Abstract
Background:
Liver resection for colorectal liver metastases offers a 5-year survival rate of 25%–58%. This study aimed to analyze whether patients with colorectal liver metastases undergo resection to an equal extent and whether selection factors play a role in the selection process.
Material and Methods:
Data were retrieved from the Swedish Colorectal Cancer Registry (2007–2011) for colorectal cancer and colorectal liver metastases. The patients identified were linked to the Swedish Registry of Liver and Bile surgery and the National Patient Registry to identify whether liver surgery or ablative treatment was performed. Analyses for age, sex, type of primary tumor and treating hospital (university, county, or district), American Society of Anesthesiologists class, and radiology for detection of metastatic disease were performed.
Results:
Of 28,355 patients with colorectal cancer, 21.6% (6127/28,355) presented with liver metastases. Of the patients with liver metastases, 18.5% (1134/6127) underwent liver resection or ablation. The cumulative proportion of liver resection/ablation was 4% (1134/28,355) of all colorectal cancer. If “not bowel resected” were excluded, the proportion slightly increased to 4.7% (1134/24,262). Around 15% of the patients with metastases were registered as referrals for liver surgery. In a multivariable analysis patients treated at a university hospital for primary tumor were more frequently surgically treated for liver metastases (p < 0.0001). Patients with liver metastases from rectal cancer (p < 0.0001) and men more often underwent liver resection (p = 0.006). A difference was found between health-care regions for the frequency of liver surgery (p < 0.0001). Patients >70 years and those with American Society of Anesthesiologists class >2 underwent liver resection less frequently.
Conclusion:
Patients with colorectal liver metastases are unequally treated in Sweden, as indicated by the low referral rate. The proximity to a hepatobiliary unit seems important to enhance the patient’s chances of being offered liver surgery.
Introduction
Colorectal cancer (CRC) is one of the most common cancers globally. In Sweden, mortality in CRC patients is decreasing (above 10/100,000 out of an incidence of 50/100,000 per year (GLOBOCAN and NORDCAN). Liver metastasis is one of the main causes of death, being detected synchronously in 14%–20% and metachronously in another 15% of the patients (1, 2).
The only curative treatment is liver resection with a 5-year survival rate of 25%–58% (3 –6). Factors of importance in decisions involving liver surgery are the extent and distribution of metastases, response to chemotherapy, estimation of future liver remnant volume, and vascular in and outflow (7). These criteria often need to be evaluated at a multidisciplinary team (MDT) conference at a hepatobiliary unit with liver surgeons present, which has been identified as a factor to increase resection rates for colorectal liver metastases (8).
The Swedish health-care system, financed through taxation, is organized to provide equal cancer treatment to all patients, regardless of regional factors.
The aim of this study was to analyze whether patients with CRLM undergo resection to an equal extent in Sweden, and if not, whether selection factors can be linked to this lack of equality.
Materials and Methods
The present registry study encompassed all registered patients diagnosed with CRC and synchronous or metachronous liver metastases in Sweden from January 2007 to December 2011. The study also included all registered patients treated with liver resection or ablation for CRLM in 2007–2012.
Data were retrieved from the Swedish Colorectal Cancer Registry (SCCR), the Swedish Registry of Liver and Bile surgery (Sweliv), and the National Patient Registry (NPR).
All patients diagnosed with CRC in Sweden are registered in the SCCR, including clinical, demographic, and treatment data. The coverage of the SCCR is 98% (9).
Sweliv is a nationwide registry for liver surgery that includes ablative therapies. The registry covers all of Sweden (10 million inhabitants) and is administrated by the regional cancer centers (RCCs) in the six health-care regions of Sweden.
The NPR is administrated by the National Board of Health and Welfare, where registration of all episodes of hospital care and diagnoses in Sweden is mandatory. Liver surgery was performed in every region at six university hospitals, whereas CRC surgery was performed at 60 hospitals during the study period.
Data regarding diagnosis of CRC in Sweden during 2007–2011 were extracted from the SCCR in June 2014. Patients with synchronous liver metastases were identified from the registration form, while patients with metachronous liver metastases were identified from the 3- or 5-year follow-up. Moreover, clinical and demographic data, region, type of hospital that performed the CRC surgery, or diagnosed the CRC (university, county, or district hospital) were collected. Type of radiology performed for metastatic detection and data on referral to a liver MDT were also retrieved.
The patients identified in the SCCR were linked to Sweliv to identify whether liver surgery or ablative treatment was performed for CRLM (Fig. 1). For completeness of liver resections, linkage to the NPR for the study period was also conducted. Coverage for Sweliv was 88% and for NPR 95% with respect to liver surgery or ablative therapy. The mandatory personal identity number ensures accurate linkage between the registries.
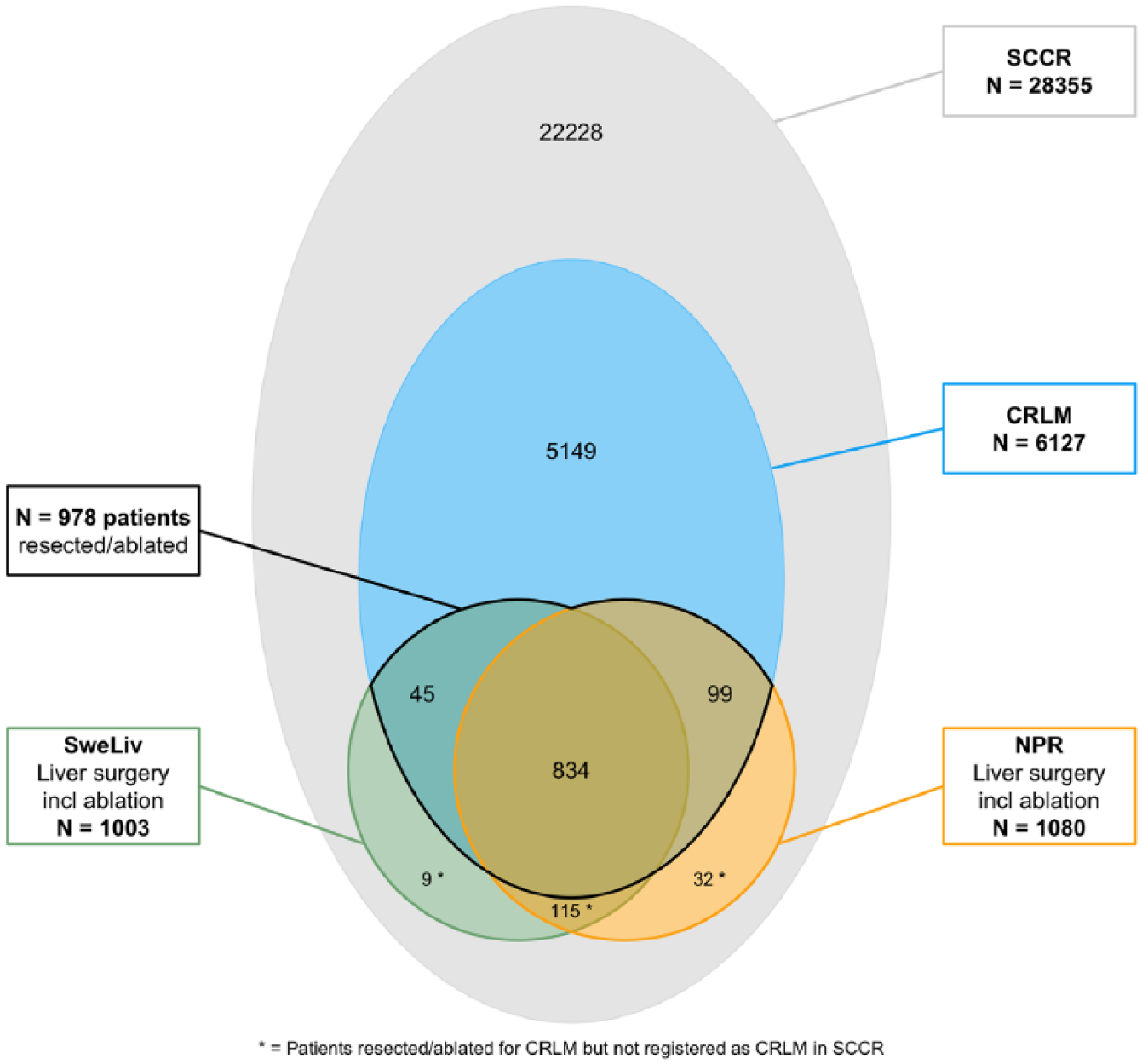
Patients registered with colorectal cancer in the Swedish Colorectal Cancer Registry in 2007–2011. Number of patients with their liver resected or ablated for liver metastases from colorectal cancer registered in the Swedish Registry of Liver and Bile surgery or the NPR in 2007–2012.
Ethical Approval
The study was approved by the local ethical board in Gothenburg (Dnr: 165-13).
Statistical Analysis
When two or more concurrent tumors were registered in the SCCR, duplicates (871/28,355) were removed by keeping the record with the earliest date of CRC diagnosis and any record containing information on liver metastases. Data were summarized in contingency tables.
Univariable logistic regression analyses were performed to assess the associations between the study variables, quantified in terms of 95% confidence intervals (CIs). To adjust for possible confounding factors of interest (type of primary tumor, age, sex, region, type of hospital, and American Society of Anesthesiologists (ASA) class) were included in a multivariable logistic regression analysis.
First-level interactions of sex and age against all other covariates were each entered into the model separately. None was found significant.
Goodness-of-fit of the final model was assessed using the le Cessie-van Houwelingen-Copas-Hosmer unweighted sum of squares test for global goodness of fit (10).
CIs and Wald tests were used to evaluate significance in the multivariable analysis. A p-value < 0.05 was considered statistically significant. Median and range were calculated when appropriate. Numbers and percentages were used as descriptive statistics.
All analyses were done using R 3.2.4 software.
Results
Demographic and clinical data for the 28,355 patients with CRC who were retrieved from the SCCR are presented in Table 1. Liver metastases were registered in 21.6% (6127/28,355) of the patients: 77% (4721/6127) were synchronous and 23% (1406/6127) metachronous. During the study period, median follow-up time from diagnosis of the primary tumor was 68.4 (20–102) months. Lung metastases were registered in 34% (2079/6127) of the patients with CRLM. A similar incidence of CRLM was seen in CRC patients. The male/female ratio was 1.26:1 in the whole cohort. Bowel resection had been performed in only 67% of the patients with CRLM compared with 91% of the patients without CRLM.
Demographic data for 28,355 patients from the Swedish Colorectal Cancer Registry (SCCR) with a CRC diagnos in 2007–2011.
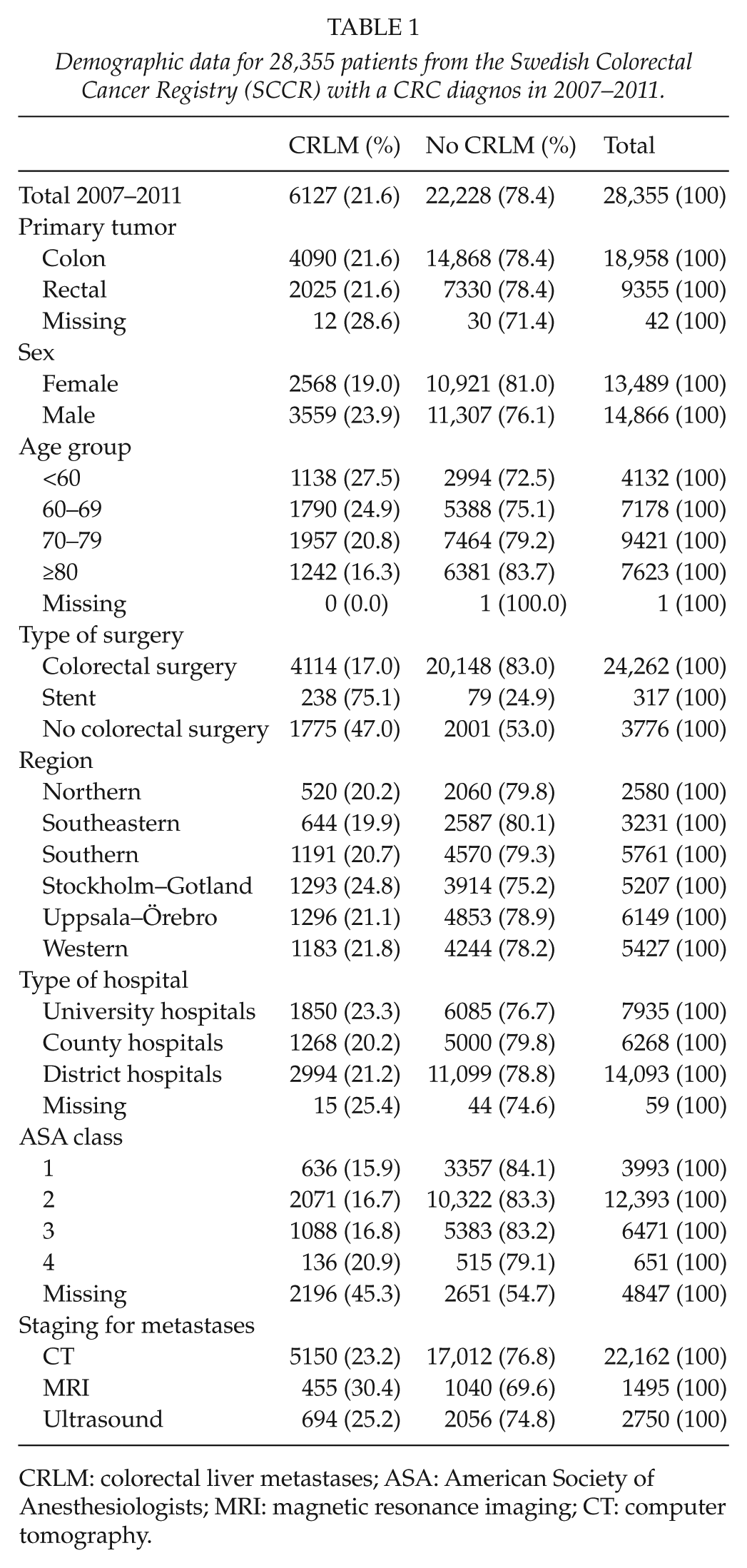
CRLM: colorectal liver metastases; ASA: American Society of Anesthesiologists; MRI: magnetic resonance imaging; CT: computer tomography.
Determination of Resectability for Metastases
According to the SCCR, 15% of the patients with CRLM were referred for evaluation of resectability of their metastases (65.6% is registered as no and 19.4% is missing data).
Patients treated for their primary tumor at a university hospital (19%) had a higher chance of liver surgery evaluation as compared with patients at county (14%) and district hospitals (13%; p < 0.0001). In addition, sex was identified, with more men (16%) being referred than women (14%; p = 0.023).
Patients with liver metastases from rectal cancer were referred more often (17%) than patients with colon cancer (14%; p = 0.001). Younger patients were also referred more often: 24% in patients <60 years, 19.7% in patients 60–69 years, 11.8% in patients 70–79 years, and 4.6% in patients >80 years (p < 0.0001).
Radiology for Liver Metastatic Detection from the Sccr
Computed tomography (CT) or magnetic resonance imaging (MRI) was performed in 82% of the patients. MRI of the liver was performed more often in men (p = 0.0001), in patients with rectal cancer (p < 0.0001), in patients <70 years (p < 0.0001), and at university hospitals (p < 0.0001).
Liver Surgery
In total, 18.5% (1134/6127) of patients with CRLM underwent liver resection (Table 2). Of these patients, ablation only was performed in 68 (5.9%) of the patients. Resection rates differed significantly between the six regions from 13% to 21% of all identified CRLM patients during the study period (p = 0.006). The cumulative proportion of liver resection or ablation was 4% (1134/28,355) of all CRCs. If patients who did not undergo bowel resection were excluded, the proportion was 4.7% (1134/24,262).
All patients registered with colorectal liver metastases in the Swedish colorectal Cancer Registry and patients who underwent liver resection or ablation.
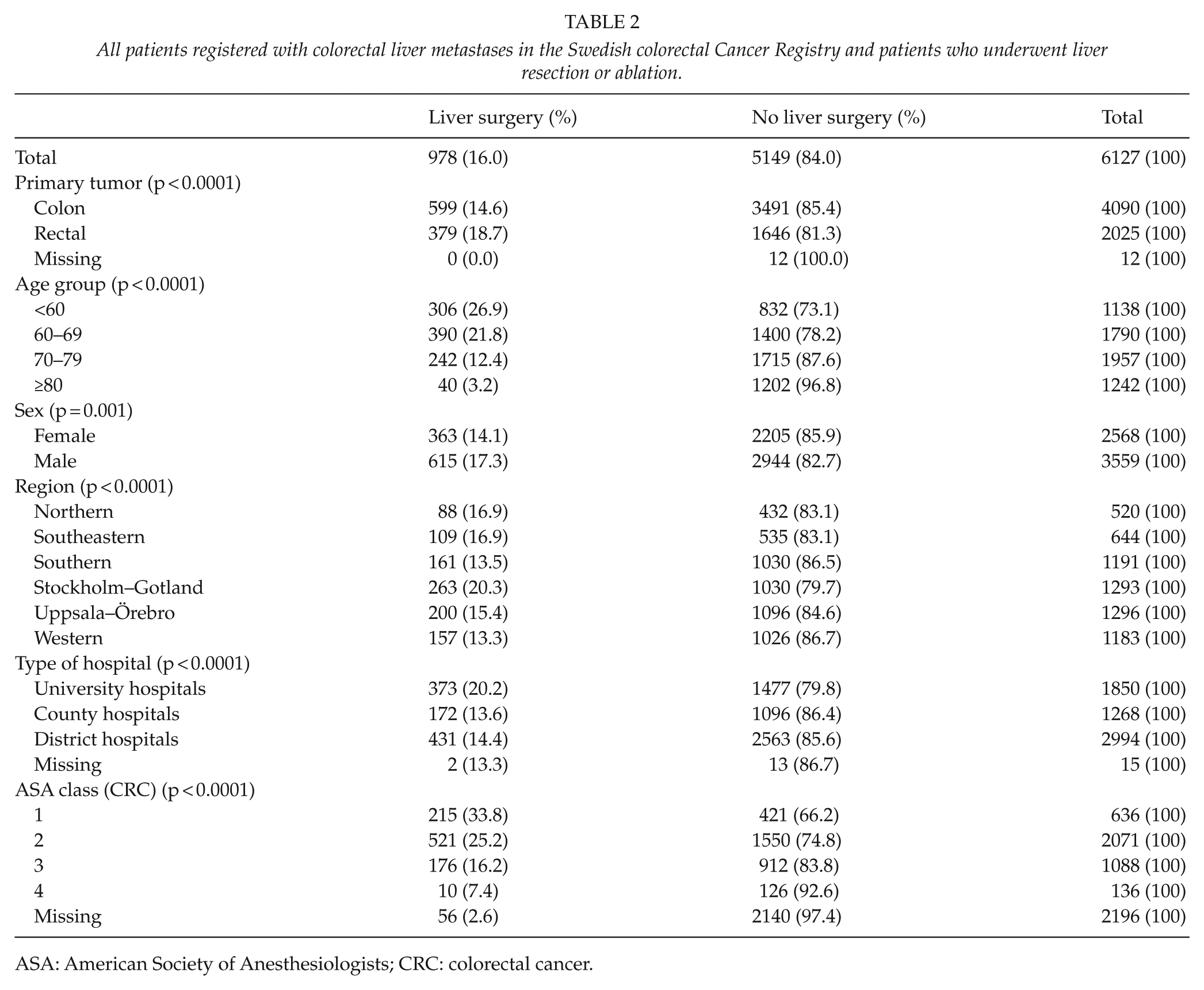
ASA: American Society of Anesthesiologists; CRC: colorectal cancer.
Single metastases were registered in 39.4% of the patients who had liver resection or ablation for liver metastases: 20.9% had two metastases, 11.7% had three metastases, 10.7% had four metastases, and 14.7% had ≥5 metastases. The number of metastases was missing in 2.6% of the cases. No differences were observed between health-care regions in the number of resected metastases. Preoperative diagnosis of lung metastases was recorded in 4.1% (40/978) of liver-resected patients.
For 156 liver-resected or ablated patients (of 1134 detected in Sweliv or NPR), no data on liver metastases were registered in the SCCR, and thus, these patients were excluded in the univariable and multivariate analyses.
Univariable and Multivariable Analyses
Variables included in the univariable and multivariable analyses for liver surgery or ablation were colon and rectal cancer, sex, age groups, region, type of hospital, and ASA. Patients treated at university hospitals for their primary tumor were more often resected for liver metastases (p < 0.0001). Liver metastases from rectal cancer (p < 0.0001) and men overall were more often liver resected (p = 0.006). A difference was also noted between health-care regions for frequency of liver surgery (p < 0.0001). Patients >70 years and patients with ASA class 2 or more underwent liver resection less frequently. Adjusted odds ratios and CIs from univariable and multivariable analyses are presented in Fig. 2 with forest plot.
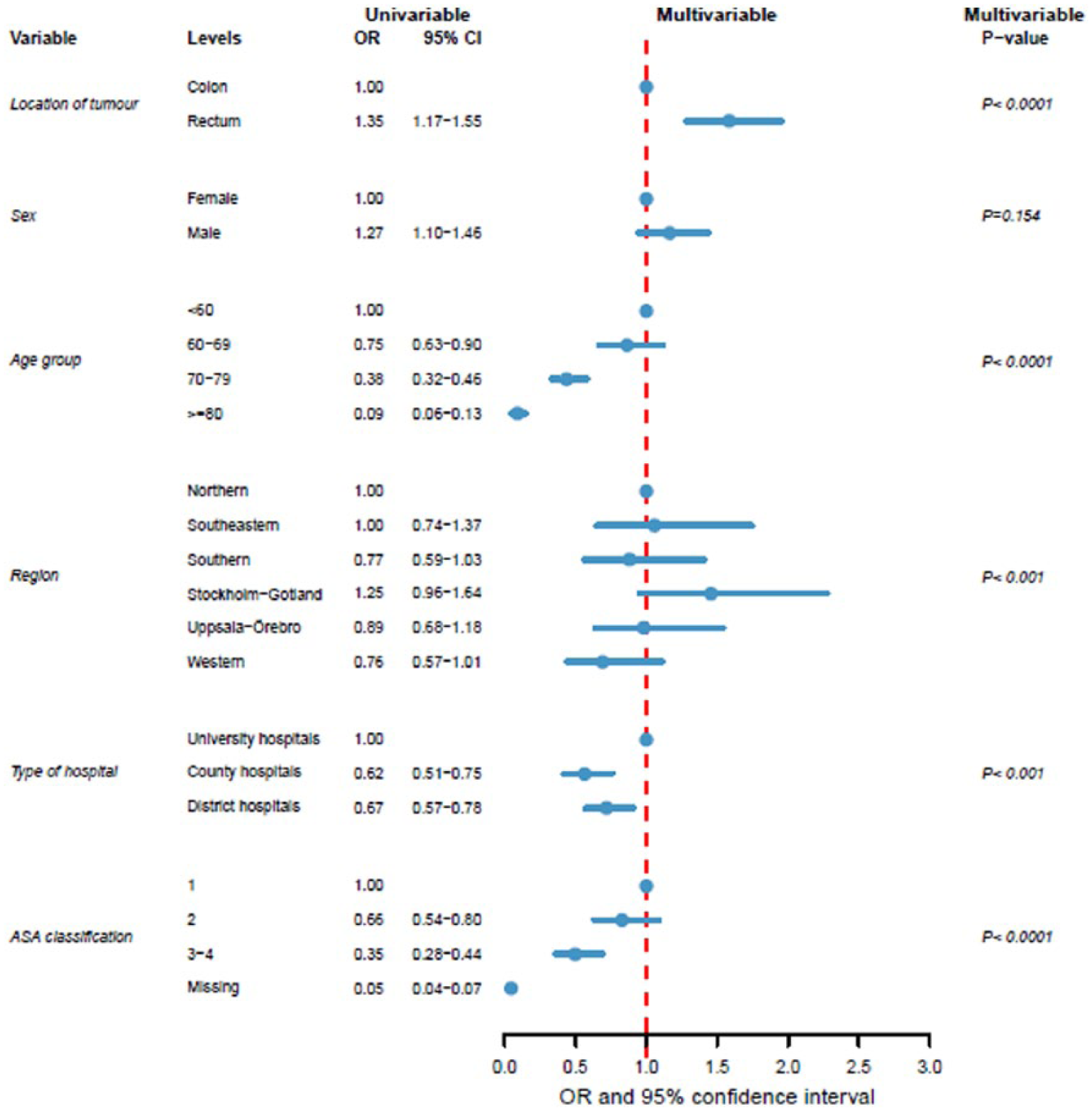
Univariable and multivariable analyses with multivariable Forest plot of patients resected/ablated for liver metastases. Analysis based on 6100 individuals with CRLM (missing data excluded).
Multivariable subgroup analysis of only the bowel-resected patients with CRLM (n = 4108) did not change the results (Supplementary Fig. 1). Furthermore, when analyzing the subgroup patients <70 years and ASA class 1 or 2, the same differences in sex, health-care region, and type of primary tumor and hospital were found (Supplementary Fig. 2).
Survival
The 5-year relative survival probability after liver resection including ablative therapies was 50.9% (95% CI = 47.5–54.5; Kaplan–Meier curve, Fig. 3).
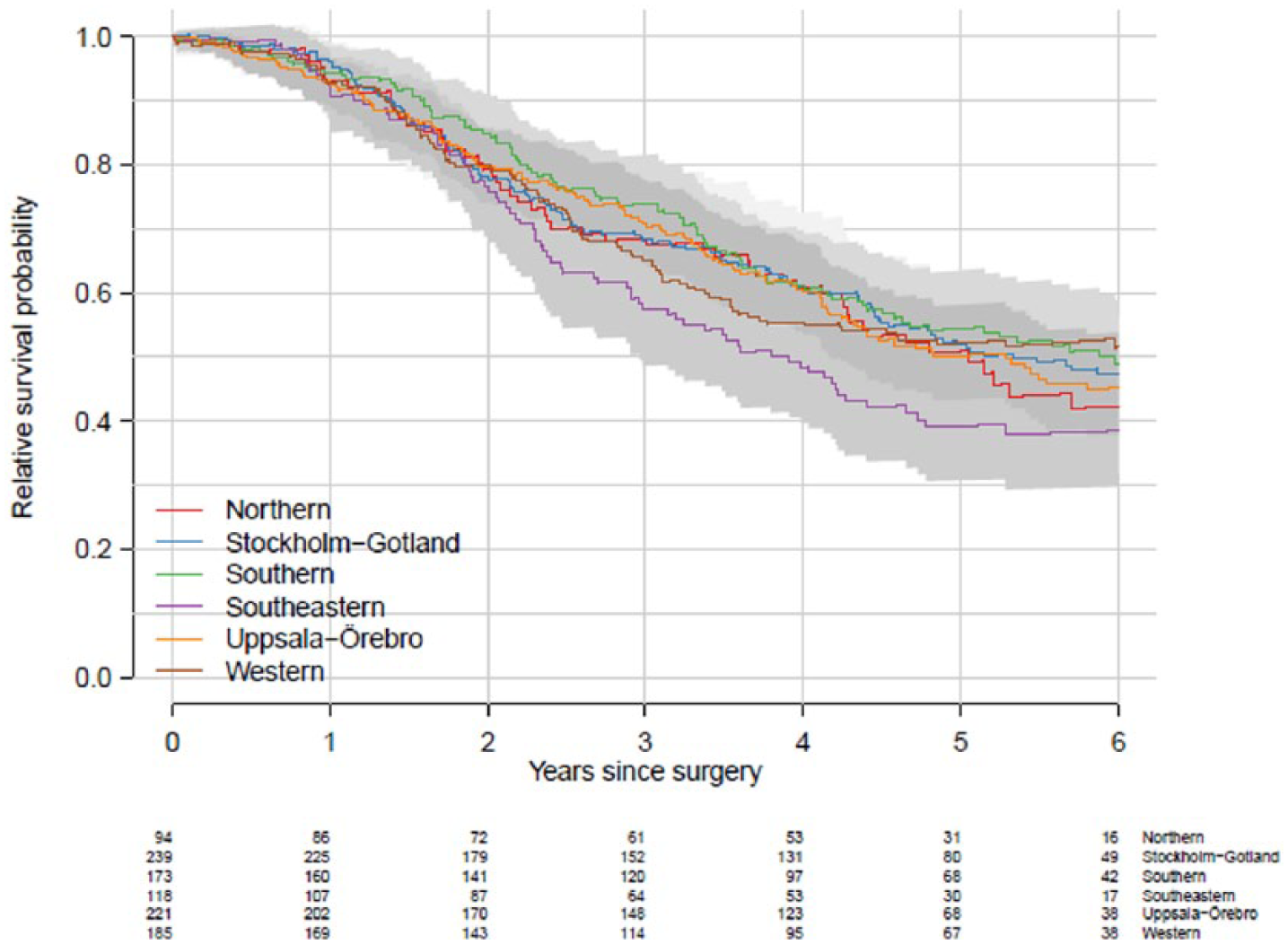
Five-year Kaplan–Meier relative survival curves after liver surgery or ablation in patients with CRLM (survival analysis was done in 2016). Note, more patients were liver resected in 2016 (n = 1030) than in 2015 in the same cohort (n = 975).
Discussion
The main findings suggest that patients with CRLM were not equally treated in the Swedish health-care system. Patients with a university hospital as their primary care source, being male, younger patients, and patients with rectal cancers were more often referred to liver specialists for evaluation and treated with liver resection. Even when excluding age >70 and ASA class >2, the same differences remained in favor of male patients, patients residing at university hospitals, and, in a subgroup analysis, patients with rectal cancer. There were also differences in the incidence of liver resection between health-care regions in Sweden.
The referral from colorectal surgeons to liver specialists could be studied because the CRC registry addressed this issue by asking about referral for metastatic surgery. The referral rate (15%) was lower than expected; the reason for this finding remains unclear. Perhaps registration for this parameter is not completely accurate or the view of surgery for metastatic disease is still negative among many colorectal surgeons. Older patients, patients from non-university hospitals, women, and colon cancer primaries were least referred. Still, no subgroup was referred at a higher rate than 25%, indicating that the main problem is referral in itself, showing that determination of liver surgical resectability is unequal in Sweden.
Surprisingly, we found that male patients were investigated more often with MRI liver assessment than female patients. Radiology for metastases was frequently often performed in men, perhaps reflecting the higher incidence of metastases in men.
The overall resection rate (including ablation) of CRLM in Sweden was 4.7% in CRC patients with primary tumor resection, which is the highest rate found in a population-based study. Other studies reported a liver resection rate of all CRCs to range from 2.7% to 3.6% (11, 12). The liver resection rate of all patients with CRLM was 18.5% in this nationwide setting. Differences were found between geographic regions in a recent Norwegian cohort study (6) in which 20% of the patients were resected for all CRLM. Similar population-based studies from earlier periods have described resection rates of CRLM from 16.5% to 24% (1, 2, 13).
The proximity to a hepatobiliary unit seems to be important to be able to receive liver surgery. We know from other studies that assessment of CRLM in a multidisciplinary setting with a liver surgeon present is important for survival (8) and assessment of resectability (14, 15).
The difference between a 3.2% liver resection rate in patients >80 years compared with 26.9% in patients <60 years (Table 2) cannot be explained by only the poorer results in the older age group (16). The complication rate correlates with both age and prognosis; however, with increased use of minimal invasive surgery and ablation, patients with poorer performance may be successfully treated as well (17).
The centralization of rectal cancer to university hospitals during the past decade might explain the difference between the liver resection rate in rectal and colon cancer. Rectal cancer patients are also younger. The earlier start of the rectal cancer registry (already started in 1995) followed by the colon cancer registry in 2007 may have put more focus on rectal cancer.
The differences between regions (13%–21%) indicate that practice differs between liver centers and referral patterns in each region. It may also indicate differences in policy between liver surgeons (18) or referrals (19). Analyses between regions in the number of liver metastases in each patient resected did not show any significant differences.
The sex difference is difficult to explain. However, one region with the highest ratio of liver surgery to number of patients with CRLM did not show sex differences.
The major strength of the study is the nationwide context and using registries with high coverage and reliable data. The major weakness of the study is the lack of staging data on metastatic spread for those not resected or ablated, which is not recorded in any registry in Sweden.
In conclusion, this study shows that a very low rate of patients is referred to liver specialists for evaluation of resectability for colorectal liver metastasis. The study also shows that when evaluated by liver surgeons, inequalities can be noted in the resection rate of CRLM between regions, types of hospitals treating the primary tumor, sex, type of CRC, and age. We predict that the greatest potential to increase liver surgery is to increase referral to liver MDT from all types of primary treating hospital. Because 5-year survival after liver surgery is high in Sweden, liver surgery should be offered to a greater extent than it is now for this diagnosis.
Awareness among patients and health professionals might increase efforts to achieve a professional consensus regarding resectability criteria that could serve to diminish inequalities in the future. To improve this situation a national treatment plan for CRLM has recently been established.
Supplemental Material
Supplementary_Figures_1_and_2 – Supplemental material for Identification Of Inequalities In The Selection Of Liver Surgery For Colorectal Liver Metastases In Sweden
Supplemental material, Supplementary_Figures_1_and_2 for Identification Of Inequalities In The Selection Of Liver Surgery For Colorectal Liver Metastases In Sweden by A. Norén, P. Sandström, K. Gunnarsdottir, B. Ardnor, B. Isaksson, G. Lindell, M. Rizell in Scandinavian Journal of Surgery
Footnotes
Declaration Of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
