Abstract
Background and Aims:
Obesity is an increasing problem in patients after total knee replacement. The aim of this study was to investigate whether a weight loss intervention before primary total knee replacement would improve quality of life, knee function, mobility, and body composition 1 year after surgery.
Material and Methods:
Patients scheduled for total knee replacement due to osteoarthritis of the knee and obesity were randomized to a control group receiving standard care or to an intervention group receiving 8-week low-energy diet before total knee replacement. Patient-reported quality of life, 6-Min Walk Test, and body composition by dual-energy X-ray absorptiometry were assessed before intervention for the diet group, and within 1 week preoperatively for both groups, and the changes in outcome from baseline to 1 year after total knee replacement were compared between groups. The number of participants was lower than planned, which might introduce a type-2 error and underestimate the trend for a better outcome after weight loss.
Results:
The analyses are based on a total of 76 patients, 38 in each group. This study showed major improvement in both study groups in quality of life and knee function, though no statistically significant differences between the groups were observed 1 year after total knee replacement. The average weight loss after 8-week preoperative intervention was 10.7 kg and consisted of a 6.7 kg reduction in fat mass. One year after total knee replacement, the participants in the diet group managed to maintain the weight reduction, whereas there was no change in the control group.
Conclusion:
The results suggest that it is feasible and safe to implement an intensive weight loss program shortly before total knee replacement. The preoperative intervention resulted in a 10% body weight loss, improved body composition, lower cardiovascular risk factors, and sustained s-leptin.
Introduction
The knee is the most common weight-bearing joint affected by osteoarthritis (OA) with symptoms of pain and loss of function (1). In addition to being a known risk factor for general health (2), obesity is by far the most important and well-documented risk factor for development of knee OA (3). The lifetime risk of symptomatic knee OA (4) as well as OA in other joints (5, 6) increases with higher body mass index (BMI).
Obesity is also an increasing problem in patients after total knee replacement (TKR). Several observational studies have shown that obesity is associated with poor health-related quality of life (QoL) (7), low physical function (8), and more pain after surgery compared with patients with a BMI <30 (9). Moreover, surgery in obese patients is associated with increased operation time, problems with wound healing, scarring (10–12), and increased morbidity and mortality (12, 13). In addition, an association between obesity and deep infection after TKR (14, 15) has been reported.
Numerous studies have shown that weight loss is very important in the treatment of knee OA (16–18). Previous results in patients using formula foods have shown lower cardiovascular risk factors (19). This is an important change in people with OA who have a higher risk of cardiovascular disease than their matched peers (20). Moreover, patients are expected to achieve a faster total weight reduction (kg) and a larger reduction in fat mass than by using the conventional hypo-caloric-diet (21–24).
Reduction in fat mass in overweight patients with knee OA before TKR is presumed to be overall beneficial to patients’ general health and outcome after TKR. Weight reduction before TKR, however, has not been investigated. The aim of the “Weight Loss Intervention before Total Knee Arthroplasty” (WITKA study) was to investigate whether weight loss interventions before primary TKR would improve health-related QoL, knee function, mobility, and total body composition 1 year after surgery. We hypothesized that it would be feasible and safe to reduce obese patients’ preoperative body weight with 5%–10% and that weight loss before TKR would improve health-related QoL, knee function, and body composition after surgery.
Patients and Methods
Study Population
The study was a single-blinded, single-center, randomized controlled trial (RCT). Patients were recruited from the outpatient clinic of the Department of Orthopedics, Hospital of Southern Jutland, Denmark, between August 2011 and April 2013. Patients with OA scheduled for primary TKR, a BMI ⩾30, and motivated for weight loss were eligible for inclusion. Exclusion criteria were rheumatoid arthritis and planned bariatric surgery. Patients operated on both knees during the project period only participated once.
Patients received both oral and written information about the study in the outpatient clinic. Within 3 days, the primary investigator (A.L.) telephoned the patients who either consented to participate or declined participation. Ethical approval was granted by Central Denmark Region Committees on Health Research Ethics (Journal number: S-201001309), and the study was registered at www.ClinicalTrials.gov (NCT01469403).
Randomization and Blinding
Patients who met the inclusion criteria and accepted participation were randomly assigned to either preoperative intensive weight loss therapy 8 weeks prior to TKR and 12 months postoperatively (diet group) or to standard care (control group). Randomization was performed in a 1:1 ratio in blocks of 10. The randomization sequence involved stratification according to gender and BMI (<35/⩾35). The randomization was performed in a project management database from Procordo.com software (DK) at inclusion into the study. The attending surgeon and the nursing staff were blinded to the randomization of patients.
Intervention
Diet group
As previously described in detail (17), patients received a low-energy liquid diet (810 kcal/day) using commercially available formula foods (Cambridge Weight Plan®, Northants, UK) and nutritional education during 8 weeks preoperatively. The patients attended weekly group sessions of 1.5 h led by an experienced dietitian.
The formula diets consisted of ready-to-use meal, bars, and sachets to mix with water or skimmed milk (7.5 dL a day) to make shakes, soups, or porridge, consumed four times a day. The program met all recommendations for daily nutrient intake of vitamins and minerals. Daily intake of protein was at least 79.7 g, fat 12.0 g, and fiber intake was at least 13.3 g a day. Patients were advised to use a fiber supplement to avoid constipation. The goal of the dietary program was to reduce body weight by 5%–10% preoperatively. The median duration of waiting time between randomization and surgery was 10.5 (minimum 8.8–maximum 61.6) weeks. The second phase of the intervention program (the maintaining phase) started right after surgery and ended 1 year postoperatively. During the second phase, regular meals were combined with one formula diet serving per day, which increased the daily calorie intake to approximately 1200 kcal, and the patients participated in approximately eight group sessions led by the study dietitian. The long-term goal was lifestyle dietary changes and modifications.
Control group
The control group followed the standard care and surgery protocol for TKR. Standard care entails no information on the benefit of losing weight before TKR. The median duration of waiting time between randomization and surgery was 6.9 (minimum 1.9–maximum 21.8) weeks. The patients in the control group did not receive any nutritional instructions from the study staff.
Measurements and Procedures
Demographic characteristics (Table 1) were recorded before the intervention (diet group) and preoperatively (control group). For all patients, body weight was measured in kilograms on the same decimal scale (Stand weight, Kern Capacity 0–200 kg, class III, approved) wearing light clothing. Body height was measured with a digital altimeter (Soehnle 5003), and BMI was calculated (kg/m2).
Demographics and baseline characteristics.
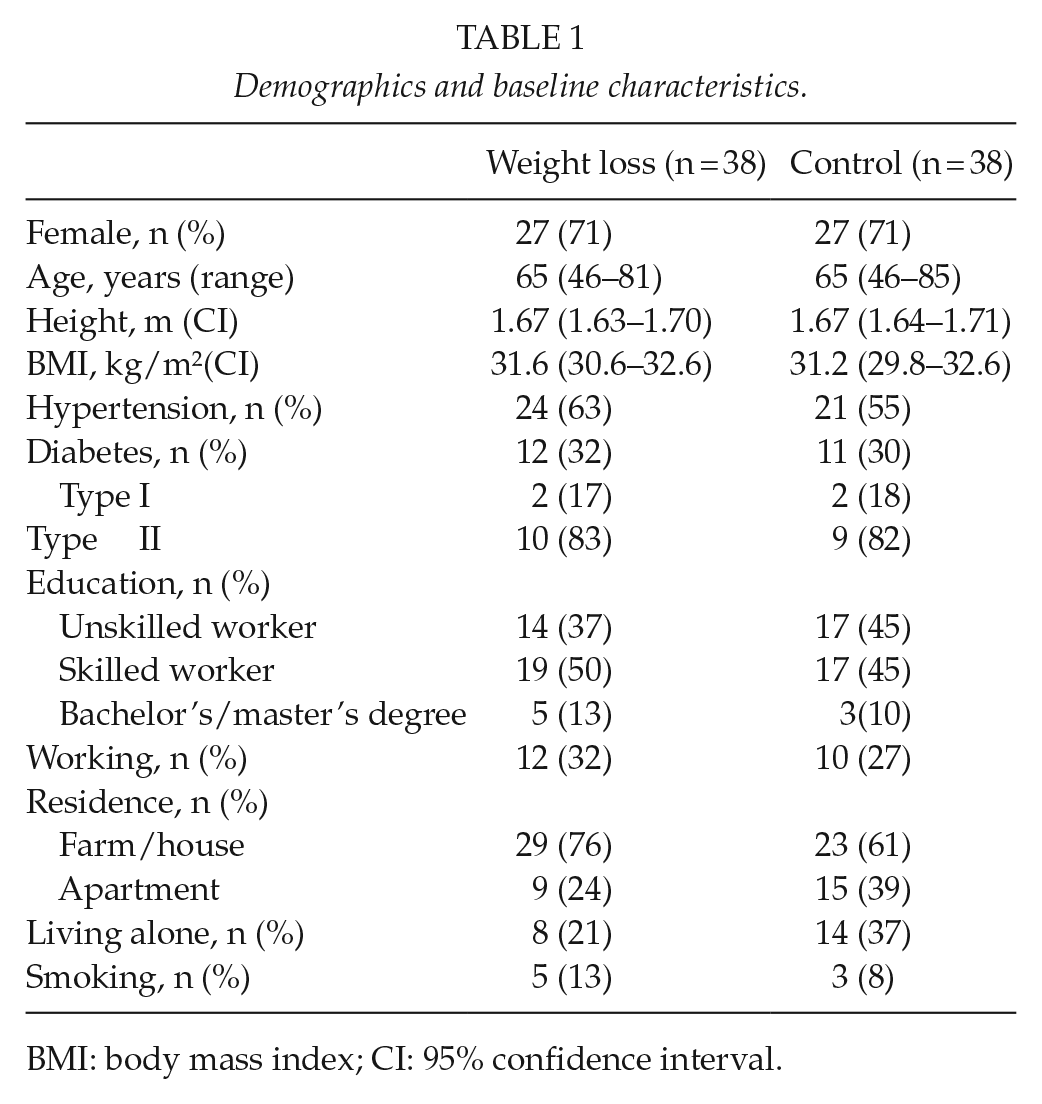
BMI: body mass index; CI: 95% confidence interval.
The patient-reported outcome measure (PROM) Short-Form 36 (SF-36) subscale Physical function Component Score (PCS) was the primary outcome. Secondary outcomes were SF-36 subscales Mental Component Score (MCS), Knee injury and Osteoarthritis Outcome Score (KOOS), and 6-Min Walk Test (6MW). All PROMs were reported electronically by patients into the project database. The 6MW was based on standard procedures on a 30-m course marked for every 5 m and led by a project physiotherapist. In addition, total body composition (fat mass, lean mass, and bone mass) and bone mineral density (BMD) were measured using dual-energy X-ray absorptiometry (DXA). DXA (Lunar Prodigy Advance) narrow-angle fan beam is a non-invasive, precise, and operator independent method that exposes the patient to a low radiation dose (25). All scans were analyzed using the enCORE Software, version 13.60 (GE Healthcare, USA). Automatic edge detection was used for scan analyses. The scanner’s calibration was checked on a daily basis before each scanning session, using the GE Lunar calibration phantom. The manufacturer’s guidelines for patient positioning and for scan acquisition were followed. To measure the variance of the total body scans, double scanning was performed on all the project participants and the coefficient of variation was 0.1%. Blood pressure and heart rate were measured using a digital sphygmomanometer (UA-852).
Blood samples (non-fasting) were collected to assess lipid status (total cholesterol (CHOL), high-density lipoprotein cholesterol (HDL), low-density lipoprotein cholesterol (LDL), and triglyceride (TGLY)). The blood samples were analyzed at the Department of Clinical Biochemistry at the Hospital of Southern Jutland, Denmark, using standardized laboratory procedures. The European recommendations of signal values for lipids (19) are CHOL < 5 mmol/L, LDL < 3 mmol/L, HDL > 1 mmol/L, and TGLY < 2 mmol/L. In addition, serum leptin concentration was measured as a biomarker of adiposity. The sample was collected, processed, stored at −80 °C, and analyzed at research laboratories at Aarhus University Hospital after the final clinical control. All outcomes were measured on all visits for both groups. The diet group had one visit more than the control group (Fig. 2) before the intervention program starting 8 weeks preoperatively. Outcomes were measured for both groups within 1 week preoperatively and after surgery at 8 weeks and 6 and 12 months. Data were collected from the patient records and included days of admission, duration of anesthesia and surgery time in minutes, mobilization (getting out of bed) (yes/no), getting help with personal care from caregivers (yes/no), and secretion from the surgical wound at the day of surgery (day 0) (yes/no).
Safety
In the weekly sessions with the project dietitian, adverse events possibly related to the low-energy diet spontaneously reported by patients were recorded for the diet group. The project nurse and the primary investigator observed if complications occurred in the waiting time between inclusion and surgery, and perioperative.
Tkr Surgery
All patients had a PFC–sigma CR knee prosthesis (DePuy Orthopaedics, Inc., USA), and the surgical procedure was midline incision with the medial parapatellar approach. Four highly experienced surgeons performed all operations.
Sample Size
The sample size of 51 participants in each group allowed a drop-out rate of 20%, which was calculated based on the primary outcome to obtain 80% power to detect an 8% difference between groups in the SF-36 physical component score 12 months after TKR (10). The significance level was set at 5% using a two-sided analysis.
Statistics
Demographics and baseline characteristics were analyzed using descriptive statistics presented as means with 95% confidence intervals (CIs) and frequency (%). Differences in preoperative outcomes between the groups were assessed using Student’s t-test. Before the t-test, the assumptions of the model were tested. Distribution of data was evaluated by a histogram, and a Bartlett test was performed for homogeneity of variances. The chi-square test was performed for the categorical variables, and Fisher’s exact test was used for the variables that had ⩽5 observations. The between-group difference in the change in primary and secondary continuous outcomes (dependent variable) after 12 months were made using analysis of covariance (ANCOVA) adjusted for baseline values, gender, and BMI. The model included the treatment group as a main effect and the baseline score as an additional covariate, with results presented as means and 95% CIs. Both the primary and secondary outcomes were analyzed in intention-to-treat (ITT) and per-protocol (PP) analyses. The ITT analyses were considered primary. The ITT population retained all randomized patients with recorded baseline outcome (n = 78). The baseline-observation-carried-forward approach was used for patients who did not complete the study (Fig. 1). All observations in the sample (n) were independent, had the same probability of events, and the sample sizes (n) were determined in advance. For the statistical analysis, the Stata 12 software was used. All p-values <0.05 were considered statistically significant.
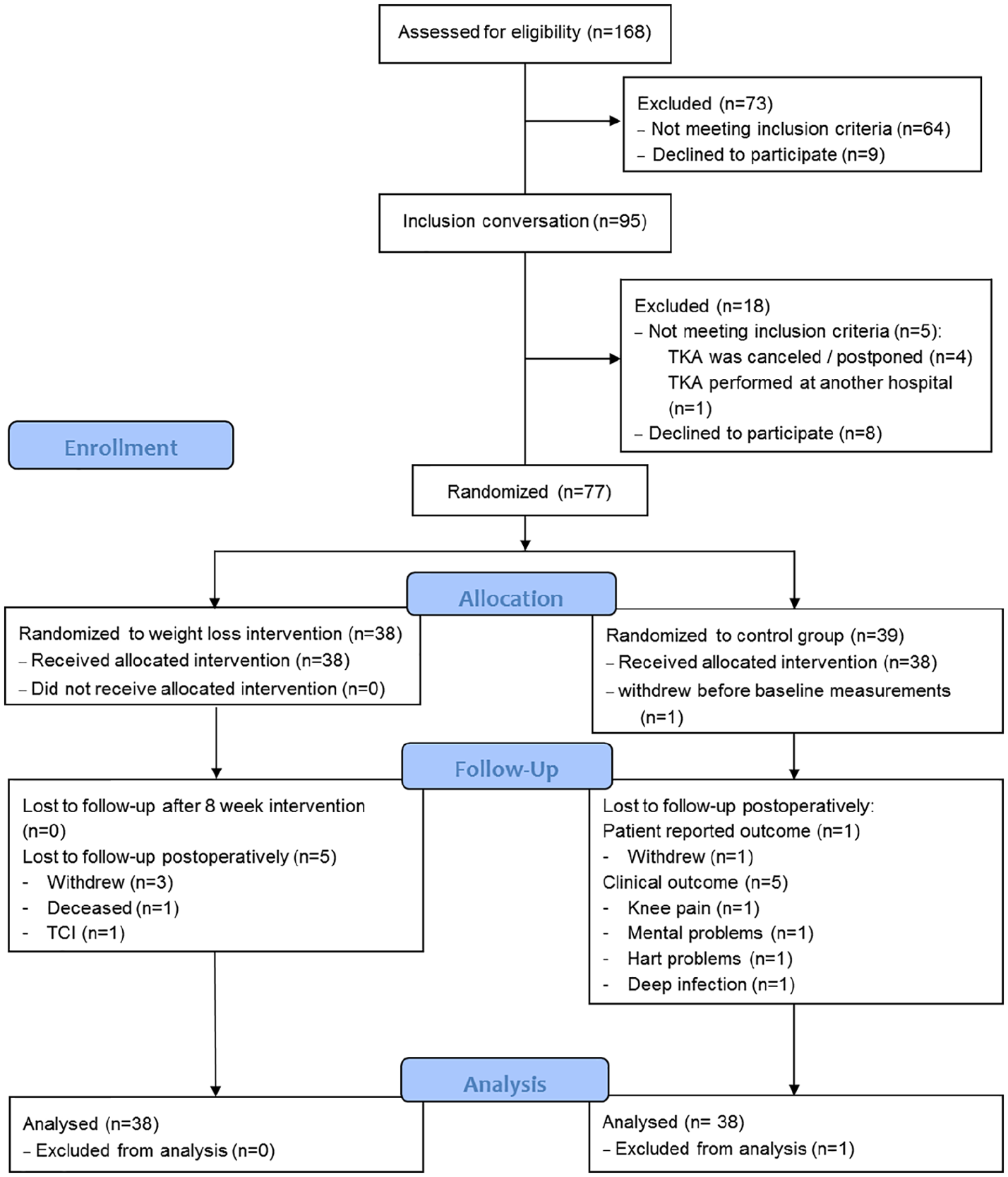
Flow of patients in the study.
Results
Patients
Of the 168 patients, eligible for enrollment, 91 were excluded (Fig. 1). A total of 77 patients underwent randomization: 38 were assigned to the 8-week weight loss intervention program before surgery (diet group) and 39 were assigned to standard TKR care (control group). Right after randomization but before baseline measurements, one patient from the control group withdrew from the study. The baseline characteristics of the two groups were similar with respect to age, gender, height, BMI (Table 1), weight, and body composition (Table 2). The patients who declined to participate were comparable to those undergoing randomization in relation to age, gender, and BMI.
Presentation of the diet group before the intervention program and comparison between the diet group after 8 weeks and the control group immediately before surgery.
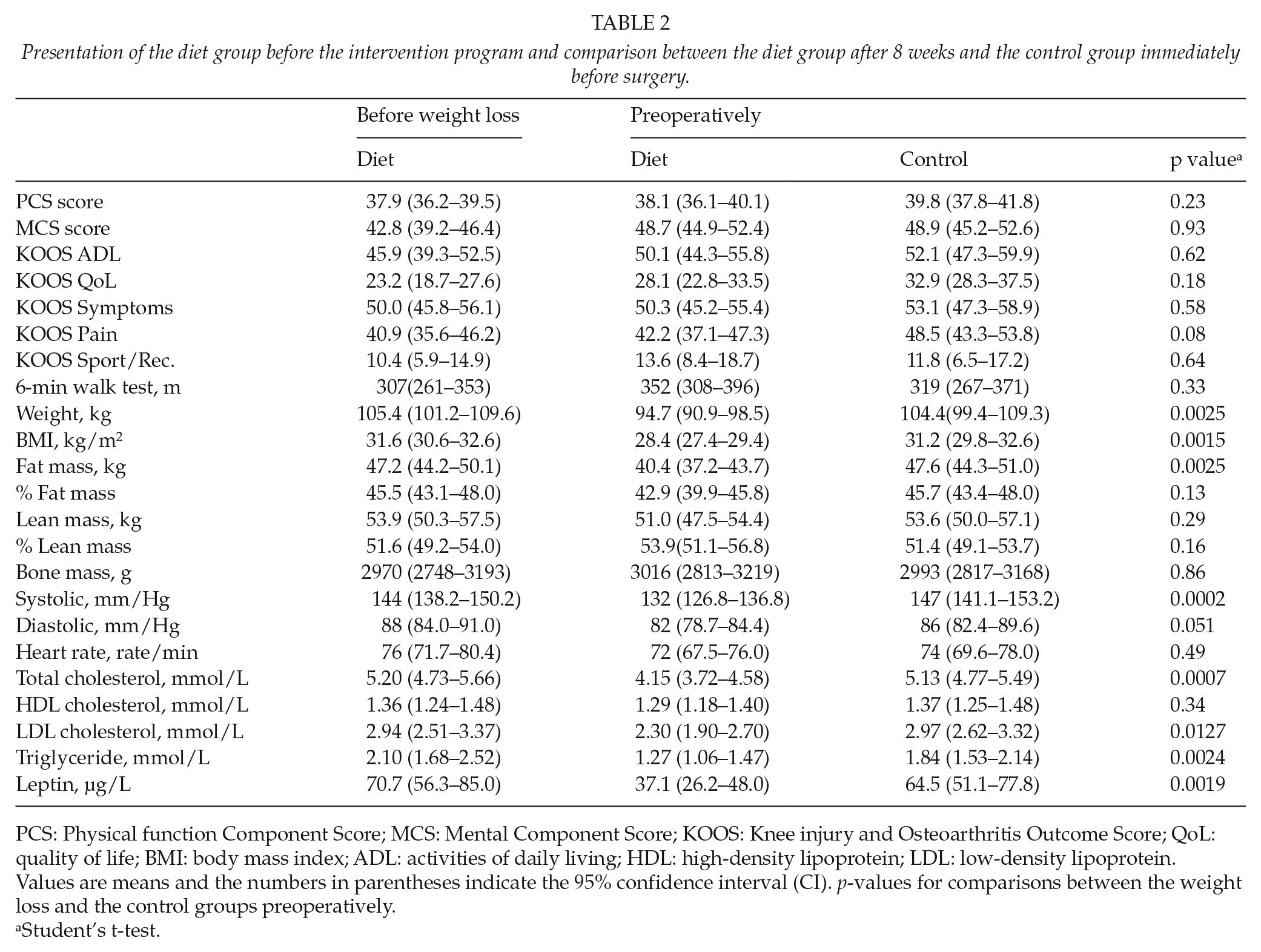
PCS: Physical function Component Score; MCS: Mental Component Score; KOOS: Knee injury and Osteoarthritis Outcome Score; QoL: quality of life; BMI: body mass index; ADL: activities of daily living; HDL: high-density lipoprotein; LDL: low-density lipoprotein.
Values are means and the numbers in parentheses indicate the 95% confidence interval (CI). p-values for comparisons between the weight loss and the control groups preoperatively.
Student’s t-test.
Perioperative and Postoperative Comparison
All baseline (before intervention) PROM subscale scores (SF-36 and KOOS) were slightly lower in the diet group than in the control group (Table 2). After 8 weeks preoperative intervention, the diet group improved in all PROM subscales, though no statistically significant differences between groups were found. The same trend was seen for the 6MW. In addition, the diet group achieved a statistically significant lower weight, BMI, fat mass, systolic blood pressure, CHOL, LDL, and TGLY (Table 2), and leptin decreased by 48% compared with the control group after the 8-week preoperative intervention. Although not statistically significant, fat percentage, diastolic blood pressure, and heart rate were also lower in the diet group. The lean percentage increased by 2.3% in the diet group in the time before intervention to the preoperative test and was 2.5% higher than in the control group. However, lean mass was reduced by 3 kg in the diet group from the time before intervention to the preoperative test, and mean lean mass was 2.6 kg lower in the diet group than in the control group.
In total, 32 patients (84%) in the diet group and 24 patients (63%) in the control group were mobilized at the day of surgery (day 0). In the control group, six patients experienced secretion from the wound compared with one in the diet group. There was no difference in the number of admission days and duration of surgery between groups.
Patient-Reported Outcome and Mobility
ITT analysis
Adjusted for baseline values, gender, and BMI, there was no statistically significant difference between groups in primary outcome SF-36 PCS (Table 3). In addition, there was no statistically significant difference between groups in secondary outcome: SF-36 MCS score, all KOOS subscale scores, and in 6MW. Both groups improved in all PROMs (SF-36 PCS combined (crude) by 7.7 units (CI: 5 to 10), and all KOOS subscale scores combined (crude) improved from 20.5 units (CI: 16.0 to 25.0) in symptoms to 32.9 units (CI: 26.4 to 37.7) in QoL, and in the 6MW combined (crude) improvement 78 m (CI: 56 to 100)).
Outcome at 1 year in the diet and the control groups.
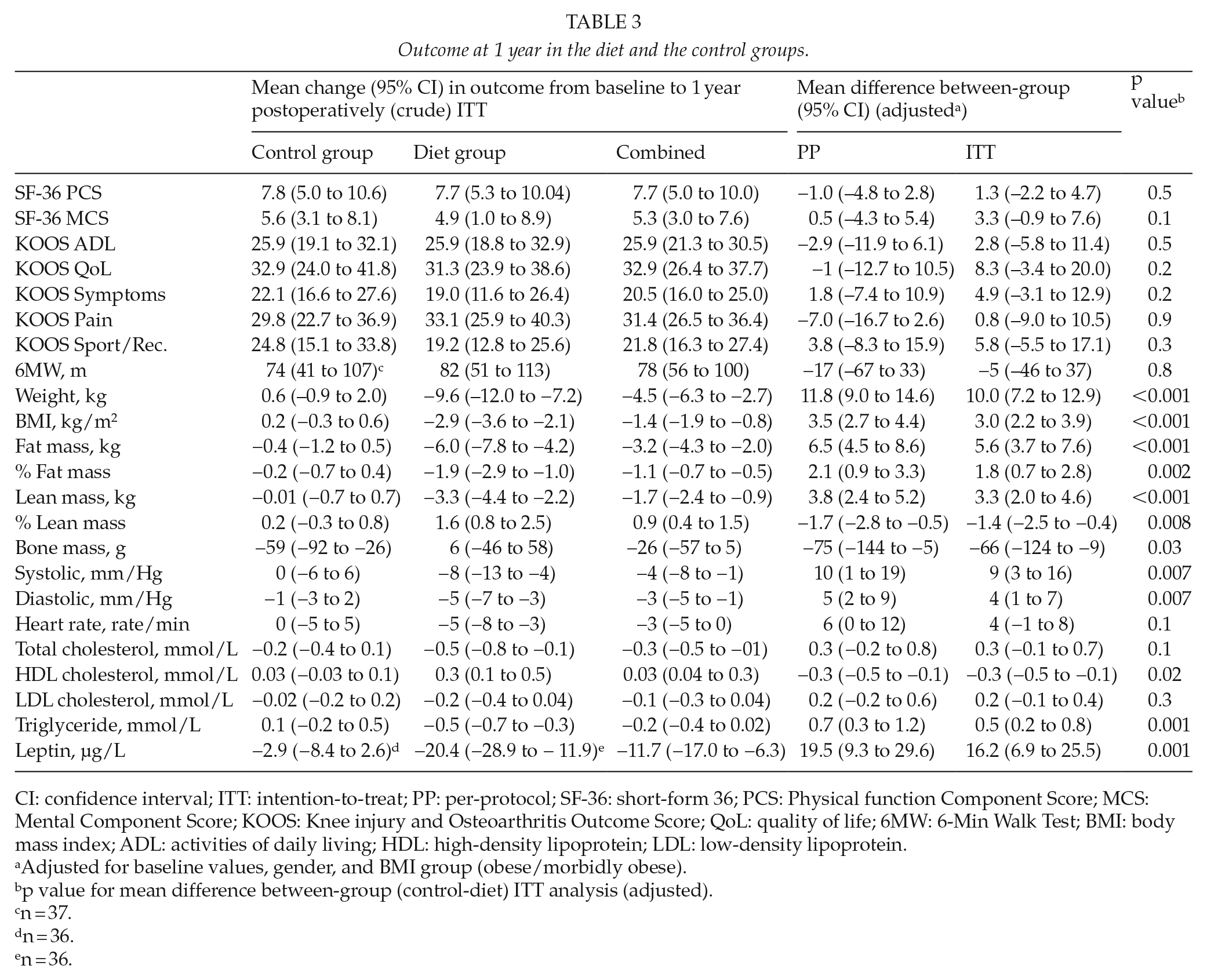
CI: confidence interval; ITT: intention-to-treat; PP: per-protocol; SF-36: short-form 36; PCS: Physical function Component Score; MCS: Mental Component Score; KOOS: Knee injury and Osteoarthritis Outcome Score; QoL: quality of life; 6MW: 6-Min Walk Test; BMI: body mass index; ADL: activities of daily living; HDL: high-density lipoprotein; LDL: low-density lipoprotein.
Adjusted for baseline values, gender, and BMI group (obese/morbidly obese).
p value for mean difference between-group (control-diet) ITT analysis (adjusted).
n = 37.
n = 36.
n = 36.
PP analysis
Although there was no significant change in the outcomes between the PP and ITT analysis, the PP analysis enhanced the differences between groups in favor of the diet group (Table 3). Fig. 2A illustrates mean SF-36 PCS improvement across the study period and shows improvement over time in both groups. Similar improvements were seen in MCS score, 6MW test, and in all KOOS subscales (not shown).
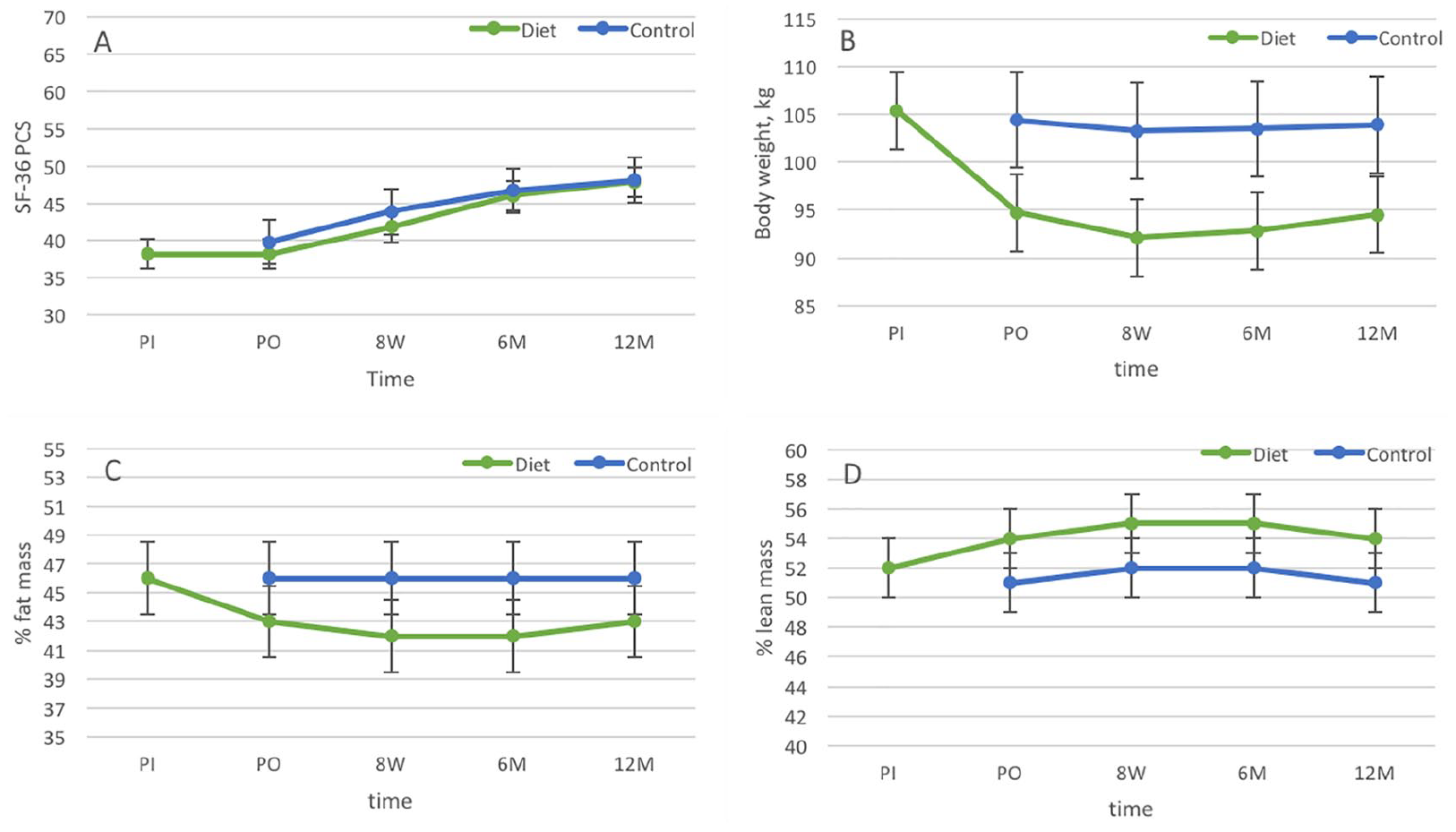
PP mean SF-36 component score, body weight, and body composition across study period.
Body Weight and Body Composition
ITT analysis
Adjusted for baseline values, gender, and BMI, the changes in weight, BMI, fat mass, % fat mass, % lean mass, and bone mass (Table 3) showed statistically significant differences between groups in favor of the diet group, while lean mass decreased significantly more in the diet group.
PP analysis
The adjusted PP analysis confirmed the finding from the ITT analysis with a statistically significant difference in all outcomes between groups. Body weight, % fat mass, and % lean mass across study period are illustrated in Fig. 2B to D and show the improvement in the diet group whereas there was no or little change in the control group.
Lipids, Leptin, and Blood Pressure
ITT analysis
The mean TGLY was reduced in the diet group and increased in the control group. The adjusted mean TGLY difference between groups was statistically significant 0.5 (CI: 0.2 to 0.8) mmol/L. In addition, leptin difference between groups was statistically significant with 16.2 (6.9 to 25.5) µg/L. The adjusted mean difference in HDL was also statistically significant. The HDL increased more in the diet group. There were no statistically significant differences between groups in CHOL and LDL.
PP analysis
The findings in the ITT analysis were confirmed by the PP analysis.
Safety and Complications
Preoperatively
We recorded only mild adverse events possibly related to the low-energy diet: three patients experienced dry skin, four experienced constipation, one was bothered by wind/flatulence, one had cramps, one felt dizzy, two experienced headaches, two suffered from sleeplessness, two were sensitive to cold, and two experienced bad breath. In addition, we recorded three serious adverse events occurring in the waiting time between randomization and surgery in both the diet group and the control group as a 70-year-old male patient had TKR postponed for 3 months due to cardiac arrhythmia. This patient had lost more than 13 kg in 8 weeks, but no adjustment was made to his dosage of antihypertensive medication. After cardiovascular examination and dosage adjustment, there was no contraindication for TKR. Another 76-year-old male patient had TKR operation postponed for 1 year due to pacemaker surgery. The treating physician did not regard this episode to be related to the weight loss intervention. Due to high blood pressure, a 63-year-old male from the control group with known hypertension had TKR postponed for 6 months. One male and one female from the diet group, both 57 years of age, chose to postpone TKR by 22 and 29 weeks, respectively, because of decreasing pain after the 8-week weight loss intervention.
In-hospital
No perioperative complications were recorded in any of the groups. Three in-hospital postoperative complications were recorded: a diet group patient was diagnosed with quadriceps microfiber blasts by ultrasonography, a control group patient was admitted to the intensive care unit and treated for urosepticemia, and a control group patient had serious surgical wound complications.
Follow-up
Five TKR-related complications were recorded: one patient in each group had manipulation under anesthesia, a patient from the control group had a dislocation of the prosthesis (no infection), one patient from each group had deep infection, which resulted in an exchange of liner for the patient in the diet group, and a total prosthesis removal for the patient in the control group. Eight patients were hospitalized in the follow-up period for reasons unrelated to TKR: in the diet group: one patient with bowel disease, one with prostate cancer, one with transient ischemic attack, one with stroke, and one died caused by cardiac arrest; in the control group, one patient underwent cataract surgery, one had enlarged prostate, and one had coronary heart disease.
Discussion
Key findings
Results from this RCT showed considerable improvement in both study groups in health-related QoL (SF-36), knee function (KOOS), and mobility (6MW), though no statistical differences between the groups were observed 1 year after TKR. Nevertheless, the diet group showed significant improvement in all general health-related outcomes compared to the control group. Furthermore, 33% more patients in the diet group were out of bed and mobilized on the day of the surgery compared to the control group.
There was no difference in the change of health-related QoL, knee function, and mobility between the diet group and the control group. This may reflect that we failed to include the planned number of participants in the study. This means that the study was slightly underpowered to detect a difference in the primary outcome measure between groups 12 months after TKR. Another explanation may be the follow-up period of 1 year that was a rather short period of time for determinations of the result of TKR. It would be interesting to investigate the outcome 5 years after the TKR (8). In addition, it is possible that the weight loss would have had a greater effect on the measured outcomes if the intervention was timed to take place at an earlier stage of knee OA. Our results on health-related QoL and knee function are comparable to findings in studies with similar interventions on knee OA patients (26, 27) where significant weight reduction and improvement in knee OA symptoms is achieved but with no difference between the groups in total Western Ontario and McMaster Universities (WOMAC) index (26) or in KOOS and SF-36 (27). Although the patients in this study achieved major improvements in health-related QoL, they did not obtain as high QoL 1 year after surgery as normal weight patients undergoing TKR (8). Our results are in concordance with a previous study (28) in a group of heavily obese patients reporting a weight loss of 5% or 10% to be insufficient to achieve clinically important changes in health-related QoL.
Preoperative Findings
The preoperative weight loss in the diet group had little effect on QoL. However, the lipid levels decreased in the diet group and fell below the recommended alert signal values (19). Furthermore, the average blood pressure and heart rate decreased after completion of the preoperative intervention program. Our preoperative results are similar to those of other RCT studies (17, 21, 26, 27) in clinical rheumatology departments.
Intervention and Safety
We did not manage to avoid that the participants in the diet group lost an average of 2.9 kg lean mass (27% of total weight loss), during the 8-week preoperative intervention program. With a reduction of 10% of the total body weight, it is natural that the lean mass is also reduced, but we might have been able to minimize the loss of lean mass if we had offered physical training as part of the intervention. However, when looking at the total change in body composition, the patients managed to increase their lean mass relative to their total body weight by 2.3%. Thus, patients had more lean mass comparative to their total weight after the weight loss.
The advantages of using low-energy formula foods for weight loss before TKR were a significant reduction in fat mass (6.7 kg), and at the same time, we ensured that the patients received adequate vitamins, minerals, and proteins corresponding to the recommendations by the Danish Health Authority (29).
In general, the intensive diet resulted in few and mild adverse events comparable to adverse events reported in similar diet programs (17). One serious adverse event presumably happened due to a too large dose of antihypertensive medication. The reduction of antihypertensive and antidiabetic medications during intensive diet represents a challenge for the treating physician (30). Finally, TKR was without any perioperative complications in all patients.
Complications
There were no perioperative complications and no difference between the groups in relation to the few reported in-hospital or postoperative complications. At 8–9 months after TKR, two patients had a deep surgical infection (one in each group). According to The Danish Knee Arthroplasty Register, the rate of deep surgical infection in Denmark in 2011 was 1.9%. In our study population, deep surgical infection was seen in 2.6%. A retrospective cohort study (31) evaluated whether obese patients losing weight before TKR (5% decrease in body weight) and maintained weight loss postoperatively were at lower risk of surgical site infection and re-admission compared with those who maintained the same weight. Similarly, there was no significant difference in deep surgical infection between patients who lost weight (1.2%) compared with those who maintained the same weight (0.9%). In a large cohort study (32) with a preoperative weight loss intervention, the risk of deep surgical infection and readmission was not significantly different in the patients who gained or lost weight preoperatively compared to those who maintained the same weight.
Limitations
The number of included participants was 7 fewer than planned to obtain the aimed 80% power. However, funding did not allow an extension of the study period, and this might introduce a type-2 error in the outcome and underestimate the trend for a better outcome after weight loss.
In conclusion, both groups obtained a large improvement in health-related QoL, knee function, and mobility 1 year after surgery, but the diet group did not achieve a greater improvement compared to the control group. Conversely, the intervention resulted in improved body composition. There were no perioperative complications and no differences between groups in relation to the reported postoperative complications. Regarding the primary outcome, the results do not support the introduction of a 10% weight loss in routine practice. An explanation for this may be due to the number of participants that was lower than planned and might underestimate the trend for a better outcome after weight loss. However, the results suggest that it is feasible and safe to implement an intensive weight loss program shortly before TKR and suggests that there are several advantages of using the waiting time for surgery for weight loss in obese patient.
Footnotes
Acknowledgements
We wish to express our special thanks to the WITAK study staff for their dedicated work; the staff in the orthopedic outpatient clinic in Sønderborg; the staff at the Clinical Orthopedic Research Unit in Aarhus, the Hospital Southern Jutland; and our sponsors: the Danish Rheumatism Association; Cambridge Weight Plan®, Northants, UK; Linak A/S; Fabrikant Mads Clausen’s foundation Danfoss; Johs. M. Klein og hustru’s foundation; Knud and Edith Eriksen’s foundation; Peter Ryholt’s foundation; and Jeppe Juhl og hustru Ovita Juhl’s foundation.
Author Contributions
A.L., J.O.L., H.B., K.S., and I.M. contributed to conception and design of study. A.L. contributed to acquisition of data. A.L. and I.M. contributed to analysis of data. A.L., J.O.L., H.B., K.S., and I.M. contributed to interpretation of data. A.L., J.O.L., H.B., K.S., and I.M. contributed to drafting, important revisions, and final approval.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.L. received travel grants to attend a scientific meeting and a research grants from the Cambridge Weight Plan, Northants, UK. H.B. has received research grants from Cambridge Weight Plan, Northants, UK.
Ethical Approval
The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki (6th revision, 2008). The Central Denmark Region Committees on Health Research Ethics approved the study (Journal number: S-201001309), and the study was registered at ![]() (NCT01469403). All participants gave written informed consent.
(NCT01469403). All participants gave written informed consent.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by The Danish Rheumatism Association; Cambridge Weight Plan®, Northants, UK; Linak A/S; Fabrikant Mads Clausen’s foundation Danfoss; Johs. M. Klein og hustru’s foundation; Knud and Edith Eriksen’s foundation; Peter Ryholt’s foundation; and Jeppe Juhl og hustru Ovita Juhl’s foundation. None of the sponsors were involved in the design of the study; in the collection, analysis, and interpretation of data; in writing of the manuscript; or in the decision to submit the manuscript for publication.
