Abstract
Background and Aims:
An adverse reaction to metal debris is a known complication after large diameter head metal-on-metal total hip arthroplasty. However, the failure rate varies depending on the implant design. Therefore, we investigated the prevalence of adverse reaction to metal debris, as well as the symptoms and risk factors after undergoing a ReCap-M2a-Magnum large diameter head metal-on-metal total hip arthroplasty.
Materials and Methods:
Between 2005 and 2012, 1188 patients (1329 hips) underwent ReCap-M2a-Magnum total hip arthroplasty at our institution. Systematic screening for adverse reaction to metal debris was arranged using the Oxford Hip Score questionnaire, hip and pelvic radiographs, and assessments of the serum chromium and cobalt ion levels. Clinical evaluation and magnetic resonance imaging were performed for the symptomatic patients, as well as those with either chromium or cobalt ion levels ⩾5 µg/L. The prevalence of adverse reaction to metal debris after ReCap-M2a-Magnum total hip arthroplasty was assessed, and the risk factors for adverse reaction to metal debris were evaluated using logistic regression. The mean follow-up time was 5.2 (0.003–9.1) years. This study was an extension of a previous study conducted at our institution with 80 patients.
Results:
In total, 33 patients (33 hips, 2.5% of all hips) required a revision operation due to adverse reaction to metal debris. Moreover, 157 hips exhibited definitive adverse reaction to metal debris, but a revision operation was not performed (157 of 1329 hips, 11.8% of all hips). Overall, 190 out of 1329 (14.3%) hips had definitive adverse reaction to metal debris. Pain, subluxation sensation, clicking, swelling, a small head size, and a fair/poor Oxford Hip Score were associated with definitive adverse reaction to metal debris.
Conclusion:
We found a high prevalence of adverse reaction to metal debris in the ReCap-M2a-Magnum total hip arthroplasty patients in this study; however, most of the patients did not require revision operations.
Keywords
Introduction
The large diameter head metal-on-metal total hip arthroplasty (LDH MOM THA) was introduced to decrease the wear induced osteolysis associated with metal on polyethylene bearings, increase the range of motion, and decrease the dislocation rate (1). Some of the short-term results have been encouraging (2, 3). The popularity of LDH MOM THA has grown rapidly, with more than one million metal-on-metal (MOM) hip implants being performed worldwide (4). In Finland, approximately 15,000 LDH MOM THA have been performed, according to the Finnish Arthroplasty Register (5).
It soon became obvious that the Articular Surface Replacement (ASR) total hip arthroplasty (THA) (Johnson & Johnson) was a poor performer, with an increased risk of early revision (6–8). In addition, recent evidence has suggested that other LDH MOM designs may have increased revision risks due to the collection of periarticular fluid, soft tissue masses, and gluteal muscle necrosis. The symptoms of these patients often include pain, swelling, clicking, and a sensation of subluxation (8–10). This condition has been called an adverse reaction to metal debris (ARMD) or adverse local tissue reaction (ALTR) (9). Some designs have been recalled by the manufacturers due to the increased risk of revision caused by ARMD (11).
The ReCap-M2a-Magnum THA (Biomet, Warsaw, IN, USA) was the most commonly used LDH MOM device in Finland from 2005 until 2012, with 6655 implantations performed (5). It was also the most commonly used total hip implant at our institution during the same time period, with a total of 1329 implantations performed. Mokka et al. (12) have previously reported the preliminary results of 80 ReCap-M2a-Magnum THA performed at our institution, in which 11 (14%) of the patients had definitive ARMD and 29 had probable ARMD. Recently, Bosker et al. (13) evaluated 626 patients (706 hips) who had undergone THA with ReCap-M2a-Magnum implants. In their study, the incidence of pseudotumors was 35%, and it increased significantly during the prolonged follow-up. The aim of this study was to assess the prevalence of ARMD using a systematic screening of the total ReCap-M2a-Magnum cohort of 1329 hips at our institution. Our hypothesis was that the prevalence of ARMD was 14%, based on our preliminary study (12).
Materials and Methods
Patient Population/Study Population
At our institution, 1188 patients (1329 hips) underwent ReCap-M2a-Magnum LDH MOM THA, and the surgeries were performed between August 2005 and April 2012. The systematic screening for ARMD consisted of an Oxford Hip Score (OHS) questionnaire, hip and pelvic radiographs, and measurements of the serum chromium and cobalt ion levels, before the end of September 2014.
Magnetic resonance imaging (MRI) was performed on those patients exhibiting symptoms and/or increased ion levels (⩾5 µg/L). An experienced orthopedic surgeon also assessed all these patients. The study population included a subset of 74 patients (80 hips) which we have described in a previous publication (12).
Overall, 989 (74%) of the patients had primary arthrosis as an indication for THA, while the other indications included hip dysplasia (7%), rheumatoid arthritis (5%), post-traumatic arthrosis (2%), secondary arthrosis (5%), avascular necrosis (3%), and fracture (3%).
Altogether, 74 hips (74 patients) were revised for reasons other than ARMD during the follow-up: 25 for periprosthetic fractures, 12 for infections, and 28 for early loosening. Dislocation was the reason for revision in three patients, and one of those had ARMD. The mean follow-up time was 5.2 (0.003–9.1) years, and 96 of the patients died during the follow-up period. In addition, 28 patients had undergone simultaneous bilateral ReCap-M2a-Magnum THA, and 113 patients had both hips operated on in separate sessions. The Biomet Bi-Metric or Reach stem and Hardinge approach were used in all of the study cases.
The ReCap-M2a-Magnum is an metal-on-metal (MOM) articulation. The bearing articulation consists of a cobalt chrome molybdenum cup (ReCap) and a cobalt chrome molybdenum head (Magnum). The head size varies from 38 to 60 mm and is considered to be a large diameter head (14).
Study Design
Metal artifact reduction sequence magnetic resonance imaging (MARS-MRI) was used to identify the ARMD changes as fluid collection and soft tissue masses around the prostheses (15, 16). MRI was performed on 352 hips (16).
The cup inclination and anteversion angle were measured using pelvic and hip x-rays. Because the measurement of the anteversion angle is relatively inaccurate in lateral hip x-rays, we categorized the cups into two subgroups for regression analysis purposes: retroverted and not retroverted. The pelvic radiographs were available for all 1199 of the hips to measure the inclination angle, and hip radiographs were available for all of the patients.
The serum cobalt and chromium ion levels were measured in 959 patients (1094 hips) (17, 18). However, the metal ion concentrations were not measured in those patients with loosening, fractures, or infections in their prostheses.
An OHS of 42–48 points was considered to be excellent, 34–41 was good, 27–33 was fair, and 0–26 was poor. Separate questions about clicking, subluxation sensation, and swelling of the hip were also asked. The OHS questionnaire was not filled out preoperatively, and the total points were available for 742 patients. Generally, those patients with bilateral Recap-M2a-Magnum THA had only one OHS questionnaire available, and we could not pinpoint which hip was of concern. Therefore, those patients with bilateral Recap-M2a-Magnum procedures were left out of the regression analyses, including their OHS data.
Definitive ARMD was established in three ways:
It was diagnosed in revision surgery.
There was a solid mass or fluid collection ⩾50 mm in the MRI.
The serum chromium or cobalt level was ⩾10 µg/L.
Probable ARMD was established with either of the following:
A collection of fluid <50 mm in the MRI.
The serum chromium or cobalt concentration was ⩾5 but <10 µg/L.
ARMD was confirmed intraoperatively if there was milky fluid, a solid pseudotumor mass, or muscle necrosis. The revision surgery was comprised of several different operations: head exchange with or without acetabular revision and stem revision with or without acetabular revision. Those patients with clearly elevated metal ion levels or pseudotumor findings did not undergo surgery if the patient refused on the basis of an asymptomatic hip or poor overall health.
The following risk factors for ARMD were assessed: age, sex, laterality, inclination angle of the cup (categorical variables: <30°, 30°–50°, and >50°), anteversion angle of the cup (categorical variables: >0° and ⩽0°), and head size (categorical variables: ⩽44, 46–50, and ⩾52 mm). The associations between the OHS score (poor, fair, or good vs excellent), pain (no, mild, moderate, or severe), symptoms (clicking, subluxation sensation, and/or swelling), and ARMD were also examined. We further assessed the same risk factors and symptoms for the occurrence of an ARMD revision.
The demographic data are presented hipwise in Table 1 and patientwise for those having a unilateral study device in Table 2.
Demographic data of the 1329 study hips presented hipwise.
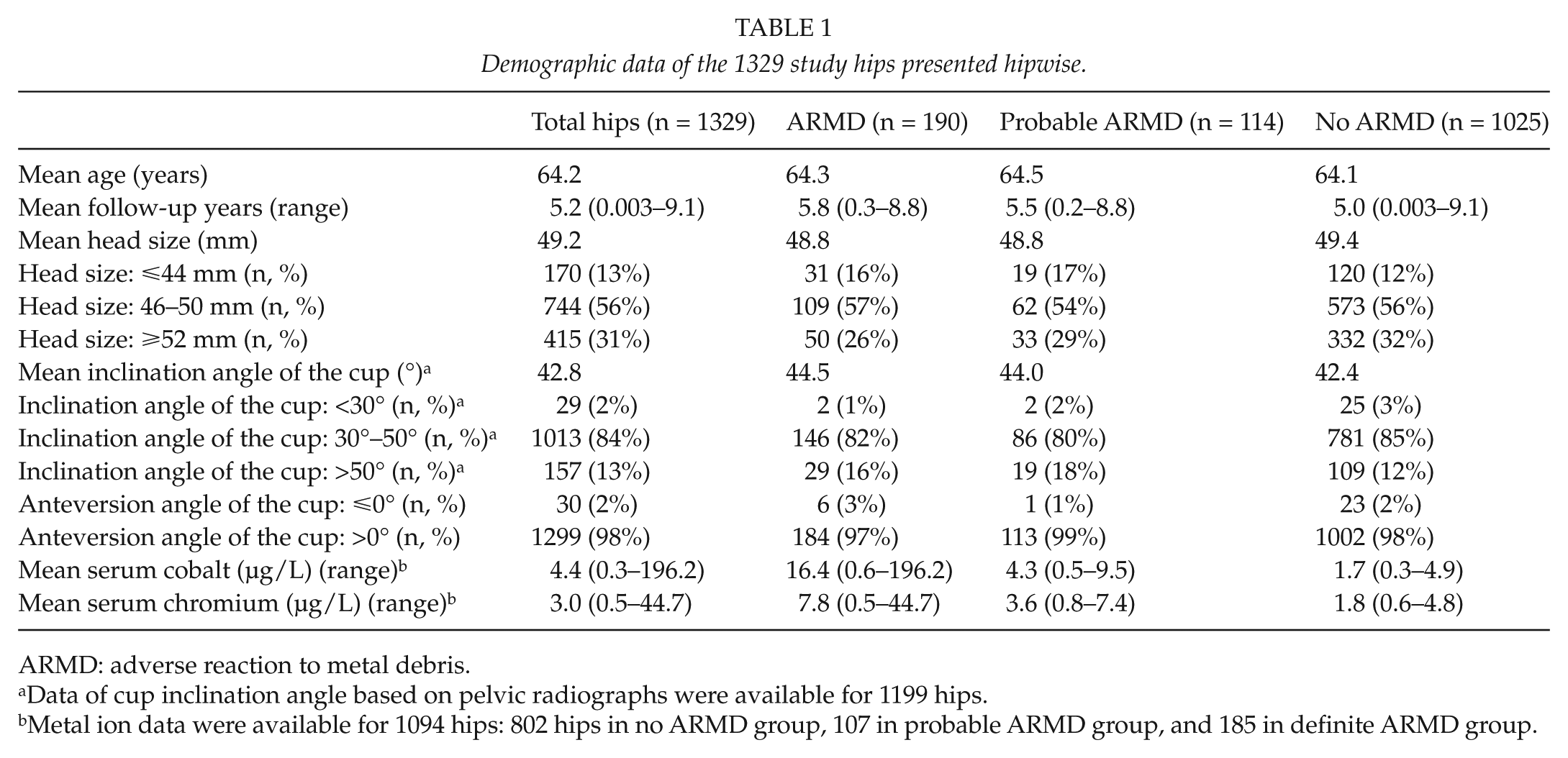
ARMD: adverse reaction to metal debris.
Data of cup inclination angle based on pelvic radiographs were available for 1199 hips.
Metal ion data were available for 1094 hips: 802 hips in no ARMD group, 107 in probable ARMD group, and 185 in definite ARMD group.
Patient characteristics for those with a unilateral arthroplasty.
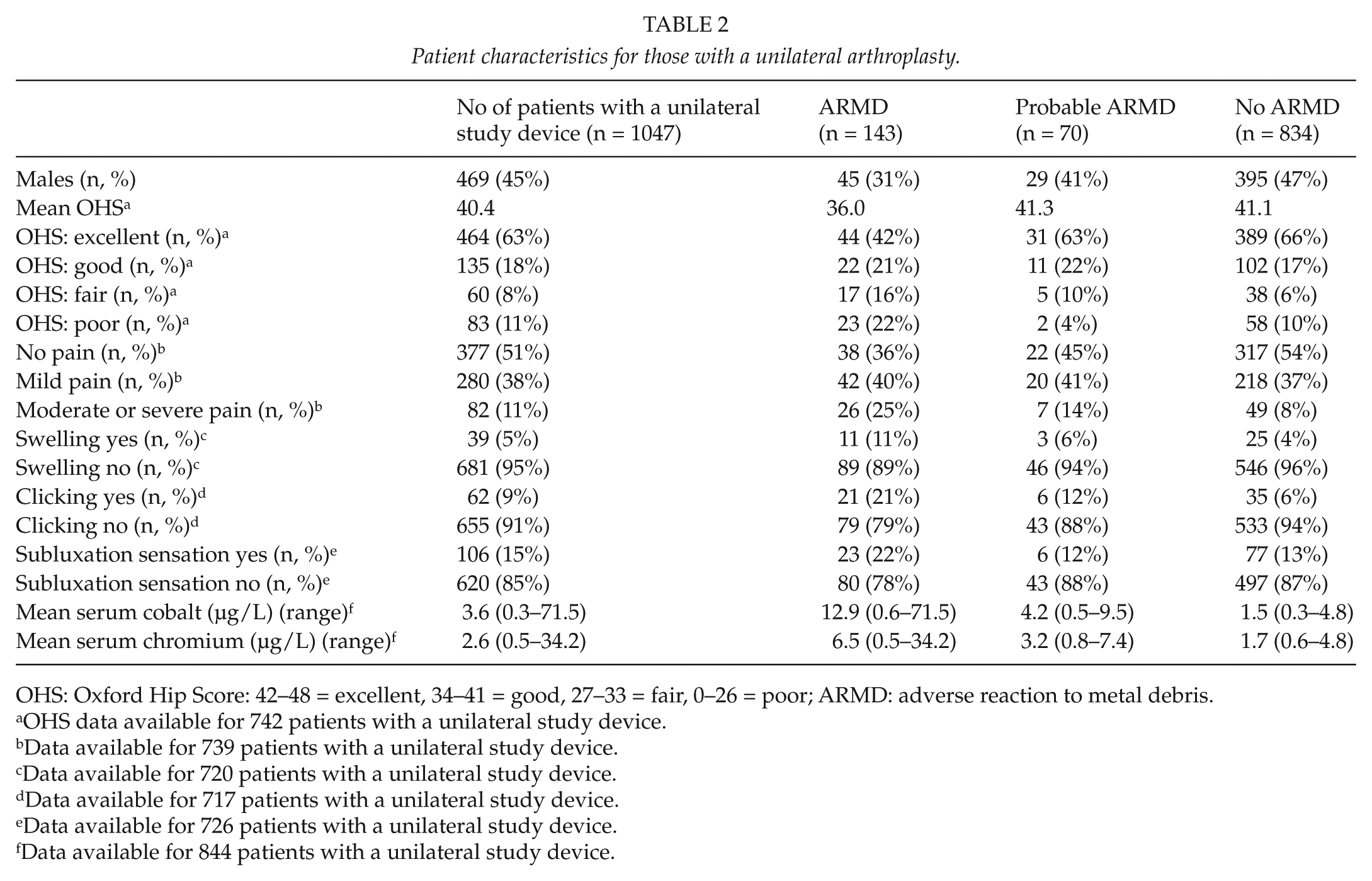
OHS: Oxford Hip Score: 42–48 = excellent, 34–41 = good, 27–33 = fair, 0–26 = poor; ARMD: adverse reaction to metal debris.
OHS data available for 742 patients with a unilateral study device.
Data available for 739 patients with a unilateral study device.
Data available for 720 patients with a unilateral study device.
Data available for 717 patients with a unilateral study device.
Data available for 726 patients with a unilateral study device.
Data available for 844 patients with a unilateral study device.
Ethics
This study was based on the national recommendations for the systematic screening of all LDH MOM THA patients provided by the Finnish Arthroplasty Society (18).
Statistical Analysis
The potential risk factors for ARMD were analyzed via univariable multinomial logistic regression. The dependent variable consisted of three groups (definitive ARMD, probable ARMD, and no ARMD), with no ARMD being used as the reference group. The results were expressed using odds ratios (ORs) with a 95% confidence interval (CI). The goodness-of-fit for the logistic regression models was evaluated with a deviance test, while the multivariable logistic model was obtained using backward elimination (inclusion criteria, p < 0.10) to examine the potential confounding effect of the other risk variables. The generalized estimating equation (GEE) was used for the hipwise data to account for the correlation between the hips from the same patient. Kaplan–Meier estimates for the revision operations (for any reason) and for the ARMD were calculated. The Cox regression analysis was used to analyze the association between the risk factors and symptoms and ARMD revision. The hipwise survival data were analyzed with a lognormal frailty model to account for the correlation between the hips from the same patient, and the results of the Cox regression were expressed using hazard ratios (HRs) with a 95% CI. The proportional hazard assumptions were evaluated with a log-cumulative hazard plot, and the assumptions were met. p-values < 0.05 were considered to be statistically significant, and the statistical analyses were carried out using SAS for Windows, version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Throughout the follow-up period, 104 patients (106 hips, 8.0% of all hips) required revision operations, while 33 patients (33 hips, 2.5% of all hips) required revision operations due to ARMD. The Kaplan–Meier survival estimate of the ReCap-M2a-Magnum THA at 5 years was 93.3% for any reason (95% CI: 91.9%–94.8%), and 98.6% (95% CI: 97.8%–99.3%) for those patients with ARMD revisions.
There was definitive ARMD in 157 hips, in which revision operations were not performed (157 of 1329 hips, 11.8% of all hips). Probable ARMD was determined in 114 hips (8.6%). In total, 190 out of 1329 (14.3%) hips had definitive ARMD, and 1025 (77.1%) hips did not have ARMD.
The univariable associations assessed using the multinomial logistic regression analysis between the risk variables/symptoms and ARMD are presented in Table 3. Pain, subluxation sensation, clicking, small head size (⩽44 vs ⩾52 mm), and fair/poor OHS scores were associated with definitive ARMD. In the multivariable model, female gender, clicking, large head size (⩾52 vs 46–50 mm), and pain (moderate/severe vs no pain) were associated with ARMD (Table 4). The results of the univariable associations between the risk factors and symptoms and ARMD revision assessed via the Cox regression analysis are presented in Table 5. Retroversion of the cup, small head size (head size: ⩽44 vs ⩾52 mm), and clicking were associated with the occurrence of ARMD revision.
Crude odds ratios (ORs) and 95% confidence intervals (CIs) of associations between risk factors and symptoms with ARMD.
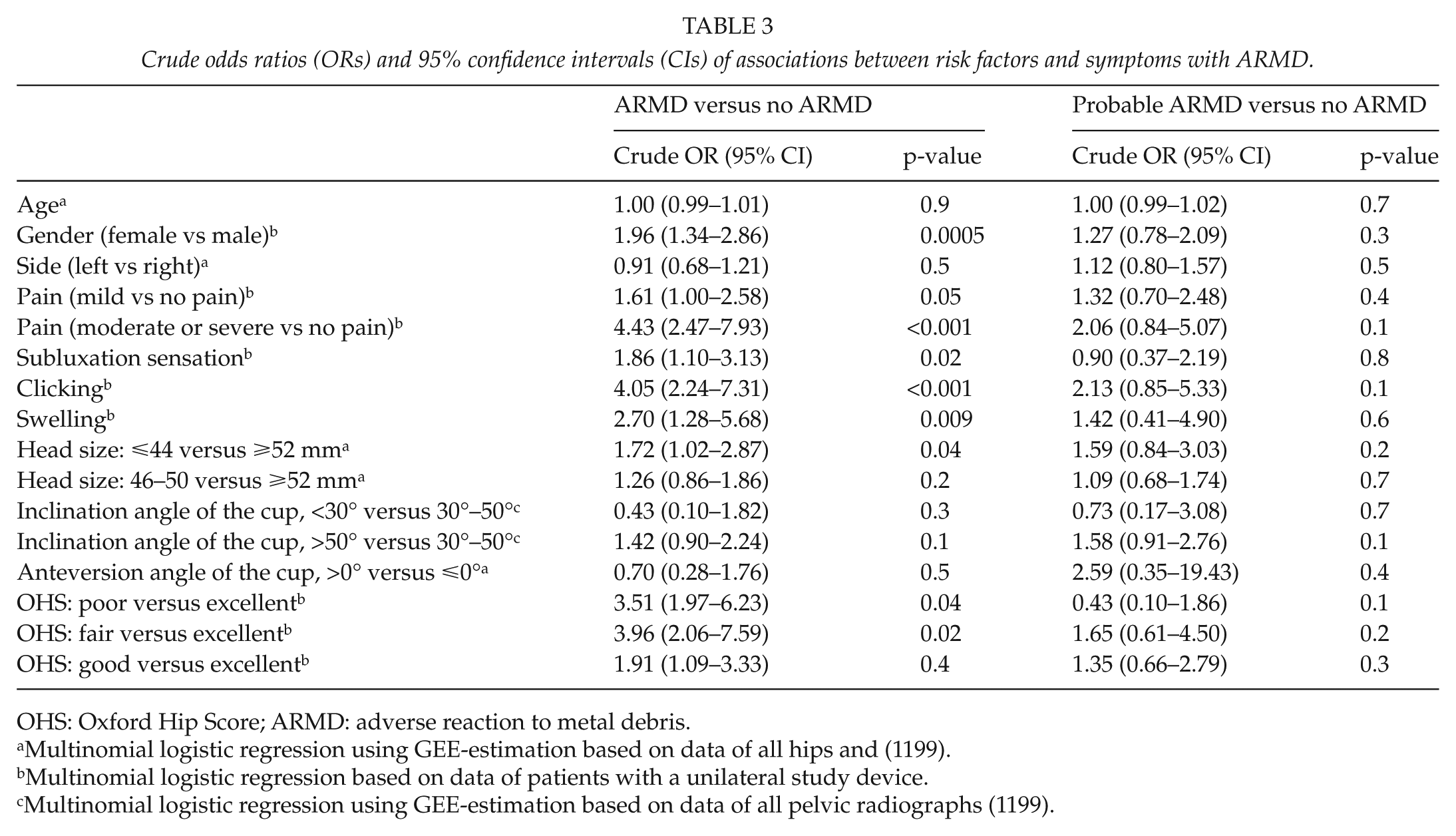
OHS: Oxford Hip Score; ARMD: adverse reaction to metal debris.
Multinomial logistic regression using GEE-estimation based on data of all hips and (1199).
Multinomial logistic regression based on data of patients with a unilateral study device.
Multinomial logistic regression using GEE-estimation based on data of all pelvic radiographs (1199).
Adjusted odds ratios (ORs) and 95% confidence intervals (CIs) of associations between risk factors and symptoms with ARMD using multiple multinomial logistic regression based on data of 714 patients with a unilateral arthroplasty.
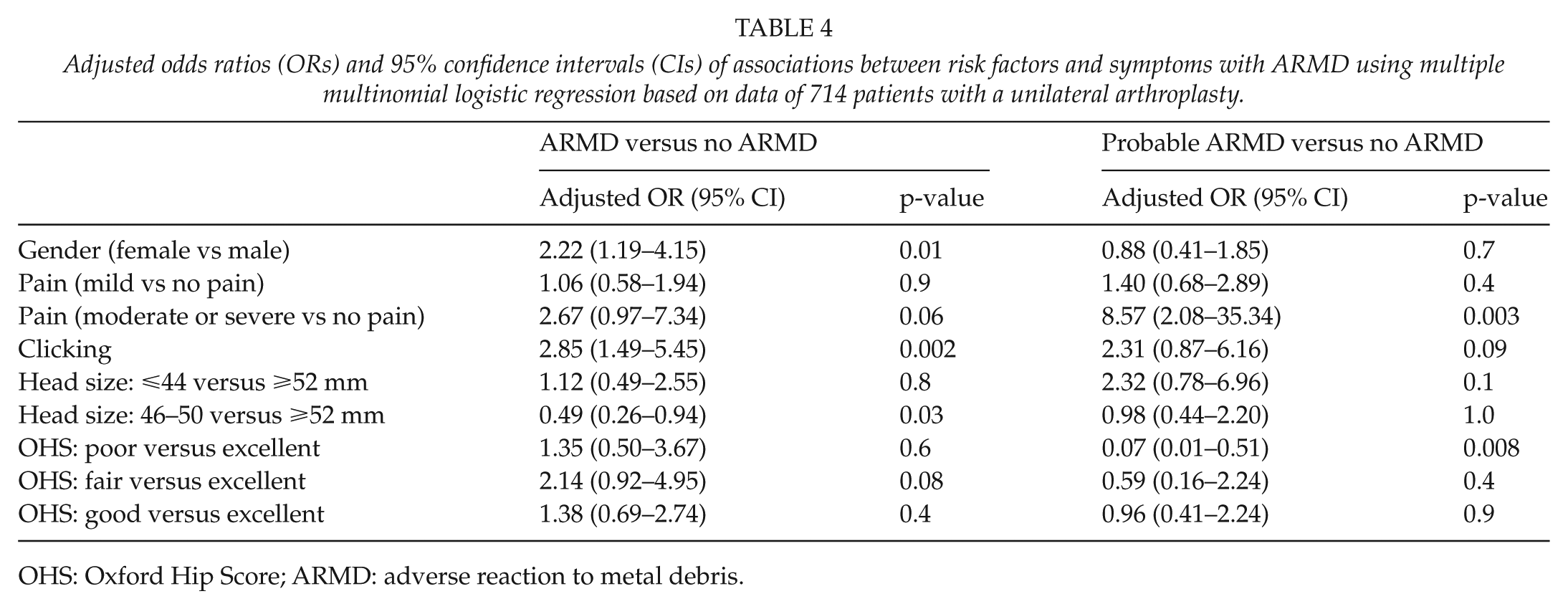
OHS: Oxford Hip Score; ARMD: adverse reaction to metal debris.
Crude hazard ratios (HRs) and 95% confidence intervals (CIs) of associations between risk factors and ARMD revisions.
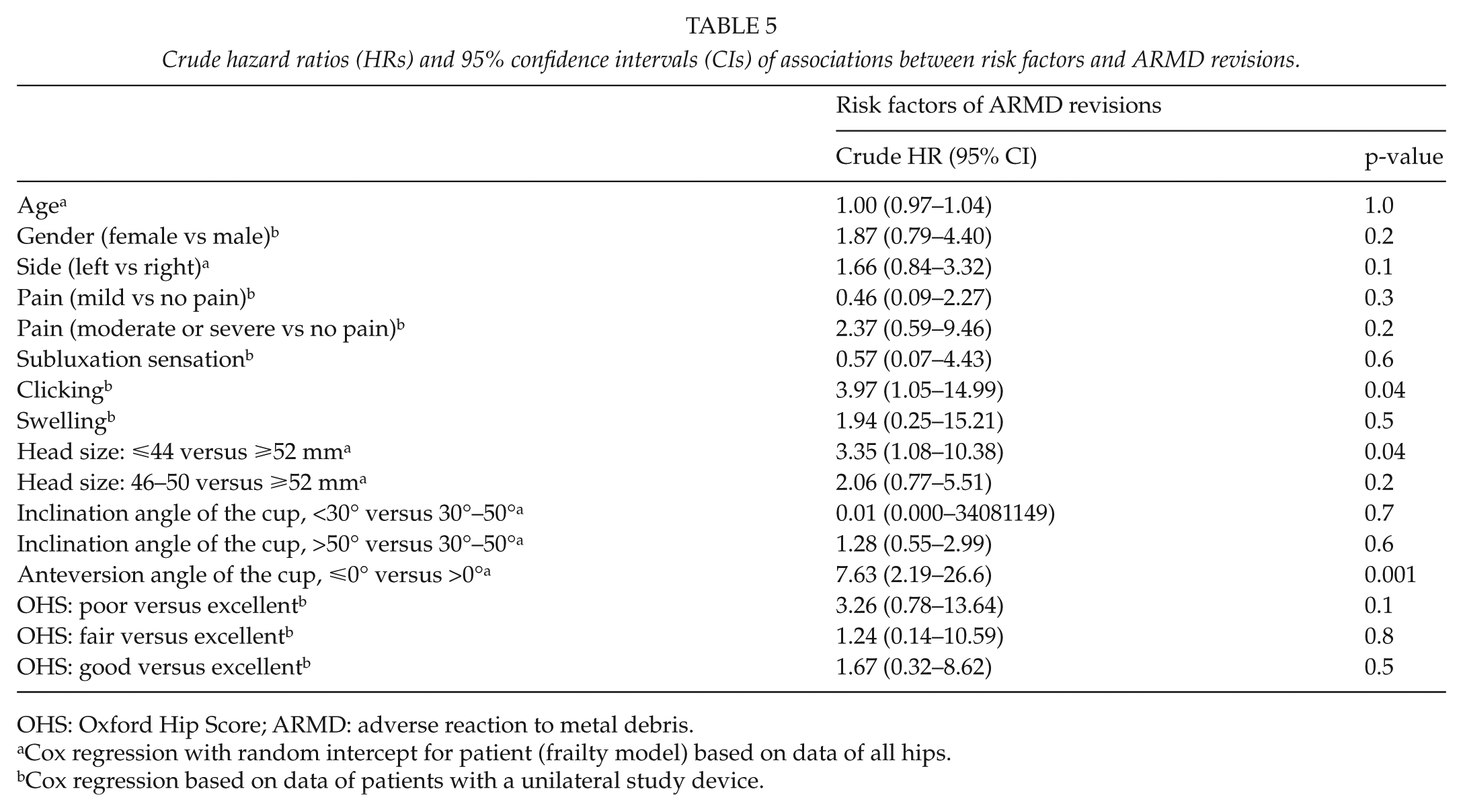
OHS: Oxford Hip Score; ARMD: adverse reaction to metal debris.
Cox regression with random intercept for patient (frailty model) based on data of all hips.
Cox regression based on data of patients with a unilateral study device.
For bilateral arthroplasty, the Co and Cr values were available for 125 patients. Mean Co was 7.2 µg/L (0.6–196.2), and mean Cr was 4.3 µg/L (0.7–44.7).
Discussion
In this study, we determined that 33 of the 1188 patients (33 of 1329 hips, 2.5%) had undergone revision operations due to ARMD. In addition, 157 hips (11.8%) were considered to have definitive ARMD during a mean follow-up time of 5.2 years but had not been revised by the end of September 2014. The prevalence of ARMD in our cohort was 14.3% (190/1329), which was similar to that of our preliminary study (12).
There were some limitations in our study that may have biased the results. For example, we concluded that patients with high ion levels (⩾10 µg/L) had definitive ARMD. ARMD is generally related to soft tissue changes around the prosthesis (9). The Medicines and Healthcare products Regulatory Agency (MHRA) (19) has recommended a cutoff level of serum cobalt and chromium of 7 µg/L. Although this shows good specificity, it shows relatively low sensitivity (20). The risk of developing a pseudotumor is four times higher with serum metal ion levels >5µg/L (21), and a cutoff level of 10 µg/L has been used previously in assessing ARMD in association with MOM hip implants (12, 22).
Another limitation was that we included patients with bilateral MOM implants, which may have biased the metal ion analyses. However, we increased the cutoff level from 8 µg/L, as suggested by Lardanchet et al. (23), to 10 µg/L due to the inclusion of the bilateral THA. We used a metal ion level of ⩾5 µg/L as a criterion for possible ARMD.
Moreover, dividing the “definite” and “probable” groups by the amount of fluid collection in the MRI is problematic. The dichotomy between MRI findings ⩾50 and <50 mm is artificial; thus, we hypothesized that a fluid collection ⩾50 mm in any dimension was a clinically significant amount of fluid with regard to the diagnosis of ARMD. This was also the way the patients were divided into the categories in the previous research conducted at our institute (12).
A further limitation of our study was that we selected MRI based on the patients’ symptoms and ion levels, and not all of the patients were scrutinized using MRI. Furthermore, the assessment of some ARMD cases was made based on the surgical findings in the medical reports, and some revisions were performed before the surgeons were familiar with the concept of ARMD.
The strengths of our study included the fact that all of the ReCap-M2a-Magnum implantations were performed at our institution. The Bi-Metric stem and Hardinge approach were used in every operation, so the stem or approach did not cause bias. In addition, to improve the assessment of the complications, we used MARS-MRI. Those cases presenting with ALTRs and periprosthetic fluid collection or soft tissue masses are not typically detectable in radiographs, or in most cases, computed tomography (CT). MRI is the best method for the evaluation of patients with MOM hip resurfacing arthroplasty because of its high soft tissue contrast and lack of ionizing radiation (24).
Concern has been raised about the high failure rate of LDH MOM THA due to ARMD. The Australian Orthopaedic Association National Joint Replacement Registry (AOANJRR) (6) first reported increased failure rates with MOM implants in 2008, while the MHRA issued a medical device alert about MOM implants in April 2010 (20). In May 2011, the American Food and Drug Administration (FDA) (25) ordered post-marketing surveillance of MOM THA from 21 companies. Furthermore, the Finnish Arthroplasty Society (18) recommended that physicians discontinue the use of LDH MOM THA in May 2012.
The short-term survival of the ReCap-M2a-Magnum THA was shown to be comparable to that of the conventional cemented THA, based on data from the Finnish Arthroplasty Register (3). The cumulative revision percentage of ReCap-M2a-Magnum THA at 7 years was 6.4% (95% CI: 4.8–8.4) according to the Australian registry data (6) and 7% (95% CI: 4.7–8.0) according to the Finnish Arthroplasty Register (5). Overall, the revision rate of ReCap-M2a-Magnum THA is higher than that of conventional THA, but lower than that of ASR THA (5, 6, 26). A hazard alert for the Biomet M2a (38 mm and Magnum) MOM devices was issued in Australia in February 2015 (27).
Bosker et al. (21) reported a pseudotumor incidence of 39% and revision rate of 12% 3.6 years after the ReCap-M2a-Magnum THA procedure. In our preliminary study of 80 ReCap-M2a-Magnum THA, we found 11 definitive ARMD cases, of which 3 had been revised at that stage (12). Recently, Bosker et al. (13) screened 706 ReCap-M2a-Magnum hips in 626 patients using CT and found 228 pseudotumors (32%) in 219 patients (35%). In addition, 76 hips (11%) were revised in 73 patients (12%) after a median of 5.3 (1.0–8.3) years. The CT-detected pseudotumor formation in their study was remarkably more common than the MRI/ion measurement–based ARMD prevalence in our study, which was based only on selective imaging. The difference between the outcomes of these two studies is understandable due to the selective MRI use in our study.
It is possible that there were asymptomatic patients in our study with low ion levels, fluid collection, or soft tissue masses in their hips. However, we believe that the clinical importance of the imaging findings of asymptomatic patients with normal ion levels may be diminutive. The overall revision rate in our study was slightly lower than that in the study by Bosker et al. (13). Most of our revisions were performed for reasons other than ARMD, such as periprosthetic fractures, lack of osteointegration of uncemented implants, and infection. The reasons for revision were not assessed in the study by Bosker et al. (13); however, we agree that the early detection of pseudotumors is important because if revision surgery is performed in patients before substantial soft tissue damage has occurred, the outcome is likely to be better. Furthermore, many of the ReCap-M2a-Magnum THA revisions in our clinic were complicated by cold-welded femoral heads and adapters, which is a unique feature of this device (28).
ARMD may occur in asymptomatic LDH MOM hips (10); however, pain, subluxation sensation, clicking, and fair/poor OHS scores were significantly associated with ARMD in our study. Pain was also associated with ARMD in the study by Bosker et al. (13). In addition, female gender and small diameter head sizes (⩽44) have been reported as risk factors for ARMD in previous studies (8); but in one multivariable model, larger head sizes were associated with ARMD when compared to medium sizes. Theoretically, it is possible that the lubrication between the bearing surfaces works best with medium sized heads. In our study, retroverted cups were scarce, but they were significantly associated with the ARMD revisions. The probability of edge loading is increased with malpositioned cups.
The theoretical health risks related to chronically elevated blood metal ion concentrations induced by abnormal wear and corrosion of MOM implants include systemic symptoms of poisoning (29). Systemic metal ion toxicity cases due to a failed hip replacement are rare; however, there have been several recent reports of systemic cobalt toxicity, including symptoms like fatigue, weakness, hypothyroidism, cardiomyopathy, polycythemia, visual and hearing impairment, cognitive dysfunction, and neuropathy (30). We are not aware of any of our patients having severe systemic symptoms of cobalt poisoning, although these symptoms were not systematically scrutinized.
We found a high prevalence of ARMD in a systematic screening of all of the ReCap-M2a-Magnum THA patients at our institution, but most of these patients did not require a revision operation. Moreover, 77% of the hips did not present with ARMD findings. Time course analysis estimates have been presented, suggesting that the development of pseudotumors continues in ReCap-M2a-Magnum THA patients (13). Therefore, annual follow-ups of all patients subjected to LDH MOM THA have been suggested for as long as the prosthesis is in situ (13). The Finnish Arthroplasty Society (31) has recommended following these patients biannually using patient questionnaires, metal ion level measurements, and imaging techniques like MRI, CT, or ultrasound when needed. Based on these results, the follow-ups should not be discontinued.
Footnotes
Acknowledgements
The authors thank Mari Järvinen and Laura Lehtikunnas for running the screening in practice; Jari Mokka and Petri Virolainen for designing the protocol, for doing the surgery, and recording the intraoperative data; Joni Hirviniemi for collecting the data; and Terhi Koivunen and Kimmo Mattila for the radiologic screening and revision of the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by Vasa Läkareförening rf.
Level of Evidence
Level III, therapeutic study.
