Abstract
Objective:
This study sought to investigate the relationship between ABO blood groups and the risk of gastric cancer as well as clinical pathological parameters and prognosis.
Methods:
Gastric cancer patient data were collected from January 1995 to January 2012 at Jilin Cancer Hospital, and the blood group information of the blood donors at Jilin City Blood Center was recorded. The relationships between ABO blood group and both clinicopathological parameters and the risk of gastric cancer were analyzed retrospectively. The impact of ABO blood type on the 5-year survival rate of patients with gastric cancer was evaluated through outpatient and telephone interviews.
Results:
(1) Compared with the healthy population, the frequency distribution of gastric cancer patients with the A blood group was significantly increased (χ2 = 4.708, P = 0.000), whereas the frequency distribution of gastric cancer patients with the AB blood group was significantly decreased (χ2 = 9.630, P = 0.002). However, there was no significant difference in the distributions of the B blood group and O blood group (P > 0.05). (2) The risk of gastric cancer in people with the A blood group was higher, whereas the risk of gastric cancer in people with the AB blood group was lower. There was no significant difference in the risk of gastric cancer between type B and type O patients (P > 0.05). (3) The ABO blood group was not related to pathological factors, including the size of the gastric tumor or the T stage or N stage of the disease (P > 0.05). (4) Univariate analysis results showed that the degree of differentiation, tumor size, T stage, lymph node metastasis, and type O blood were factors affecting the 5-year survival rate of gastric cancer patients (P < 0.05). Multivariate analysis results showed that tumor size, T stage, lymph node metastasis, and O blood group were independent prognostic factors. The 5-year survival rate for gastric cancer was significantly better in patients with type O blood (hazard ratio = 0.97, 95% confidence interval = 1.67–3.92).
Conclusion:
(1) The risk of gastric cancer was higher in patients with the A blood group and lower in those with the AB blood group. (2) The ABO blood group showed no significant effect on the clinicopathological parameters of gastric cancer. (3) The O blood group may be a prognostic factor for gastric cancer patients.
Gastric cancer is one of the most common malignant tumors of the digestive system (1). In China, the rate of detection of early gastric cancer is low, and many patients are in the advanced stage of disease at the initial visit and have thus passed the best time for treatment (2). Therefore, the overall prognosis of patients with gastric cancer is poor. Understanding of the risk factors associated with the development and progression of gastric cancer may lead to the development of better preventive measures and more rational treatment options for patients. The occurrence and development of gastric cancer are caused by the interaction of genetic factors and environmental factors (3). ABO blood grouping is one of the most stable genetic factors, and it was the first human blood group system discovered, as reported by Karl Landsteiner in 1990 (4). ABO blood typing is a routine preoperative examination for patients. Many researchers have explored the association between this blood group system and malignancies (5). For example, Lennon et al. (6) demonstrated that the risk of pancreatic cancer is associated with genetic changes in the ABO blood group genes. Gates et al. (7) studied clinical data from 49,153 women and found that ABO blood type was associated with the incidence of epithelial ovarian cancer. This large prospective study concluded that the presence of the B antigen was positively associated with the incidence of ovarian cancer, whereas individuals with type A blood were not at risk. Therefore, the association between ABO blood group antigens and malignancy has been demonstrated. However, firm conclusions regarding the relationship between the ABO blood groups and gastric cancer cannot be made, as the findings of different research centers are conflicting. For example, certain researchers have proposed that differences in ABO blood group have an impact on the prognosis of patients with gastric cancer, with certain blood groups showing a protective effect on the prognosis of such patients (8). However, other studies have concluded that ABO blood type has no effect on the prognosis of patients with gastric cancer (9). Therefore, the purpose of the present study was to explore the relationship between the ABO blood groups and the clinical pathological parameters of patients with gastric cancer as well as the incidence and prognosis of the disease, with the goals of providing both additional reference indicators for the clinical prognosis of patients and possible clues for further study of the pathogenesis of gastric cancer.
Materials and Methods
Study Population
From January 1995 to January 2012, pathological data for patients with gastric cancer treated at Jilin Cancer Hospital were collected. The inclusion criteria were as follows: (1) gastric adenocarcinoma treated with underwent subtotal or total gastrectomy with full D2 lymphadenectomy, (2) no preoperative radiotherapy or neoadjuvant chemotherapy, (3) pathological specimens confirmed by pathological diagnosis, and (4) being diagnosed with gastric cancer for the first time. The exclusion criteria were as follows: (1) other malignancies or a history of malignancy, and (2) an Rh-negative blood type. According to the above criteria, a total of 897 patients with gastric cancer were enrolled, including 613 males and 284 females, with a male-to-female ratio of 2.1:1.0 and an age range of 29–96 years (median age, 54 years).
Data Collection
The blood group information of 7880 healthy individuals was collected during the physical examination of patients at Jilin Cancer Hospital and from donors at the Jilin City Blood Center. Patients with ABO blood type information were identified by the hospital blood transfusion section or laboratory. The clinicopathological data included age, sex, tumor site, tumor size, tumor differentiation, T staging, lymph node metastasis, TNM stage, serum carcinoembryonic antigen (CEA) level, and serum CA19-9 level.
Statistical Analysis
Statistical analysis was carried out using SPSS20.0. Chi-square tests were used to compare categorical variables. The predictors of overall survival tested by univariate analysis were assessed by multivariate analysis using the Cox proportional hazards model. A P value <0.05 was considered statistically significant.
Results
Blood Type Distribution between Patients with Gastric Cancer and Healthy Subjects
Of the 897 patients with gastric cancer, 311 patients had type A blood (34.7%), 216 patients had type B blood (24.1%), 280 patients had type O blood (31.2%), and 90 patients had type AB blood (10%). Among the 7880 healthy subjects, there were 2348 cases of type A blood (29.8%), 1993 cases of type B blood (25.3%), 2411 cases of type O blood (30.6%), and 1128 cases of type AB blood (14.3%). Compared with the number in the healthy population, the number of gastric cancer patients with type A blood was significantly higher (χ2 = 4.708, P = 0.000), whereas the AB blood type distribution frequency among the gastric cancer patients was significantly lower (χ2 = 9.630, P = 0.002). However, there was no significant difference between the distributions of the B blood type and the O blood type (P > 0.05). Regarding the risk of gastric cancer, blood type A increased the risk in the population (odds ratio (OR) = 1.211, 95% confidence interval (CI) = 1.059–1.546), whereas people with blood type AB showed a lower risk of gastric cancer (OR = 0.846, 95% CI = 1.202–1.451). There was no significant effect of type B or type O blood on the risk of gastric cancer (P > 0.05) (Table 1).
Distributions of the ABO blood groups in patients with gastric cancer and a healthy population.
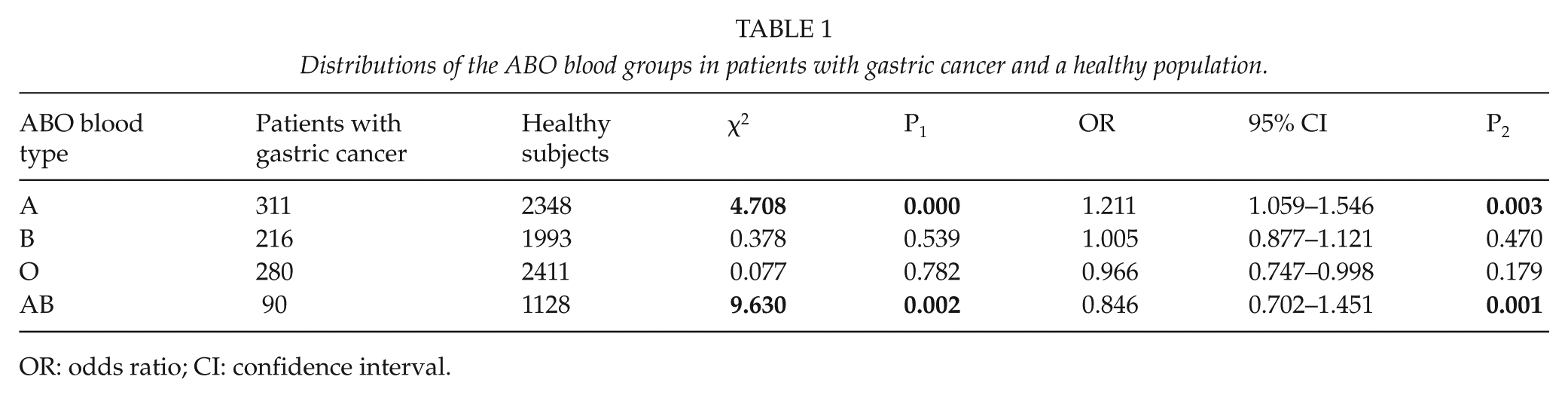
OR: odds ratio; CI: confidence interval.
Association between ABO Blood Groups and The Clinicopathological Characteristics of Gastric Cancer Patients
The results showed that for all clinical parameters examined in this study, including age, gender, tumor location, tumor size, degree of differentiation, T staging, lymph node metastasis, serum CEA, and serum CA19-9, the ABO blood group distribution showed no statistically significant association.
Association between The Clinical Prognosis of Gastric Cancer Patients and ABO Blood Groups
The median follow-up period of the 897 gastric cancer patients was 94 months, with 487 (54.3%) alive and 410 (45.7%) deceased at the final clinical follow-up. Univariate analysis showed that the degree of differentiation, tumor size, T stage, lymph node metastasis, and type O blood were factors influencing the 5-year survival rate of gastric cancer patients (P < 0.05). Moreover, the significant parameters in the univariate analysis were further analyzed in a multivariate analysis. The results showed that tumor size, T stage, lymph node metastasis, and type O blood were independent prognostic factors (Table 2).
Relationship between ABO blood type and the 5-year survival rate in patients with gastric cancer.
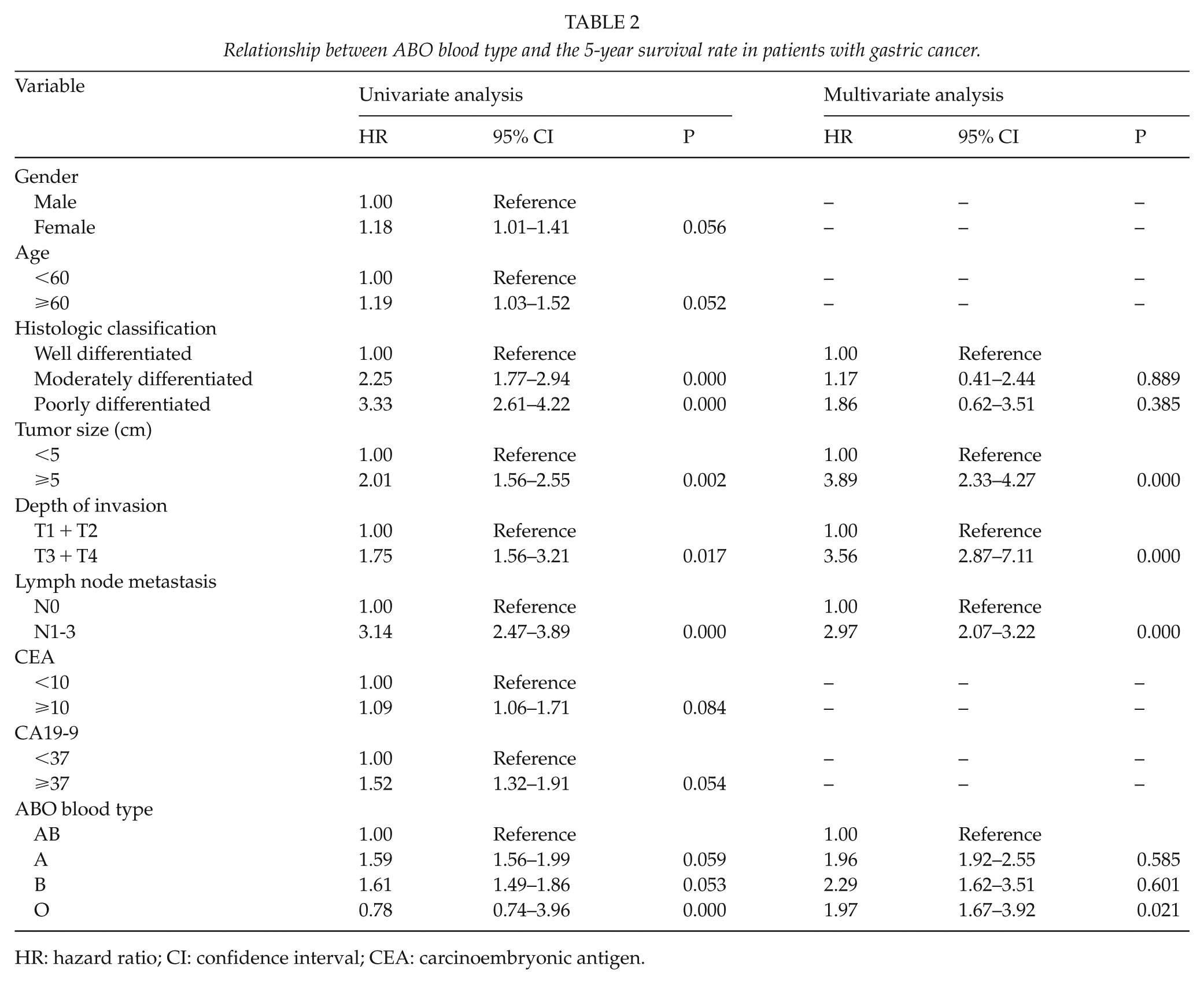
HR: hazard ratio; CI: confidence interval; CEA: carcinoembryonic antigen.
Discussion
Over the past half century, the incidence and mortality of gastric cancer have decreased continuously worldwide, particularly in developed countries (10). However, Roder (11) found that China, Japan, Eastern Europe, and parts of the Middle East showed a higher incidence of gastric cancer relative to Northern Europe, North America, Australia, India, and New Zealand. In particular, over the past 20 years, southern Australia’s gastric cancer mortality rates have decreased by approximately 40%. The reason for this decrease may be that with the development of the economy, the standard of living has significantly improved. For instance, people now eat more vegetables and fruit and lower amounts of pickled and smoked foods (4). However, the role of genetic factors in the development of gastric cancer cannot be ignored. Many previous studies have confirmed the correlation between ABO blood groups and the occurrence of malignant tumors (12, 13). However, in studying the relationship between ABO blood groups and gastric cancer patients, many diametrically opposed conclusions have been reported by different centers. Therefore, our study sought to further clarify the relationship between ABO blood types and gastric cancer.
A total of 897 patients who underwent gastrectomy were included in this study. We first established a risk prediction model for the ABO types and gastric cancer and then analyzed the relationship between blood type and the risk of gastric cancer. The results showed that there were 311 (34.7%), 216 (24.1%), 897 (24.1%), 280 (31.2%), and 90 (10%) patients with A, B, O, and AB type blood, respectively. Of 7880 healthy subjects, there were 2348 cases (29.8%), 1993 cases (25.3%), 2411 cases (30.6%), and 1128 cases (14.3%) of A, B, O, and AB type blood, respectively. Compared with the frequencies in the healthy group, the frequency of gastric cancer patients with type A blood was significantly higher (χ2 = 4.708, P = 0.000), whereas the frequency of patients with the AB blood type was significantly lower (χ2 = 9.630, P = 1.146). However, there was no significant difference between the distributions of patients with B type blood and O type blood (P > 0.05).
The risk of developing gastric cancer was significantly higher in patients with type A blood than in patients with other blood types, suggesting that type A blood is likely to be a factor in the pathogenesis of gastric cancer. We hypothesize that this finding may be related to higher levels of substance A in the saliva among type A blood subjects. Substance A in the saliva and gastric juices can act on the gastric mucosa, leading to an increased likelihood of a malignant change in the gastric mucosa. According to this hypothesis, people with the B blood type and O blood type should have a lower gastric cancer risk, although the results do not support this theory. In our study, blood types B and O showed no significant difference in the risk of gastric cancer (P > 0.05). Meanwhile, the risk of developing gastric cancer was lower in people with type AB blood (OR = 0.846, 95% CI = 1.202–1.451) than in people with other blood types, suggesting that the AB blood type may be a protective factor in the development of gastric cancer. This finding suggests that in addition to substance A, there are other potential mechanisms that affect the development of gastric cancer through the blood group system.
Univariate analysis showed that the distribution of the ABO blood groups was not significantly affected by the patient’s age, gender, tumor location, tumor size, degree of tumor differentiation, T staging, lymph node metastasis, serum CEA level, or serum CA19-9 level (P > 0.05). This finding is consistent with most other researchers’ conclusions. However, certain scholars have noted that as an important cell receptor or signaling substance, ABO antigens may play a critical role in the invasion and metastasis of malignant tumors. However, according to a large sample of studies, although the ABO blood groups show a clear effect on the biological behavior of gastric cancer, this effect is not dominant and therefore is not easily observed or is nearly negligible. The role of the ABO blood groups in the biological behavior of gastric cancer thus remains to be further studied.
The relationship between the ABO blood groups and the prognosis of patients with gastric cancer remains controversial. In the present study, the degree of differentiation, tumor size, T stage, and lymph node metastasis affected the prognosis of gastric cancer patients, such that a high degree of differentiation, a larger tumor size, worse T staging, and lymph node metastasis were associated with a poor prognosis. The prognosis of patients with type O blood was significantly better than that of patients with other blood types (hazard ratio (HR) = 0.78, 95% CI = 0.74–3.96). Multivariate analysis confirmed that tumor size, T stage, lymph node metastasis, and type O blood were independent prognostic factors. Of particular note is that type O blood showed a significant protective effect on the prognosis of patients.
Sammuel et al. concluded that type O patients with renal cell carcinoma had higher overall survival than non-type O patients, whereas Michela et al. found that ABO blood type was not associated with prognosis in patients with renal cell carcinoma. Cao et al. (14) designed a study with a large sample size and performed a retrospective analysis with a long follow-up period, and the results showed that the AB blood group was an independent and favorable prognostic factor for colon cancer patients; this finding is similar to our results. However, there has been little research on the relationship between ABO blood type and prognosis in gastric cancer patients. In one study, Qiu and co-authors showed that blood type cannot be used as a prognostic indicator in patients with gastric cancer. Currently, the posited relationship between ABO blood type and gastric cancer prognosis is thus based on a retrospective study, suggesting a lack of experimental support. However, many previous investigations have employed large sample sizes, and although the conclusions are not consistent, most studies have suggested that ABO blood group has an impact on the prognosis of patients with malignancies, which warrants further investigation.
In summary, the present study revealed a relationship between ABO blood type and the risk and prognosis of gastric cancer. In particular, type A blood patients are more likely to develop gastric cancer, and the prognosis is relatively good for patients with type O blood. However, the biological mechanism is worthy of further investigation, which may provide new information that can explain the biological behavior, incidence and prognosis of malignant tumors. In particular, whether Helicobacter pylori and other key factors are related to the ABO blood group in the occurrence of gastric cancer are also worthy of further study.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
