery. SRS can be used for follow-up if it was initially positive. Then the clinical exam and abdominal imaging should be performed every 6–12 months for 5 years, then every 12–24 months for 10 years then every 5 years. Biological markers are not recommended during follow-up of patients, unless if it was initially elevated.
For patient with G3-PNETs: Clinical exam and conventional imaging should be performed every 2 months for 6 months, then every 3 months for 1 year and then every 6 months.
2. For the Commonwealth Neuroendocrine Tumor Collaboration (CommNETS) (68):
Summary of recommended follow-up for PNETs after curative treatment.

PNET: pancreatic neuroendocrine tumor; CT: computed tomography; MRI: magnetic resonance imaging; SRS: somatostatin receptor scintigraphy; NCCN: National Comprehensive Cancer Network; ENETS: European Neuroendocrine Tumor Society; ESMO: European Society for Medical Oncology; US: ultrasound.
Follow-up should be performed every year for the first 3 years, then every 1 to 2 years for a total of 10 years. At 10 years, the necessity of surveillance should be discussed with the patient. The routine use of biomarkers is not recommended or only if the patient had a functional PNET. This working group analyzed and identified two groups of patients according to recurrence risk:
Tumors with low risk of recurrence require no follow-up: “G1-PNETs, node-negative tumors smaller than 2 cm; and patients with completely resected insulinomas of any size with either no suspected nodal activity or nodal activity formally classified as N0 based on nodes removed.”
Tumors with a high risk of recurrence (Ki-67 index of >5%, positive LNs) require a dedicated follow-up, that is, every 6–12 months for 3 years, then every 1–2 years for at least 10 years).
3. For National Comprehensive Cancer Network (NCCN) guidelines, clinical review with biomarkers and CT/MRI were performed at 3–12 months after surgery and then review 6–12 monthly for a maximum of 10 years. Patients with G3-PNETs must be followed every 3 months (69).
4. For European Neuroendocrine Tumor Society (ENETS) guidelines, patients with G1 or G2-PNETs need to be followed every 3 to 9 months with measurement of biochemical markers and conventional imaging as CT and/or MRI. An exception was identified: this interval of this follow-up can be increased if this patient had an “indolent” G1-PNET defined as well-differentiated tumors with Ki67 of <3%. SR imaging if positive should be repeated every 2 years or earlier if progression is suspected (70). For patient with localized R0/R1 resected G3-PNET, clinical exams and conventional imaging (CT and/or MRI) should be performed every 3 months during the first 2–3 years after surgical resection, and every 6–12 months up to 5 years following surgery. In patients with advanced disease, clinical exams and conventional imaging should be performed every 2–3 months while on active therapy. The biomarkers are recommended if they were positive initially (80).
5. For European Society for Medical Oncology (ESMO), a patient with R0/R1 resected G1 or G2-PNETs may be followed up every 3–6 months (biochemical markers CgA first or NSE, CT or MRI) and every 2–3 months for patients with G3-PNET. Concerning the SR imaging, it should be recommended after 18–24 months if positive (71).
Given the absence of consensus, the risk of excess radiation exposure, and the financial burden of follow-up, it appeared necessary to develop prognostic score for disease recurrence to guide individually tailored surveillance strategies (Table 7). In a recent article, Zaidi et al. divided the patients with “primary non-functioning, non-metastatic well/moderately differentiated PNET who underwent curative-intent resection” into three groups according to a RRS. This RRS ranging from 0 to 10 was calculated according to prognostics factors, which were assigned to a score:
Asymptomatic tumors size < 2 cm = 0 versus symptomatic tumor = 1,
Tumor size > 2 cm = 2;
Ki-67 < 3% = 0 versus Ki-67 3% to 20% = 1 versus Ki-67 > 20% = 6;
LN negative = 0, LN positivity = 1 (48).
Summary of predictive recurrence score.

PNET: pancreatic neuroendocrine tumor; LN: lymph node; RRS: recurrence risk score; CgA: chromogranin A; LNRS: lymph node risk score.
Patients were divided into low-risk group (RRS = 0–2), intermediate-risk group (RRS = 3–5), or high-risk group (RRS = 6–10). The recurrence risk at 2 years was 2%, 14%, and 33% for the low-risk group (RRS = 0–2), intermediate-risk group (RRS = 3–5), or high-risk group (RRS = 6–10), respectively. According to these results, they proposed a follow-up interval based on this score: 12, 6, and 3 months for low, intermediate, and high RRS patients, respectively (48).
The RRS has not included perineural invasion, which it is known as a prognostic factor. This factor was used in the scoring system described by Genç et al. (72). This score was used specifically for G1/G2-PNETs. Moreover, studies have suggested that biochemical markers as CgA may have clinical utility but Zaidi et al. (48) did not use this factor in this score. Fisher et al. (73), using CgA (>5 upper limit of normal), grade (2 or 3), surgery for tumor recurrence, and tumor size (⩾4.0 cm) identified three groups: low (score = 0), intermediate (score = 1), and high (score ⩾ 2) risks. 5-year recurrence-free survival (RFS) rate was 39%, 63%, or 96%, respectively. In the study by Ausania et al. (74), among three factors (tumor size (>20 mm), LN metastasis, and Ki-67 of >5% or mitotic index (MI) of >2), patients with two factors had a significantly lower 5-year disease-specific survival compared to low-risk patients (70% versus 100%).
All these scores can be used after curative surgery of PNETs but many studies have proposed observation for PNETs of <2 cm (75, 76). For this selected patients, Lopez-Aguiar et al (77) described a new lymph node risk score (LNRS) based on tumor localization (distal = 1, proximal = 4) and Ki-67 (<3% = 1, ⩾3 = 3). This score categorized patients into three groups: low (LNRS 1–2), intermediate (LNRS 3–4), and high (LNRS 5–7) risks. The lymph recurrence was 3.2%, 13.8%, and 20.5%, respectively.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.