Abstract
Background:
During abdominal surgery, traction of the mesenterium provokes mesenteric traction syndrome, including hypotension, tachycardia, and flushing, along with an increase in plasma prostacyclin (PGI2). We evaluated whether postoperative complications are related to mesenteric traction syndrome during esophagectomy.
Methods:
Flushing, hemodynamic variables, and plasma 6-keto-PGF1α were recorded during the abdominal part of open (n = 25) and robotically assisted (n = 25) esophagectomy. Postoperative complications were also registered, according to the Clavien-Dindo classification.
Results:
Flushing appeared in 17 (open) and 5 (robotically assisted) surgical cases (p = 0.001). Mean arterial pressure was stable during both types of surgeries, but infusion of vasopressors during the first hour of open surgery was related to development of widespread (Grade II) flushing (p = 0.036). For patients who developed flushing, heart rate and plasma 6-keto-PGF1α also increased (p = 0.001 and p < 0.001, respectively). Furthermore, severe postoperative complications were related to Grade II flushing (p = 0.037).
Conclusion:
Mesenteric traction syndrome manifests more frequently during open than robotically assisted esophagectomy, and postoperative complications appear to be associated with severe mesenteric traction syndrome.
Introduction
Gastrointestinal cancer is associated with a low level of long-term survival (1). The global incidence of gastro-esophageal junction cancer has increased by about 50% (2), and although surgery is the most significant treatment for long-term survival (3), postoperative complications remain a challenge (4, 5).
During abdominal surgery, organ manipulation and traction of the mesenterium provoke a triad of symptoms consisting of hypotension, tachycardia, and flushing—a mesenteric traction syndrome (MTS) (6, 7)—related to the release of prostacyclin (PGI2) (8, 9). MTS has an incidence from 32% to 85% in patients undergoing upper-gastrointestinal, pancreatic, and major abdominal vascular surgical procedures (6, 8, 10–12), with a lower incidence during minimally invasive surgery (12). It is unclear whether the incidence of MTS is equally low during robotically assisted laparoscopic surgery and whether postoperative complications relate to the development of MTS.
The current report evaluated if complications after esophagectomy relate to the development of MTS during the abdominal part of the operation, since postoperative morbidity is related to intraoperative hypotension (13). Our hypothesis is that robotically assisted laparoscopic surgery results in a lower incidence of MTS and intraoperative hypotension than does open surgery, which would attenuate the incidence of postoperative complications.
Methods
This article reports on secondary outcomes from a study investigating the gastric microcirculation during open versus robotically assisted hybrid esophagectomy. The protocol was approved by the Ethical Committees, Capital Region, Denmark (No. H-2-2013-101) and registered at clinicaltrials.gov (ID: NCT02077673). Adult patients with gastro-esophageal junction cancer scheduled for an open or robotically assisted Ivor-Lewis procedure were screened for eligibility. Patients suspected of disseminated disease were excluded, as were patients treated with nonsteroidal anti-inflammatory drugs (NSAIDs) or glucocorticoids at the time of surgery. Both oral and written informed consent were obtained from the patients at least 1 day prior to surgery.
Anesthesia and Monitoring
Patients were anesthetized according to a standardized regime. After the establishment of peripheral venous access, an epidural catheter was placed at the seventh to eighth or eighth to ninth intervertebral space. Correct location of the epidural catheter was tested with 3 mL lidocaine with adrenaline (komb. 2%, SAD, Amgros I/S, Denmark), followed by bolus bupivacaine (4 mL, 5 mg/mL, SAD). For intraoperative analgesia, infusion of 4 mL/h bupivacaine/morphine (komb. 2.5 mg/50 µg/mL, SAD) was activated before starting the surgical procedure.
General anesthesia was induced with remifentanil (0.5 µg/kg/min) and propofol (2.0 mg/kg). Placement of a double-lumen endobronchial tube was facilitated by cisatracurium (0.10–0.15 mg/kg). Anesthesia was maintained with remifentanil (1.75–2.25 mg/h) and propofol (5–10 mg/kg/h), and patients were ventilated to maintain an end-tidal CO2-tension of 28–32 mmHg (Dräger CATO; M32040, Lübeck, Germany). After induction of anesthesia, a catheter was placed in the radial artery of the nondominant arm for hemodynamic monitoring as continuously recorded by Nexfin® (BMEYE B. V., Amsterdam, The Netherlands) (14). A central venous catheter was also placed through the right internal jugular vein for infusion of fluids, and vasopressors were used if mean arterial pressure (MAP) was lower than 60 mmHg. Ephedrine was provided as bolus injections, and phenylephrine could be provided both as bolus injections and as infusion. Warmed, lactated Ringer’s solution was provided at approximately 3 mL/kg/h during surgery. Patients could be supplemented with 5% human albumin, if needed. Blood loss was replaced with allogeneic blood if hemoglobin decreased below 4.5 mmol/L or below 5.5 mmol/L if the patient had cardiopulmonary disease.
Surgical Procedures
The Ivor-Lewis (15) procedure involves both laparotomy and thoracotomy. An upper laparotomy mobilized the stomach, and a right thoracotomy at the sixth intercostal space removed the tumor en bloc, along with adjacent tissue and lymph nodes. Gastric continuity was re-established using a tube formed from the remaining stomach. A robotically assisted laparoscopic procedure was used in the abdominal part of the Ivor-Lewis procedure (hybrid esophagectomy), in half of the patients. This robotically assisted technique has been performed once a week since 2013, reflecting the capacity of the robot at the department. Both techniques were standard procedures during the study period.
Scoring of Flushing
Flushing was evaluated according to Nomura et al. (12). The nurse anesthesiologist confirmed the assessment, with any conflicting scores settled by discussion. Grade 1 flushing only involved the forehead and cheeks, while Grade II represented flushing of the whole head, eventually involving the shoulders. The highest score during the first hour of surgery was noted.
Determination of Plasma 6-Keto-Pgf1α
During the abdominal part of surgery, predefined time points were marked in the Nexfin® file: (a) after induction of anesthesia (baseline), (b) after opening the abdomen/establishing pneumoperitoneum, (b) after 15 min of surgery, (d) following mobilization of the stomach, and (e) after closing the abdomen/desufflation. At each time point, blood samples were collected in chilled EDTA tubes and centrifuged at 3000 r/min for 10 min with plasma stored in cryotubes (CryoPure 1.8 mL, Sarstedt, Nürnbrecht, Germany) at −80 °C for analyses of 6-keto-PGF1α.
Plasma 6-keto-PGF1α, a stable hydrolysis product of PGI2, was determined, due to the short half-life of PGI2. Enzyme-linked immunosorbent assay (ELISA) kits from two manufacturers (Enzo Life Science, Lörrach, Germany, ADI-900-00 and Cayman Chemicals, Ann Arbor, MI, USA, 515211.96-CAY) were tested for eligibility for human EDTA plasma according to the manufacturers’ instructions.
Postoperative Complications
Postoperative morbidity was independently registered by two investigators, according to the Clavien-Dindo classification (16), and classified as moderate (Clavien-Dindo Grade 1–2) or severe (Grades 3–5). Disagreements were settled by discussion among the surgeons.
Statistics
Hemodynamic data are reported as mean over 30 s after each marker in the Nexfin® file. Intragroup changes were analyzed using repeated-measures analysis of variance (ANOVA) with Bonferroni correction for multiple comparisons and inter-group comparisons using one-way ANOVA. Demographic and intraoperative characteristics were tested with chi-square test or Fisher’s exact test, while 6-keto-PGF1α data were analyzed using Friedman’s test and Mann–Whitney U test. To correlate the demographic and intraoperative data to postoperative complications, characteristics with a significant correlation in univariate analysis were considered in a multivariate analysis. The level of significance was <0.05 and data were presented as mean ± standard deviation (SD). Statistical analyses were performed using SPSS (IBM SPSS statistics for Windows, Version 22.0. Armonk, NY), and graphs were prepared using Graph Pad (Graph Pad Software Inc., USA).
Results
A total of 50 patients were consecutively included in the protocol, of which 25 were operated upon, using the robotically assisted, minimally invasive technique for the abdominal part. No patient was excluded due to preoperative use of NSAIDs or glucocorticoids. The demographic and intraoperative data are summarized in Table 1, showing no significant differences between the two groups of patients (open versus robotically assisted surgery) in regard to age, sex, American Society of Anesthesia (ASA) score, body mass index, length of surgery, or fluid balance after surgery. However, the blood loss was less during robotically assisted surgery (p < 0.001).
Intergroup comparison of demographic and intraoperative data (mean ± SD).
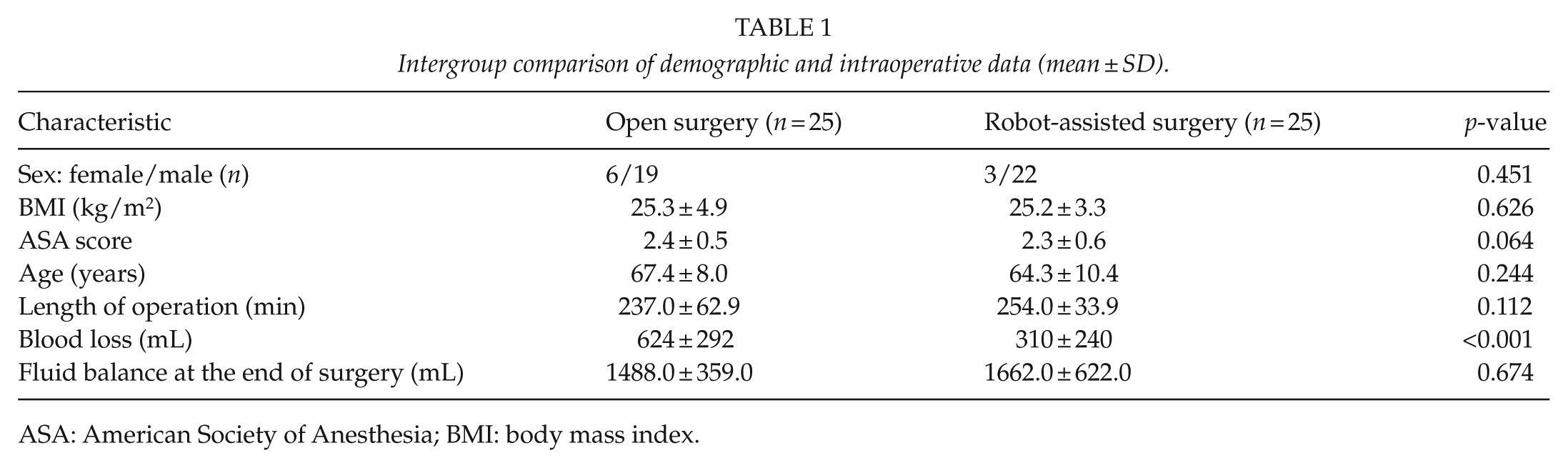
ASA: American Society of Anesthesia; BMI: body mass index.
Flushing
Flushing was observed in 17 patients (68%) during open surgery, but only in 5 patients (20%) during robotically assisted surgery (p = 0.001). Of the patients with flushing during open surgery, eight were registered as Grade II (32%), while no Grade II flushing was registered during robotically assisted surgery (Table 2).
Flushing and postoperative complications after open and robotically assisted hybrid esophagectomy.
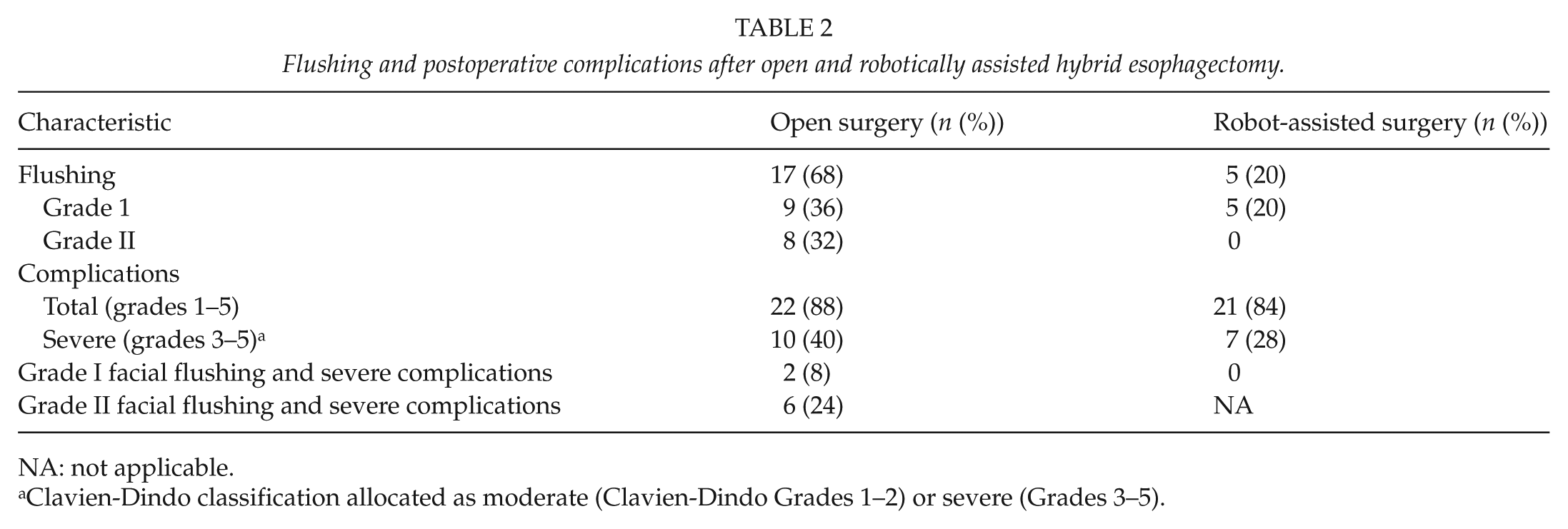
NA: not applicable.
Clavien-Dindo classification allocated as moderate (Clavien-Dindo Grades 1–2) or severe (Grades 3–5).
Hemodynamics
In the open-surgery group, MAP remained stable (p = 0.583) (Fig. 1), while heart rate (HR) increased after 15 min of surgery (p = 0.001) and returned to baseline values after closing the abdomen (p = 1.000). In parallel, systemic vascular resistance (SVR) decreased from baseline to start of surgery (p = 0.032) and decreased further after 15 min of surgery (p = 0.005), but normalized following mobilization of the stomach and closing the abdomen (p = 1.000). Likewise, patients with flushing demonstrated an increase in HR from baseline to 15 min of surgery (p = 0.005) that was higher than for the patients without flushing (p = 0.015) (Fig. 2). However, there were no significant differences in MAP or SVR between the patients with and without flushing during open surgery (p > 0.05).
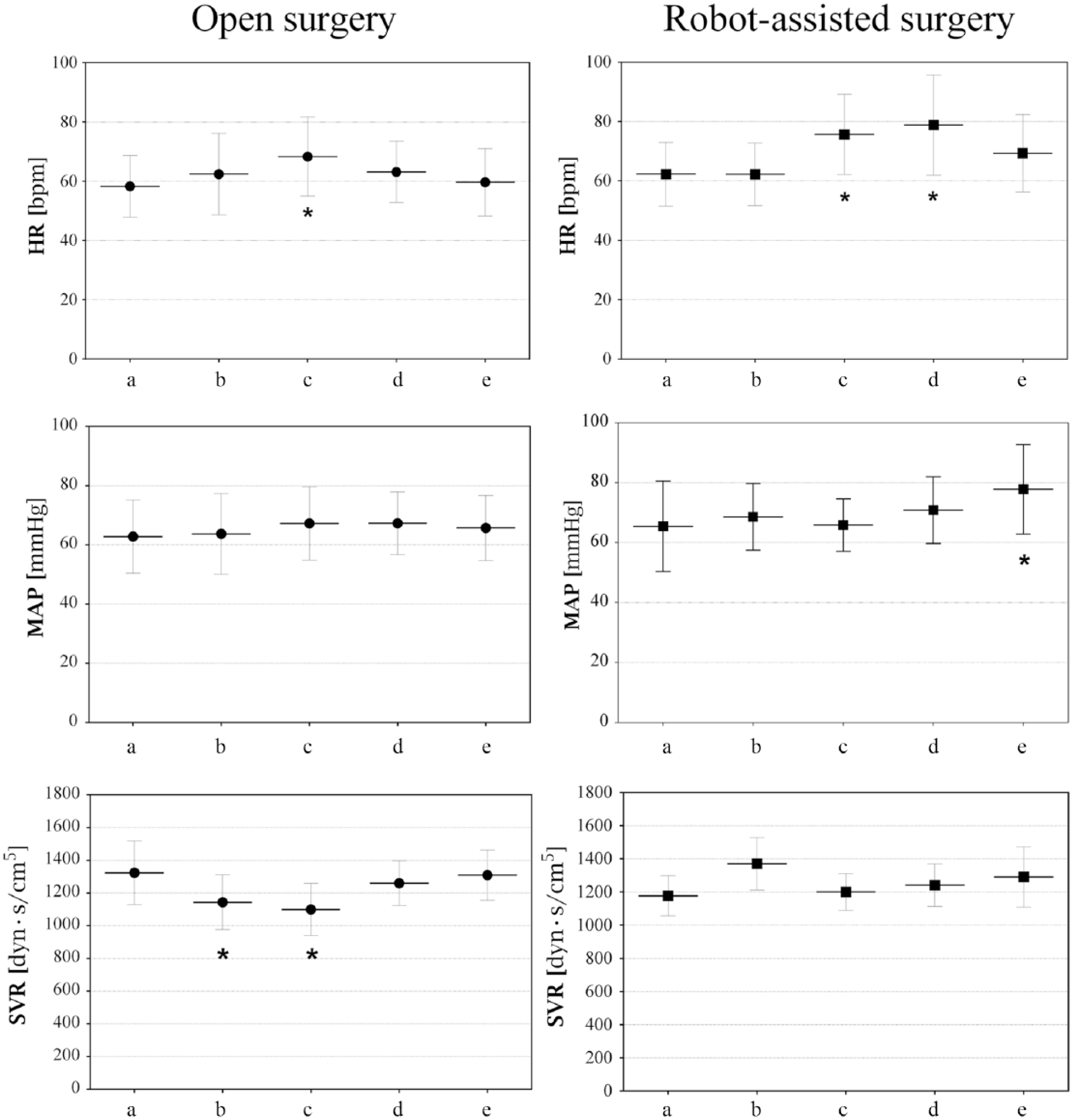
Hemodynamic variables during the abdominal part of open and robotically assisted esophagectomy (mean ± SD). MAP: mean arterial pressure; HR: heart rate; SVR: systemic vascular resistance. Time points for hemodynamic measurements: (a) baseline, (b) after opening the abdomen/establishing pneumoperitoneum, (c) after 15 min of surgery, (d) following mobilization of the stomach, and (e) after closing the abdomen/desufflation.
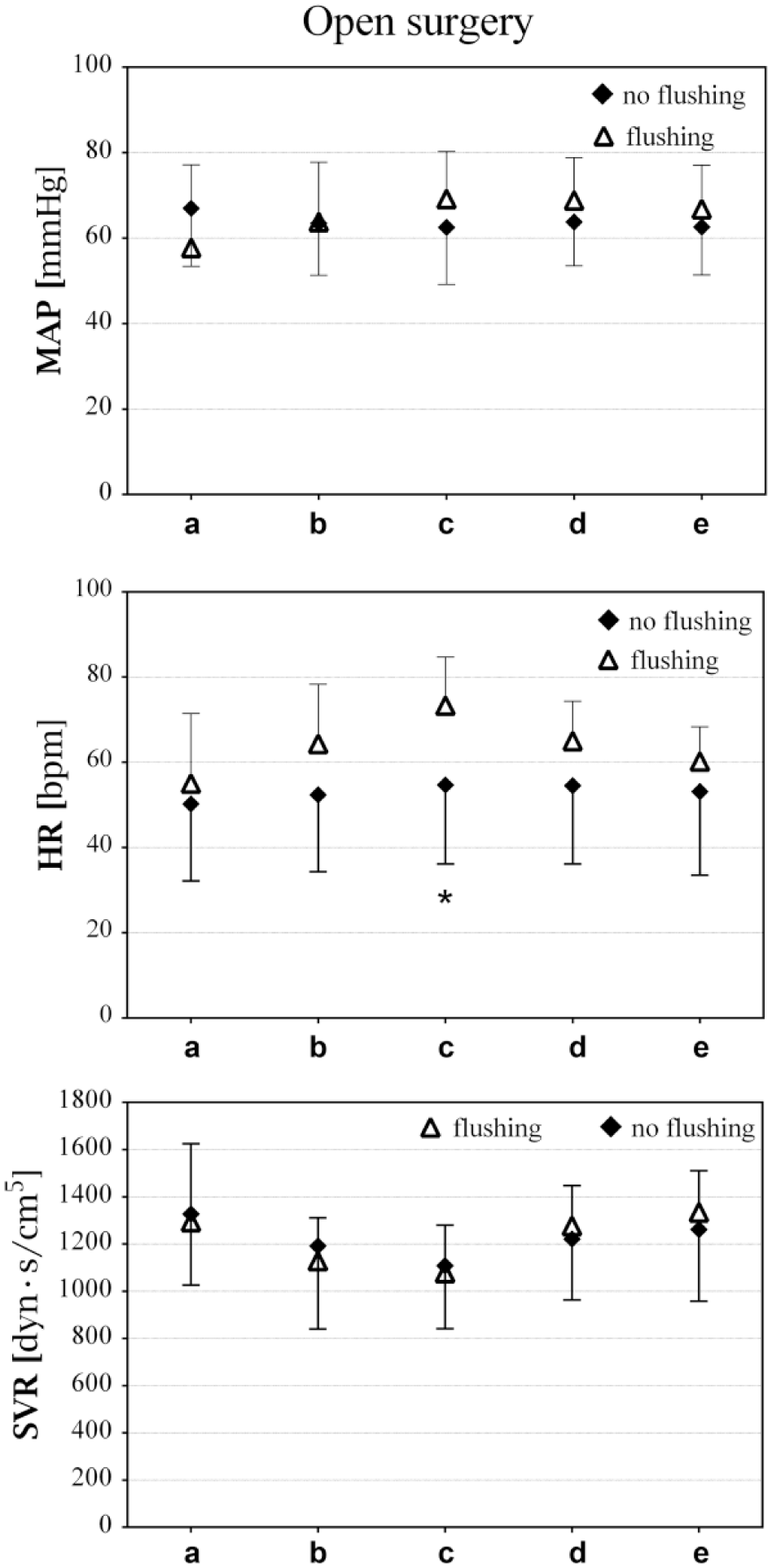
Hemodynamic variables of patients with and without facial flushing during open surgery, presented as mean ± SD. MAP: mean arterial pressure; HR: heart rate; SVR: systemic vascular resistance. Time points for hemodynamic measurements: (a) baseline, (b) after opening the abdomen/establishing pneumoperitoneum, (c) after 15 min of surgery, (d) following mobilization of the stomach, and (e) after closing the abdomen/desufflation.
MAP also remained stable during robotically assisted surgery, but increased after desufflation of the abdomen (p = 0.018). HR also increased after 15 min of surgery (p = 0.003) and remained high until after desufflation, while no changes in SVR were observed (0.452).
Flushing and Intraoperative Use of Vasopressors
There was a correlation between infusion of vasopressors during the first hour of open surgery and Grade II flushing (p = 0.036). No correlation was found between the infusion of vasopressors and severe postoperative complications (p = 1.000). However, continuous infusion of vasopressors during robotically assisted surgery was not correlated with flushing or severe postoperative complications (p = 0.307 and p = 0.856, respectively).
Intraoperative 6-Keto-Pgf1α
Using both samples with expected high and low concentrations of 6-keto-PGF1α, the ELISA kits from Cayman Chemicals showed little variation, while the kits from Enzo Life Science displayed the expected variation (Fig. 3), so were used for the present evaluation.
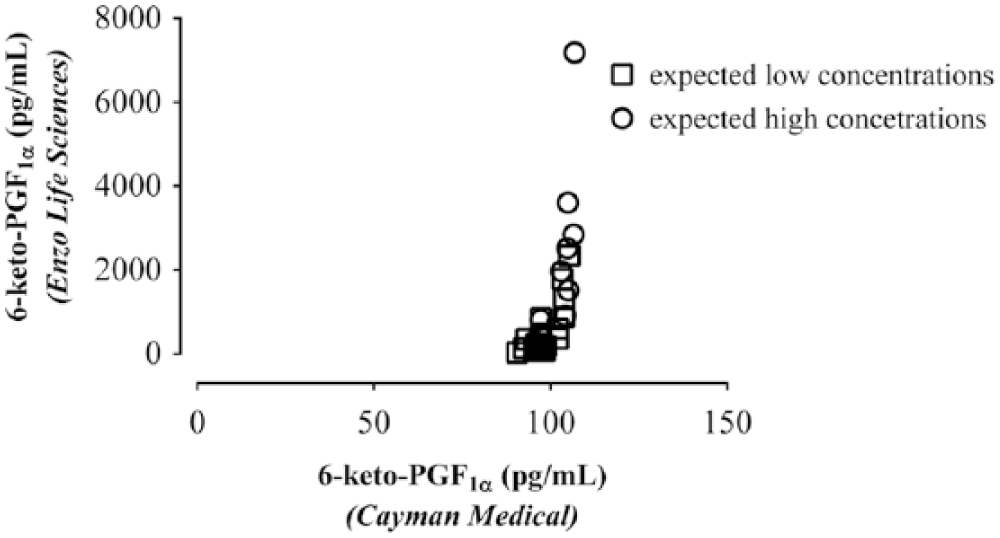
Scatter plot of plasma 6-keto-PGF1α, determined in the same samples with ELISA kits manufactured by Enzo Life Science (y-axis) and Cayman Chemicals (x-axis). The Enzo kits displayed an expected variation, while no variation was seen using the kits from Cayman Chemicals.
Plasma 6-keto-PGF1α increased both during open and robotically assisted surgery (p < 0.001), but the increase was greater after opening the abdomen and 15 min of surgery (p = 0.003 and p = 0.013, respectively) (Fig. 4A). For all patients, changes in plasma 6-keto-PGF1α were higher after 15 min of surgery in patients with flushing (Grades I and II) compared with patients who did not flushed (p = 0.029) (Fig. 4B). Furthermore, patients with Grade II flushing had a greater increase in 6-keto-PGF1α after opening of the abdominal wall than did patients with Grade I or no flushing (p = 0.002).
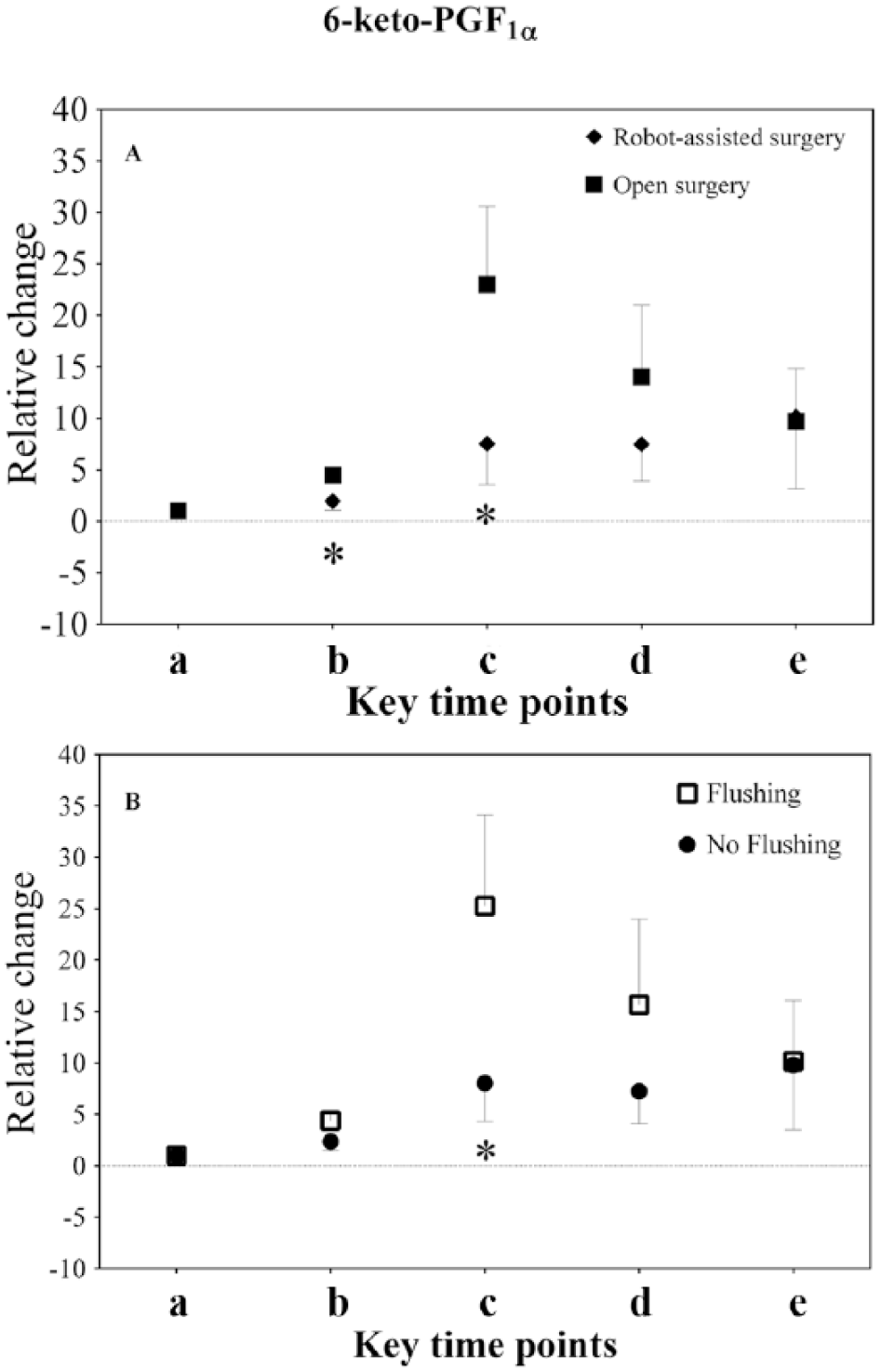
Relative changes in plasma 6-keto-PGF1α (mean ± SD) during surgery: (a) baseline, (b) after opening the abdomen/establishing pneumoperitoneum, (c) after 15 min of surgery, (d) following mobilization of the stomach, and (e) after closing the abdomen/desufflation. (A) Difference between open and robotically assisted surgery. (B) Changes in 6-keto-PGF1α between patients with and without flushing.
Flushing and Postoperative Complications
There was no difference in the incidence of overall (p = 0.687) or severe (p = 0.243) postoperative complications (Clavien-Dindo ⩾ 3) between the groups. In the open-surgery group, 10 patients had 19 severe complications, with seven patients having more than one complication. In the robotically assisted group, seven patients had 15 severe complications (five patients with more than one complication) (Table 3).
Total number of severe complications (Clavien-Dindo Grade ⩾ 3) for 10 and 7 patients after open and robotically assisted hybrid esophagectomy, respectively.
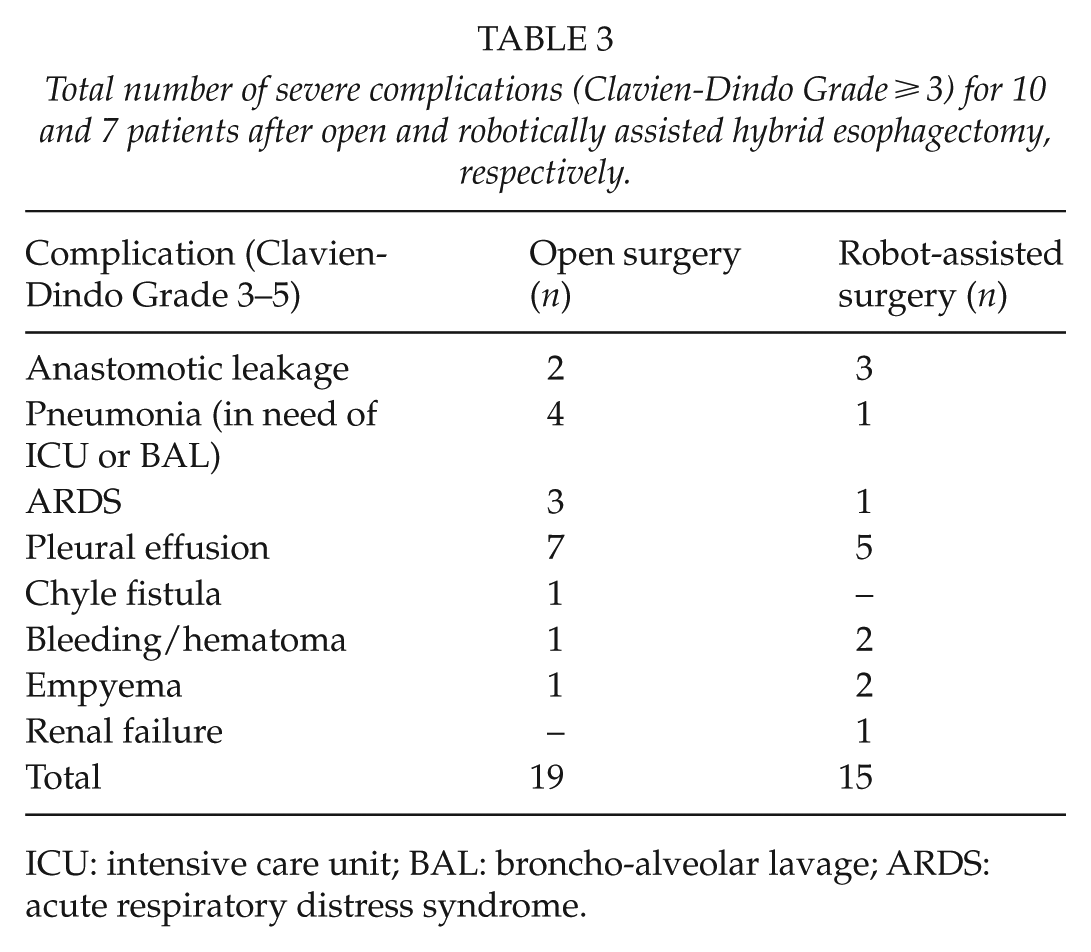
ICU: intensive care unit; BAL: broncho-alveolar lavage; ARDS: acute respiratory distress syndrome.
In the open-surgery group, flushing (Grades I and II) was not associated with overall (p = 0.170) or severe complications (p = 0.294). However, Grade II flushing was correlated with severe postoperative complications (p = 0.037), but not with other variables (age (p = 0.410), sex (p = 0.227), or ASA status (p = 0.561)). However, flushing was not correlated with postoperative complications in the robotically assisted surgery group (p = 0.119) (Table 2).
Discussion
We registered only mild and few incidents of flushing during robotically assisted esophagectomy. Moreover, serious postoperative complications following open surgery seemed related to Grade II, but not Grade I, flushing.
Grade II flushing (severe MTS) manifested in eight patients during the first hour of open surgery. These patients also demonstrated increased HR and plasma 6-keto-PGF1α. Severe MTS was also associated with serious postoperative complications. MTS develops in almost all patients if they are exposed to planned traction of the mesenterium (17), explaining the different frequencies of MTS during open and robotically assisted hybrid esophagectomy. We speculate that MTS may be part of the inflammatory response that restores homeostasis to the injured patient (18). However, any significant exaggeration of the response could predispose the patient to harmful consequences (19). The occurrence of severe MTS may have been part of, or even the triggering factor for, an exaggerated stress response, resulting in more severe postoperative complications in the open-surgery group (20).
Although the incidence of MTS varied among the groups, and severe MTS showed a correlation with severe complications after open surgery, the overall incidence of postoperative complications did not significantly differ between the groups. This indicates that dissimilar mechanisms are responsible for development of complications among patients exposed to open or minimally invasive surgery. Minimally invasive surgery is believed to offer advantages over open surgery, including less pain, attenuated humoral immunological response, and early mobilization (21). However, this study was not powered to determine the incidence of postoperative complications between open and robotically assisted esophagectomy. A possible difference could be masked by the fact that the thoracic part of the operation used the open technique in both groups. Nevertheless, the incidence and severity of MTS were lower during robotically assisted surgery. Correspondingly, attention should be on MTS as an intraoperative risk factor. Since NSAIDs inhibit the occurrence of MTS (11, 22), pre- or intraoperative treatment with a single dose may positively affect postoperative complications.
Intraoperative hypotension correlates with postoperative morbidity and mortality (13, 23). However, perioperative use of vasopressors is associated with increased risk of gastrointestinal anastomotic dehiscence (24) and may be due to the vasoconstricting effect of these agents (25), since impaired microcirculation affects surgical outcome (26, 27). In this study, infusion of vasopressors was correlated with Grade II MTS, but no correlation between vasopressor infusion and severe postoperative complications was observed. This was likely due to the small number of patients. Therefore, avoiding MTS may reduce both the need for vasopressors during surgery and the incidence of postoperative complications related to ischemia. Flushing is believed to be the key factor when detecting MTS, while hemodynamic variables are subject to regulation and manipulation. However, visual accuracy is dubious (28), and quantification of flushing would likely improve the diagnosis. Noninvasive techniques with the capacity to record facial blood flow (increased flow = flushing) or assessing the degree of redness would be ideal for detecting MTS.
One limitation of this study is the nonrandomized protocol, with the risk of selection bias. Yet, consecutive patients reduced that risk, supported by the nonsignificant differences in preoperative data. Also, few participants may inherently increase the risk of Type 2 error. However, the purpose of this study was not to compare complications between the two groups of patients, but to record the incidence of MTS. There seemed to be enough patients involved in the study for that evaluation. Another limiting factor is the subjective scoring of flushing, with the risk of overlooking redness in darker skin. During the study period, no patient with darker skin was operated upon, and flushing was scored by two persons to reduce that risk.
Conclusion
MTS manifests frequently during open esophagectomy, and Grade II MTS correlates with the development of severe postoperative complications. MTS may be a harmful event that needs attention. A quantitative evaluation of flushing could strengthen its possible association with postoperative complications. Also, it should be evaluated if perioperative administration of NSAIDs and/or glucocorticoids affects MTS and postoperative outcome.
Footnotes
Acknowledgements
The authors thank Dr Hans-Jørgen Frederiksen for his guidance and help during acquisition of data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by grants from the Danish Cancer Society (No. R90-A6132-14-S2) and the Aase and Ejner Danielsen Foundation.
Trial Registration
Scientific Ethical Committees, The Capital Region, Denmark, No. H-2-2013-101; ClinicalTrials.gov. ID: NCT02077673.
