Abstract
Background and Aims:
Large-diameter head total hip arthroplasty and hip resurfacing arthroplasty were popular in Finland from 2000 to 2012 for the treatment of hip osteoarthritis. The aim of this retrospective study was to investigate the mid-term survival of large-diameter head total hip arthroplasty patients operated on in three university hospitals and to compare these results to the survival of hip resurfacing arthroplasty patients.
Material and Methods:
A total of 3860 hip arthroplasties (3029 large-diameter head total hip arthroplasties in 2734 patients and 831 hip resurfacing arthroplasties in 757 patients) were operated on between January 2004 and December 2009. The mean follow-up was 4.3 years (range: 0.3–8.0 years) in the total hip arthroplasty group and 5.1 years (range: 1.7–7.9 years) in the hip resurfacing arthroplasty group. Cox multiple regression model and Kaplan–Meier survival analysis were used to study the survival of the total hip arthroplasties and the hip resurfacing arthroplasties. Intraoperative complications and reasons for revisions were also evaluated.
Results:
In Cox regression analysis, the hazard ratio for revision of hip resurfacing arthroplasty was 1.5 compared with large-diameter head total hip arthroplasty (95% confidence interval: 1.0–2.2) (p = 0.029). The cumulative Kaplan–Meier survival rate was 90.7% at 7.7 years for the large-diameter head total hip arthroplasty (95% confidence interval: 86.8–94.6) and 92.2% at 7.6 years for hip resurfacing arthroplasty (95% confidence interval: 89.9–94.6). There were a total of 166/3029 (5.5%) intraoperative complications in the large-diameter head total hip arthroplasty group and 20/831 (2.4%) in the hip resurfacing arthroplasty group (p = 0.001). Revision for any reason was performed on 137/3029 (4.5%) of the arthroplasties in the large-diameter head total hip arthroplasty group and 52/831 (6.3%) in the hip resurfacing arthroplasty group (p = 0.04).
Conclusion:
The mid-term survival of both of these devices was poor, and revisions due to adverse reactions to metal debris will most likely rise at longer follow-up. There were more intraoperative complications in the large-diameter head total hip arthroplasty group than in the hip resurfacing arthroplasty group.
Introduction
Large-diameter head (LDH) metal-on-metal (MoM) total hip arthroplasty (THA) and hip resurfacing arthroplasty (HRA) became widely accepted alternatives to conventional metal-polyethylene bearing THA in the late 1990s for the treatment of hip osteoarthritis (OA) (1–3). One reason for the increased use of LDH THA and HRA was the modest long-term results associated with the metal-on-polyethylene bearing due to wear and osteolysis, especially among young and active people. Other reason for the popularity of LDH THA and HRA was based on the supposed increased functional ability as a consequence of the larger head size compared to smaller head size used in conventional THA. The number of LDH THA and HRA increased rapidly in the late 2000s and represented approximately 10% of all implanted hip devices (4). HRA offers several potential advantages over LDH THAs including preservation of own proximal femur, reduced risk of dislocation, retaining hip anatomical biomechanics, and easy revision to THA if needed (5). Intraoperative and post-operative complication profiles and revision rates of LDH THAs is different than HRAs based on implant surgery technique, implant design, and patient selection (5, 6).
After widespread use, it soon became obvious that there are specific problems related to MoM bearings, like periarticular fluid collections and even soft-tissue masses or gluteal muscle necrosis (5, 7). These findings were termed adverse reactions to metal debris (ARMeD). The British Orthopedic Association issued in April 2010 a statement to halt the use of MoM bearing surfaces (8). This was followed by a statement by the Finnish Arthroplasty Association in June 2012 not to use LDH THA or HRA (9). Despite these recommendations, some centers have still elected to use MoM bearings in selected patient groups.
The aim of this study was to investigate mid-term survival of LDH THA and HRA. In addition, intraoperative and early complications of these devices were evaluated. Our hypotheses were that LDH THAs have more intraoperative complications and also higher risk of revisions than HRAs do.
Material and Methods
Study Design
This is a retrospective, multicenter study. The participating university hospitals were Helsinki University Hospital, Kuopio University Hospital, and Turku University Hospital. Primary outcome of this study was survival rate comparison of LDH THAs over HRAs. Secondary outcome was comparison of complication and revision rates between LDH THA and HRA.
Patients
A total of 3860 hip arthroplasties in 3491 patients were performed between January 2004 and December 2009. There were 3029 stemmed THAs in 2734 patients and 831 HRAs in 757 patients. Of these hips, 2103 were males (54.5%) and 1757 females (45.6%). Among the patients, there were more males in both groups. In the HRA group, there were 570/831 (68.6%) males, and in the THA group, there were 1610/3208 (50.2%) males. The mean follow-up of study patients was 4.4 (range: 0.3–8.0 years) years. In the THA group, follow-up was 4.3 years (range: 0.3–8.0 years), and in the HRA group, 5.1 years (range: 1.7–7.9 years; standard deviation (SD) 5.1).
Demographic data (age, gender, side, indication for surgery, date of surgery) and surgical details (surgical approach, intraoperative complication, type, and name of components used) were collected from the medical records (Table 1 and Appendix 1). There were 16 different THA femoral components, 14 different THA acetabular components, and 6 different HRAs (Appendix 1). THAs and HRAs were performed according to the manufacturers’ instructions. Reasons for revision were evaluated, and the revision risk of LDH MoM THA was compared to revision risk of HRA.
Baseline demographic data.
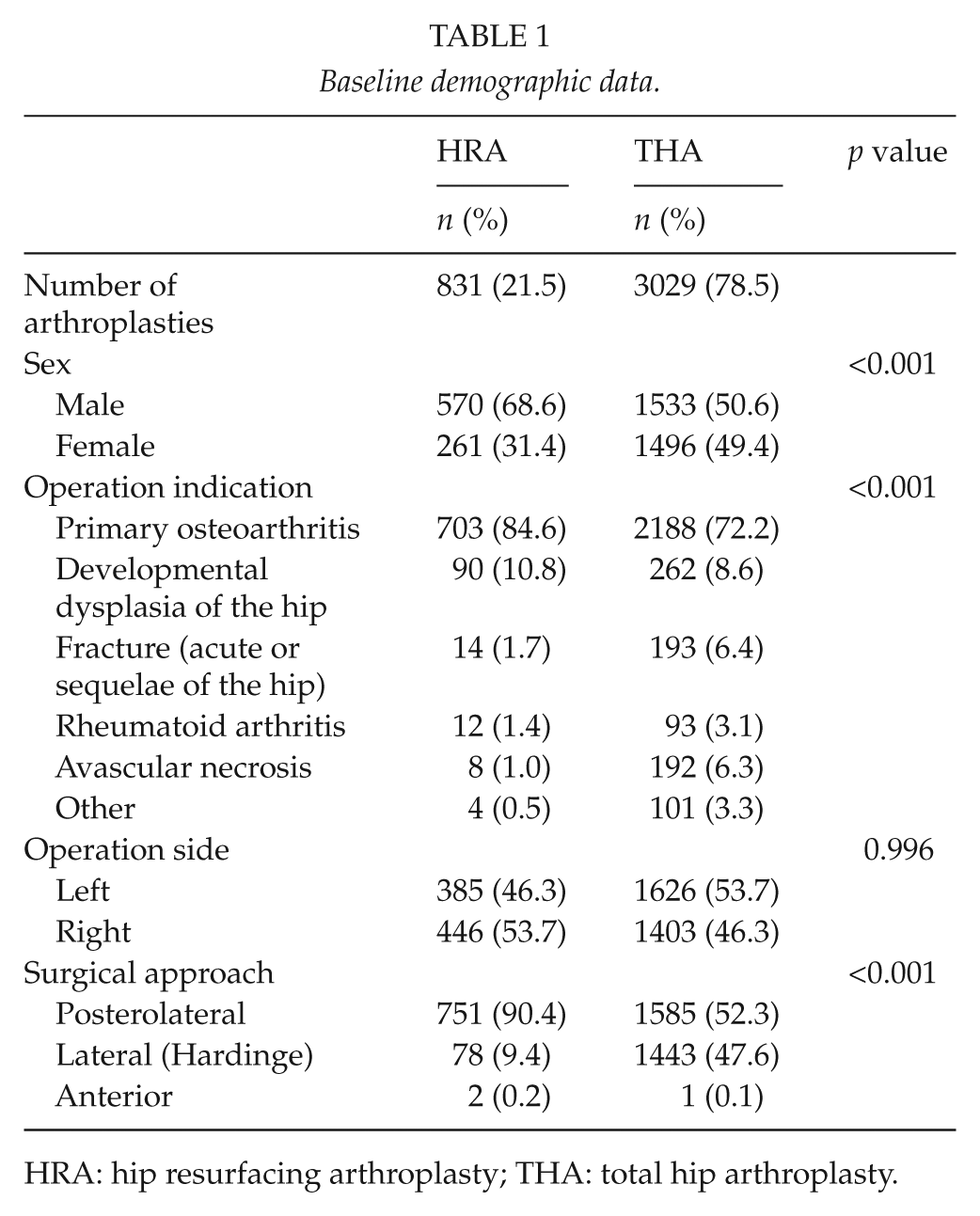
HRA: hip resurfacing arthroplasty; THA: total hip arthroplasty.
THAs with a head size of 38 mm or larger were included to this study, and THAs with smaller head sizes were excluded. The rationale for this selection was to standardize the effect of head size on the survival analysis of LDH THAs and HRAs.
Radiological Analysis
Radiological assessments were conducted from the anteroposterior (AP) full-weight-bearing and true-lateral post-operative radiographs. Anteversion and inclination angles of the acetabular component were measured according to the method described by Widmer (10). Picture archiving and communication systems (PACS) were used in every participating hospital. AGFA IMPAX (ver. 6.5.2.657) PACS was used at Helsinki University Hospital, and Sectra Workstation IDS7 (ver.15.1.8.5) PACS were used at Kuopio University Hospital and Turku University Hospital.
Statistical Analysis
Kaplan–Meier analysis was used to study implant survival. The outcome was any revision surgery, defined as removal or exchange of at least one of the prosthetic components. A Cox multiple regression model was used to assess revision risk (hazard ratio (HR)) with a 95% confidence interval (CI) and with adjustment hazard ratio (aHR) for age at surgery, gender, operated side, and diagnosis. Comparison of continuous data was carried out using a Mann–Whitney U-test. For categorical data, a chi-square test was used. A dot plot chart was used to evaluate the cup positioning of the revised components (anteversion and inclination angles). All p values ≤0.05 were considered statistically significant. All data were analyzed using SPSS (SPSS Inc., Chicago, IL, USA. Ver. 21.0.0, IBM).
Results
The cumulative Kaplan–Meier survival rate was 90.7% at 7.7 years for LDH THA (95% CI: 86.8–94.6) and 92.2% at 7.6 years for HRA (95% CI: 89.9–94.6) (p = 0.210) (Fig. 1).
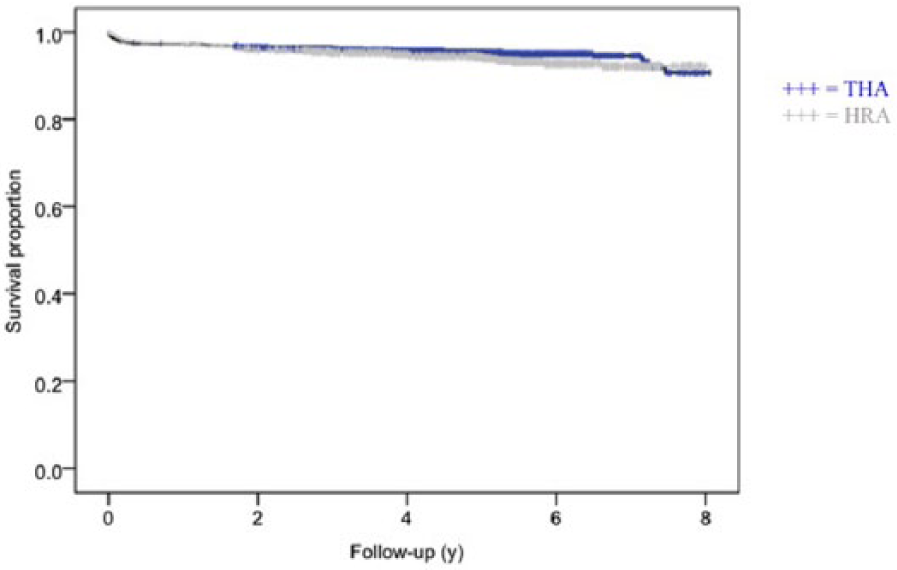
The cumulative Kaplan–Meier survival rate was 90.7% at 7.7 years for LDH THA (95% CI: 86.8–94.6) and 92.2% at 7.6 years for HRA (95% CI: 89.9–94.6) (p = 0.210). THA: total hip arthroplasty; HRA: hip resurfacing arthroplasty.
Mean time to revision was 1.2 years (range, 1 day–7.4 years) in the THA group and 1.9 years (range, 1 day–6.5 years) in the HRA group (p = 0.004). Revision for any reason was performed for 137/3029 (4.5%) hips in the THA group and for 52/831 (6.3%) hips in the HRA group (p = 0.040) (Table 2). There was a trend towards a higher incidence of periprosthetic fractures and component loosening in the HRA group compared with the THA group even though there was no statistically significant difference between the groups (p = 0.078). ARMeD was a reason for revision in the THA group for 17/3029 (0.6%) hips and in the HRA group for 3/831 (0.4%) hips (p = 0.078). However, if revisions due to ARMeD were excluded, revision was performed for 120/3012 (4.0%) hips in the THA group and for 49/828 (5.9%) hips in the HRA group (p = 0.016). Revisions were performed for 78/1496 (5.7%) females in the THA group and for 23/261 (8.8%) females in the HRA group, whereas in the THA and HRA groups, males had 59/1533 (3.8%) and 29/570 (5.1%) revisions, respectively. Overall, females had more revisions (5.7%) than males (4.2%) (p = 0.025).
Demographic data of revision surgeries.
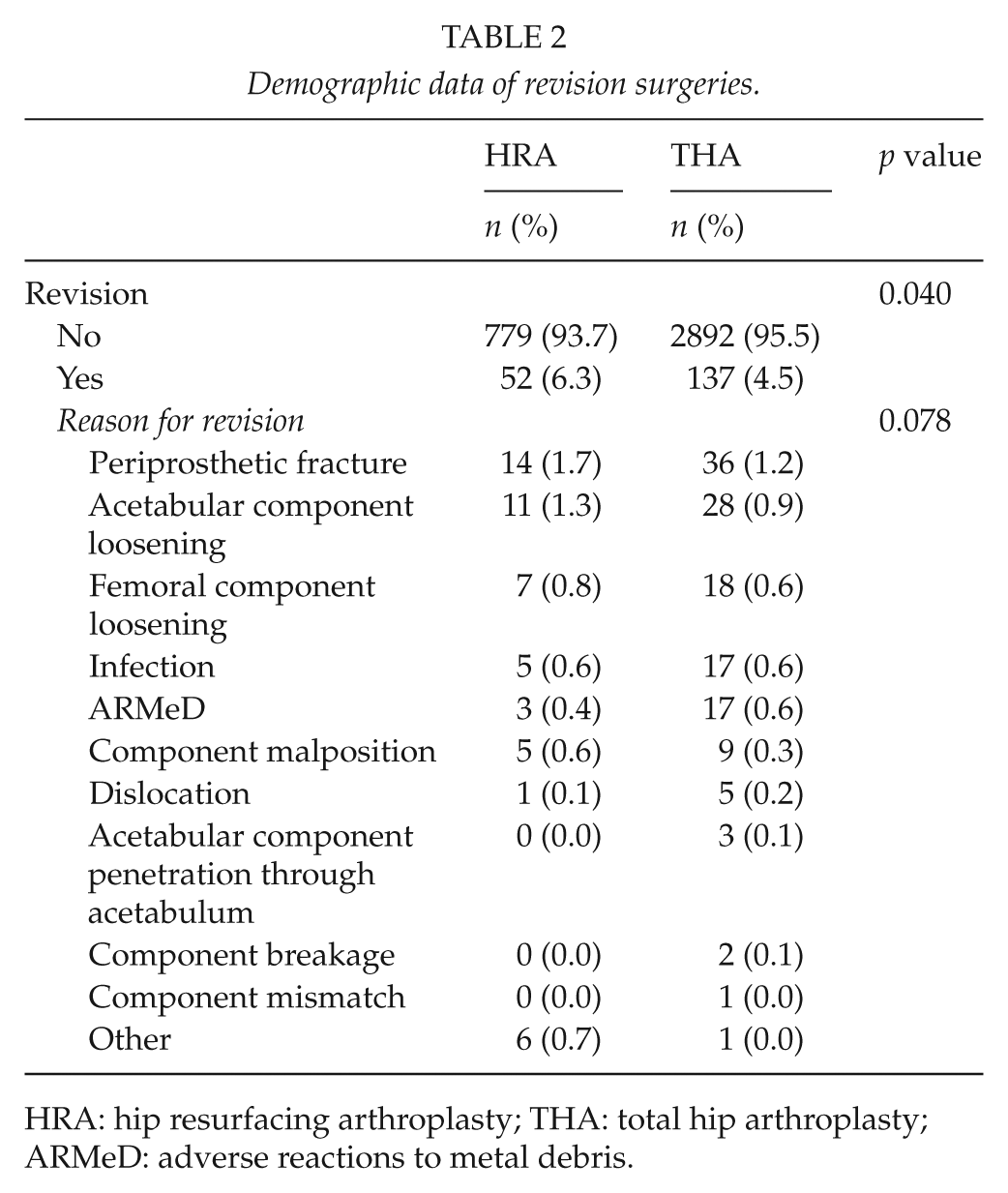
HRA: hip resurfacing arthroplasty; THA: total hip arthroplasty; ARMeD: adverse reactions to metal debris.
In the LDH THA group, there were 166/3029 (5.5%) intraoperative complications, whereas in the HRA group there was a total of 20/831 (2.4%) complications (p = 0.001) (Table 3). The most common intraoperative complication in the LDH THA group was femoral fracture at the calcar region (3.5%) and in the HRA group, it was nerve damage (1.3%) (p = 0.001) (Table 3).
Intraoperative complication data.
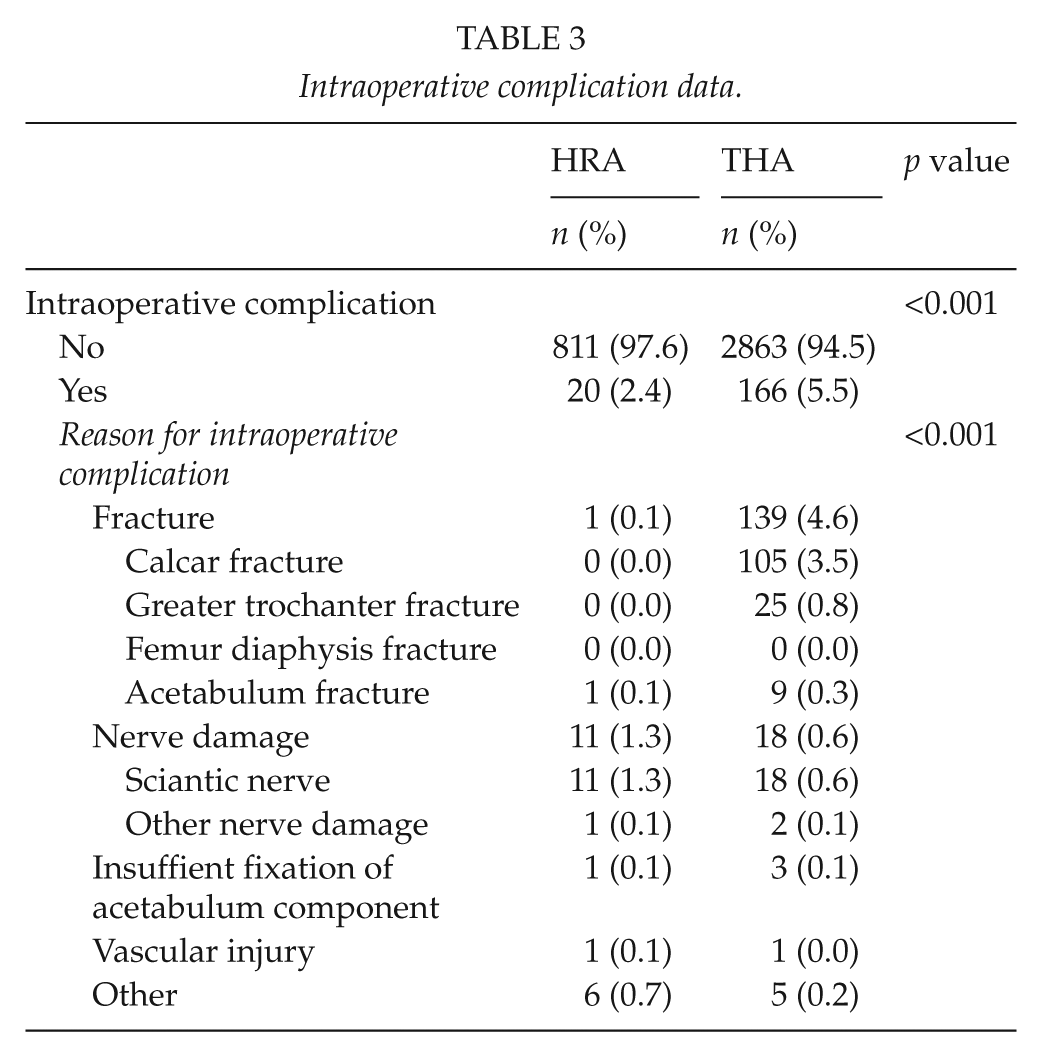
In the multivariable Cox regression model adjusted for age, sex, operation side, and operation diagnosis, HRA (aHR = 1.5 (95% CI: 1.0–2.2), p = 0.029) and female sex (aHR = 1.5 (95% CI: 1.1–2.0), p = 0.009) were associated with a higher risk of revision. The only significant operation diagnosis was fracture (aHR = 1.8 (95% CI: 1.1–3.1), p = 0.023) compared with primary OA. Age (aHR = 1.0) was not associated with risk of revision.
Specific brands of LDH THAs and HRAs were compared to determine whether there was any difference between the components in terms of the risk of having intraoperative complications or revision; there were no statistically significant differences between the components in either group (Appendix 1).
In the radiological analyses, the anteversion and inclination angles of the acetabular component were measured. In the THA group, mean anteversion angle was 20.8° (SD: 11.2, range: –35.0 to 60.0) and mean inclination was 44.6° (SD: 8.1, range: –17.0 to 91.0). In the HRA group, mean anteversion of the acetabular component was 19.6° (SD: 11.1, range: –29.4 to 59.0) and mean inclination was 45.4° (SD: 8.6, range: 5.0–93.0). There were statistically significant differences in the radiological measurements of the revised THA compared with non-revised THA patients. Mean anteversion angle of the acetabular component of the revised patients was lower (17.6°, range: –35.0 to 59.0; p = 0.001) and inclination angle was higher (47.8°, range: –17.0 to 93.0; p = 0.001). Dispersion of survived and revised acetabular component inclination and anteversion angles are shown in the dot plot (Fig. 2).
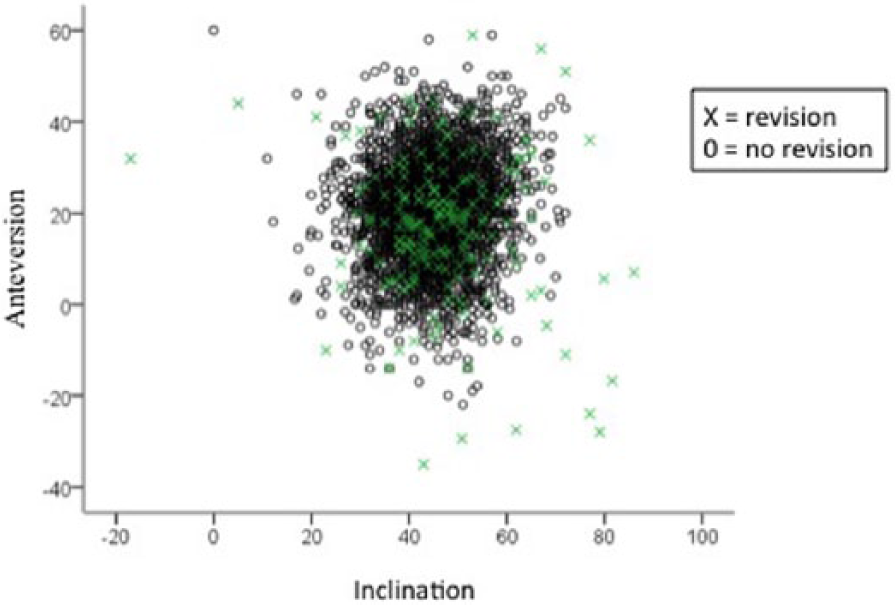
Dot plot showing anteversion and inclination angles of all acetabular components (n = 3860) measured from the 3-month follow-up radiographs.
Discussion
This study explored the mid-term survival of LDH THA and HRA. Intraoperative and early complications were also evaluated. The results showed that HRAs had 1.5 times higher HR for revision than LDH THAs. More intraoperative complications were noted in the LDH THA group compared to the HRA group.
The use of modular LDH THAs and HRAs with MoM bearings became very popular in 1990s and early years of 2000s (1–4). Short- and mid-term results of LDH MoM bearings were excellent, and the increased jumping distance from the large head gave inherent stability of the articulation (11, 12). However, studies with longer follow-up have shown high failure rates of MoM bearing THAs (13–17). Although both LDH THA and HRA have large head size MoM bearing, LDH THAs have had higher revision rates due ARMED, and this is probably caused by corrosion at the taper junction (18). In a recent register study by Junnila et al., short-term revision risk of LDH THA was not increased compared to analogous HRA in two of three studied device (Synergy/BHR THA vs BHR HRA and Bimetric/ReCap THA vs ReCap HRA). However, HRA had higher risk of revision in one of three devices (Corail and Summit/ASR THA vs ASR HRA) they studied (6).
The spectrum of the intraoperative complication is different in HRAs compared to THAs. Common reasons for failure and high revision rate are mechanical failures, such as femoral neck fracture in the HRAs and bearing-related problems such as soft-tissue reactions and osteolysis (19, 20). In LDH THA group, the incidence of the calcar fracture was 3.5% which is similar to previous studies where the incidence has been varied between 0.4% and 5.4% (21, 22). In our study, there were significantly more intraoperative femoral fractures in LDH THA group compared to HRA group. The reason for this is the difference in the surgical technique. When implanting the HRA, femoral component is cemented on the femoral head. When implanting the conventional THA, the femoral canal is broached, and stem is implanted using press-fit concept. There were two times more nerve injuries in HRA group compared to LDH THA group. In a previous study, incidence of nerve injury after HRA was 0.5%, which is smaller than our findings showed 1.3% (23). Nerve injury is most likely related to the more demanding acetabular exposure as the femoral neck is not osteotomized, thus increasing the susceptibility of stretching of the sciantic nerve. One major reason for THAs higher intraoperative complication rate is more heterogeneous operative indications compared to HRAs.
The revision rate was higher in the HRA group (6.3%) than in the THA group (4.5%). Our mid-term revision rates are comparable to those of previous studies in which the prevalence of revision for any reason varied from 1.7% to 4.1% (23, 24). In a recent study based on data from the Nordic Arthroplasty register at 6-year follow-up, the risk rate of revision for any reason was 1.5 times higher for MoM bearings compared with metal-on-polyethylene bearings (15). ARMeD has been the most common reason for revision of LDH MoM THA and HRA at 8-year follow-up in Australia and also in other studies with smaller sample sizes (4, 13–17). In our study, the most common reason for revision in the THA group was periprosthetic fracture (1.2%). The number of periprosthetic fractures was comparable between ours and other previous studies in which the incidence of post-operative fracture varied between 0.07% and 18% (23, 25). In our study, the second most common reason for revision was aseptic loosening either component (1.7%). Our results are similar to those of the Nordic Arthroplasty Register Association (NARA) study, in which aseptic loosening was the reason for revision in 1.9% of cases (15).
Fracture of the femoral neck was the most common reason for revision in the HRA group, with a prevalence of 1.7%. In previous studies, the prevalence of femoral neck fracture varied from 0.96% to 1.98% (5, 19, 23). In general, revisions in both groups were performed more often on females than on males. Previously, it has been shown that female gender is a risk factor for early revision in MoM arthroplasties (24). The specific component brand had no effect on the risk of revision or intraoperative complication in the THA or the HRA group (Appendix 1).
In the radiological analysis, we noticed that revised THAs and HRAs had smaller anteversion angles and larger inclination angles than survived THAs and HRAs. Previously, it was shown that component orientation is important to ensure optimal survivorship of THA patients, and our results support this finding (1). Measurements of the anteversion and inclination angles include all studied components, as well as early loosened acetabular components, which causes dispersion of the results and can be seen in the dot plot chart (Fig. 2). Acetabulum component malposition is a factor that contributes to a higher dislocation rate, edge loading, excessive metal surface wear, and pseudotumors (5, 26). Increased inclination and anteversion angles have also been shown to correlate with higher serum metal ion levels following MoM arthroplasties (5, 16, 27).
We are not aware of any published studies with the same size population and as detailed in radiographic measurements and patient demographic data as our study. However, our study had several limitations. This was a retrospective study based on patient record data of three university hospitals. Co-morbidity data were not included. Mean follow-up of our study was quite short, 4.3 years in the THA group and 5.1 years in the HRA group. Previously, it has been shown that a longer follow-up will reveal more problems related to MoM bearings, so a longer follow-up is needed in the future to see how revision rates between the groups develop. Furthermore, the strict definition of ARMeD at the time of these revisions were made was not clear. We did not measure whole blood metal ion levels, neither did we take hip magnetic resonance images for this study; this might underestimate the amount of ARMeD as a reason for revision. We did not include to our study THAs head sizes <38 mm because the complication profile of small head size THAs is different as well as survival rates are known to be poorer (28).
Conclusion
THAs have more intraoperative complications than HRAs. HRAs have higher mid-term revision rate than THAs; however, time to revision was longer. Reasons for a higher revision rate of the HRAs are female gender and a different operation technique in HRAs compared with THAs. The overall HR of HRAs was higher than that of THAs. Problems related to MoM bearings have evoked in recent years, and complications and revision rates are going to significantly increase with longer follow-up.
Footnotes
Appendix 1
Names and types of total hip arthroplasty components used.

| n (%) | |
|---|---|
| Conventional total hip arthroplasty | 3029 (78.5) |
| Femur components | |
| Fit-and-fill type | 2605 (86.0) |
| Biomet Bi-Metric (Biomet, Warsaw, IN, USA) | 2278 (75.2) |
| Synergy (Smith & Nephew, Memphis, TN, USA) | 321 (10.6) |
| Biomet Integral (Biomet, Warsaw, IN, USA) | 5 (0.2) |
| Mitch Symax (Stryker, Mahwah, NJ, USA) | 1 (0.0) |
| Tapered | 380 (12.5) |
| Conserve Profemur TL (Wright Medical Technology, Arlington, TN, USA) | 187 (6.2) |
| Metasul CLS (Zimmer, Warsaw, IN, USA) | 145 (4.8) |
| M/L Taper (Zimmer, Warsaw, IN, USA) | 46 (1.5) |
| Accolade (Stryker, Mahwah, NJ, USA) | 2 (0.1) |
| Other | 44 (1.5) |
| Biomet Reach (Biomet, Warsaw, IN, USA) | 31 (1.0) |
| Biomet CDH (Biomet, Warsaw, IN, USA) | 6 (0.2) |
| Metalic | 5 (0.2) |
| Biomet Balance (Biomet, Warsaw, IN, USA) | 1 (0.0) |
| Biomet Head/Neck (Biomet, Warsaw, IN, USA) | 1 (0.0) |
|
|
|
| Biomet ReCap (Biomet, Warsaw, IN, USA) | 1852 (61.1) |
| Biomet M2a38 (Biomet, Warsaw, IN, USA) | 469 (15.5) |
| BHR (Smith & Nephew, Memphis, TN, USA) | 246 (8.1) |
| Conserve (Wright Medical Technology, Arlington, TN, USA) | 188 (6.2) |
| Durom Cup (Zimmer, Warsaw, IN, USA) | 184 (6.1) |
| R3 (Smith & Nephew, Memphis, TN, USA) | 75 (2.5) |
| MMC Cup (Zimmer, Warsaw, IN, USA) | 6 (0.2) |
| Metalic | 5 (0.1) |
| Mitch TRH (Stryker, Mahwah, NJ, USA) | 3 (0.1) |
| Morscher Cup (Sulzer/Zimmer GmbH, Winterthur, Switzerland) | 1 (0.0) |
| Hip resurfacing arthroplasty | 831 (21.5) |
| BHR (Smith & Nephew, Memphis, TN, USA) | 467 (56.2) |
| Biomet ReCap (Biomet, Warsaw, IN, USA) | 157 (18.9) |
| Conserve Plus (Wright Medical Technology, Arlington, TN, USA) | 120 (14.4) |
| Cormet (Corin, Tampa, FL, USA) | 44 (5.3) |
| Durom (Zimmer, Warsaw, IN, USA) | 23 (2.8) |
| ASR (DePuy Orthopaedics, Warsaw, IN, USA) | 20 (2.4) |
| Total | 3860 (100) |
Acknowledgements
The authors thank PhD Inari Laaksonen for her assist and comments for this study. The authors also thank MD Henri Miettinen for his assist for this study. All authors participated in the planning and design of the study and in the interpretation of results. H.H. participated in planning the study protocol, performing statistical analyses, and preparing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The ethical review committee of University of Turku gave permission (ETMK: 78/1801/2013) for this study.
Funding
This study was made possible by support from the Finnish Arthroplasty Society, Finnish Research Foundation for Orthopedics and Traumatology, Research Foundation of Kuopio University Hospital, Finnish Cultural Foundation, North Savo Regional fund, and The Finnish Medical Foundation Duodecim.
Informed Consent
No consent was required.
Level of Evidence
Level III: a retrospective cohort study.
