Abstract
Background and Aims:
Endocrine surgeons and pathologists alike are well aware of the diagnostic predicament that follicular thyroid tumors impose in the clinical setting, best exemplified by the current inability to preoperatively assess the malignant potential of each individual lesion. As the proper recognition of a follicular thyroid carcinoma lies in the histopathological identification of invasive behavior, preoperative cytology alone is not yet sufficient to identify malignant tumors eligible for a total thyroidectomy upfront. Numerous auxiliary markers have been proposed as discriminating markers between follicular thyroid carcinomas and follicular thyroid adenomas, although many have proven suboptimal in terms of sensitivity, specificity, or overall clinical practicality. Of late, recurrent promoter mutations in the telomerase reverse transcriptase gene have been intimately coupled to subsets of well-differentiated thyroid cancer specimen with aggressive clinical characteristics as well as less differentiated forms of thyroid cancer with exceedingly poor prognosis. The mutations are thought to enhance the telomerase reverse transcriptase gene expressional output and cause immortalization through telomerase-associated mechanisms.
Materials and Methods:
In this review, the current value of telomerase reverse transcriptase promoter mutations is detailed from a clinical angle—as well as the possible future application of additional telomerase reverse transcriptase gene aberrations as adjunct markers for the proper recognition of malignant potential.
Results:
Telomerase reverse transcriptase promoter mutations are found in subsets of follicular thyroid carcinomas and follicular tumors of uncertain malignant potential while exceedingly rare in recurrence-free follicular thyroid adenomas. Collectively, these aberrancies are suggested as possible diagnostic and prognostic discriminators of follicular thyroid tumors.
Conclusions:
Telomerase reverse transcriptase gene analyses greatly facilitate the clinical assessment of follicular thyroid tumors, and pinpoints cases at risk of future recurrences. High-volume, tertiary thyroid centers are therefore recommended to implement the mutational screening in clinical routine.
Introduction
A follicular thyroid tumor is the most commonly encountered primary neoplasia in the clinical setting (1). Each year, over 100 cases are surgically removed at the Karolinska University Hospital—to date the largest tertiary center for thyroid cancer in the Nordic region. The diagnosis is regularly put forward following a fine-needle aspiration biopsy (FNAB), in which micro-follicular patterned thyrocyte aggregates are detected in routine stainings. However, the distinction between follicular thyroid adenoma (FTA) and follicular thyroid carcinoma (FTC) is not possible to establish using conventional light microscopy alone—prompting most patients to surgery for a ipsilateral lobectomy and a definite diagnosis following histopathological examination. The pathologist demand unequivocal evidence of malignancy to put forward an FTC diagnosis, demonstrated by capsular and vascular invasion, criteria that in turn demands attentiveness to macroscopic findings already at the gross examination stage (1). If invasive properties are not seen, the tumor is assumed to be benign, that is, an FTA. However, in a few tumors, the tumor cells display an uncertain relationship to blood vessels and/or the capsule (e.g. tumor cells within, but not through the capsule) suggestive of malignant properties. This tumor type is classified as a “follicular tumor of uncertain malignant potential” (FT-UMP). In Sweden, patients diagnosed with FT-UMPs are subject to follow-up as opposed to being declared disease-free, but the degree of follow-up is debatable.
A plethora of histological and expressional markers have been suggested to aid in the identification of FTCs and predict patient outcome for specific case series, but many of these display suboptimal sensitivity/specificity, others are not sufficiently reproduced or simply just too experimentally cumbersome and expensive for reflex testing and therefore not yet standardized in clinical routine (2, 3). Although there still is a considerable lack of well-defined molecular markers used in the daily practice of thyroid cancer patients, one inexpensive, cheap, and easy-to-interpret candidate method showing great promise as a combined diagnostic and prognostic marker is telomerase reverse transcriptase (TERT) gene promoter mutational testing of tumor DNA from formalin-fixated paraffin-embedded (FFPE) material.
Tert Aberrancies in Cancer—A Brief Functional and Mechanistic Overview
A key hallmark of malignant tumors is augmented proliferation, and thyroid cancer is no exception to this rule. To counteract the successive shortening of vital, chromosomal material that follows excessive replication, eukaryotic chromosomes are capped with telomeres—repetitive, non-coding sequences at the chromosomal ends. These telomeres in turn are maintained by telomerase, an enzyme partly built up by both RNA and protein components, the latter encoded by TERT (4). In human cancer, increased TERT expression and telomerase activity have been demonstrated as recurrent events, leading to telomerase activation, bypassed senescence, and subsequent immortalization (4). Even so, the underlying reason for the TERT up-regulation has been obscure. In recent years, two recurrent point mutations of the TERT promoter (C228T and C250T) were shown in sporadic and familial forms of malignant melanoma and have since been found in various tumor types, including subsets of thyroid cancer (5–9). The mutations are thought to confer increased TERT expression thought altered regulation of the promoter region, and are therefore believed to confer a selective advantage in tumors with marked proliferation. In thyroid cancer, TERT promoter mutations are tightly coupled to more advanced forms of the disease such as poorly differentiated thyroid carcinoma (PDTC) and anaplastic thyroid carcinoma (ATC), as well as in well-differentiated cases of papillary thyroid carcinoma (PTC) and FTC with adverse clinical parameters, such as older age at surgery, extrathyroidal extension, lymph node/distant metastases, and recurrences (7–14). The C228T mutation is the most commonly encountered alteration in thyroid cancer, occurring in almost 9 out of 10 cases with a TERT promoter mutation (15). The reason for this selectivity is not known, but could in theory stem from transcription-factor specific expressional patterns showing a predilection for the C228 position in thyroid cancer. Other TERT promoter mutations besides C228T and C250T are very uncommonly found, as are coding mutations in TERT exons (16).
In addition to the promoter mutations described above, other TERT gene aberrancies have since been discovered as recurrent events in human cancer. Specifically, copy number gain of the TERT gene locus at chromosome 5p15 as well as aberrant methylation of two different regions within the TERT promoter have been shown to correlate with altered TERT gene expression, and these events have been demonstrated for various cancers, including malignant thyroid tumors (Fig. 1) (17–21). High levels of methylation (compared to normal tissues) at positions ‒578 to ‒541 base pairs (bp) of the TERT promoter (entitled “region A”) has been recognized as a recurrent event in medullary thyroid carcinoma (MTC), and this phenomenon correlated with poorer patient outcome (18). In addition, low methylation levels within a promoter region located ‒162 to ‒100 bp of the transcriptional start site (entitled “region B”) was recently demonstrated as necessary for TERT expression in vitro—although such a methylation profile has yet to be proven in thyroid cancer (Fig. 1 and Table 1) (17).
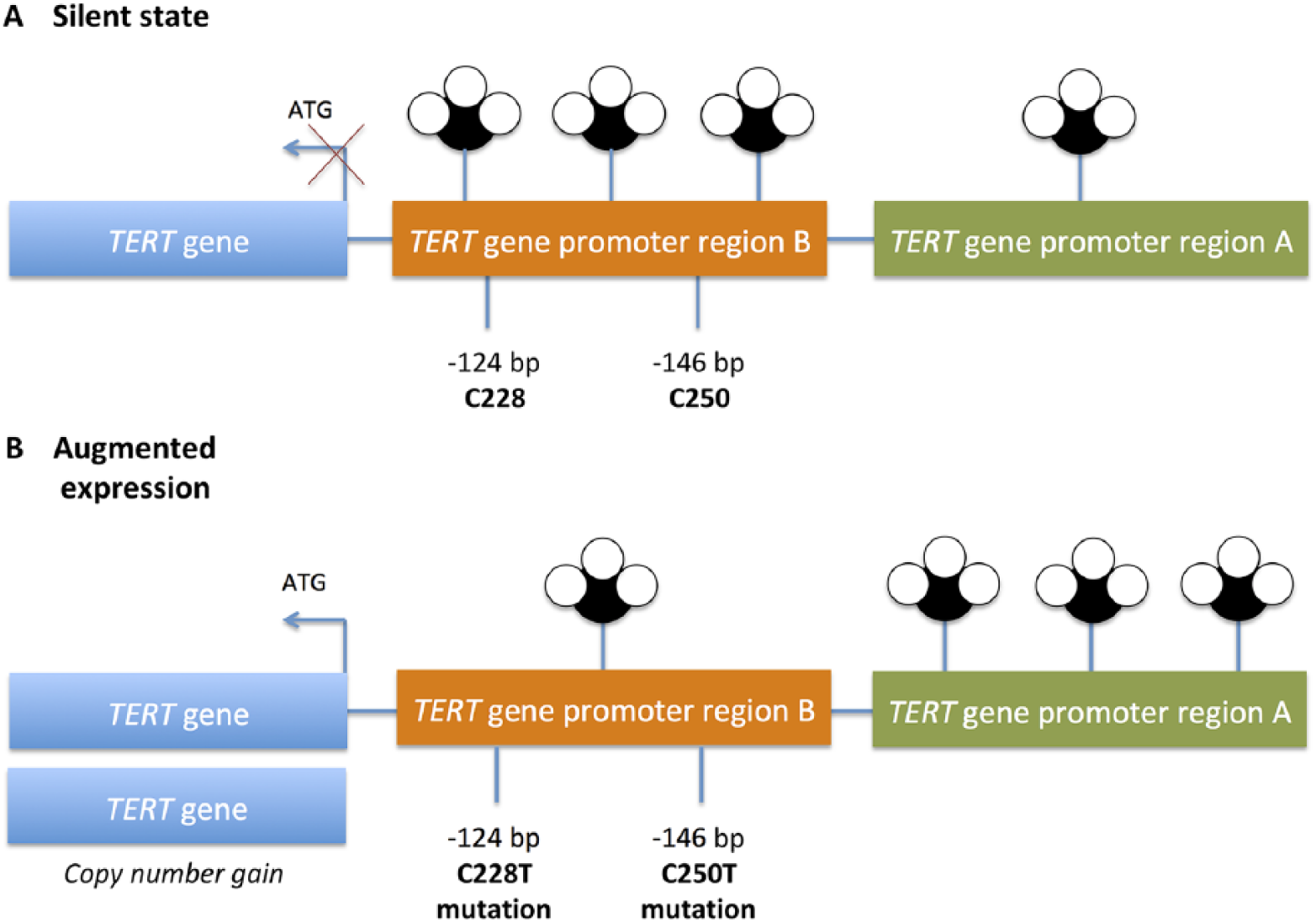
Schematic overview of the TERT gene regulation and various TERT gene aberrancies. (A) Theoretical normal state in a non-neoplastic thyrocyte, depicting absence of TERT gene expression. The promoter region is devoid of promoter mutations at positions C228 and C250 (at positions 124 and 146 base pairs upstream of the transcriptional start site) and the promoter region A is hypomethylated while region B is hypermethylated. (B) TERT gene expression is initiated through one or several of the following TERT gene aberrancies: TERT copy number gain (additional gene copy(ies)); TERT promoter mutations C228T and C250T; and promoter hypermethylation of region A. Hypomethylation of region B has yet to be demonstrated in thyroid tumors.
Tert gene aberrancies in follicular thyroid tumors.

TERT: telomerase reverse transcriptase; FTA: follicular thyroid adenoma; FT-UMP: follicular tumor of uncertain malignant potential; FTC: follicular thyroid cancer.
+++ = Very common (>50%); ++ = Common (25%–50%); + = Observed (5%–25%); – = Not commonly observed (<5%); and ? = Not investigated.
Tert Gene Aberrancies as a Diagnostic Categorizer for Follicular Thyroid Tumors
In a recent study, Paulsson et al. (19) investigated a large series of follicular thyroid tumors for TERT expression, copy number, promoter mutations as well as promoter methylation levels in region A, and the findings were then coupled to clinical parameters and patient outcome. Around 40% of FTCs and FT-UMPs displayed TERT gene expression, in contrast to a small fraction (14%) of FTAs. The majority of FTCs and FT-UMPs carried at least one TERT gene aberration, and TERT-expressing cases almost always exhibited either region A promoter hypermethylation, promoter mutations, or copy number gain of the TERT gene locus. The survival analysis for FTC patients showed a shorter time to relapse in cases with any TERT aberrancy, and an increased risk for relapse was observed in FTCs with TERT gene expression, TERT copy number gain, or TERT promoter methylation index > 18% after adjusting for covariates. Interestingly, the only FT-UMP in the cohort that later recurred as a full-blown FTC displayed TERT expression, region A promoter hypermethylation as well as TERT copy number gain, providing further evidence that a clinical screening for TERT aberrancies could detect small subsets of FT-UMPs which are probably misclassified FTCs (11, 19, 22).
For follicular thyroid tumors, TERT promoter mutations alone display high specificity toward FTC and, however, show a reduced sensitivity in the sense that only 10%–20% of FTCs display these mutations (Table 1) (15, 19). Therefore, the positive predictive value of a mutation is close to 100%, whereas the negative predictive value is too low to make a balanced conclusion in terms of diagnosis. At our department, clinical routine TERT promoter mutational screening was implemented for FT-UMPs in 2014. The finding of a mutation in tumor DNA extracted from FFPE material leads to more vigilant follow-up and sometimes also the amendment of adjuvant treatment modalities, as these tumors are considered to display malignant potential although not yet demonstrable through conventional light microscopy. The outcome of this algorithm will be fully evaluated in the coming years. Testing for additional TERT gene aberrancies besides promoter mutations has not yet been routinely performed at our unit, partly due to the scarce amount of studies covering these alterations in follicular thyroid cancer and partly because the methodology is less straightforward than routine Sanger sequencing—but could indeed be considered if fully reproduced in an independent material.
Tert Gene Aberrancies as Prognostic Markers for Follicular Thyroid Cancer
Besides the diagnostic value of assaying TERT in follicular thyroid tumors, there are numerous studies conducted that also pinpoint the prognostic value in thyroid cancer (8–10, 12, 13, 15, 23). This is perhaps best illustrated by the exceedingly high frequencies of TERT promoter mutations in advanced forms of thyroid cancer such as ATCs, with series reporting this mutation in up to 95% of tumor specimen investigated (24). In well-differentiated thyroid cancer, the bulk of data stems from PTCs, in which the finding of a TERT promoter mutation predicts worse clinical outcome—which is also true for FTCs with this genomic alteration. Indeed, in the study by Paulsson et al. (19), TERT promoter mutational screening was a strong predictor of subsequent tumor recurrences in FTCs—with a negative predictive value of 81%, suggesting that the main bulk of FTCs without TERT promoter mutations seldom recur.
Current Dilemmas of Clinical Screening for Tert Aberrancies
In the postoperative setting, molecular testing of TERT promoter mutations is currently based on the acquisition of tumor DNA from FFPE material, in which the pathologist identifies a representative block for cutting. For conventional Sanger sequencing, a cut-off of at least 10% tumor cells is usually recommended. There are no consensus guidelines as to which area of the tumor should be sampled, or if a genetic heterogeneity exists with mutation-positive and mutation-negative clones in the same follicular tumor. Indeed, TERT promoter mutations have been shown to display regional heterogeneity in meningiomas (25). If the same is true for subsets of follicular thyroid tumors, then the sampling technique becomes a factor to consider also for the endocrine pathologist—and a multi-regional sampling approach might be a reasonable way of increasing the sensitivity in these cases. Future studies will reveal whether or not regional TERT aberrancy disparities play a role in thyroid cancer.
As the presence of TERT gene expression is tightly coupled to underlying TERT gene aberrancies in follicular thyroid tumors, the question arises whether the visualization of the TERT protein through conventional immunohistochemistry (IHC) might be a cheap and effective alternative to genetic testing. However, studies conducted suggest an unexpected cytoplasmic expression rather than the anticipated nuclear localization, as well as little or no correlation between TERT mRNA expression levels and TERT immunoreactivity (26, 27). These studies, although few, suggest that there is yet no reliable antibody available with data supporting that IHC could replace genetic screening in the clinical setting. In this aspect, an alternative approach detecting TERT mRNA using in situ hybridization should not be overlooked (28).
Discussion
When diagnosing thyroid lesions in clinical routine practice, the main benefit of employing TERT promoter mutational screening is mostly attributable to the high specificity in detecting poor-prognosis cases at risk of disseminated disease. Several studies have shown that the presence of such a mutation in thyroid cancer predicts worse patient outcome, as this genetic event is tightly coupled to the development of distant metastases and shorter disease-specific survival (8, 9, 11, 12, 19, 23, 24). In this context, TERT promoter mutations could be considered a valuable prognostic marker when assessing differentiated forms of thyroid cancer (PTC and FTC)—as these mutations are overrepresented among cases that are radioiodine refractive and subsequently metastatic. For more advanced forms of the disease (PDTC and ATC), TERT promoter mutations are much more common, but given the apparent dismal prognosis irrespective of mutational status, the current value of this methodology is debated. From a diagnostic standpoint, the key strength of the methodology lies in diagnosing follicular thyroid tumors at risk of disseminated disease.
The main aim of a clinical marker is to introduce a novel method that changes how we diagnose, prognosticate, or treat our patients. The obvious end-point should therefore be to investigate if screenings for TERT gene aberrancies could identify FTCs already pre-operatively. Indeed, several studies have investigated indeterminate thyroid nodules using TERT promoter mutational screening of fine needle aspiration biopsies, and the results indicate that the marker could be of value for identification of poor-prognosis cases (23, 29). However, no focused study investigating the value of this marker in relation to the distinction between FTA and FTC/FT-UMP pre-operatively exists yet. A proposed model of the implementation of TERT gene aberrancies in the clinical workup of follicular thyroid tumors is presented in Fig. 2. This model is based on the diagnostic “rule in” properties of TERT gene aberrancies to identify malignant tumors as well as the prognostic “rule out” function of the same aberrations in histologically proven FTCs (lack of TERT aberrancies indicates lower risks of future recurrences). Although in dire need of validation in a prospective setting, the model could serve as a schematic outline as to how TERT gene aberrancies will be interpreted in the clinical setting.
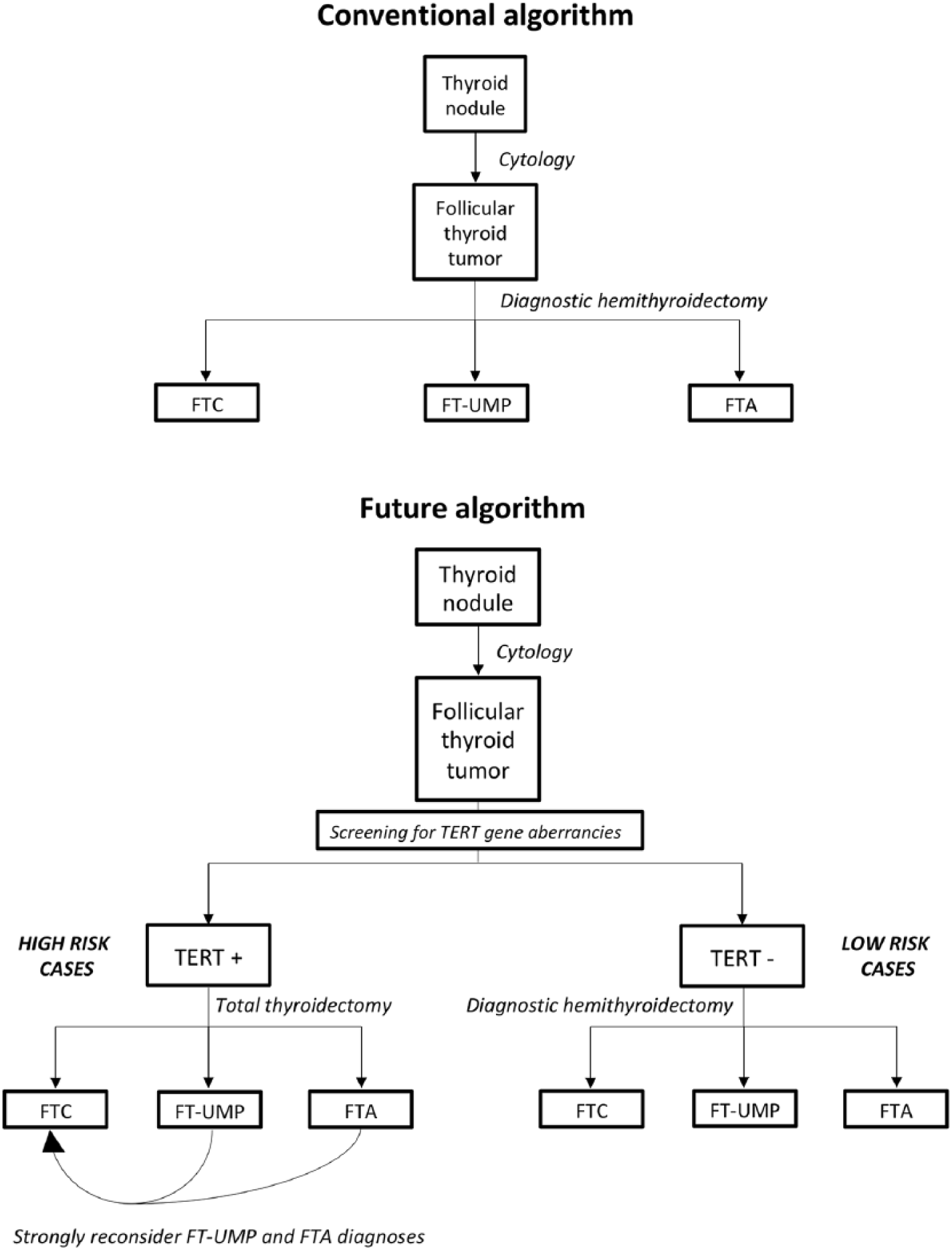
Theoretical model for the clinical implementation of screening for TERT gene aberrancies in follicular thyroid tumors. Top: conventional algorithm used when assessing follicular thyroid tumors in the clinical setting. A follicular thyroid tumor is diagnosed using routine cytological investigations, and a diagnostic hemithyroidectomy is performed. The final diagnosis is based on the histopathological assessment of excised tumor material. Bottom: alternative algorithm employing molecular testing of TERT gene aberrancies (most notably promoter mutations) at the level of preoperative cytological investigation. Cases with TERT gene aberrancies are denoted “high risk cases” given the coupling between these genetic events and poor prognosis, and a total thyroidectomy is performed up-front. Any cases of FT-UMPs or FTAs in this pool need to be subjected to critical revision of the diagnosis. Cases without TERT aberrancies have an estimated low risk of future recurrences, and a diagnostic hemithyroidectomy is preferred.
As reflex molecular testing of tumor material in the clinical setting becomes more and more common, surgical pathologists inevitably have to adapt—as the possibilities to merge histology with molecular biology allow for an immense improvement of the diagnostic and prognostic accuracy when assessing neoplasia. Although future molecular testing probably will incorporate global gene profiles of individual tumors, TERT gene aberrancies serve as a simplistic model of the benefits of incorporating molecular testing in clinical routine practice.
To conclude, TERT gene aberrancies are gaining ground as collective predictors of poor outcome in thyroid cancer. Specifically, TERT promoter mutational screening is a cheap, cost-effective, and easy-to-interpret method that should be incorporated in clinical routine assessment of follicular lesions at all high-volume tertiary centers, as the occurrence of such a mutation strongly indicates malignant potential, whereas wild-type sequences identify cases with low risk of future recurrences.
Footnotes
Acknowledgements
The author is a Junior Clinical Investigator of the Swedish Cancer Society.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
