Abstract
Background and aims:
Today, a significant proportion of solid abdominal organ injuries, whether caused by penetrating or blunt trauma, are managed nonoperatively. However, the controversy over operative versus nonoperative management started more than a hundred years ago. The aim of this review is to highlight some of the key past observations and summarize the current knowledge and guidelines in the management of solid abdominal organ injuries.
Materials and Methods:
A non-systematic search through historical articles and references on the management practices of abdominal injuries was conducted utilizing early printed volumes of major surgical and medical journals from the late 19th century onwards.
Results:
Until the late 19th century, the standard treatment of penetrating abdominal injuries was nonoperative. The first article advocating formal laparotomy for abdominal gunshot wounds was published in 1881 by Sims. After World War I, the policy of mandatory laparotomy became standard practice for penetrating abdominal trauma. During the latter half of the 20th century, the concept of selective nonoperative management, initially for anterior abdominal stab wounds and later also gunshot wounds, was adopted by major trauma centers in South Africa, the United States, and little later in Europe. In blunt solid abdominal organ injuries, the evolution from surgery to nonoperative management in hemodynamically stable patients aided by the development of modern imaging techniques was rapid from 1980s onwards.
Conclusion:
With the help of modern imaging techniques and adjunctive radiological and endoscopic interventions, a major shift from mandatory to selective surgical approach to solid abdominal organ injuries has occurred during the last 30–50 years.
Introduction
A rational approach to nonoperative management of abdominal injuries requires appropriate imaging techniques that were not available several decades ago. Thus, nonoperative management of both penetrating and blunt abdominal trauma was based on clinical presentation and plain x-rays. Only after the emergence of ultrasonography (US) and computed tomography (CT), could injuries to solid abdominal organs be evaluated accurately without surgical exploration.
Aims
Nonoperative management has become a standard management technique in stable patients initially with renal and later on with liver and spleen injuries. Although the controversy regarding when to operate and when not started already more than a hundred years ago, modern imaging techniques and the general trend favoring less invasive management techniques has elevated the discussion—and practice—to a new level. It is important to be familiar with the past developments in order to understand how current guidelines were formulated. The aim of this review was to highlight some of the key past observations in managing solid abdominal organ injuries and summarize the current knowledge and guidelines in managing these injuries.
Methods
A non-systematic search through historical articles and references on the management practices of abdominal injuries was conducted utilizing early printed volumes of major surgical and medical journals from the late 19th century onwards. The first part deals with general evolution in managing abdominal injuries and is followed by organ-specific development milestones. The current role of nonoperative management is summarized in the latter part of the article.
Management of abdominal injuries
Until the late 19th century, the standard treatment of penetrating abdominal injuries consisted of starvation and opium although isolated reports of operatively treated patients with intestinal perforation or evisceration were published (1, 2). Apparently, the first well-recorded trauma laparotomy was performed by George E. Goodfellow on 13 July 1881, who treated a miner suffering an abdominal gunshot wound. He had altogether six holes in the small and large intestine that were treated by suturing. The patient survived (3). In 1881, Sims in his review of recent advances in peritoneal surgery advocated formal laparotomy for the treatment of abdominal gunshot wounds to prevent death from shock, hemorrhage, or septicemia (4).
In 1882, professor Kinloch from Charleston explored a patient with an abdominal gunshot wound and sutured five intestinal wounds but the patient died. Successful suturing of stomach and intestinal wounds were carried shortly thereafter by Kocher and Bull, respectively, and in 1885, Smart extracted a bullet from the right lobe of the liver through a subcostal incision (5, 6). However, the controversy over nonoperative versus operative management of abdominal gunshot wounds continued during the Boer and Russo-Japanese wars, and even at the end of World War I, when operative management was again more in favor. It should be pointed out that in those days the mortality rates for penetrating abdominal injuries was about 50% (7).
In 1922, Mason presented a paper emphasizing the importance of hemorrhage associated with mortality of gunshot and other abdominal wounds and advised the use of blood transfusions, and in 1930 Hauer published a report of 334 patients treated for abdominal injuries at the Cincinnati General Hospital with a mortality rate of 50% for gunshots and 13% for stab wounds (6). In World War II, the mortality rate for abdominal wounds began to fall and ultimately reached 25% (7).
During the latter half of the 20th century, several critical observations and techniques started to challenge the policy of mandatory laparotomy especially for penetrating abdominal trauma. The high non-therapeutic laparotomy rates with associated morbidity and costs, the evolving diagnostic techniques (diagnostic peritoneal lavage, US, CT, natural orifice, and cavitary endoscopy), and non-surgical interventions (percutaneous drainage, endoscopy, interventional radiology) increased the possibility of selective nonoperative management (8–10).
To reduce the more than 50% non-therapeutic laparotomy rate in patients with anterior abdominal stab wounds undergoing mandatory exploration, suggestions of selective nonoperative management and the introduction of diagnostic peritoneal lavage in the 1960s moved the pendulum toward nonoperative management (6, 9, 11). The first randomized study comparing mandatory and selective surgical exploration for anterior abdominal stab wounds was published in 1996 (12). In abdominal gunshot wounds, selective nonoperative management was adopted in the 1990s as reflected in a prospective study of 309 patients where eventually 30% of the patients were managed successfully nonoperatively (13). In patients with blunt trauma and especially blunt solid abdominal organ injuries, the evolution of nonoperative management depended more on the imaging capability of specific organs.
Liver
In 1870, Burns resected a lacerated portion of the liver after a gunshot wound, and in 1879, Buckhardt used packing to arrest hemorrhage from a liver injury (14). In 1885, a formal laparotomy was performed to remove a bullet from the right lobe of the liver and the patient made excellent recovery (5). Perihepatic packing that became a crucial part of damage control surgery later on was first described by J. Hogart Pringle from the Glasgow Royal Infirmary in 1908. He reported eight patients treated for hepatic injuries. Three of them died immediately post-admission and one refused surgery; all four died. Another four patients were operated; two exsanguinated on the table and one undergoing liver suture and packing died postoperatively on the same day. The last patient had injuries to the right lobe of the liver and right kidney. Mobilization of the liver resulted in more bleeding, and Pringle decided to pack the liver and the kidney. The patient died 4 days later of pulmonary embolism, but at autopsy, the bleeding seemed to have been controlled by the packs (15).
Over time, surgical management became the standard for managing liver injuries, and this trend continued for the next 50–70 years up to the late 1980s. Lucas and Ledgerwood analyzed the management of liver injuries over three time periods (1969–1970, 1981–1982, and 1997–1998) and showed that laparotomy was done in all patients during the first two time periods, whereas in 1997–1998 most blunt hepatic injuries were managed nonoperatively with only 9/65 patients (14%) requiring hemostasis (16).
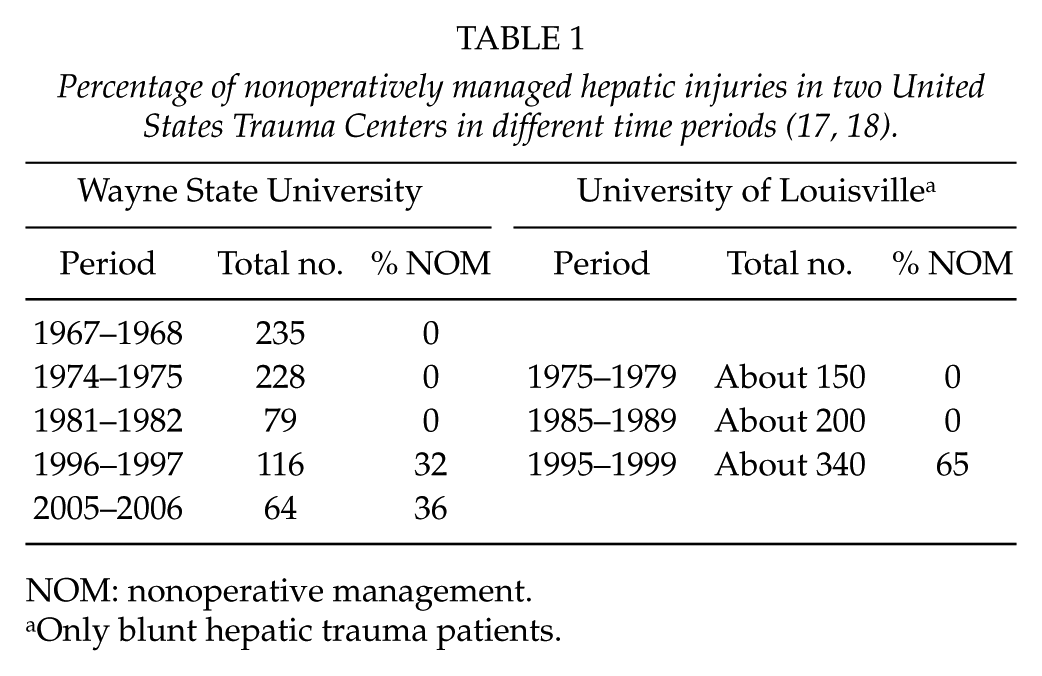
A significant change in the management strategies of liver injuries occurred after the late 1980s (16). As shown in Table 1, there was a marked increase in the percentage of nonoperatively managed blunt hepatic trauma patients in two major United States Trauma Centers from mid-1990s onwards (17, 18). Paralleling the management of blunt and penetrating abdominal trauma in general, selective nonoperative management of blunt hepatic injuries became the standard, and only patients with specific indications underwent operative management. A key component of the selective approach was the ability to diagnose and assess the severity of liver and other associated injuries with CT. Before the more selective approach, it was a common finding at laparotomy that bleeding from the liver injury had ceased spontaneously, in about 40% after both stab wounds and low-velocity gunshot wounds, leading to a non-therapeutic laparotomy (19, 20). The high success rate of nonoperative management was further improved with several alternative methods for hemorrhage and bile leak control, such as angioembolization (AE) and endoscopic biliary drainage, respectively, used as adjuncts to nonoperative management (21).
NOM: nonoperative management.
Only blunt hepatic trauma patients.
Spleen
For about 100 years, the standard treatment of splenic injury was splenectomy. Although already in 1590 Rousset described what may have been the first splenectomy for trauma, the first well-documented case of survival after splenectomy for trauma was reported by Reigner in 1893 (22). In 1912, Bland-Sutton described probably the first case of successful nonoperative management of a splenic rupture. A woman fell from a window, had a femur fracture, and died 10 days later. At post-mortem examination, it was seen that her spleen had been torn cross, and the torn surfaces had united with an encapsulated collection of blood around the organ (23).
The earliest reference to delayed rupture of the spleen is by Evans in 1866. He treated a 63-year-old dustman who suffered a crushing injury and died 5 days later. Autopsy examination revealed hemorrhage from a deep laceration of the spleen, the capsule “which was extensively separated in parts from the splenic substance.” Evans concluded, “Although the blood continued to be effused into the spleen during the subsequent days, it was probably not until the evening of the fourth day … that its capsule gave away” (24). In 1931, McIndoe described a patient with delayed manifestations of bleeding from spleen and collected 45 similar cases from the literature choosing an arbitrary minimum latency period of 48 h (25). While in most cases it is probably more of a delayed presentation of continuous bleeding from a splenic injury, a true delayed rupture apparently exists but is less common than previously thought (26).
Although already in 1919 Morris and Bullock described the increased risk of infection after splenectomy, a 1952 report by King and Schumacher describing lethal post splenectomy sepsis led gradually to a better understanding of the role of the spleen in the prevention of invasive bacteremia and its immunological functions in general (27). Preserving splenic function became important and paved the way for splenic salvage and nonoperative management. Richardson divided the efforts at splenic salvation into three different areas: operative attempts at maintaining splenic function (from 1980s), embolization of the splenic artery (1995), and nonoperative management (late 1980s in adults) (28). Today, more than 80% of adult patients with blunt splenic trauma are managed nonoperatively.
Pancreas
The first pancreatic injury was reported in the modern literature in a woman hit by a stagecoach wheel in 1827, and in 1868, Kleberg described a case with resection of a portion of pancreas protruding through the stab wound (29, 30).
In his 1903 review of the surgery of the pancreas, Mickulitz identified 45 cases of pancreatic trauma in the literature (30). Of the 12 gunshot wounds, five were operated, and three of them survived. All seven that underwent nonoperative management died. All of the nine patients with stab wounds were operated, eight survived. Of the 24 blunt injuries, 13 were not operated and all died. Of the 11 operated patients, seven recovered.
Post-traumatic pseudocysts and fistulas were known to surgeons more than 100 years ago. In 1882,Kulenkampff described a pancreatic pseudocyst after blunt trauma, and in 1905 Korte reported a case with pancreatic fistula after transection; it was managed nonoperatively and the patient survived (31).
The key element in selecting patients with pancreatic injuries for nonoperative management includes not just the detection of the injury itself but also the accurate assessment of the potential injury and its location in the main pancreatic duct. Failure to treat a main duct injury usually leads to delayed complications that can manifest even decades after the initial injury (32).
Kidney
Nonoperative management of renal injuries was the standard well into the 20th century even though already in 1883 three nephrectomies for renal trauma were reported (33, 34). In 1950, Sargent and Marquardt treated 206 patients with renal trauma, of which eight patients underwent early surgery with three deaths and further six with shattered kidneys had the diagnosis made at autopsy. The remaining 192 patients were managed nonoperatively with no deaths, but delayed nephrectomy was needed in two cases (35).
Routine nonoperative management was challenged by Nation and Massey in 1963 who analyzed 258 patients and recommended operative treatment for patients with continuing hemorrhage, persistent nonfunction, or perirenal contrast extravasation (36). In a series of 69 blunt renal trauma patients treated in 1976–1985, 16 underwent immediate surgery, whereas of the 53 patients undergoing radiological evaluation, six were operated. Overall, of the 47 patients (68%) managed nonoperatively, one required delayed surgery (37).
Diagnostic and therapeutic adjuncts
The first CT scanner was developed by Sir Godfrey Hounsfield already in 1967, but the first CT scans (of the head) were performed in 1971 (38). A more widespread use of CT especially in blunt trauma patients was encouraged in the early 1980s by Federle and coworkers (39). CT has become the main diagnostic tool for hemodynamically stable abdominal trauma patients and has replaced the historical diagnostic peritoneal lavage.
First angiographic experiments in the animals in the early 20th century were hindered by the lack of nontoxic contrast media when injected intra-arterially, but in the late 1920s an organic, water-soluble, iodinated contrast agents initially developed as a therapeutic agent for staphylococcal infections of the gall bladder was modified for use in angiography (40). Another critical step toward therapeutic angiography was the description of the transfemoral catheter angiography by Seldinger in 1953 (41). A landmark paper by Sclafani and coworkers in 1995 on nonoperative salvage of CT-diagnosed splenic injuries managed with AE paved the way to the current role of interventional radiology in managing solid abdominal organ injuries (42).
Current management guidelines
Based on an International Consensus Conference held in Milan and its results published in 2018, the role of nonoperative management of solid abdominal organ injuries has been described (43). In general, patients who are hemodynamically unstable have generalized peritonitis on clinical examination or have associated abdominal injuries requiring surgery are not candidates for nonoperative management. The diagnosis and severity grading are based on CT with intravenous contrast (CE-CT). Nonoperative management should only be attempted in institutions that can provide intensive monitoring and have an operating room always available. Institutions without these capabilities should transfer these patients. A summary of the recommendations is presented in Table 2.
Summary of consensus guidelines of nonoperative management of solid abdominal organ injuries (43).
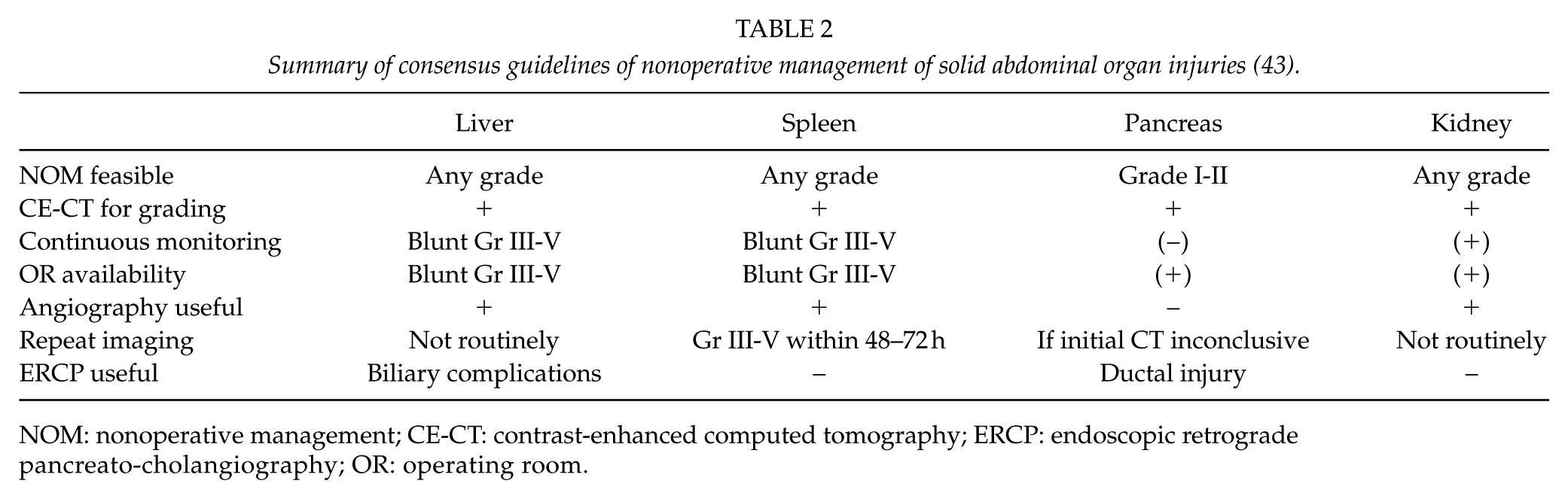
NOM: nonoperative management; CE-CT: contrast-enhanced computed tomography; ERCP: endoscopic retrograde pancreato-cholangiography; OR: operating room.
Liver
Patients with ongoing instability should undergo surgery and are not candidates for AE unless performed in hybrid environment. If contrast extravasation or a pseudoaneurysm can be seen on CT, AE should be considered.
The risk of biliary complications increases with increasing liver injury grade. Endoscopic retrograde cholangio-pancreatography (ERCP) with sphincterotomy and biliary stenting for bile leaks, percutaneous drainage of bile collections, and open or laparoscopic surgical interventions can be used to manage biliary complications.
Spleen
In stable patients with blunt splenic injury regardless of grading, AE should be performed early if active bleeding or a nonbleeding vascular injury is seen on CT, and even without these findings in grade IV-V injuries. Proximal AE after the origin of dorsal pancreatic artery is preferred over distal embolization.
Pancreas
Because diagnostic delay of main pancreatic duct injury increases the risk of mortality and morbidity, CE-CT performed during the portal venous phase is the best initial examination even though CT is not sensitive for detecting main ductal injury. Repeated CT improves accuracy, but the first choice of secondary imaging in patients with suspected main ductal injury is magnetic resonance cholangiopancreatography (MRCP). The most sensitive technique to detect a main ductal injury is ERCP that can also be used to treat a partial main ductal injury (side fistula) with a stent.
Nonoperative management is safe in grade I-II pancreatic injuries, whereas in grade III-V injuries operative management is preferred unless endoscopic management (grade III-IV) is feasible. Endoscopic or percutaneous drainage can be used to manage pancreatic complications, such as peripancreatic fluid collections, pseudocysts, or persistent pancreatic fistulas.
Kidney
CE-CT scan should include an excretory phase. In patients with renal parenchymal injury, AE can be used as an adjunctive tool to improve the success of NOM.
Although most urinary leaks heal spontaneously, persistent urinary leaks require open surgery, nephrostomy, or ureteral stenting. If traumatic renal artery thrombosis or dissection is not treated within 4 h of warm ischemia time, the risk of kidney loss is high.
Failed Nonoperative Management
The failure rate of nonoperative management of solid abdominal organ injuries is low when appropriate indications are used. In a prospective observational study of 206 patients with blunt liver, spleen, or kidney injuries, the failure rate was 9% for liver, 33% for splenic, and 11% for kidney injuries, respectively (44). The most common reasons were continuous or recurrent bleeding and missed bowel injuries.
Today, more than 80% of blunt liver injuries in adults are managed nonoperatively. In a systematic review of NOM of blunt liver trauma, the pooled failure rate of NOM was 9.5% (45). In a study of 144 patients with blunt grade III-V liver injuries, the initial management was nonoperative in 65%, and it failed in 8 (9%) of the 94 patients. Shock on admission and associated high-grade splenic injury predicted failure of NOM (46). Nonoperative management of high-grade blunt hepatic injuries is associated with significant morbidity. In a series of 230 nonoperatively managed patients with blunt Grade III-V hepatic injuries, a total of 25 patients (11%) had 37 hepatic-related complications including bleeding, liver abscesses, biliary leaks and duct disruptions, and abdominal compartment syndrome (47). Most bile leaks and intra-abdominal abscesses were amenable to percutaneous drainage. Persistent bilious drainage often requires ERCP and a placement of a biliary stent (40). In most cases, the bile leak will resolve within 10 days from the ERCP (48).
In an EAST multi-institutional study of 1488 adult patients with blunt splenic injury, 62% underwent initial NOM out of which 11% failed, more than half within 24 h. Successful NOM was associated with higher blood pressure and hematocrit, quantity of hemoperitoneum, and less severe injury grade. The failure rates for grades I, II, III, IV, and V were 5%, 10%, 20%, 33%, and 74%, respectively (49). In a study of 712 patients with blunt splenic injury, including mandatory AE for grade IV-V injuries and selective AE for grade I-V with contrast blush, the failure rate of NOM decreased from 4% to 1% in all patients and from 19% to 3% in grade IV-V injuries (50).
In patients with pancreatic trauma, the success rate of nonoperative management is about 50%–75%, even in high-grade injuries, but is associated with significant morbidity (51). Out of 120 patients with blunt pancreatic trauma, 35 with grade I-II pancreatic injury detected with CT were selected for NOM. It failed in five patients due to missed bowel injury in two and pancreatic abscess in three, out of which one developed a pancreatic fistula (52). In another study, six patients with grade III injury was treated nonoperatively, out of which one developed a peripancreatic abscess and another a pancreatic fistula, both managed successfully without surgery (53).
In a National Trauma Data Bank study of 19,572 patients with renal injury, 83% were managed nonoperatively, and NOM failed in 2.7%. In addition to penetrating trauma mechanism and overall trauma burden, increasing renal injury grade was associated with increasing risk of failed NOM (54).
Conclusion
It seems that with proper patient selection, accurate grading based on CT, and active use of adjunctive procedures such as AE and ERCP, NOM of blunt solid abdominal organ injuries is safe, especially in low-grade injuries. However, NOM should only be used with close monitoring and the ability to perform acute surgery, if NOM fails.
It can be anticipated that in the future, the polarization of two extreme management techniques increase, that is, more and more patients can be safely managed nonoperatively utilizing adjunctive interventional radiological and endoscopic techniques, whereas damage control surgery becomes a dominant surgical intervention approach for severely injured patients with multiple injuries including those with solid abdominal organ injuries.
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.