Abstract
Background and Aims:
We present an open retromuscular mesh technique for incisional hernia repair, the modified peritoneal flap hernioplasty, where the fascia is sutured to the mesh and the hernia sac utilized for anterior mesh coverage. The aim was to describe the modified peritoneal flap hernioplasty technique and to compare it to a retromuscular repair, without component separation, regarding short-term complications, patient satisfaction, abdominal wall complaints, and recurrent incisional hernia.
Materials and Methods:
Consecutive patients operated electively with modified peritoneal flap hernioplasty technique (December 2012–December 2015) or retromuscular technique (Jan 2011–Oct 2014) were included in a retrospective single-center cohort study. Outcomes were evaluated from the Swedish Ventral Hernia Registry, by chart review, physical examination, and an abdominal wall complaints questionnaire.
Results:
The modified peritoneal flap hernioplasty group (n = 78) had larger hernias (mean width 10.4 vs 8.5 cm, p = 0.005), more advanced Centers for Disease Control classification (p = 0.009), and more simultaneous gastrointestinal-tract surgery (23.1% vs 11.5%, p = 0.041) than the retromuscular group (n = 96). No difference in short-term complications was seen. Incisional hernia recurrence was lower in the modified peritoneal flap hernioplasty group (1.4% vs 10.3%, p = 0.023), and patients were more satisfied (93.8% vs 81.7%, p = 0.032). Follow-up time was shorter in the modified peritoneal flap hernioplasty group (614 vs 1171 days, p < 0.001).
Conclusion:
This retrospective study showed similar rates of short-term complications, despite more complex hernias in the modified peritoneal flap hernioplasty group. Furthermore, a lower incisional hernia recurrence rate for the modified peritoneal flap hernioplasty technique compared with the retromuscular technique used in our department was found. If this holds true with equally long follow-up remains to be proven.
Keywords
Introduction
Incisional hernia (IH) is the most frequent long-term complication after midline laparotomy with incidences between 11% and 25%, and causes considerable morbidity (1–4).
Surgical techniques for IH-repair have evolved over the last decades with improved results related to the use of mesh (5–7). A frequently experienced problem with the retromuscular (RM) technique, without component separation (CS), used at our department for many years was the inability to cover the mesh with fascia, leaving the mesh exposed to subcutaneous tissue with increased risk for seroma formation and wound/mesh infection (8–12). An anterior component separation (ACS) (13) demands an extensive subcutaneous dissection with risk of related wound complications. In 2012, the minimally invasive ACS was sparsely reported and not adopted at our department. The posterior component separation (PCS) (14) with division and release of the transverse abdominis muscles might be considered technically demanding for surgeons not specialized in hernia surgery.
An alternative technique for anterior mesh coverage, without CS, was developed at our department in 2012. In 2014, Malik et al. (15) described a similar technique, the peritoneal flap hernioplasty, in which flaps of the hernia sac are created by entering the hernia sac in the midline and the RM space through the posterior rectus fascia on one side and the anterior fascia on the other side. The flaps are then used to close the abdominal cavity and to cover the mesh subcutaneously by suturing the flaps to the fascial edges. Similar methods, known as sandwich techniques, have been described earlier in small case series and were recently summarized in two reviews (16–20). Our technique differs in the way the anterior closure is achieved (Fig. 1). In this modified peritoneal flap hernioplasty (MPFH) technique, the incised fascial edges are sutured together when possible and to the mesh anteriorly when not. The remaining hernia sac is used to cover any exposed part of the mesh. In this way, the better suture bearing capacity of the fascia, compared with the hernia sac, is utilized when the fascial edges are advanced and sutured as medially as possible in order to minimize the distance needed to be bridged.
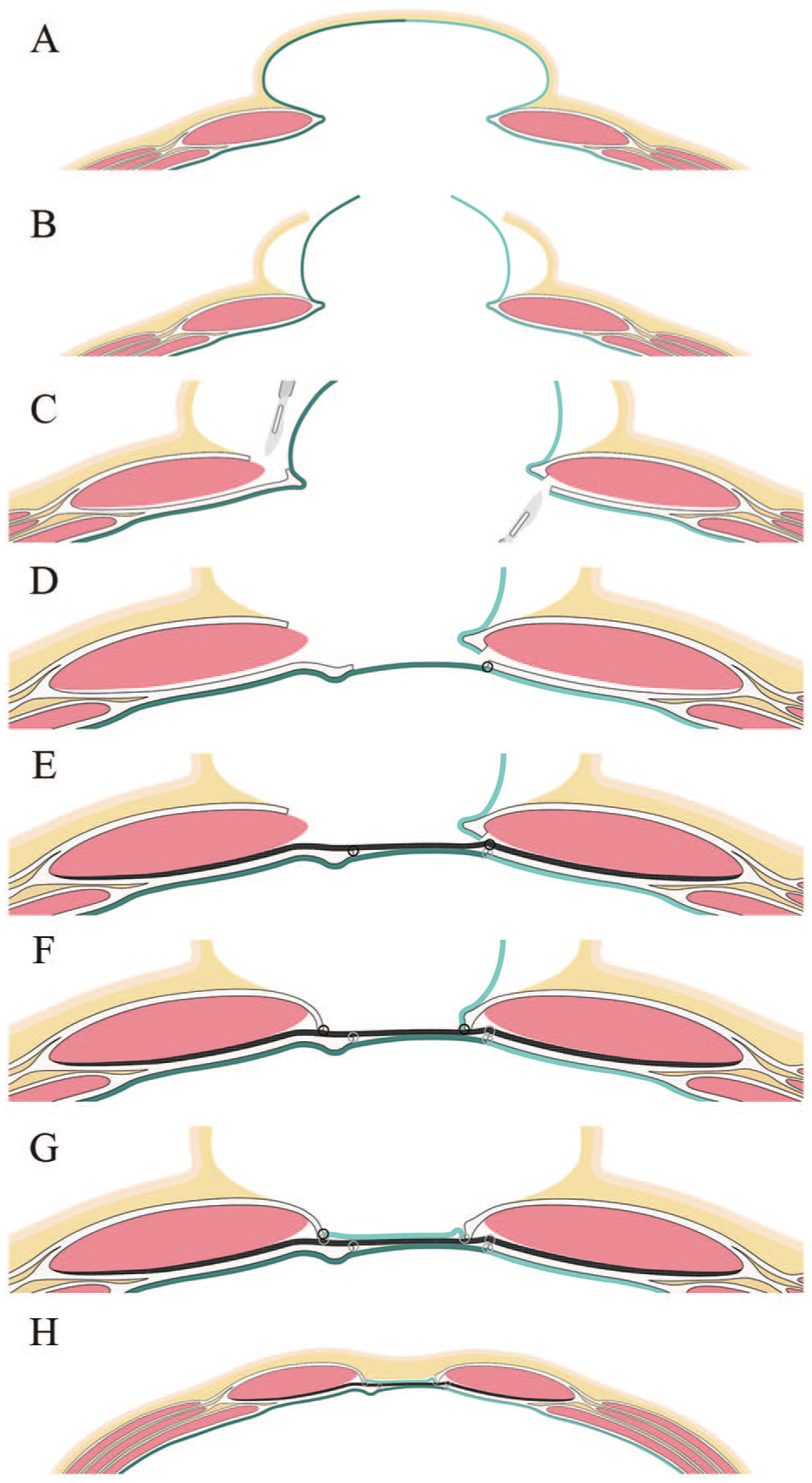
Schematic figure of the steps for the modified peritoneal flap hernioplasty technique (MPFH): (A) the initial incisional hernia (IH); peritoneum and the hernial sac is colored dark green (left side) and light green (right side), respectively; (B) the skin is incised, and the old scar and redundant skin is excised. The hernial sac is isolated and entered in the midline; (C) the retromuscular space is entered through the anterior fascial sheath on the left side, and through the posterior sheath on the right, creating one posterior and one anterior flap; (D) the abdominal cavity is closed by suturing the posterior flap to the posterior rectus fascia on the right side. Before this is made, the rectus muscles are approximated as much as possible; (E) the retromuscular mesh is positioned and fixated by suture to the fascial edge; (F) the anterior fascial edges are sutured to the mesh as close to the midline as possible; (G) the anterior flap is used to cover the mesh and sutured to the fascial edge on the left side; and (H) subcutaneous tissue and skin is closed.
The aim was to describe the MPFH technique and to compare it to the previously used RM technique regarding short-term outcomes, long-term patient satisfaction, abdominal wall complaints, and hernia recurrence. Finally, to evaluate if mesh covered with either fascia or hernia sac was advantageous to un-covered subcutaneously exposed mesh, two other groups were identified. Patients with covered mesh from both the MPFH and the RM groups were compared with patients with un-covered meshes.
Materials and Methods
Study Design
This is a retrospective single-center cohort study of consecutive patients electively operated at Skåne University Hospital in Malmö, Sweden, January 2011–December 2015 for midline IH with a width of ⩾4 cm, corresponding to W2-W3 in the European Hernia Society (EHS) classification for IHs (21). The patients were identified by searching diagnosis- and operation-registers.
MPFH Versus RM Comparison
The study evaluates two RM mesh techniques for IH repair, both without CS. The RM technique had been used for many years at our department. The purpose of the MPFH technique was to enable anterior coverage of the mesh with part of the hernia sac if midline fascia-to-fascia closure was not possible. The MPFH technique was implemented gradually, starting in December 2012 and by October 2014, the RM technique had been phased out and abandoned. The operative technique used for each patient was dependent on whether the surgeon in charge had adopted the new MPFH technique or not. Patients operated with the RM technique between January 2011 and October 2014 or the MPFH technique between December 2012 and December 2015 were included in the study. Surgeons performing the operations were all specialized in hernia surgery. For comparison, the MPFH and RM groups were evaluated for short- and long-term outcome.
Covered Versus Un-Covered Mesh Comparison
The theory was that covered mesh was less prone to complications than un-covered mesh, which was the incitement and hypothesis when developing the MPFH technique. In order to evaluate this, a new grouping of the patients was carried out. A covered mesh is either covered by fascia or by hernia sac, and patients in this group were recruited from both operative techniques (RM and MPFH, where fascia-to-fascia closure of the midline was achieved, or MPFH, where hernia sac was used for complete mesh coverage). Un-covered mesh patients were, with a single exception, recruited from the RM group (RM, where fascia-to-fascia was not possible, and one patient from the MPFH group, where the hernia sac became ischemic and had to be excised).
Perioperative Routines and Surgical Techniques
Non-absorbable polypropylene meshes (Parietene Light, Covidien, Dublin, Ireland or Bard Soft Mesh, C.R. Bard Davol Inc., RI) or polyvinylidene fluoride mesh (PVDF, DynaMesh-CICAT, FEG, Textiltechnik mbH Aachen, Germany) were used. Polypropylene sutures (Prolene 2-0, Ethicon, Somerville, NJ) were used for mesh fixation and fascial suturing. A single-dose antibiotic prophylaxis with cloxacillin (Ekvacillin, Meda AB, Solna, Sweden) was administered. Subcu-taneous drain was optional. If utilized, prolonged prophylaxis with flucloxacillin (Heracillin, Meda AB, Solna, Sweden) was given until the drain was removed. There were no changes in the pre- or perioperative routines during the study period.
MPFH
The technique is demonstrated in Fig. 1. The old scar and redundant skin is excised and the hernia sac is dissected from subcutaneous fat and scar tissue. The hernia sac is entered in the midline and adhesiolysis is performed on the side offering the easiest intra-abdominal access to the posterior rectus fascia and muscle. The RM space is then entered through the posterior rectus fascia on this side and through the anterior rectus fascia as medial as possible on the other side. This way, two flaps of circulated hernia sac are created, one anterior and one posterior. After dissecting the RM space, the abdominal cavity is closed by suturing the posterior fascial edges together when possible. The posterior flap of the hernia sac is used in parts where fascia-to-fascia closure is not possible. The mesh is placed in the RM space and sutured to the closed posterior fascia in the midline or to both sides of the fascial edges in patients where the hernia sac is used for posterior closure. The anterior fascial edges are then sutured together in the midline when possible and if not, the fascial edges are sutured to the mesh as medially as possible. This is done under some tension to minimize the bridged distance. Finally, the anterior peritoneal flap is used to cover the bridging part of the mesh to avoid subcutaneous exposure of the mesh.
RM Mesh Repair
Initial steps are the same as for the MPFH technique. The anterior rectus fascia is freed, the rectus fascia incised anteriorly as medial as possible on both sides, and the RM space dissected bilaterally. The posterior fascial layer is sutured and tightened after either invagination or excision of the hernia sac. The mesh is placed in the RM space and sutured to the closed posterior fascia in the midline. In patients where the anterior fascial edges are not possible to join in the midline, they are sutured to the mesh as medially as possible, leaving a part of the mesh without fascial coverage and thereby exposed to subcutaneous tissue.
Technique Adjustments in Patients With a Stoma
In order to create enough space for adequate 5 cm mesh overlap around stomas, we have utilized a localized PCS and transverse abdominis release lateral to the stoma in these patients. The craniocaudal distance for this dissection is usually 10 cm and is not aimed for medial advancement of the posterior fascia but solely for creating an extension of the RM plane lateral to the stoma.
The Standardized Outpatient Follow-Up
A wound control is scheduled 7–10 days after discharge and performed by a wound-specialized nurse. The evaluation and actions taken are registered in a standardized wound chart. If the first control reveals treatment-demanding complications, further controls are initiated, and the patient is followed until healed. An outpatient visit to the operating surgeon is performed after 3 months. If no treatment-demanding complication is present at this time, no further clinical follow-up is scheduled.
Study Protocol
Chart Review and Swedish Ventral Hernia Register
A chart review was performed, and data retrieved according to Tables 1 to 4 and Supplementary Table 1. Complementary data were retrieved from the prospective national Swedish Ventral Hernia Register. If the patient had been examined with computed tomography (CT)-scan or ultrasound (US) postoperatively, the images were evaluated for a recurrent IH. The hernia and mesh areas were calculated as quadrants using width and length.
Patient demographics.
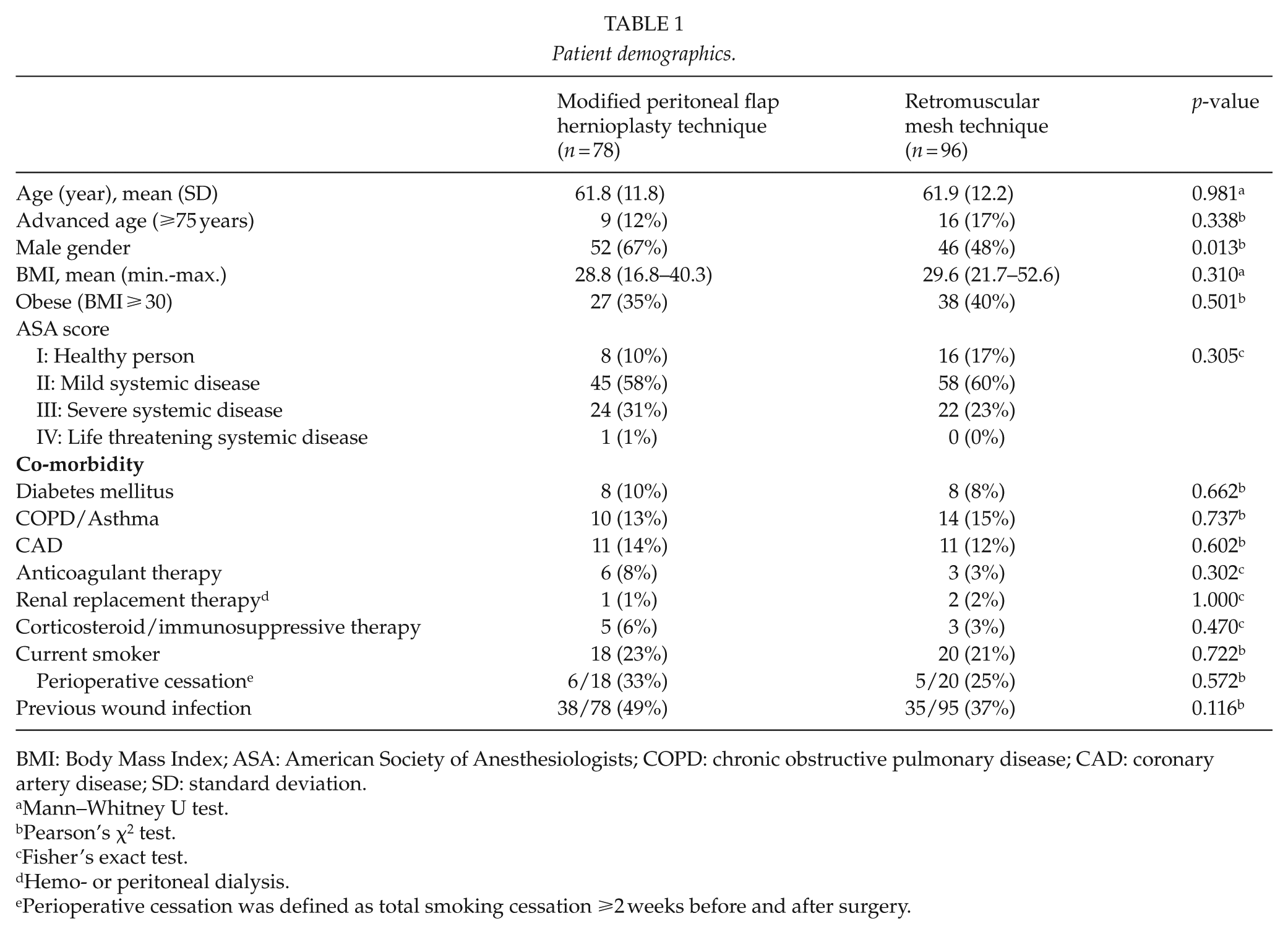
BMI: Body Mass Index; ASA: American Society of Anesthesiologists; COPD: chronic obstructive pulmonary disease; CAD: coronary artery disease; SD: standard deviation.
Mann–Whitney U test.
Pearson’s χ2 test.
Fisher’s exact test.
Hemo- or peritoneal dialysis.
Perioperative cessation was defined as total smoking cessation ⩾2 weeks before and after surgery.
The incisional hernia repair operation characteristics.
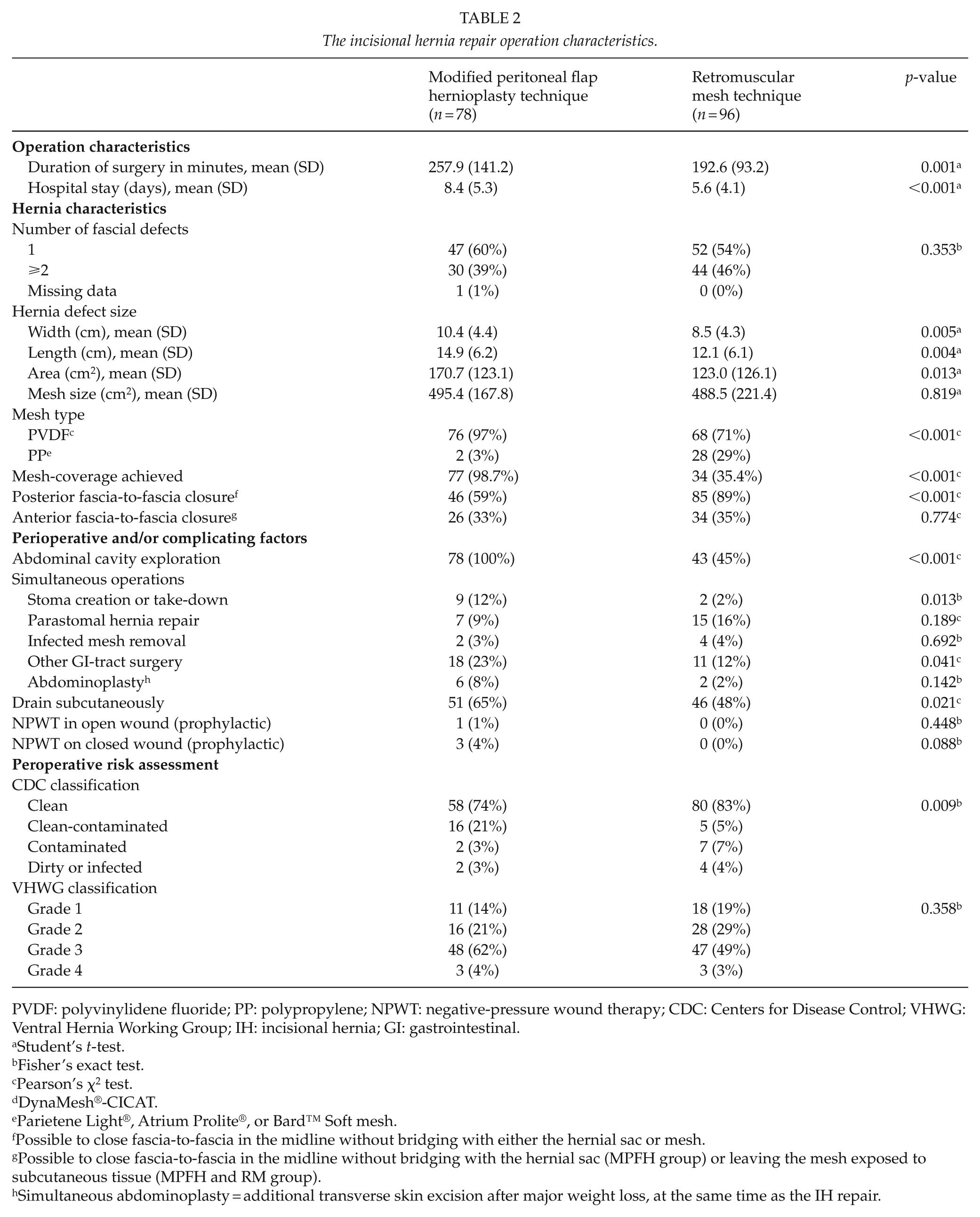
PVDF: polyvinylidene fluoride; PP: polypropylene; NPWT: negative-pressure wound therapy; CDC: Centers for Disease Control; VHWG: Ventral Hernia Working Group; IH: incisional hernia; GI: gastrointestinal.
Student’s t-test.
Fisher’s exact test.
Pearson’s χ2 test.
DynaMesh®-CICAT.
Parietene Light®, Atrium Prolite®, or Bard™ Soft mesh.
Possible to close fascia-to-fascia in the midline without bridging with either the hernial sac or mesh.
Possible to close fascia-to-fascia in the midline without bridging with the hernial sac (MPFH group) or leaving the mesh exposed to subcutaneous tissue (MPFH and RM group).
Simultaneous abdominoplasty = additional transverse skin excision after major weight loss, at the same time as the IH repair.
Postoperative short-term complications, Clavien-Dindo classification, long-term outcome, and selected questions with affirmative answers from the modified Ventral Hernia Pain Questionnaire.
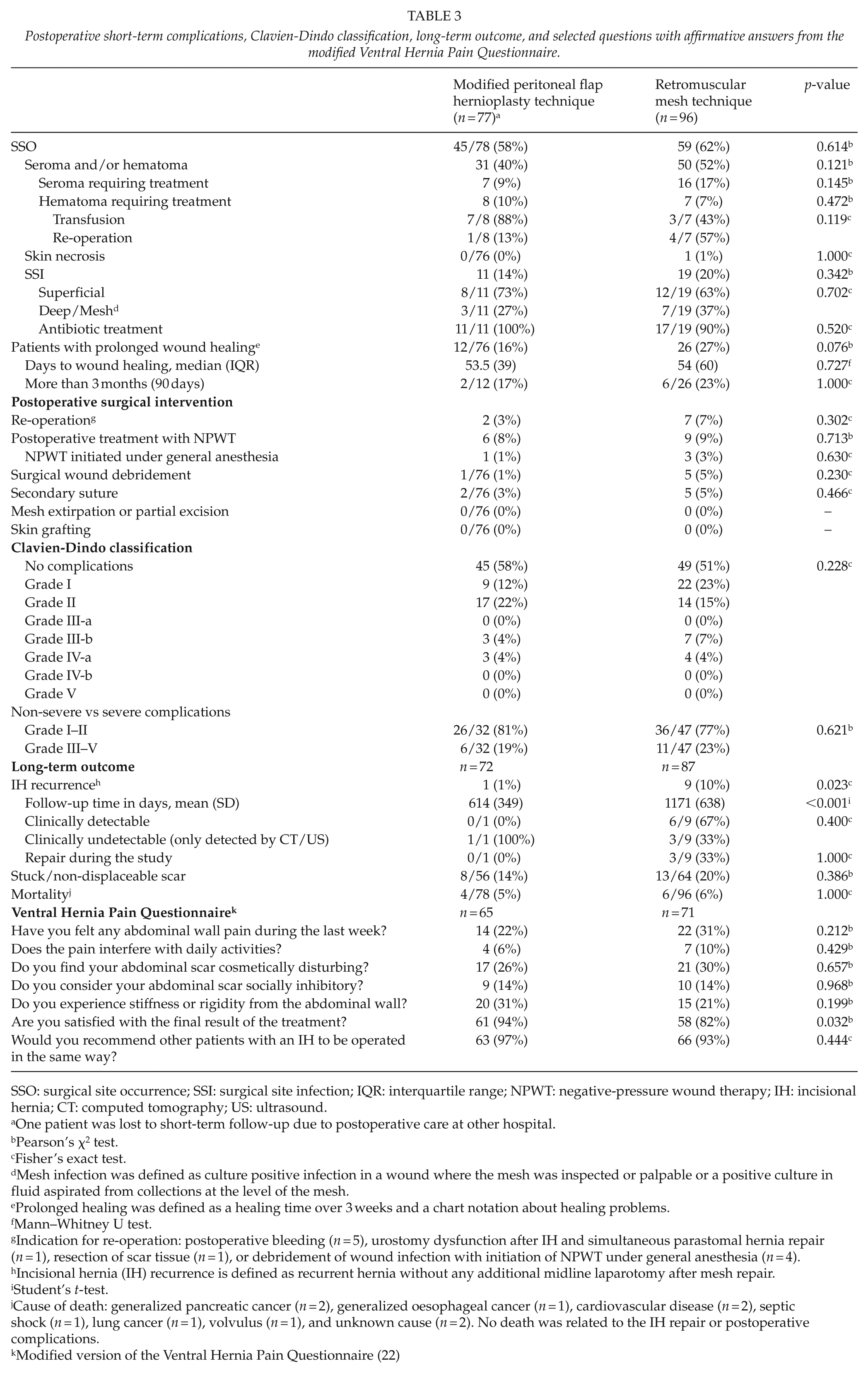
SSO: surgical site occurrence; SSI: surgical site infection; IQR: interquartile range; NPWT: negative-pressure wound therapy; IH: incisional hernia; CT: computed tomography; US: ultrasound.
One patient was lost to short-term follow-up due to postoperative care at other hospital.
Pearson’s χ2 test.
Fisher’s exact test.
Mesh infection was defined as culture positive infection in a wound where the mesh was inspected or palpable or a positive culture in fluid aspirated from collections at the level of the mesh.
Prolonged healing was defined as a healing time over 3 weeks and a chart notation about healing problems.
Mann–Whitney U test.
Indication for re-operation: postoperative bleeding (n = 5), urostomy dysfunction after IH and simultaneous parastomal hernia repair (n = 1), resection of scar tissue (n = 1), or debridement of wound infection with initiation of NPWT under general anesthesia (n = 4).
Incisional hernia (IH) recurrence is defined as recurrent hernia without any additional midline laparotomy after mesh repair.
Student’s t-test.
Cause of death: generalized pancreatic cancer (n = 2), generalized oesophageal cancer (n = 1), cardiovascular disease (n = 2), septic shock (n = 1), lung cancer (n = 1), volvulus (n = 1), and unknown cause (n = 2). No death was related to the IH repair or postoperative complications.
Modified version of the Ventral Hernia Pain Questionnaire (22)
Comparison between patients with covered and un-covered mesh.
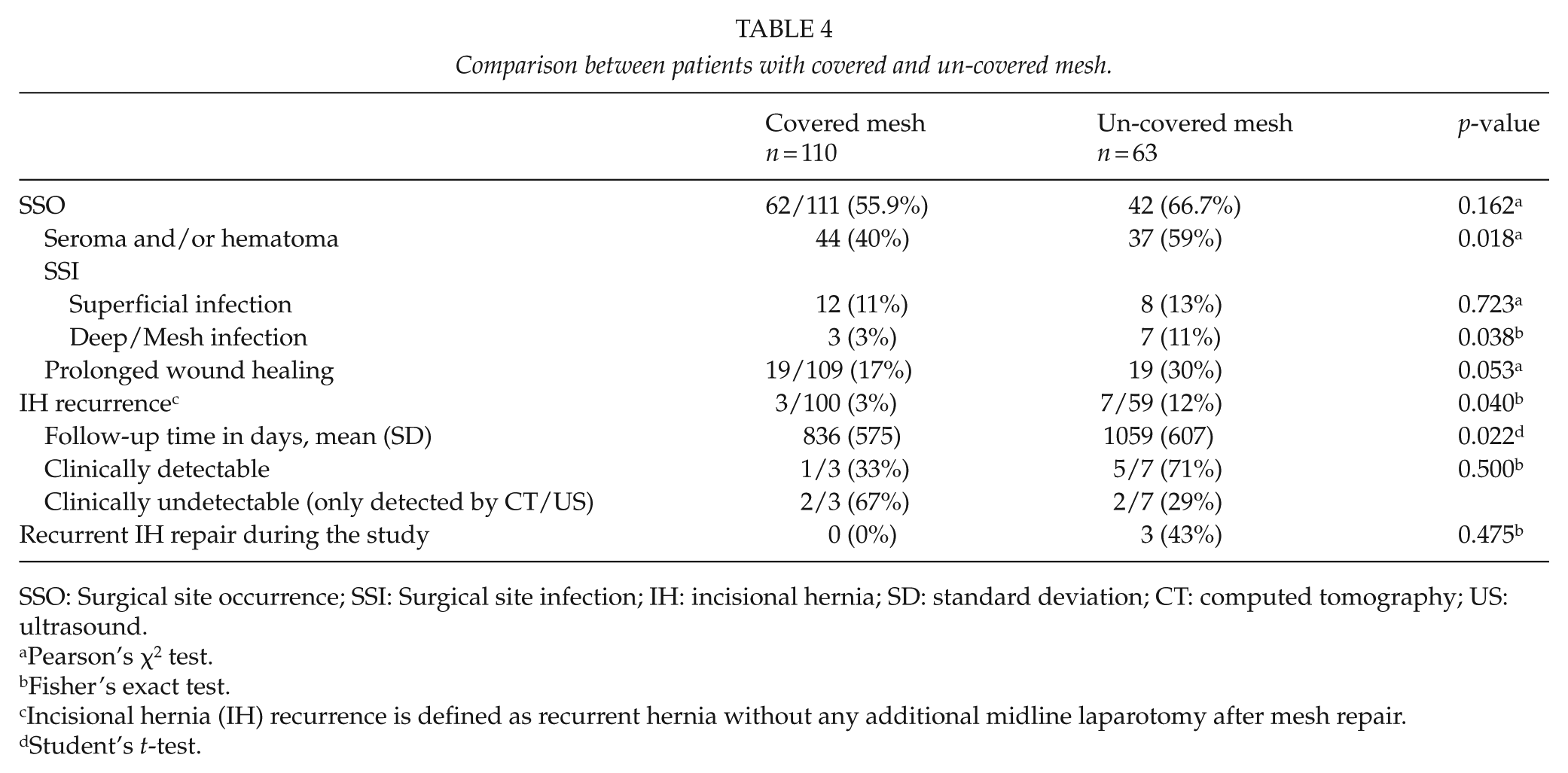
SSO: Surgical site occurrence; SSI: Surgical site infection; IH: incisional hernia; SD: standard deviation; CT: computed tomography; US: ultrasound.
Pearson’s χ2 test.
Fisher’s exact test.
Incisional hernia (IH) recurrence is defined as recurrent hernia without any additional midline laparotomy after mesh repair.
Student’s t-test.
Recurrent IH was defined as “any abdominal wall gap with or without a bulge in the area of a postoperative scar perceptible or palpable by clinical examination or imaging” (21, 23). Follow-up time was defined as the time between operation and the study clinical follow-up, CT/US examination or last chart notation commenting on the presence or absence of an IH. Postoperative complications were categorized as surgical site occurrences (SSO) including seroma/hematoma, skin necrosis, and surgical site infections (SSI) (24, 25). Wound healing was defined as completely healed skin at the surgical site. Prolonged healing was defined as chart notations revealing healing problems and a healing time exceeding 3 weeks.
Perioperative Risk Assessment and Postoperative Classification of Surgical Complications
The risk for surgical site complications was assessed according to the Ventral Hernia Working Group (VHWG) (9) and the Centers for Disease Control (CDC) classification for prevention of surgical wound infections (26). Patients with enterocutaneous fistula were defined as contaminated or dirty/infected.
Postoperative complications were graded and presented according to the Clavien-Dindo (C-D) classification of surgical complications (27). C-D grades I–II were defined as non-severe and III–V as severe complications (28). Initiation of negative-pressure wound therapy (NPWT), aspiration of seroma, and bedside secondary suture was defined as C-D grade I.
Superficial wound infection was defined as local signs of infection with or without wound discharge, fever or systemic signs of inflammation. Deep/Mesh infection was defined as culture positive infection in a wound where the mesh was exposed or being palpable, or a positive culture in fluid aspirated from collections at the level of the mesh. Treatment-requiring postoperative bleeding was defined as needing transfusion or re-operation and treatment-requiring seroma/hematoma needing aspiration or insertion of a drain.
Clinical Long-Term Follow-Up and Patient Reported Outcome
Patients were invited to an outpatient visit in 2017. The visit included physical examination for recurrent IH in upright and supine position during relaxation, straining, and coughing. It was noticed whether the scar was stuck/non-displaceable to the underlying fascia or freely moveable. Patients without hernia recurrence, operated with a new midline laparotomy, were excluded from the clinical long-term follow-up. Follow-up time in these patients was defined as time from the IH-repair to the succeeding operation.
Patients answered a modified version of the validated Ventral Hernia Pain Questionnaire (VHPQ), approved for use in its current form by the inventors (29). The questionnaire contains 24 questions designed to reflect the patient’s current experience of pain, wound healing at follow-up, cosmesis, scar-inflicted social limitations, and if the patient is content with the result. Patients not able to attend the follow-up were asked to answer the questionnaire by telephone or by mail. Exclusion criteria for the questionnaire were dementia and/or patients operated with a new midline incision or for an IH recurrence after the IH repair.
Ethical Approval and Statistics
The study was approved by the Regional Ethics Committee at Lund University, Sweden. Quantitative variables were expressed as mean and standard deviation (SD) or median and interquartile range (IQR). Comparison between groups was calculated with the Student’s t-test, Mann–Whitney U test, Pearson’s χ2 test, or Fisher’s exact test, as appropriate. The p ⩽ 0.05 was considered significant. Statistical analyses were performed using Statistical Package for Social Sciences (SPSS®) version 24 (SPSS, Chicago, IL, USA).
Results
Patient Demographics and Operative Characteristics
A total of 78 patients consecutively operated with the MPFH technique and 96 patients with the RM technique were included. Patient demographics are presented in Table 1. Type of index operation is presented in Supplementary Table 1. A difference with predominance of male patients in the MPFH group was seen (p = 0.013).
Operation characteristics are shown in Table 2. The MPFH patients had larger hernia width (mean 10.4 vs 8.5 cm, p = 0.005); longer operation time (257.9 vs 192.6 min, p = 0.001); and longer hospital stay (8.4 vs 5.6 days, p < 0.001).
The MPFH technique enabled anterior coverage of the mesh in 98.7% compared with 35.4% in the RM group (p < 0.001). Stoma creation or take-down (11.5% vs 2.1%, p = 0.013), other gastrointestinal (GI)-tract surgery (23.1% vs 11.5%, p = 0.041) and higher CDC classification (p = 0.009), was more frequent in the MPFH group. There was a difference in mesh type with more frequent use of PVDF in the MPFH group (97.4% vs 70.8%, p > 0.001). Subcutaneous drainage was used more often in the MPFH group (65.4% vs 47.9%, p = 0.021). The posterior fascia was possible to close without hernia sac-bridging less frequent in the MPFH group (59.0% vs 88.5%, p < 0.001). No difference regarding anterior fascia-to-fascia closure was seen (MPFH 33% vs RM 35%, p = 0.774).
Postoperative Complications and Long-Term Follow-Up
Short- and long-term complications are presented in Table 3. There were no differences between the groups regarding short-term complication and C-D classifications. Hernia recurrence rate was significantly lower in the MPFH group (1.4% vs 10.3%, p = 0.023), and follow-up time was significantly shorter compared with the RM group (614 vs 1171 days, p < 0.001). In total, 104 of the eligible 155 patients (67.1%) attended the clinical long-term follow-up, with no difference between groups (MPFH 67.1% vs RM 67.1%, p = 0.995). The reasons for not attending were: patient living far away (n = 10), declining participation due to co-morbidity (n = 6), declining participation for other reasons (n = 24), and not possible to reach (n = 11). There was no difference between groups regarding mortality.
Patient Reported Outcome
Selected questions with affirmative answers are presented in Table 3. Answers were obtained from 136 of the 156 eligible patients (87.2%), with no difference between the MPFH and RM group (87.8% vs 86.6%, p = 0.815). The reasons for not participating were dementia (n = 1), denying participation (n = 5), and not possible to reach (n = 14).
The patients operated with the MPFH technique were more satisfied with the result (93.8% vs 81.7%, p = 0.032). Excluding patients with IH recurrence did not alter the result for patient satisfaction between groups (p = 0.036). A total of 10% of patients, equally distributed between groups, had experienced pain during the last week that interfered with daily activities while 22% of the MPFH and 31% of the RM patients (p = 0.212) had experienced pain of any kind.
Covered Versus Un-Covered Mesh Comparison
Comparison between patients with covered and un-covered (i.e. subcutaneously exposed) mesh, regardless of operative technique, is presented in Table 4. There was a difference favoring patients with covered mesh regarding seroma/hematoma (40.0% vs 58.7%, p = 0.018); deep/mesh infection (2.7% vs 11.1%, p = 0.038); and IH recurrence (3.0% vs 11.9%, p = 0.040).
Discussion
This retrospective single-center cohort study showed similar incidence of short-term complications in the MPFH group and the earlier used RM technique, despite larger hernias, longer duration of surgery, and more advanced CDC classification and more frequent simultaneous GI-tract surgery in the MPFH group. Significantly lower IH recurrence rate was found after MPFH repair but the shorter follow-up time in this group must be taken into account when interpreting the results. It was possible to cover the mesh with fascia or the anterior peritoneal flap in all but one of the MPFH patients. When grouping patients, from both the MPFH and the RM groups, according to anterior coverage of the mesh or not, fewer seromas, mesh infections, and IH recurrences were found for covered meshes compared with those with un-covered meshes.
The MPFH technique was developed with the intention to reduce wound complications. The aims were to retrospectively evaluate the MPFH technique compared with the former RM technique used for many years, regarding short- and long-term outcomes. There was a difference in patient characteristics with larger hernias and more simultaneous GI-tract surgery in the MPFH group, indicating technically more challenging conditions with a higher risk for complications. This implicates that prerequisites for comparison of groups were not optimal, which might obscure possible differences between groups. Despite the larger hernias in the MPFH group, it was possible to avoid subcutaneous mesh exposure by either fascia-to-fascia closure or anterior flap coverage in all patients but one. In this respect, that is, coverage of the mesh, the new technique was sufficient for most hernia repairs, without the need of CS, also pointed out by Malik et al. (15) for the peritoneal flap hernioplasty.
The theory behind and incitement for developing the MPFH technique was that covered mesh was less prone to complications than un-covered mesh and therefore the primary aim of the study was to evaluate postoperative wound complications. In this respect, we did not find any significant differences between the groups. However, a somewhat lower frequency of prolonged wound healing was found in the MPFH group (p = 0.076). Considering the differences in patient’s characteristics with more complicated hernias, the MPFH technique performs equally or possibly better than the RM technique in relation to postoperative short-term complications. A reasonable explanation is the difference in mesh coverage between groups. The longer hospital stay after MPFH may be explained by the greater complexity in this group. The incidences of treatment demanding SSO and SSI in this study are comparable to reports by others (12, 15, 28).
Other aims with the study were to evaluate long-term IH recurrence and abdominal wall complaints. There was a significantly lower incidence of IH recurrence using the MPFH technique. The shorter follow-up time in the MPFH group might account for part of this difference, but the MPFH group still had a relatively long follow-up time of 614 days and only one recurrence detected. The 10.3% recurrence rate in the RM group was somewhat disappointing but is still in accordance with other reports, while 1.4% noted in the MPFH group is in the lower range of earlier reported frequencies for RM IH repair (5, 8, 11, 12). Equally, low recurrence rates, 2.8% after a mean follow-up of 75 months, were reported recently for 251 peritoneal flap hernioplasties performed in Edinburgh (22). We did not find any significant differences among the possible risk factors for recurrence between the MPFH and the RM group that could explain the higher recurrence rate in the RM group. On the contrary, several possible risk factors, that is, male gender, longer duration of surgery, larger width and length of hernias, more stoma take-down, and other GI-tract surgery and higher CDC classification, were significantly more common in the MPFH group (30).
Patients operated with the MPFH technique were significantly more satisfied with the result of their operation than patients in the RM group, also when patients with recurrences were excluded. Despite not being fully satisfied, more than half of the non-satisfied patients would recommend someone else to undergo the same operation. This implicates that the advantages with surgery outweigh the disadvantages for those patients. Long term pain interfering with daily activities during the last week was reported by almost 10%, a not negligible frequency that patients ought to be informed of preoperatively.
Anterior coverage of the meshes were achieved in all but one of the MPFH patients and in one third of patients in the RM group. A larger proportion of coverage for RM techniques may be achieved by modifying the technique we have used. If the hernia sac is dissected from the fascial border, the peritoneum freed from the posterior fascia and the retrorectus space entered from behind, some anterior fascia will be gained. However, the MPFH technique enables coverage of larger distances.
In order to test the hypothesis that covered mesh perform better, we compared patients with covered and un-covered mesh from both groups and found significantly higher incidences of seroma/hematoma, deep/mesh infection, and hernia recurrence in patients with un-covered meshes. In two thirds of the MPFH patients, the anterior peritoneal flap was used for mesh coverage. Suturing the anterior fascia to the mesh is recommended to be done under some tension to minimize the bridged distance to prevent mesh bulging. Consequently, the mesh will functionally act as part of the abdominal wall. It seems as covered meshes, regardless if fascia or hernia sac is used, perform better both in regard of SSO and IH recurrences (8–12). A RM mesh technique combined with lateral release is another way to achieve mesh coverage.
This retrospective chart review suffers from the disadvantages related to the study design. The somewhat disappointing recurrence rates for the RM technique, the low frequency of fascia covered meshes and the risk for a wound infection to reach the level of the mesh, which was seen in 11% in this group, indicate that there are drawbacks with the technique and room for technical improvement. The results found in this cohort reflects the conditions that applied to this study and is not necessarily generalizable. Due to the lack of protocol-based regulation of the pre-, per-, and postoperative course, differences in management might influence the results. The risk of bias might to some extent be counteracted by the organization at our department where all IH patients are managed by the hernia team according to shared treatment and clinical follow-up routines. The few patients lost to follow-up were equally distributed between the groups, hopefully minimizing distortion of the results.
In conclusion, this retrospective study single-center cohort study showed similar incidences of postoperative short-term complications, despite more complex hernias in the MPFH group compared with the RM technique without CS that was applied at our department earlier. Furthermore, a significantly lower IH recurrence rate was found in the MPFH group, but it is essential to emphasize the shorter follow-up time in this group. We conclude that the MPFH is a technique with encouraging results, especially for repair of larger IHs, and consider it to be a good alternative or adjunct to CS. On these grounds, it has become the standard technique for IH repair at our department.
Supplemental Material
Supplementary_table_1_MPFH_190115 – Supplemental material for Modified Peritoneal Flap Hernioplasty Versus Retromuscular Technique for Incisional Hernia Repair: a Retrospective Cohort Study
Supplemental material, Supplementary_table_1_MPFH_190115 for Modified Peritoneal Flap Hernioplasty Versus Retromuscular Technique for Incisional Hernia Repair: a Retrospective Cohort Study by P. Petersson, A. Montgomery and U. Petersson in Scandinavian Journal of Surgery
Footnotes
Declaration Of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
