Abstract
Background and Aims:
The use of mesh repair in a small- or middle-sized umbilical hernia remains controversial, and evidence is based on only few and small heterogeneous randomized trials. The primary aim was to assess differences, if any, in recurrence (clinical and reoperation), and secondary aim was to assess differences in infections, seroma formation, hematomas, chronic pain, cosmetic result, and quality of life.
Method:
A systematic review (predefined search strategy) and meta-analyses were conducted based on pre-study strict and well-defined methodology. The literature search was completed on 1 January 2018. The study protocol was registered in PROSPERO.
Results:
Five randomized controlled trials were identified (mesh repair, n = 326 versus non-mesh sutured repair, n = 330) and 602 records were excluded. Randomized controlled trials included patients with defect diameters of ⩾1 to 4 cm. Mesh repair reduced the risk of recurrence compared with sutured repair with a relative risk of 0.28 (95% confidence interval = 0.13–0.58, I2 = 0%, number needed to treat = 13 patients). Additional analyses found no differences between the two surgical techniques regarding infection (relative risk = 0.80, 95% confidence interval = 0.36–1.79), seroma formation (relative risk = 1.38, 95% confidence interval = 0.57–3.32), or hematomas (relative risk = 0.55, 95% confidence interval = 0.23–1.30). Lack of sufficient data precluded meta-analysis evaluating risk of seroma formation, hematomas, chronic pain, cosmetic result, and quality of life.
Conclusion:
Mesh repair is recommended for umbilical hernia of ⩾1 to 4 cm. More evidence is needed for the optimal placement of the mesh (sublay or onlay) and the role of mesh in patients with an umbilical hernia <1 cm.
Introduction
Open umbilical repair is one of the most commonly performed minor surgical procedures. The incidence is approximately 1500 repairs in Denmark (1) and 175,000 repairs in the United States each year (2). In particular, recurrence remains a concern, but also the risk of chronic pain has drawn increasing attention, although only scarcely investigated (3–6). The controversy concerning the use of mesh in these repairs is not settled despite being a high volume and minor surgical intervention (1).
Two previous systematic reviews compared open umbilical or epigastric hernia repair with or without mesh reinforcement including only one and three randomized controlled trials (RCTs), respectively (7, 8). Both analyses contained mainly retrospective older cohort studies, a mixture of elective and emergency repairs, and umbilical and epigastric hernia repairs. Included studies did not discriminate between recurrences as primary or secondary outcomes. Moreover, two recent meta-analyses (8, 9) compared a mixture of elective and emergency procedures including a variety of types of ventral hernias (i.e. incisional and primary (umbilical) hernia repair) with or without liver cirrhosis and did not discriminate between recurrence as primary or secondary outcome. Although it is well accepted that reoperation for recurrence severely underestimates clinical recurrence (10), the four previous systematic reviews (7–9, 11) and previous RCTs (12–15) did not discriminate clinical recurrence or reoperation for recurrence. In addition, a large high-quality Dutch RCT (n = 300) was recently published (17) and was not included in the previous meta-analyses. Finally, and perhaps most important, none of the previous systematic reviews and meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines for meta-analysis conductance (18).
For the above-mentioned reasons, it was regarded important to present an updated systematic review based exclusively on RCT data on strict inclusion criteria including only adult patients undergoing elective open umbilical hernia repair. The primary outcomes of this meta-analysis includes exclusively primary outcome results from RCTs. Before study start, it was decided that the primary aim was to assess the difference, if any, in recurrence defined as clinical, reoperation, and combined assessment. Secondary aims were to appraise differences, if any, in surgical site infections, seroma formation, hematomas, chronic pain, cosmetic result, and quality of life (QoL).
Methods
This systematic review was based on a registered protocol in PROSPERO (https://www.crd.york.ac.uk/prospero/display_record.php?RecordID=46845) and results are reported according to the PRISMA guidelines (18).
Search Strategy (End of Search 1 April 2018)
We searched Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, EMBASE, and Science Citation Index Expanded. We used the Cochrane highly sensitive search strategies for identification of clinical RCTs (Table 1) (19). The search string was ((“Hernia, Ventral” (Mesh) OR “ventral hernia”) AND (umbilical AND (surgery OR herniotomy OR herniorrhaphy OR hernioplasty)) AND (recurrence OR “chronic pain” OR “surgical site infections” OR “wound infections” OR “seromas” OR “quality of life” OR cosmesis)). In the manual searches, we scanned reference lists of relevant articles and proceedings from meetings. We also wrote to the principal authors of RCTs for information about any ongoing trials and searched the online trial registries, ClinicalTrials.gov (clinicaltrials.gov/) and World Health Organization (WHO) International Clinical Trial Registry Platform (www.who.int/ictrp), for ongoing or unpublished trials. Two authors (T.B. and L.L.G.) scrutinized searches, performed additional manual searches, listed potentially eligible RCTs, and collected data. Contrary opinions were resolved through consensus before analyses. All authors read the potentially eligible RCTs and participated in the final selection of RCTs. We described the characteristics of included trials in summary tables and described reasons for exclusion for all excluded trials. Extracted data included study design, type of surgical techniques, number of participants, hernia defect size, length of follow-up, and outcomes as defined above.
Characteristics of randomized controlled trials (RCTs) included in the meta-analyses.
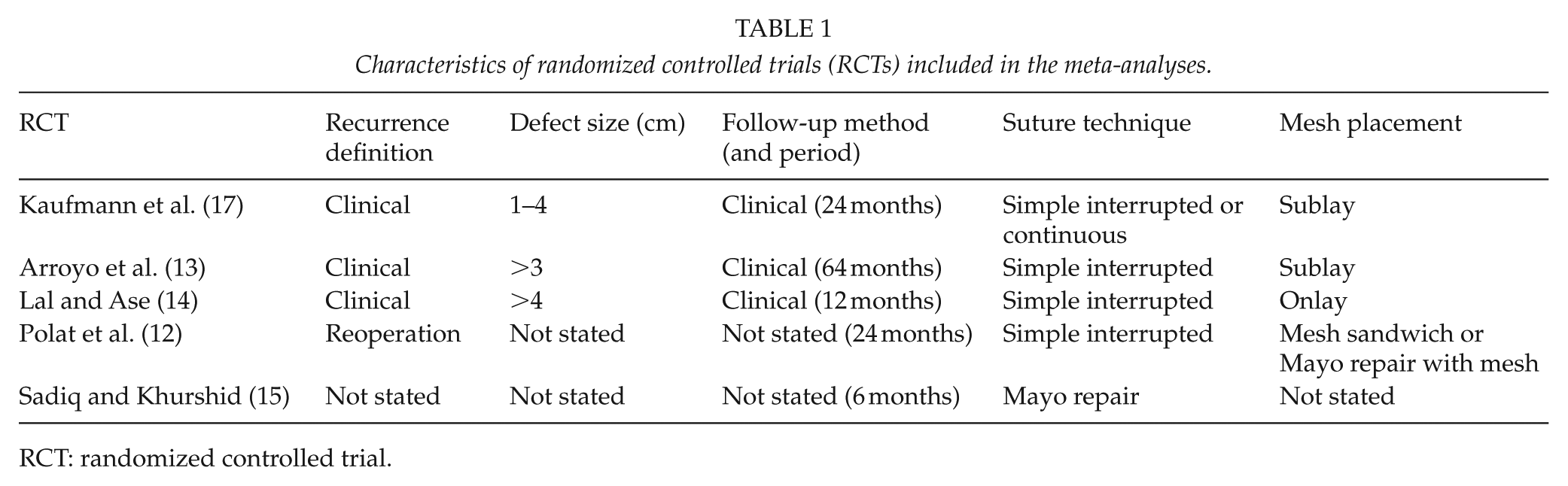
RCT: randomized controlled trial.
We included RCTs regardless of their publication status (published manuscript or abstract). Inclusion of studies was limited to the English language. The assessed interventions were elective umbilical hernia repair with either open mesh repair or non-mesh repair. We excluded patients with liver cirrhosis, patients undergoing emergency repairs, other types of ventral hernia repair (i.e. epigastric, Spigelian, parastomal, or incisional hernia repairs), and repairs during concomitant surgery. To secure robustness of recurrence outcomes, we excluded RCTs where recurrence was not the primary outcome as mentioned above (20).
Types of Outcome Measures
Study outcomes were predefined before the study was conducted. The primary analysis of the meta-analysis was differences, if any, in clinical recurrence, non-clinical assessment (reoperation and unclear defined recurrence), and total recurrence (clinical and not clinically defined). Secondary analyses were also performed on other outcomes such as differences, if any, in surgical site infections, seroma formation, hematomas, chronic pain, cosmetic result, and QoL after mesh repair or sutured repair. We assessed all outcomes at the maximum duration of follow-up.
Bias and Quality Assessment
Bias control was assessed using the Cochrane domains. Due to the nature of the intervention, we only included an assessment of blinding of outcome assessors (21). The domains were combined into an overall assessment and graded RCTs as low risk of bias if none of the individual domains were classified as unclear/high risk of bias.
The GRADEpro system (21) was used to evaluate the quality of the combined evidence for outcomes reported in the review, considering the within-study risk of bias (methodological quality), inconsistency, imprecision, indirectness, and publication bias.
Statistical Analyses
Meta-analyses were conducted and results reported as relative risks (RR) with 95% confidence intervals (CIs). Trial Sequential Analyses were conducted to evaluate the risk of error and futility. For our primary outcome, we calculated the number needed to treat (NNT) based on 1/risk difference. We expressed heterogeneity as I2 values using the following thresholds: 0% to 40% (unimportant), 40% to 60% (moderate), 60% to 80% (substantial), and >80% (considerable). We initially conducted random-effects and fixed-effect meta-analyses. The estimates of the random-effects and fixed-effect meta-analyses were similar for all analyses. We chose to report the random-effects models, which provided the most conservative estimate of the intervention effect. We did not conduct planned subgroup analyses evaluating bias because only one RCT had a low risk of bias (17). A Trial Sequential Analysis was performed in the assessment of primary outcome. The required information size was defined as the number of participants needed to detect or reject an intervention’s effect based on the relative risk reduction (RRR) and assumed control risk (ACR). We defined evidence as established if the Z-curve crossed the monitoring boundary before reaching the required information size. We performed the analyses with alpha set to 5%, power to 80%, and model based diversity. Based on previous evidence (7, 8), we set the RRR to 46% and the ACR to 10%. We performed the analyses in Review Manager 5 (Nordic Cochrane Centre, Copenhagen, Denmark), STATA (version 14, Philadelphia, USA), and Trial Sequential Analysis (Copenhagen Trial Unit, Copenhagen, Denmark).
Results
The electronic searches revealed 767 potentially eligible references. The manual searches identified additional 13 references (Fig. 1). We excluded RCTs evaluating patients with incisional hernias, liver cirrhosis, or emergency surgery, and a total of 602 records were excluded. In total, we included five RCTs (full paper articles published in English language) in our meta-analyses (12–17). The RCTs were published between 2001 and 2018, and varied clinically in respect of hernia defect size, follow-up method (clinical or non-clinical assessment (reoperation and unclear defined recurrence)), and total recurrence (clinical and non-clinical assessment), follow-up period, and mesh placement (Table 1). The duration of follow-up varied between 6 and 64 months.
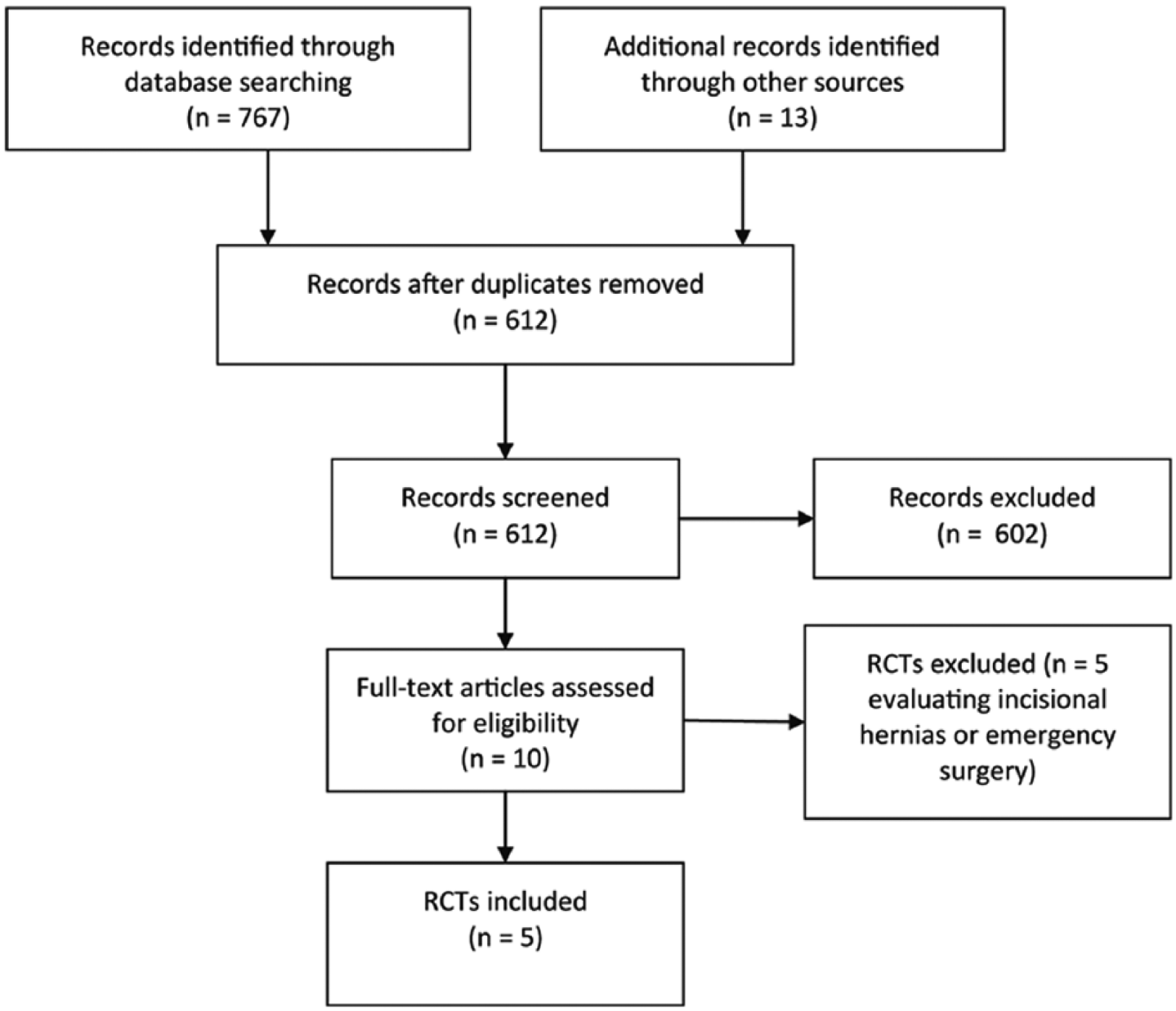
PRISMA flowchart of studies for the analysis.
Bias Assessment
Two RCTs reported the allocation sequence generation and one reported the allocation concealment (Table 2). None of the trials employed blinded outcome assessment. Only one of the RCTs had a low risk of bias in the overall assessment (17).
Risk of bia in included RCT.
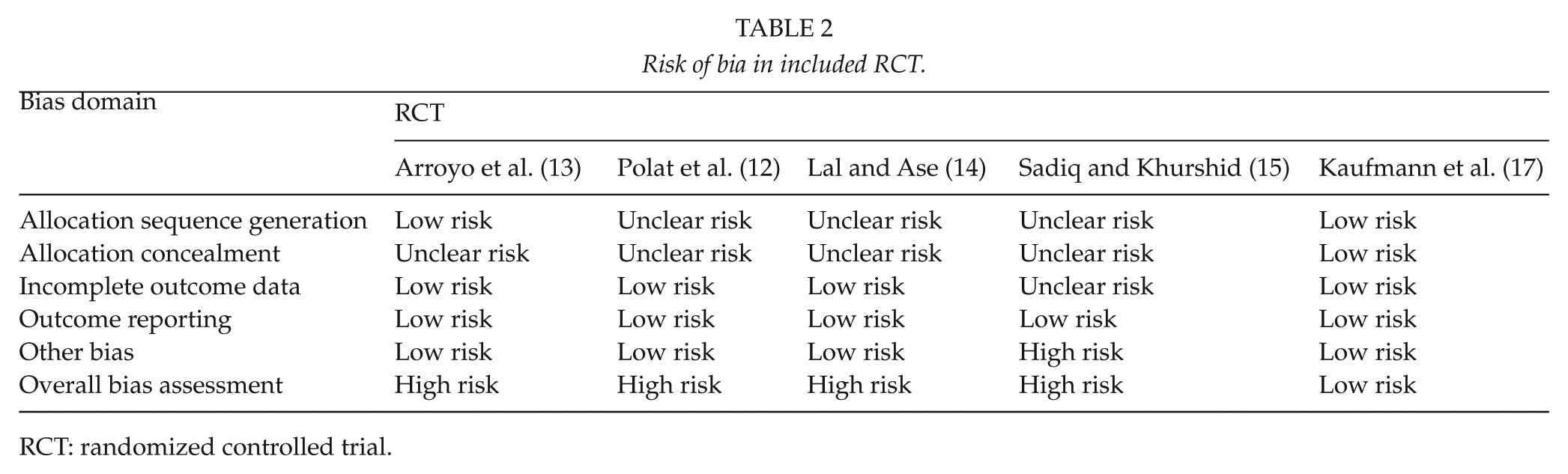
RCT: randomized controlled trial.
Meta-Analyses
We were able to gather data on recurrence from all five RCTs (12–17) (Fig. 1). In total, 326 patients were randomized to a mesh and 330 to sutured repair. Overall, mesh repair reduced the risk of recurrence with a relative risk (RR) of 0.28 (95% CI = 0.13–0.5; I2 = 0%; NNT = 13 patients; i.e. for every 13 patients undergoing elective repair for an umbilical hernia, use of mesh repair will prevent one hernia recurrence) (Fig. 2).
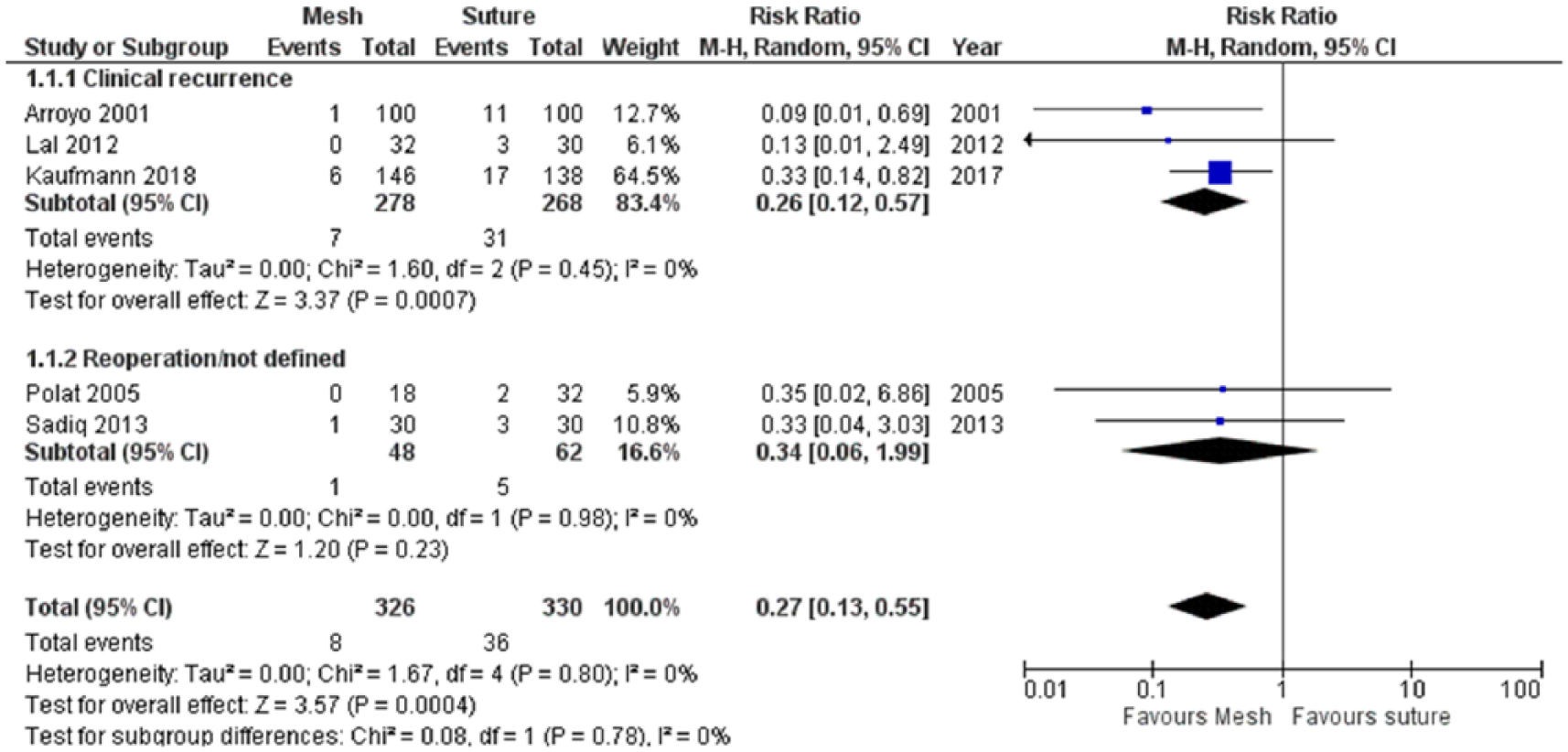
A Forest plot for risk of recurrence in favor of mesh versus suture repair by subtype using clinically assessment or not assessed clinically (reoperation for recurrence or non-defined recurrence) or combined total events.
In a sub-analysis of RCTs using either a clinically well-defined classification of recurrence or an unclear definition of recurrence, there was a significant difference between mesh repair or no mesh repair (RR = 0.24 (95% CI = 0.10–0.57) and RR 0.32 (95% CI = 0.05–2.03), test for subgroup differences, P = 0.06). In Trial Sequential Analysis, the Z-curve crossed the monitoring boundary after inclusion of all RCTs, suggesting that the result of the meta-analysis did not reflect random or systematic error.
As shown in Table 3, there were no differences between the two surgical techniques regarding infection (RR = 0.80, 95% CI = 0.36–1.79), seroma formation (RR = 1.38, 95% CI = 0.57–3.32), or hematomas (RR = 0.55, 95% CI = 0.23–1.30).
Relative risks of surgical outcomes in randomised controlled trials (RCTs) comparing mesh versus suture.
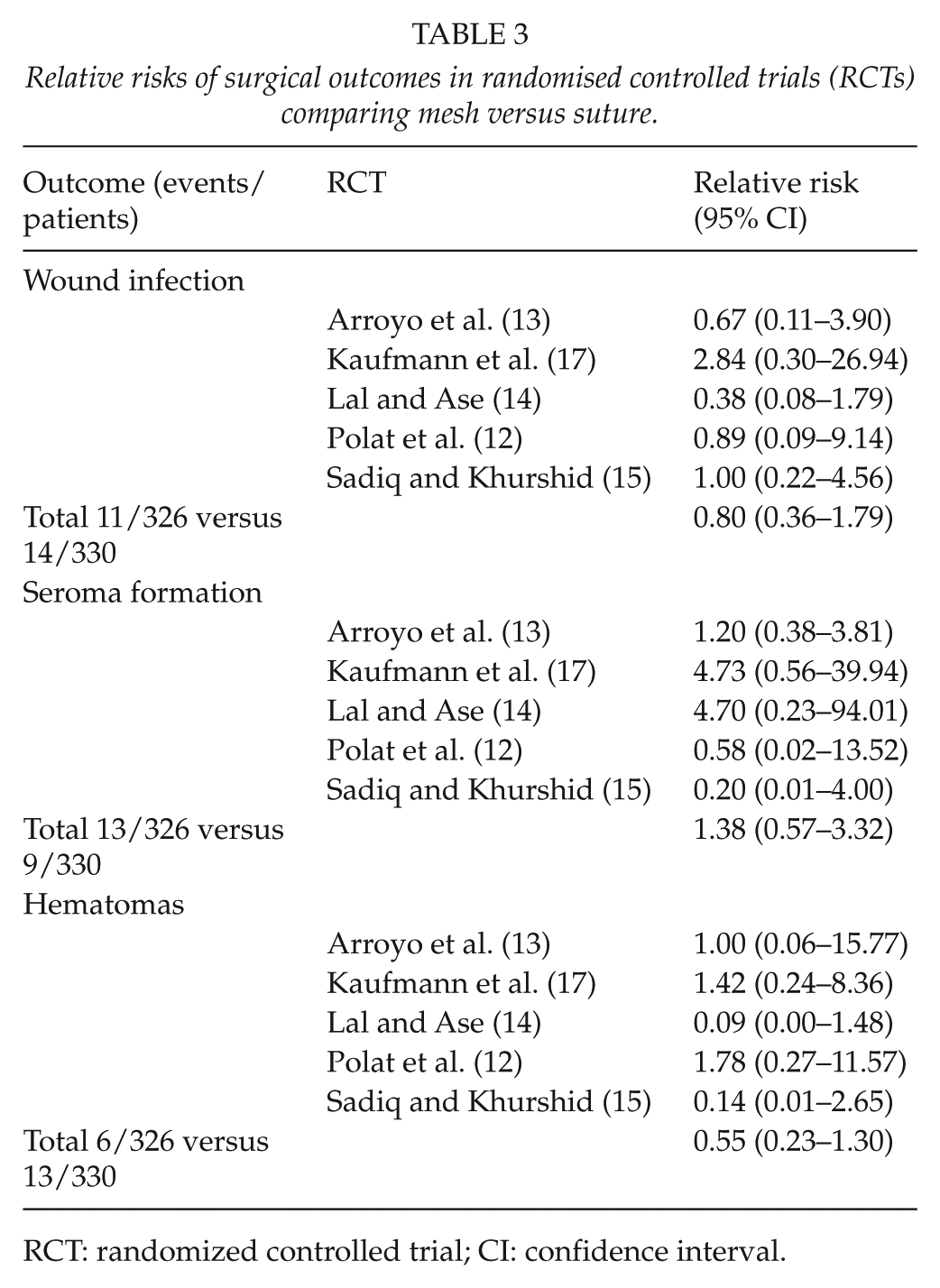
RCT: randomized controlled trial; CI: confidence interval.
Only one RCT reported data on acute and chronic pain and QoL (17). No significant difference was found in postoperative pain between mesh and suture repair during all time points. Two weeks after the operation, 102 patients (74%) in the suture repair group and 111 patients (76%) in the mesh group were free of pain (P = 0.57). After 2 years, 129 patients (93%) in the suture repair group and 138 patients (95%) in the mesh group were free of pain (P = 0.45). QoL was evaluated using the MOS SF-36 health survey (23) and the EQ-5D-5L (24). There was no significant difference at baseline preoperatively and 12 months after umbilical repair (P > 0.05).
Discussion
Based on the updated literature search, five RCTs were identified (12–17). The current analysis of the data revealed that mesh repair of umbilical hernia protected significantly against recurrence compared with a non-mesh sutured repair. The use of mesh repair did not increase risk of surgical site infection, seroma formation, hematomas, or chronic pain.
RCTs and on meta-analyses based exclusively on RCTs with low heterogeneity provide the highest level of evidence for the effects of treatment (25–27. However, surgical research questions, including hernia surgery, cannot always be answered through RCTs. Registry-based cohort studies tend to reflect the daily clinical practice (high external validity) but can be criticized due to risk of selection bias and possible confounding (28). In the present systematic review, all included RCT studies reported significantly lower risk of recurrence of umbilical hernia after mesh repair compared with a sutured repair. The findings are in line with the results from a recent regional cohort study reflecting daily surgical life (nationwide hernia databases are prone to provide high external validity opposed to high internal validity from RCTs) (5) by comprising 1313 patients from the Danish Ventral Hernia Database. The study demonstrated that mesh reinforcement for an umbilical hernia significantly lowered the risk of recurrence (5). On the other hand, a single-center study on 162 patients undergoing sutured repair for epigastric hernias performed by two dedicated surgeons found that a simple suture technique leads to acceptable low clinical recurrence rate of 6% (29). As mentioned above, previous published systematic reviews (7–9, 11) used methodology of problematic quality, including several mainly observational prospective and retrospective studies, emergency repairs, patients with liver cirrhosis, and a mixture of repairs for epigastric, incisional, and other types of ventral hernias. Furthermore, these meta-analyses included studies that did not clearly define primary outcomes in terms of recurrence. The consequence was therefore probably lack of robust quality and unclear definition of recurrence (20). Due to the RCTs of moderate or even lower methodology quality (7–9, 11), final conclusions from these meta-analyses are not possible. A high-quality RCT has recently been published and was therefore not included in previous systematic reviews (17). Kaufmann et al. (17) included 300 patients with an umbilical hernia, diameter 1 to 4 cm, and a 24- to 30-month follow-up with clinical examination. The findings from the Kaufmann study were in accordance with an earlier nationwide study with subgroup analyses of hernia defect size in relation to recurrence (>0–1 and >1–2 cm) (5).
Mesh position (onlay, sublay, etc.) has been suggested to be a risk factor for recurrence in incisional hernia repair (30). Recent evidence supports that anatomic position of mesh affects the risk of recurrence after incisional hernia repair (30), but this has been contradicted by a later long-term follow-up from the same study cohort (31). A previous nationwide cohort study on umbilical hernia repair (different mesh positioning compared with sutured repair; n = 4786) (32) and RCT data (onlay versus sublay mesh position; n = 80 (33)) reported no significant difference regarding mesh position for risk of recurrence, surgical site infection, and seroma formation in the onlay mesh position (33). In addition, the onlay mesh repair resulted in significantly shorter operation time (33). It may therefore be concluded that onlay mesh position for umbilical hernia repair is probably safe, efficient, and associated with comparable low risk of complication and recurrence rates although final conclusion is awaiting more solid data. Also, the necessary mesh overlap is not evidence-based.
In contrast to clinical results after groin hernia repair, chronic pain after umbilical hernia mesh repair has not been an often patient-reported outcome. In the recent RCT by Kaufmann et al. (17), 93% patients in the suture repair group and 95% in the mesh group were free of chronic pain 2 years after repair (P = 0.45). These findings were confirmed by the smaller Dalenbäck retrospective Single-center = USA Single-centre = UK language (n = 162) (29). Two other retrospective studies (n < 232) (3–5) and one prospective regional study (n = 1313) (5) found that an open umbilical or epigastric hernia repair induces chronic pain in 5% of the patients but with no difference between mesh or sutured repair. Christoffersen et al. (5) found that recurrence was the only independent risk factor for chronic pain and a previous retrospective study concluded that chronic complaints can, in part, be explained by recurrence (3). However, the overall lack of chronic pain as the primary outcome makes final conclusions difficult.
QoL has exclusively been reported by Kaufmann et al. using the MOS SF-36 health survey (17) and the EQ-5D-5L (24) preoperatively and at 12 months postoperatively. The QoL was without significant difference after umbilical repair.
The included RCTs in the present meta-analysis (12–17, 33) found seroma prevalence varying from 0% to 8% in the mesh group and 0% to 7% in the suture group after varying follow-up period. The inconsistencies of the results are comparable with previous literature and may be explained by the lack of definition and different diagnostic procedures. Thus, final conclusions on seroma formation with or without mesh repair are not possible.
There are several limitations of the present systematic review. Only one RCT of high quality was included (17). Furthermore, most studies included only few patients, and recurrence outcome was not based on a statistical power analysis with the risk of statistical type I and II errors. Another limitation is that several of the studies have included different surgical techniques in both groups, which may introduce some possible confounders. The literature search was limited to the English language, which may introduce a possible language bias. However, it has previously been shown that this restriction will probably have only limited impact on final conclusions (34). In addition, the extensive literature search across different databases limited the risk of omitting relevant literature. However, the most important limitation of the present analysis is the lack of high methodologic quality in the majority of the included studies and that studies, except one (17), were probably underpowered and comprised no prior statistical power analysis.
The strength of the present systematic review and meta-analysis is—in contrast to previous systematic reviews and meta-analyses—the coherence to a strict meta-analysis methodology to reduce risk of bias and subsequently an overestimation of intervention effects impeding final conclusions (16). The present analysis included exclusively patients undergoing elective umbilical hernia repair. A previous study has shown that emergency repair has significantly worse outcome than elective repair with up to 15-fold higher mortality, reoperation, and readmission rates (35). Therefore, the results on emergency and elective repairs are not comparable.
Future long-term high quality RCTs with a long-term follow-up with recurrence as primary outcome and chronic pain as secondary are awaited before final recommendations can be given on routine use of mesh repair in patients undergoing umbilical for defects ⩽1 cm. Until then, and based on convincing data from a large nationwide clinical database study (not included in the present meta-analysis), defects even ⩽1 cm may be closed using mesh repair.
In conclusion, mesh repair is probably safe and can be recommended for routine use to reduce the risk of recurrence after a small- and middle-sized umbilical hernia repair. More data are warranted for the optimal placement of mesh (sublay or onlay) and indication for mesh patients with an umbilical hernia defects <1 cm.
Footnotes
Acknowledgements
The paper is not based on any previous communications.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.B. reports support from Bard BD Nordic, Medtronic Danmark A/S, Robot Intuitive, B. Braun Denmark, Baxter Denmark, Cook Medical Denmark, Ethicon, Johnson & Johnson, Stryker Denmark, and Gore International, during the conduct of the study. None of the other authors report any disclosures.
