Abstract
Background and Aims:
Several studies and guidelines are questioning routine preoperative laboratory tests in surgical and endoscopic procedures. Their effect in endoscopic retrograde cholangiopancreatography is not currently known. This study was carried out to evaluate the risk of adverse effects in endoscopic retrograde cholangiopancreatography and their association with preoperative lab tests.
Materials and Methods:
A single-center, prospective observational study on all 956 patients undergoing 1196 endoscopic retrograde cholangiopancreatographies in the Endoscopy Unit of Helsinki University Central Hospital from 1 March 2012 to 28 February 2013. Routine preoperative laboratory test results (basic blood count, creatinine, potassium, sodium, international normalized ratio/thromboplastin time, and amylase), health status, medication, and demographic information of all patients were analyzed in relation to adverse effects related to endoscopic retrograde cholangiopancreatography and procedural sedation.
Results:
Multivariate analysis showed post–endoscopic retrograde cholangiopancreatography pancreatitis (43 cases, 3.6%) to have no association with abnormal routine preoperative laboratory tests. Respiratory depression caused by sedation (128 cases, 11%) was not associated with abnormal routine preoperative laboratory tests, and anemia was found to be a slightly protecting factor. Cardiovascular depression caused by sedation was associated with thrombocytopenia (odds ratio = 1.87, p = 0.025) and, in male patients, hyponatremia (odds ratio = 3.66, p < 0.001). Incidence of other adverse effects was too low for statistical analysis.
Conclusion:
Routine universal preoperative lab testing was not found to be successful in predicting adverse effects in endoscopic retrograde cholangiopancreatography procedures. Laboratory testing should be done focusing on each patient’s individual needs.
Keywords
Introduction
Routine preoperative laboratory testing (RPLT) is a practice of ordering a given set of laboratory tests to every patient before a procedure regardless of the health status of individual patients. Traditionally, routine laboratory tests have been part of the protocol for endoscopic retrograde cholangiopancreatography (ERCP) in our institution. Several studies and guidelines are questioning such policy for surgery and endoscopy (1–3). ERCP is one of the most technically demanding endoscopic procedures with a risk for serious adverse effects (4). To our knowledge, no studies have been made to assess the effect of RPLT on patient outcomes and adverse effects for ERCP. Because of the policy of RPLT in our institution, we had a large database of preoperative laboratory results we could compare with adverse effects associated with ERCP.
This study was carried out in order to assess the association of RPLT before ERCP with adverse effects and mortality in the Endoscopic Unit of Meilahti Hospital, a tertiary university clinic where over 1200 ERCP procedures are performed annually.
Material And Methods
This study was approved by the Institutional Ethics Committee of Helsinki University Central Hospital (Ethics Committee, Department of Surgery, Biomedicum Helsinki 2C, Tukholmankatu 8C, PL 705, 00029 Helsinki, Finland).
A total number of 1228 ERCPs were attempted at the hospital during the 12-month period from 1 March 2012 to 28 February 2013. All the adult patients who were treated in the Endoscopy Unit were included in the study.
The following data were registered on each patient: age, weight, height, American Society of Anesthesiology (ASA) physical status classification, RPLT results (basic blood count (BBC), electrolytes (P-Na and P-K), thromboplastin time (P-TT) and international normalized ratio (P-INR), amylase levels before the procedure and 4 and 24 h after the procedure), indications of ERCP, cannulation method, performed procedures, duration of the procedure, ERCP adverse events (pancreatitis, bleeding, perforation, etc.), sedation-related adverse effects (cardiovascular depression—defined as systolic blood pressure <90 mm Hg, respiratory depression—defined as oxygen saturation SpO2 < 90%, need for mask ventilation, need for intubation), comorbidities (diabetes treated with insulin, diabetes treated with oral medication, electrocardiogram (ECG) analysis, hypertension, cardiovascular disease, cerebrovascular disease, atherosclerosis, chronic obstructive pulmonary disease, other pulmonary disease, kidney failure, hypothyreosis/other endocrinological disease, malignancy, bleeding disorders, other), regular medication (opioids, diuretics, beta blockers, other cardiological medication, corticosteroids, anticoagulants, benzodiazepines, other psychopharmacological drugs, and chemotherapy within the last 30 days), smoking and alcohol consumption, and 1-day and 30-day mortality were recorded.
Primary end points were adverse effects related to ERCP, sedation, or both.
The results are reported as medians and interquartile ranges (median (IQR)) or number of patients and percentages. Logistic regression analysis was used to assess the risk of adverse effects. Forward stepping was used with p < 0.05 inclusion and p < 0.1 removal criteria (preselection from univariate with p < 0.1); however, age and gender were included in all the models. Statistical calculations were generated using IBM SPSS Statistics 21 (International Business Machines Corporation, Endicott, NY, USA).
Results
We excluded 15 ERCPs performed in the operation theater and 17 ERCPs performed on patients under 18 years of age. In total, 956 patients with 1196 ERCPs were left in our analysis. Two gastroenterologists performed 253 ERCPs for cases with suspected or diagnosed primary sclerosing cholangitis (PSC) and 4 gastrointestinal surgeons performed 943 ERCP procedures with other indications. All patients were in prone position during the procedure.
Demographics
Median age of the 956 patients was 59.0 [25] years (range 18–100 years) and median body mass index (BMI) 24.8 [6.2] kg/m2, 529 (55%) were male and 427 (45%) female. The median length of the procedure was 23 [19] min. There were 299 (25%) procedures regarded as emergency ERCPs.
Comorbidities
ASA classification of the patients was as follows (number of patients (percentage)): ASA 1: 35 (2.9%), ASA 2: 417 (34.8%), ASA 3: 585 (48.7%), ASA 4: 160 (13.4%), and ASA 5: 2 (0.2%). Major comorbidities were as follows: diabetes: 256 (21.4%), chronic hypertension 404 (33.8%), cardiovascular disease 204 (17.1%), and liver disease 331 (27.7%). Prevalence of other diseases was less than 10%. 191 (16.0%) of the patients were smokers.
Ercp Details
ERCP was considered successful if the level of papilla or anastomosis was reached and the intended duct was successfully cannulated. Attempted ERCP was unsuccessful in 33 out of 1196 procedures. In 20 procedures, papilla was not reached or found (postoperative state (gastric resection, pancreaticoduodenectomy) (n = 5), obstruction proximal to papillary region (n = 6), edema of the descending duodenum (n = 8)) and one ERCP could not be performed due to patient’s allergy to sedatives. In 13 cases, papilla was reached, but the desired duct could not be cannulated. The endoscopic procedures included 1844 biliary tract interventions (biliary cytology, biliary sphincterotomy, biliary dilatation, or stent application) and 503 pancreatic procedures (pancreatic cytology, pancreatic dilatation, or stent application). Majority of the patients (787, 82%) had only one ERCP, but 116 (12%) had two, 42 (4%) had three, and 11 (2%) had four or more ERCPs performed on them during the study period.
Laboratory Test Results
Laboratory test results, listed here as median (IQR), followed by reference values were given as follows: hemoglobin (g/L): female 122 (20), 117–155, male 135 (29.5), 134–167. Platelet count (10E9/L): 243 (126), 150–360; potassium (P-K, mmol/L), 3.9 (0.53), 3.3–4.9; sodium (P-Na): 139 (4), 137–145; thromboplastin time (P-TT, %): 95 (41), 70–130; and international normalized ratio (P-INR): 1.1 (0.2), 2–3 (patients on anticoagulants). Hypokalemia was found on 62 patients, most of which was mild and required no intervention. Hypokalemia was treated with intravenous potassium chloride before the procedure on 20 patients. P-INR or P-TT levels needed to be normalized with prothrombin complex concentrate on 44 patients before the procedure.
Adverse Effects—Ercp
There were altogether 315 adverse events in 956 patients. Post-ERCP pancreatitis was defined as pain in the stomach, rise in the plasma amylase by three times the upper range limit, and the patient had to be hospitalized for 2 days because of pancreatitis. Pain in the stomach was experienced by 118 (9.9%) of patients. This was associated with already hospitalized patients (p = 0.012). Post-ERCP pancreatitis developed after 43 out of 1196 ERCPs, (3.6%). 24 of them were categorized as mild, 14 moderate, and 5 severe (5). Multivariate analysis (Table 1) showed that abnormalities in none of the RPLTs were associated with increased risk of post-ERCP pancreatitis.
Univariate and multivariate analysis of risk factors of post-ERCP pancreatitis.
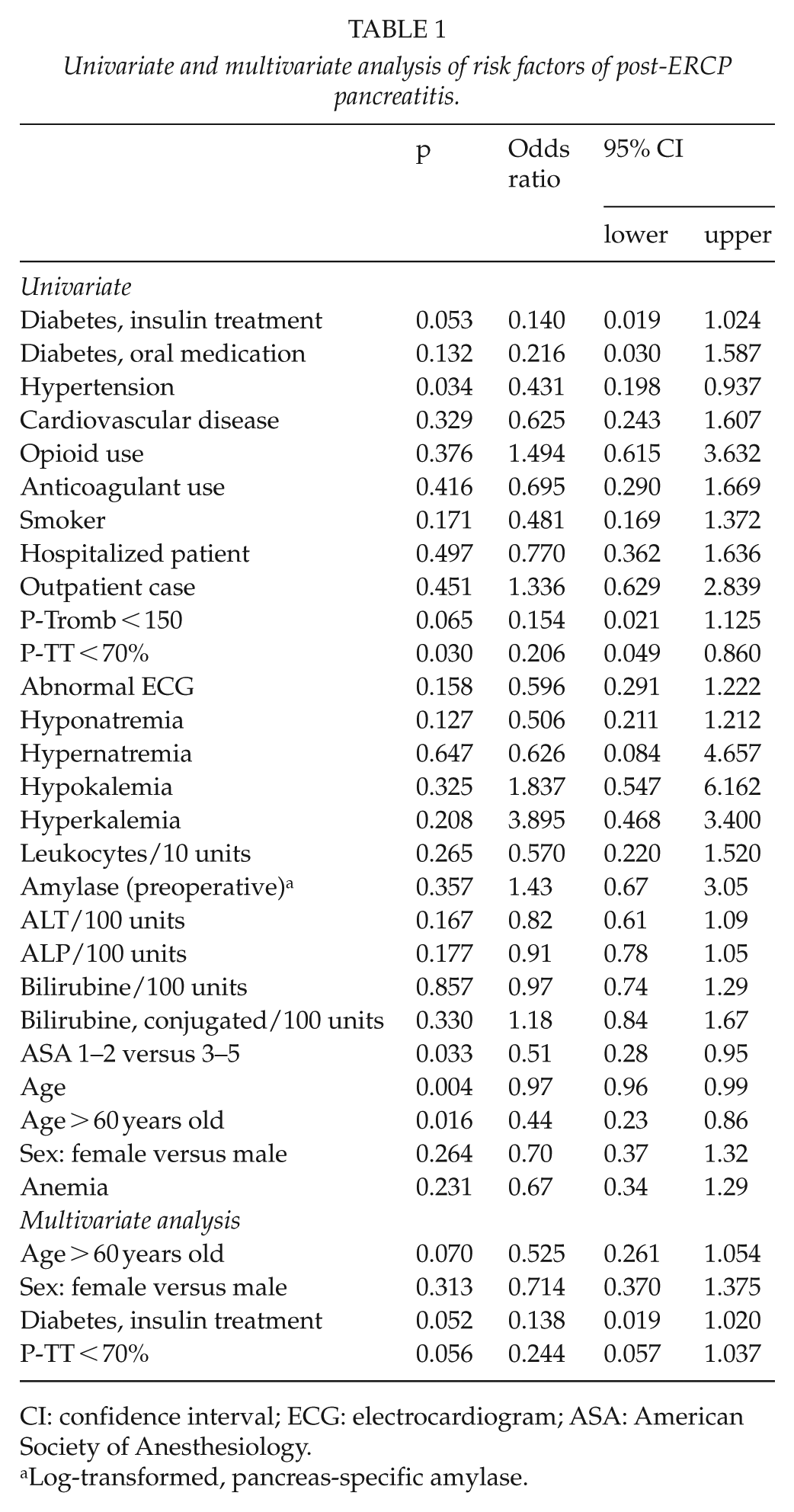
CI: confidence interval; ECG: electrocardiogram; ASA: American Society of Anesthesiology.
Log-transformed, pancreas-specific amylase.
Post-sphincterotomy bleeding (17 cases, 1.4%) and periampullary perforations (6 cases, 0.5%) were treated conservatively. No luminal perforation occurred. In addition, 12 patients (1%) with guide-wire-induced perforations received antibiotics. 17 (1.2%) patients had post-ERCP cholangitis, of which 2 patients (0.2%) were treated with antibiotics. In addition, 19 miscellaneous adverse events (1.6%; pseudocyst infection, stent migration, stent rupture, peptic ulcer bleeding, air embolism, etc.) and 5 cardiopulmonary adverse events (0.4%; myocardial infarction and pulmonary embolism) occurred.
Two patients died the same day ERCP was performed (one due to air embolism caused by gas insufflation during endoscopy on a patient with a transplanted liver and one elderly patient because of acute myocardial infarction). In total, 10 patients (1%) died within a week after the procedure, 8 patients with end stage cancer, 1 patient suffering from complications of cholecystectomy died of pulmonary embolism 5 days after ERCP, and 1 elderly patient died of myocardial infarction on the third day after ERCP. The 30-day mortality was 44 (4.6%).
Adverse Effects—Sedation
Respiratory depression occurred with 11% of patients (128 of 1196). The multivariate analysis of respiratory depression (Table 2) showed no association with preoperative laboratory tests. There was an interaction between anemia and cardiovascular disease in the statistical model, and upon further analysis, anemia was found to be a stronger protective factor (p < 0.001, odds ratio (OR) = 0.26), if the patient had no cardiovascular disease.
Univariate and multivariate analysis of risk factors of respiratory depression.
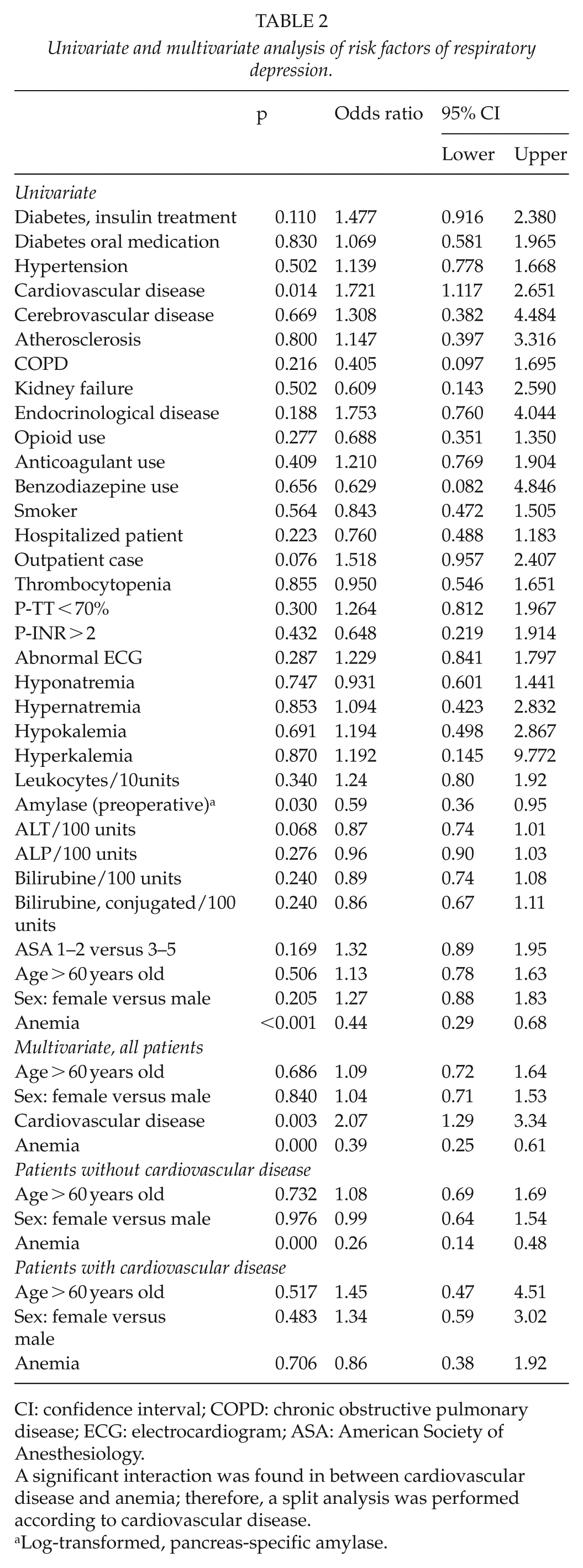
CI: confidence interval; COPD: chronic obstructive pulmonary disease; ECG: electrocardiogram; ASA: American Society of Anesthesiology.
A significant interaction was found in between cardiovascular disease and anemia; therefore, a split analysis was performed according to cardiovascular disease.
Log-transformed, pancreas-specific amylase.
Cardiovascular depression occurred in 7.2% of patients (86 of 1196). The multivariate analysis of cardiovascular depression (Table 3) showed that the risk of cardiovascular depression was increased if the patient had hyponatremia, thrombocytopenia, or was over 60 years old. There was an interaction between gender and hyponatremia in this statistical model, according to which hyponatremia was associated slightly more with cardiovascular depression on men than women.
Univariate and multivariate analysis of risk factors of cardiovascular depression.
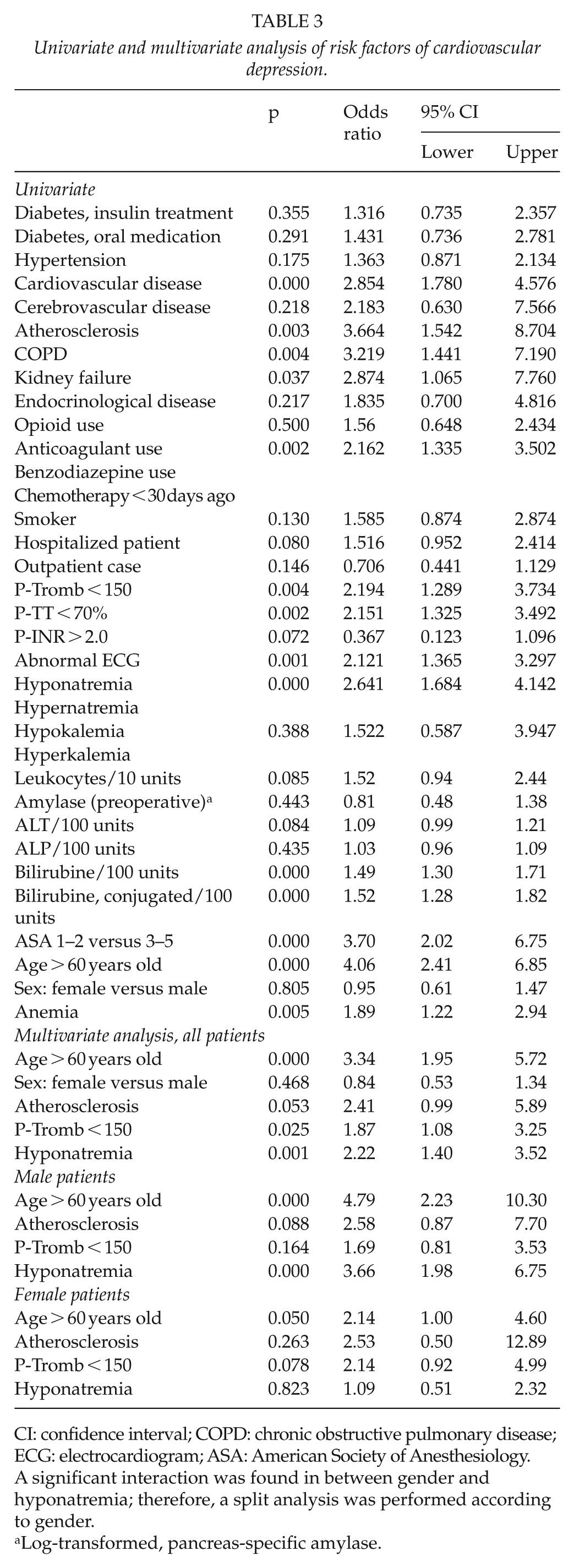
CI: confidence interval; COPD: chronic obstructive pulmonary disease; ECG: electrocardiogram; ASA: American Society of Anesthesiology.
A significant interaction was found in between gender and hyponatremia; therefore, a split analysis was performed according to gender.
Log-transformed, pancreas-specific amylase.
In total, 20 (1.7%) patients had both cardiovascular and respiratory depression as defined in the study. The procedure had to be paused due to mask ventilation 20 times (1.7% of patients). 3 patients were intubated (one case of air embolism leading to resuscitation, one case of laryngobronchospasm, and one ICU patient with septic cholecystitis, already intubated upon arrival to endoscopy room). Other sedation-related adverse events were not observed. Statistical analysis of the risk of mask ventilation or intubation was not feasible due to the small number of cases.
Discussion
It has been estimated that ERCP is needed in about 50–100 per 100,000 persons per year. As more therapeutic options become available, the need will most likely rise (6, 7). As the need for ERCP rises, the economic burden of preoperative testing will also rise. In our institution, the RPLT package costs about US$49 (€41) on one elective patient. In case of emergency patients, the costs of same package are significantly higher, about US$79 (€66). If we had refrained from taking RPLTs from patients not considered to be at risk for adverse effects or abnormal laboratory test results based on clinical judgment, based on the data gathered for this study, we would not have taken RPLTs from 805 patients, which translates to savings of at least US$40,000 (€33,000), assuming they were elective procedures. Systematic preoperative laboratory screening will most likely lead to excessive testing without any true impact on clinical decisions as has been shown by numerous studies and guidelines (1–3, 8). While most such studies are focused on surgery, comparable results are also found on studies on endoscopy, even though they are fewer in number. Also, one could assume that an endoscopic procedure such as ERCP would cause less stress on the patient and therefore require even less preoperative routine testing than patients undergoing surgery. Some RPLT results did indeed have an impact on the treatment of these patients, such as correcting hypokalemia or coagulation levels with prothrombin complex concentrate. However, the vast majority with only a few exceptions, these patients had a health condition or medication that would have warranted laboratory testing regardless of RPLT.
The rate of adverse effects of ERCP (9, 10) and sedation (11, 12) in this study was similar to other studies on the subject. This would lend credence to the notion that the results of this study should be applicable outside the Endoscopy Unit, in which this study was conducted.
There are some limitations to our study. First, being a single-center study, its applicability to other institutions with less experience with treating these patients may be limited. Also, the relatively small number of adverse effects prevents making solid conclusions on the matter, but does provide a foundation for possible further studies. There were 15 ERCPs performed in the operating theater instead of the Endoscopy Unit. The reason for this was because the patients were considered too unstable for sedation and required general anesthesia and more stringent monitoring than was available in the endoscopy unit. Naturally, this will create a possibility for bias, but the total number of these procedures is low, and the effect should be minimal.
We did not differentiate between acute and elective setting of ERCP. The rationale behind this was that RPLT is more relevant in the elective setting since patients having acute ERCP are already having laboratory testing done on them because of the underlying medical condition. When considering this—and the fact that patients receiving acute ERCP are more prone to adverse effects and abnormal laboratory test results—we should not expect the results to be biased against RPLT when considering all the patients as a single group, since RPLT is the focus of this study.
This study found that none of the RPLTs that were used were directly associated with any adverse effects for ERCP. There was an association with RPLT and adverse effects related to sedation with thrombocytopenia and hyponatremia and cardiovascular depression. However, the causal link between these findings is unclear and does warrant further studies. Both hyponatremia and thrombocytopenia are usually caused by some other health-related issues, and it is these root causes which are most likely responsible for hypotension during sedation. Simply correcting the values would most likely not alleviate the problem. Also, a clinical assessment of the patient before the procedure would probably inform the anesthesiologist of the risk of cardiovascular depression even without the preoperative laboratory testing. Therefore, it would seem that the policy of RPLT is not positively affecting the patients’ treatment and care and is not economically sound. That is not to say preoperative testing should be abandoned completely, but should be focused on those patients that would benefit from them. Interestingly, anemia was found to be a protecting factor for respiratory depression caused by sedation, especially on patients without cardiovascular disease. It could be that anemia causes sensitization to hypoxemia and thus leads to pronounced reactivity and increased respiratory drive when the patient is sedated, and respiratory center is affected by the sedative agents. However, clinical utilization of this finding, should it prove to be factual in possible further studies, is somewhat difficult, since anemia is something to be avoided when possible in clinical practice.
In conclusion, while it is not possible to give precise recommendations on RPLTs based on our study, we would venture to suggest the following general rules based on clinical experience and generally accepted medical practices as shown in Table 4. Other laboratory tests should be prescribed by the treating physician’s deliberation based on the patient’s individual health status.
Suggested laboratory tests before ERCP.

ASA: American Society of Anesthesiologists physical status classification, BBC: basic blood count, ECG: electrocardiogram, P-Na: plasma sodium, P-K: plasma potassium; P-INR: international normalized ratio.
Footnotes
Acknowledgements
J.J. and S.I. contributed equally to this paper. This work was carried out in the Endoscopy Unit, Helsinki University Hospital, Helsinki, Finland.
Declaration Of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
