Abstract
Colorectal cancer is the second most common cause of cancer-related death worldwide, and liver metastases are the leading cause of mortality in these patients. Surgical innovations combined with modern systemic chemotherapy have improved 5-year overall survival rates. This review aims to equip non-hepatobiliary surgeons with a comprehensive toolbox for the diagnosis and management of colorectal liver metastases. It covers the current landscape of diagnostic workup, including imaging modalities and biomarkers, and reviews the surgical and nonsurgical treatment options. This guide attempts to enhance multidisciplinary care for patients with colorectal liver metastases by integrating evidence-based practices with innovative strategies, which can improve outcomes and survival.
Keywords
Context and relevance
Colorectal liver metastases represent a major determinant of prognosis in patients with colorectal cancer. As therapeutic strategies become increasingly sophisticated, there is a growing need for non-hepatobiliary surgeons to be familiar with current diagnostic and management principles. This review addresses that need by providing a structured overview of contemporary approaches, with the goal of enhancing multidisciplinary coordination and promoting evidence-based clinical decision-making. In doing so, it aims to support improved oncologic outcomes within a complex and evolving treatment landscape.
Introduction
Colorectal cancer is the second leading cause of cancer-related death worldwide, with liver metastases being the predominant cause of death in this patient group. 1 Since the 1950s, increased understanding of hepatic functional capacity and anatomy, combined with the implementation of fluid-restrictive strategies in perioperative and postoperative care, paved the way for systematic liver resections. 2 Improvements in surgical techniques and modern systemic chemotherapy regimens have improved the 5-year overall survival (OS) rate for patients with colorectal liver metastases (CRLM) undergoing hepatic resection from 20% to 30% in 1970s to 40% to 50%.3,4
From the 1980s onwards, modern imaging techniques such as computed tomography (CT) and magnetic resonance imaging (MRI) have enabled preoperative anatomical, while intraoperative ultrasound facilitated the identification of tumors and critical anatomical landmarks. The challenge, however, remains in correlating tumor characteristics with long-term outcomes, prompting a shift toward more aggressive treatment strategies. 5 Furthermore, innovative approaches in the surgical management of CRLM have challenged traditional definitions of unresectable disease.6,7 In this review, we present a comprehensive toolbox for non-hepatobiliary surgeons regarding the diagnostic workup and treatment of patients with CRLM (Fig. 1).
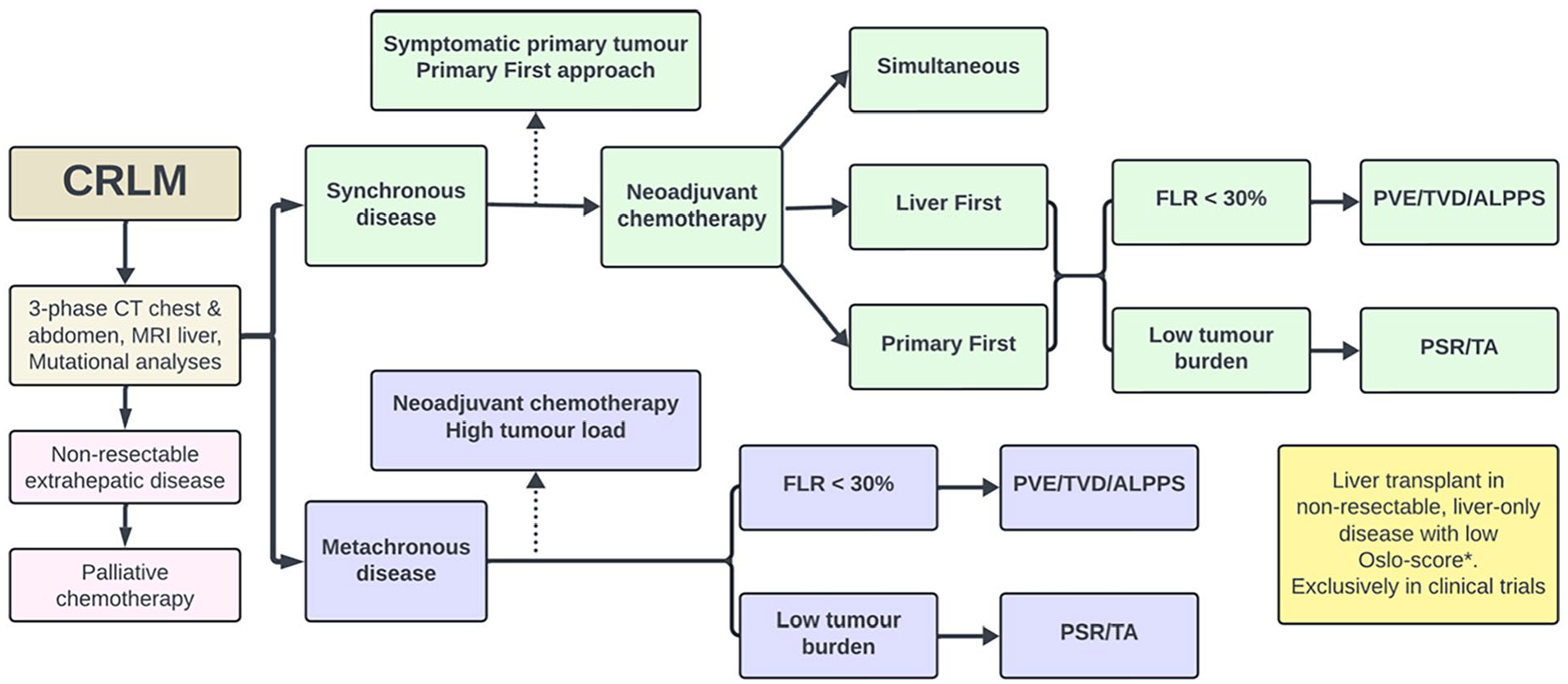
Flowchart of treatment options for patients with colorectal liver metastases.
Diagnostic workup
Multiple imaging techniques are utilized in the management of CRLM. Diagnostic preoperative imaging should include multiphase CT scans of the chest, abdomen, and pelvis, as well as MRI with liver-specific contrast agents. CT is preferred for initial staging, providing a preliminary evaluation of resectability and assessment of extrahepatic disease, while MRI is more sensitive than CT in detecting liver metastases, especially lesions <10 mm. 8 In addition, MRI can identify the presence of hepatic steatosis. Although ultrasound has low sensitivity for lesions smaller than 10 mm, it can help identify patients with diffuse secondary liver involvement who may not benefit from further hepatic imaging. Occasionally, contrast-enhanced ultrasound is used to evaluate liver lesions. Positron emission tomography-CT (PET-CT) may be employed to clarify any extrahepatic disease and avoid futile resections. 9 Liver biopsy is only indicated in cases of diagnostic uncertainty and generally avoided if surgery is planned due to the risk of tumor seeding. Bone and central nervous system metastases are rare, and bone scans and brain imaging are unnecessary for asymptomatic patients. Serum levels of carcinoembryonic antigen (CEA) and cancer antigen 19-9 (CA19-9) can help monitor treatment response but cannot confirm or refute the presence of CRLM. In the future, PET-MRI may become a valuable diagnostic tool, combining MRI’s high accuracy for CRLM detection with PET’s effectiveness in evaluating extrahepatic metastases. 10
The impact of genetics on selection and surgery
Patient selection is crucial, with personalized treatment strategies emerging based on molecular markers. In medical oncology, genetic markers are essential for predicting the efficacy of targeted treatments, such as identifying RAS wild-type status before anti–epidermal growth factor Receptor (EGFR)-targeted therapy or determining dMMR (deficient DNA mismatch repair) status before PD-1 blockade therapy. In surgical oncology, it is likely that genetic biomarkers will play a role in selecting the appropriate surgical strategy when it comes to timing and type of intervention.
RAS mutations have gained attention for their prognostic value predicting long-term outcomes, such as survival and site of recurrence, after resection of CRLM. 11 RAS mutations, in combination with other factors, can influence patient selection and impact surgical techniques, determining the necessity for extended resection margins. 12 While retrospective studies face limitations in generalizing findings to clinical practice, recent prospective studies have validated the association between RAS mutations and poor outcomes, strengthening their role as biomarkers. 13 Although BRAF mutations are less common in CRLM and associated with poor survival postresection, some patients with BRAF mutations still achieve acceptable outcomes. 14 In a retrospective multicenter study utilizing propensity score matching, BRAF-mutated patients with resectable liver-only CRLM experienced better outcomes with resection compared to systemic therapy. 15 Therefore, it remains challenging to use BRAF mutations to exclude patients from surgery. Combined mutational analysis is suggested to better predict patient outcomes and aid in patient selection. 16 Intra-tumoral and inter-tumoral heterogeneity pose challenges in treatment response, potentially requiring sophisticated testing methods like liquid biopsy. 17 Modern chemotherapy may influence molecular profiles, affecting treatment outcomes and selection biases. Currently, molecular biomarkers are assessed in line with other factors of survival such as response to preoperative chemotherapy, size, and the number of metastases in the workup of patients with CRLM. Personalized medicine is rapidly evolving, and with further research, genetic analyses may improve patient selection and treatment strategies for CRLM patients.
Perioperative systemic treatment
Preoperative treatment
Although surgery is the primary treatment modality for CRLM, most patients initially present with technically unresectable lesions. Chemotherapy can potentially convert these metastases to a resectable state. 18 Data from the international LiverMetSurvey registry, involving almost 25,000 patients, shows a notable 5-year OS of 33% among approximately 4500 initially unresectable patients. 19 The addition of a targeted agent to a cytotoxic doublet or triplet (fluoropyrimidine, oxaliplatin, irinotecan) is the most effective treatment and should be considered based on the molecular tumor profile and the patient performance status. 20
The EORTC trial demonstrated that perioperative chemotherapy for patients with initially resectable liver metastases resulted in improved progression-free survival; 21 however, there was no significant difference in OS. 22 Nonetheless, a subgroup analysis suggested that patients with elevated CEA levels might derive some benefit from perioperative chemotherapy. 23
Systemic chemotherapy often serves as neoadjuvant treatment in scenarios characterized by unfavorable predictors such as high CEA levels, multiple lesions, synchronous metastases, or bilobar disease. In this context of “biologically advanced” disease, the recommended regimen typically includes a combination of fluoropyrimidine and oxaliplatin. Surgery should be avoided in cases of disease progression during chemotherapy, as these patients exhibit poorer survival compared to those showing partial response or stable disease. 24
Postoperative treatment
The benefit of chemotherapy following liver resection remains unclear. While several randomized controlled trials have demonstrated statistically and clinically significant improvements in disease-free survival (DFS), this has not translated into improved OS. 25 These trials, however, are small compared to trials in adjuvant therapy in colon cancer. The ongoing randomized trial “OPTIMISE” investigates the utility of a ctDNA-guided approach in selecting patients for intensified adjuvant chemotherapy based on ctDNA positivity. 26 The results of this trial may provide further insights into the optimal use of adjuvant chemotherapy after liver resection.
Surgical treatment
Increased understanding of critical parenchyma volume (future liver remnant [FLR]) and of portal and liver vein circulation has transformed liver resection into a safe procedure, with perioperative mortality reported as low as 1%. 2 In a Norwegian cohort of 2,118 liver resections (2012–2016), the 90-day mortality rate was 2.1%. 27 All CRLM patients with reasonable physiological reserves and acceptable performance status should be considered for liver resection. Time to functional recovery following open liver surgery is reported to be 4–5 days in randomized trials, which is slightly longer than after minimally invasive liver surgery (MILS). 28
Laparoscopic MILS
Laparoscopic minimally invasive liver surgery (L-MILS) is an alternative to open liver surgery (Fig. 2A). Technical difficulties, survival benefit, postoperative quality of life, and costs should be considered when choosing the appropriate approach. Maneuvering within the confined abdominal space and achieving adequate exposure and vascular control can be more demanding than in open surgery. In MILS, certain liver segments are technically challenging to access due to their anatomical location, vascular supply, and proximity to critical structures. 29 Laparoscopic ultrasound can be helpful in these situations. Survival is equivalent for appropriately selected patients undergoing minimal invasive and open liver surgery. 30 Long-term data on oncological outcomes are, however, still being evaluated. Studies have shown superior short-term outcomes, such as reduced postoperative pain, hospital stay, and recovery time with L-MILS compared to open surgery. 31 While the initial costs for minimally invasive techniques might be higher due to equipment and training, potential benefits in postoperative care include reduced hospital stays and faster recovery times. 28
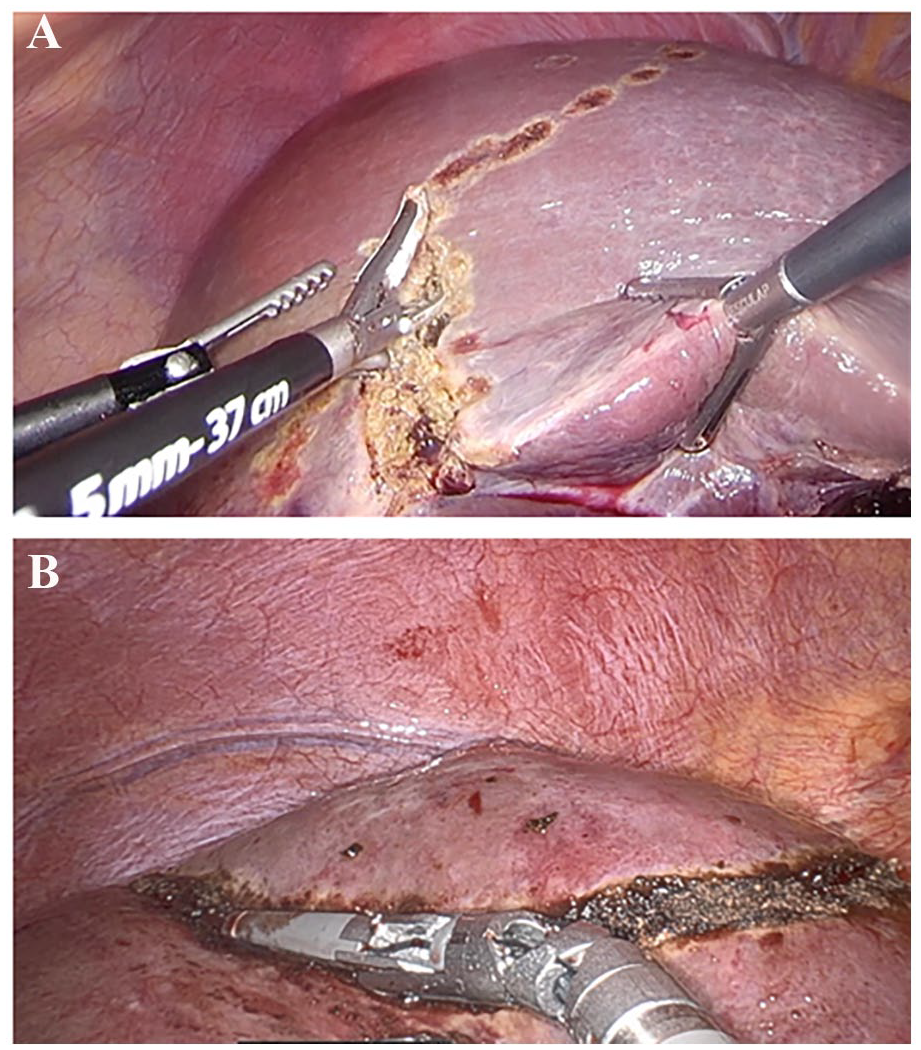
Intraoperative images of minimal invasive liver surgery (MILS). (A) Laparoscopic MILS with resection along the Cantlie’s line. (B) Robot-assisted MILS of liver segment 8.
Robot-assisted minimally invasive liver surgery
Robot-assisted minimally invasive liver surgery (R-MILS) for CRLM is less studied but increasingly adopted. 32 The benefits are primarily due to the articulation of the instruments, tremor filtration, easier suturing, improved camera techniques, and ergonomics. 33 The possible added value compared to the conventional laparoscopic approach has been suggested for resection in the posterior–superior liver segments and when hilar dissection is needed (Fig. 2B). 34 The learning curve for R-MILS appears shorter. 35 A large international multicenter study involving over 10,000 patients demonstrated higher rates of textbook outcomes in liver surgery with R-MILS compared to L-MILS (78.3% vs 71.8%, p < 0.001). 36 Other studies have indicated comparable results between R-MILS and L-MILS. 37 Overall, evidence is conflicting, and the question of whether the increased costs of R-MILS can be justified by some advantages over L-MILS remains unanswered.
Strategies to increase the FLR
Successful surgical resection of CRLM requires complete removal of tumor tissue while ensuring that the remaining liver remnant has both the volume and the quality to maintain adequate function. When preoperative evaluations indicate insufficient FLR, several methods can augment FLR volume.
Volume augmentation by venous manipulation
In the 1980s, Makuuchi and colleagues demonstrated that portal vein embolization (PVE) could induce hypertrophy of the FLR. 38 PVE is recommended when the FLR volume is insufficient, enhancing liver regeneration and preventing posthepatectomy liver failure. PVE-related complications are rare, but there is a risk of tumor progression during the hypertrophy period. Recently, simultaneous manipulation of both venous inflow and outflow, often referred to as portal vein embolization/hepatic vein embolization (PVE/HVE) or “total venous deprivation” (TVD) has gained significant interest. 39 Combined occlusion of both the portal inflow and drainage through the hepatic veins may yield volume expansion both in terms of growth rate and volume gained with lower complication rates compared to associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) (section “ALPPS”). 40 A prospective multicenter trial assessing the safety and feasibility of PVE/HVE was recently completed and will be published soon (NCT04272931). 41
Two-stage liver resection
Two-stage hepatectomy (TSH) is considered when the FLR volume is insufficient for a single operation. The first stage involves resection of metastases in the FLR, followed by PVE to induce hypertrophy of the remaining liver. After sufficient hypertrophy, the second stage includes resection of deportalized hemi-liver. TSH can achieve high resection rates, although it requires careful patient selection and management due to the risk of complications and disease progression between stages.
ALPPS
An alternative method termed the Associating Liver Partition and Portal vein ligation for Staged hepatectomy (ALPPS), first published in 2012, 42 combines portal vein occlusion with in situ liver partition to achieve rapid hypertrophy of the FLR. ALPPS has shown remarkable hypertrophy rates, with significant increases in FLR volume within 7–10 days. 6 Although some studies showed improved favorable outcome following ALPPS compared to PVE, 43 the method is now seldom applied as the primary method, due to relatively high complication rates.
Transarterial techniques for volume augmentation
Selective internal radiation therapy (SIRT; described in detail in section “SIRT”) of the diseased liver is another method for augmentation of FLR. 44 SIRT results in radiation-induced cell death of the diseased liver followed by contralateral volume expansion. The volume expansion is slower than seen in both PVE, ALPPS, and TVD but may halt tumor growth or even induce tumor shrinkage.
Application of the various techniques
If the FLR is >20%, PVE is usually sufficient to achieve adequate volume within 2–4 weeks. If adequate volume is not reached by PVE, one may wait for further expansion, providing the initial kinetic growth rate (KGR) is satisfactory. When initial KGR is low, rescue procedures, either by ALPPS 45 or additional venous deprivation, may be applied. TVD or ALPPS can also be considered as the initial method if the FLR is very small (<20%).
Simultaneous or staged resection
The optimal timing for surgical resection of synchronous liver metastases has been debated for decades. Various strategies have been proposed, yet high-level evidence remains limited. Simultaneous resection of the primary tumor and liver metastases has been examined in numerous retrospective studies and meta-analyses. 46 Potential advantages of simultaneous resections include the elimination of the tumor burden in a single procedure, reduced overall procedure time, shorter hospital stays, and anticipated improvements in quality of life, as well as decreased use of healthcare resources compared to staged procedures. However, concerns persist regarding the accumulation of complications and the impact on survival outcome. The optimal selection criteria for patients who would benefit from simultaneous resections remain unclear. 47
A randomized controlled trial found no significant difference in major complications between simultaneous and delayed resections, although recruitment challenges and logistical issues were highlighted. 48 The trial was underpowered to definitively determine the best approach. Given the current evidence, simultaneous resection should be considered primarily for patients with a limited hepatic disease. Additional studies are needed to assess quality of life, complication rates, survival outcomes, and the overall impact on healthcare resources associated with simultaneous resections. 47 In Europe, two randomized trials are investigating the role of simultaneous surgery, LIVACOR (NCT05138094) and SYLMET (NCT06200831).
Local ablative therapy
Local ablative therapy has increasingly been utilized for treating CRLM. Various methods are being used, including radiofrequency ablation (RFA), microwave ablation (MWA), cryoablation, and irreversible electroporation (IRE). 49 These procedures involve placing an energy applicator in or near the tumor, guided by imaging techniques such as ultrasound or CT.
Cryoablation destructs tumor tissue by freezing but is less commonly used for liver tumors, where RFA and MWA are more prevalent. 50 IRE is a non-thermal ablation modality that induces apoptosis by creating nanopores in cell membranes, preserving the collagen matrix and tissue structures. 51 IRE is particularly suitable for CRLM located near vital structures, such as central bile ducts, where thermal ablation is not feasible. MWA and RFA, collectively referred to as thermal ablation (TA), destruct tumors by heating the tissues. At temperature between 60 °C and 100 °C, they induce rapid protein coagulation and necrosis. While surgery remains the preferred treatment for resectable CRLM, studies have reported comparable survival rates between patients treated with partial hepatectomy and TA. The MAVERRIC study 52 indicated similar survival outcomes and a lower complication rate for TA. In addition, a randomized trial (COLLISION study) 53 comparing resection and TA is expected to be published soon. Two more randomized trials, NEW-COMET (NCT05129787) and HELARC (NCT02886104), are still recruiting, and their results may contribute to significantly impact future guidelines and clinical practices.
Hepatic arterial infusion pump therapy
Hepatic Arterial Infusion Pump (HAIP) therapy, pioneered by the Memorial Sloan Kettering Cancer Center (MSKCC)-group, suggested this to be an attractive modality in treating CRLM. 54 The preferred drug for this therapy, floxuridine (FUDR), achieves a 97% first-pass effect, significantly limiting systemic side effects and allowing high concentrations in the liver parenchyma and tumor tissue.
The efficacy of induction HAIP is currently being tested in multiple trials, including a multicenter trial in the United States (NCT05863195), a national trial in Norway (NCT04898504), and a multicenter trial in Europe (PUMP-IT, NCT04552093). Adjuvant HAIP, which aims to reduce liver recurrence rates, involves placing a pump immediately after the surgical resection of all known CRLM. This approach demonstrated benefits in a single-center randomized control trial in 1999 55 and was recently supported in a propensity score-matched study. 56 In addition, a non-randomized prospective cohort trial for patients with recurrent liver-only colorectal metastases has recently completed accrual (CCMO NL71691.078.19).
SIRT
SIRT, also known as transarterial radioembolization (TARE), involves infusing radioactive microspheres into the hepatic artery via a transfemoral or transradial catheter. This technique can target small, specific areas with a distal catheter position or treat larger regions such as a hemi-liver with a more central catheter position (Fig. 3). The primary radionuclides used are Yttrium-90 (Y-90) and Holmium-166, with Y-90 being the most studied and widely used.
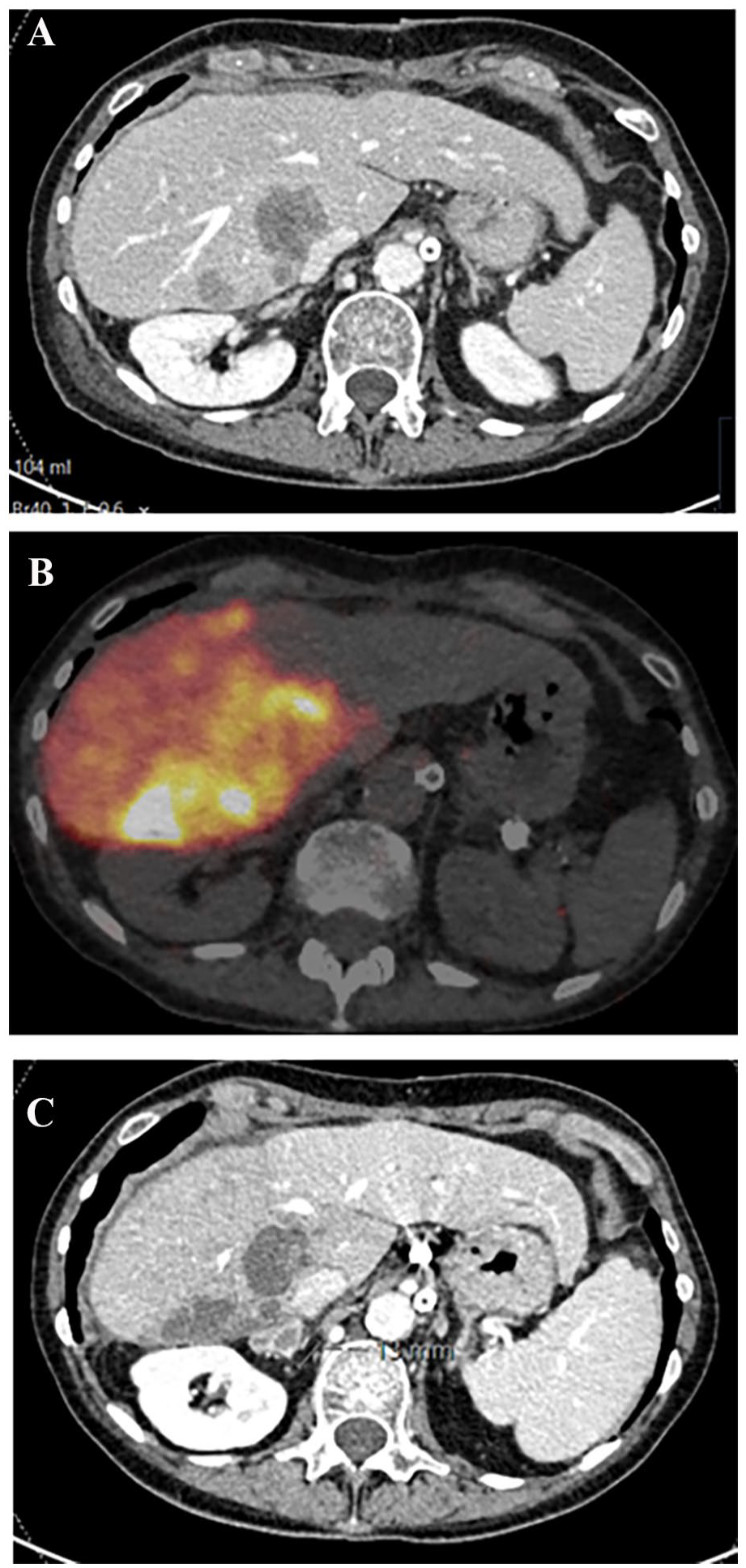
Selective internal radiation therapy (SIRT) in a patient with right-sided colorectal liver metastases. (A) Axial CT scan displaying the tumors adjacent to the caval vein in the right hemi-liver. (B) Axial PET-CT scan postinfusion of 3 GBq to the right hemi-liver. (C) Axial CT scan 2 months post-treatment, demonstrating response and intrahepatic disease control, with atrophy of the right lobe and hypertrophy of the left lobe. In addition, a new lesion indicating extrahepatic progression is observed in the right adrenal.
Three randomized controlled trials have compared FOLFOX combined with SIRT versus FOLFOX alone as first-line treatment. 57 A pooled analysis of these trials indicated a higher response rate with SIRT addition (72% versus 63%), but no significant difference in overall progression-free survival (PFS) and OS, both at 23 months. This lack of improvement in survival is likely due to the progression of extrahepatic disease, along with an increase in adverse events in the SIRT group. Consequently, most clinical guidelines do not recommend SIRT as a first-line treatment for CRLM, and SIRT is mainly reserved for patients who are either failing or intolerant to chemotherapy, or in salvage setting (Table 1).
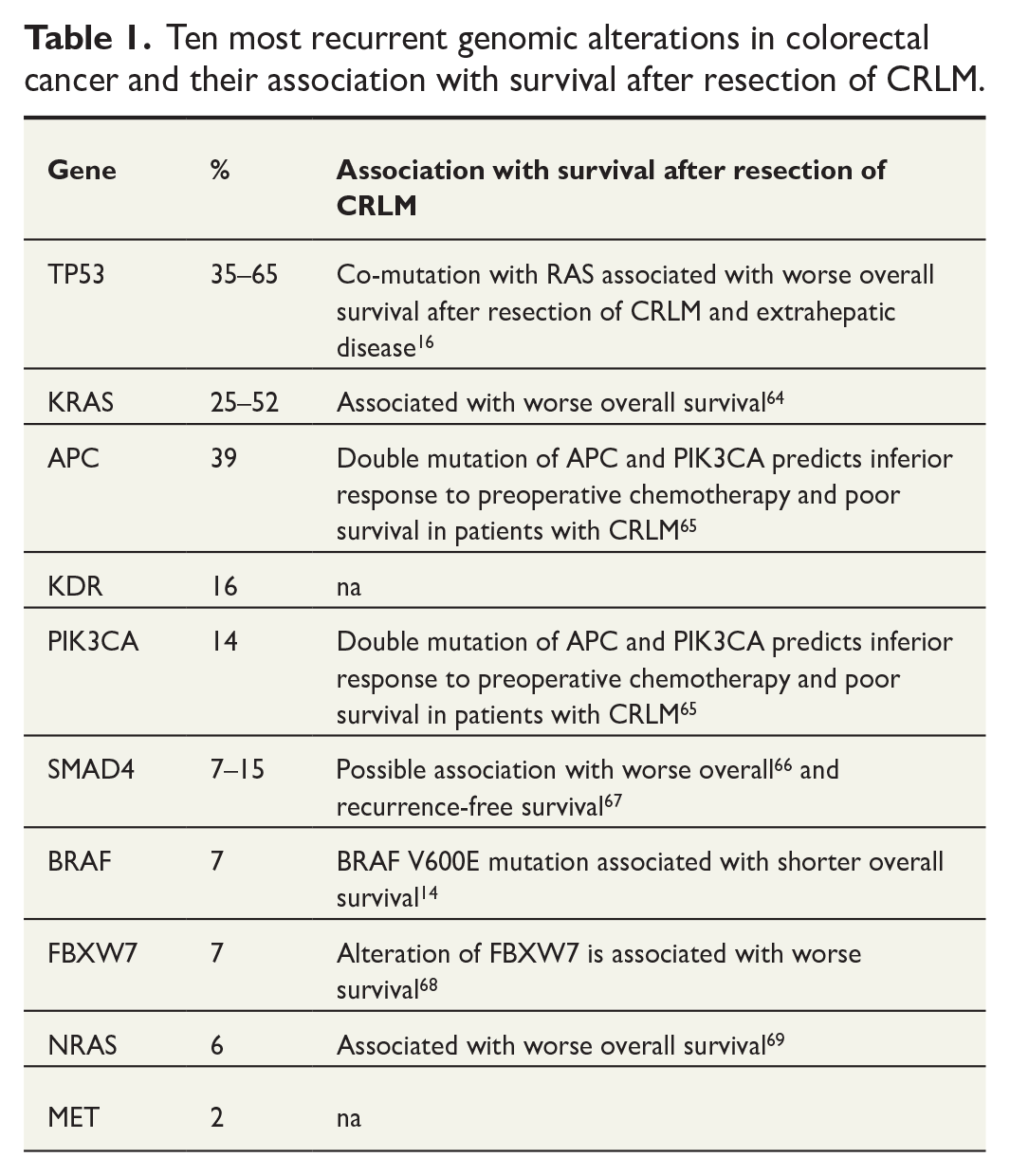
Ten most recurrent genomic alterations in colorectal cancer and their association with survival after resection of CRLM.
Future research should focus on proper dosimetry and refined patient selection to optimize the benefits of SIRT.
Liver transplantation
Transplantation for non-resectable CRLM was initially attempted during the early phase of liver transplantation (LT) but was abandoned due to poor survival outcomes. The improvements in imaging, oncological treatment, and surgical technique have revived interest in this concept during the last 15 years. Studies have demonstrated a 5-year survival rate of 60%–80% in highly selected patients. 58
For patients with CRLM to be eligible for LT, the disease must be confined to the liver, and the post-transplant immunosuppressive regimen should not negatively impact cancer-related outcomes. While recurrence after transplantation is common, it predominantly manifests as lung metastases, accounting for 50%–60% of relapses. Importantly, the growth rate of lung metastases is not worsened by immunosuppression, and many pulmonary lesions can be surgically resected with curative intent. 59
Significant negative predictive factors for post-transplant survival are incorporated into the Oslo-Score, which includes no response to chemotherapy, a pre-transplant CEA >80 μg/mL, the largest lesion >5.5 cm in diameter, and a time interval from diagnosis to transplant <2 years. 60 In addition, a metabolic tumor volume of less than 70 cm³ on pre-transplant PET-CT is a strong predictor of high 5-year survival probability. 61
LT for CRLM has gained increased international traction, and consensus guidelines for management were published in 2021. 62 The TRANSMET trial, 63 a randomized controlled study comparing LT plus adjuvant chemotherapy to chemotherapy only, demonstrated a significantly improved OS for the LT plus chemotherapy group. These findings suggest that LT should be a treatment option for highly selected patients with unresectable liver-only metastases.
Conclusion
The management of CRLM has evolved significantly, with advancements in surgical techniques, perioperative strategies, and systemic treatments contributing to improved patient outcomes. Surgical resection remains the treatment of choice in resectable disease and offers a possible curative option. Personalized treatment strategies based on genetic markers and innovative surgical approaches continue to enhance the care of patients with CRLM, offering new hope for extended survival and improved quality of life. Ongoing research and clinical trials will further refine these approaches, advancing the field toward more effective and individualized therapies for CRLM.
Footnotes
Author contributions
S.Y. contributed to the conception or design of the work. S.Y., A.L., K.L. contributed to drafting the article. S.Y., A.L., J.-H.A., K.W.B., U.C., B.E., Å.A.F, D.J.G., P.-D.L., B.I.R., T.M.S., E.S., T.S., O.V., and K.L. contributed to the critical revision of the article. S.Y., A.L., J.-H.A., K.W.B., U.C., B.E., Å.A.F., D.J.G., P.-D.L., B.I.R., T.M.S., E.S., T.S., O.V., and K.L. contributed to the final approval of the version to be published.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
