Abstract
Introduction:
This study used data from a prospectively maintained trauma database to assess the level of systolic blood pressure at which mortality rates for trauma begin to increase and to compare systolic blood pressure with base deficit as a predictor of outcome.
Methodology:
The Pietermaritzburg Metropolitan Trauma Service maintains a prospective digital trauma registry. All trauma patients admitted to the service for the period January 2012–January 2015 were included. Analysis was performed on systolic blood pressure relative to a number of selected markers of outcome and a variety of physiological parameters.
Results:
Out of an original data set of 2974 trauma patients, a total of 169 elective patients, 799 patients with isolated traumatic brain injury, and 27 patients with incomplete data were excluded to leave a sample size of 2148 patients. Of these, 1830 (85.2%) were males and 318 (14.8%) were females. The mean age (standard deviation) was 31.8 (12.0) years. The median age (interquartile range) was 29 (23–37) years. There were 89 deaths in this cohort (4.1%). The median systolic blood pressure (interquartile range) was 123 (112–136) mmHg. The median base deficit was −1.4 (interquartile range: −4.5 to 1). The inflection curves below with fitted non-linear curve clearly show the upward change in mortality frequency around a systolic blood pressure of ⩽110 mmHg as well for a base deficit below −5. A cutoff of <110 for systolic blood pressure yields a high sensitivity and very high positive predictive value of 82% (95% confidence interval: 81–84) and 98% (95% confidence interval: 97–98), but low specificity (56%) and negative predictive value (12%), respectively. Similar optimal cutoff analysis for base deficit versus mortality suggests base deficit >4.8 as a good predictor area under the curve (0.82; 95% confidence interval: 0.75–0.88). This cutoff yields a high sensitivity of 80% (95% confidence interval: 78–82), moderate specificity of 75% (95% confidence interval: 62–85), very high positive predictive value of 98% (95% confidence interval: 97–99) but low negative predictive value of 17% (15–28).
Conclusion:
The data suggest that traumatic shock starts to become manifest at a systolic blood pressure of 110 mmHg and that a systolic blood pressure reading of 90 mmHg represents an advanced state of shock. Systolic blood pressure by itself is a poor predictor of mortality and outcome. Base deficit appears to be a far better predictor of mortality than systolic blood pressure. Future models to categorize shock will have to combine vital signs with biochemical markers of hypoperfusion.
Introduction
The presence of shock in a trauma patient is associated with increased mortality and morbidity. The focus of trauma resuscitation is on the early identification of shock and its appropriate treatment. In the field and in most hospitals, shock is diagnosed by a combination of clinical signs and by the use of standard physiological parameters (1–7). The two most widely used physiological parameters to assess shock are heart rate and systolic blood pressure (SBP). For the purposes of education and clinical usefulness, a number of classifications of shock have been popularized. The most ubiquitous is the Advanced Trauma Life Support (ATLS) classification (1, 2). This breaks shock down into four classes based on the pulse rate, the SBP, the estimated volume of blood loss, and the level of consciousness of a patient. Currently, stage III shock is diagnosed at a SBP of 90 mmHg (1, 2). In addition, a number of other international groups and standard text books use a systolic blood pressure (BP) of <90 mmHg as the definition of shock.
The development of large electronic trauma registries and databanks has allowed for analysis of large cohorts of patients. Using data from these registries may allow for a more scientific basis for the selection of a number which can be used clinically. Several studies have been published over the last decade from North American, European, and military databases which suggest that the choice of SBP reading of <90 mmHg to diagnose shock is inappropriate and that shock begins at much higher SBP of around 100–110 mmHg (2–7). In light of this, there had been a move away from an emphasis on a specific SBP reading as indicating shock. The most recent ATLS manual states that “no vital sign and no laboratory test can diagnose shock.” The manual goes on to reiterate that an undue focus on SBP as an indicator of shock may well result in delayed recognition of the condition. Our service has recently developed a hybrid electronic medical record system, which allows us to capture clinical data in real-time and incorporate it into a relational database (8, 9). Our service is in a low-middle income country (LMIC) and is plagued with long transfer times, inadequate pre-hospital services, and a high burden of penetrating trauma (10, 11). This makes our patient cohort different from cohorts of other high-income countries (HIC) and from military cohorts. In light of these differences, the findings in the literature from international trauma services and our rapidly expanding clinical database, we elected to replicate these studies and see whether their findings applied equally to a cohort of civilian trauma patients in our environment. Based on our data, we hoped to generate a simple clinical model which would assist clinicians working in our service to accurately identify and categorize shocked trauma patients.
Setting
The Pietermaritzburg Metropolitan Trauma Service (PMTS) maintains a prospective digital trauma registry, which captures data at our institutions. Ethical approval to maintain the registry has been obtained from the Biomedical Research Ethics Committee (BCA221/13 BREC) of the University of Kwa Zulu-Natal and from the Research Unit of the Department of Health. This digital registry is somewhat unique and has been discussed in the literature. The clerking medical staff enters the data onto an electronic pre-prepared clerking sheet. This process is the clerking process for all new admissions, so the clinical data are entered in real-time. As the data are entered, it is then directly incorporated into the registry. The completed pre-prepared clerking sheet is then printed out and becomes the patient’s clinical records. At operation and at discharge, a similar process is followed. This system combines the functions of a medical registry and a medical record system. It also combines an electronic system with a paper-based system and has been called the Hybrid Electronic Medical Registry (8, 9).
Management
All trauma patients are received in the emergency unit and are assessed and resuscitated, according to standard ATLS protocols, by staff of the department of surgery and of the emergency unit. Automated BP cuffs are used to measure SBP in all trauma patients. Routine radiographic studies are obtained as indicated and routine blood tests, including electrolytes, glucose, and full blood count, and arterial blood gas are obtained as indicated. These specimens are taken by the junior medical staff and analyzed by the laboratory service based in the hospital.
Methodology
All trauma patients older than 15 years of age admitted to the service for the period January 2012–January 2015 were included. For the purposes of this study, a methodology similar to those used in the North American and European civilian series and in the single military series was used and modified where appropriate. As in these previous studies, traumatic brain injury (TBI) was excluded by removing all patients with both a Glasgow Coma Score <8 and base deficit (BD) <5. Analysis was performed on SBP relative to overall mortality, mortality excluding brain injury, number of complications, number of ventilator days, number of intensive care unit (ICU) days, total length of hospital stay, age, and gender. Subset analysis was performed on blunt compared to penetrating trauma and on patients who presented with a delay greater than 6 h from time of injury.
Statistical Methods
Data were analyzed using Stata 13.0 and R statistical package 3.0.3 (12–14). Several methods or criteria for selecting optimal cutoffs have been implemented using receiver operating characteristic (ROC) curves. The commonly used Youden index was used to identify the optimal breakpoint for low systolic BP (shock) and BD for the optimal classification of mortality. From an optimization point of view, this approach is identical to the method that maximizes the sum of sensitivity and specificity and to other criterion that maximizes concordance. This approach yields a high sensitivity and positive predictive value (PPV), that is, few missed deaths based on this cutoff and few false positives. We deemed this preferable to maximizing specificity. The discriminatory power was evaluated by the area under the ROC curve (AUC). An AUC value of 0.5 indicates no discriminative ability, while an AUC exceeding 0.8 suggests good to excellent predictive capability. Sensitivity and specificity based on the optimal cut-point were calculated along with 95% confidence intervals (CIs). Comparison of mortality frequency and other categorical characteristics by shocked classification (systolic BP <110 versus ⩾110) was performed using Pearson’s chi-square (χ2) test or Fisher’s exact test (if expected cell count with fewer than five observations). Differences in mean base excess and other continuous characteristics by shocked status were performed using the standard t-test. A p-value of <0.05 was considered statistically significant.
Results
Out of an original data set of 2974 trauma patients, a total of 169 elective patients, 799 patients with isolated TBI, and 27 patients with incomplete data were excluded to leave a sample size of 2148 patients. Fig. 1 summarizes this breakdown. Of these, 1830 (85.2%) were males and 318 (14.8%) were females. There were 825 patients who sustained blunt trauma and 1247 sustained penetrating trauma and 76, a combination of blunt and penetrating trauma. Time from injury to presentation was recorded in 1277 patients. At total of 1067 took more than 6 h to reach hospital and 210 arrived within 6 h. The mean age (standard deviation (SD)) was 31.8 (12.0) years. The median age (interquartile range (IQR)) was 29 (23–37) years. There were 89 deaths in this cohort (4.1%). The median systolic BP (IQR) was 123 (112–136) mmHg. The median BD was −1.4 (IQR: −4.5 to 1). Note that there were 1229 non-missing observations for BD. Fig. 2 summarizes these data. The inflection curves below with fitted non-linear curve clearly shows the upward change in mortality frequency around a SBP of 110 mmHg and lower as well for a BD below −5. A cutoff of <110 for SBP yields a high sensitivity and very high PPV of 82% (95% CI: 81–84) and 98% (95% CI: 97–98) but low specificity (56%) and negative predictive value (NPV) (12%), respectively. Similar optimal cutoff analysis for BD versus mortality suggests BD >4.8 as a good predictor AUC (0.82; 95% CI: 0.75–0.88) (see Fig. 3). This cutoff yields a high sensitivity of 80% (95% CI: 78–82), moderate specificity of 75% (95% CI: 62–85), very high PPV of 98% (95% CI: 97–99) but low NPV of 17% (95% CI: 15–28). Combining SBP and BD in the same predictive model did not significantly improve on the AUC observed for BD alone, suggesting no additive effect of the two predictors. The combined AUC was 0.824. Fig. 3 shows the inflection curves, and Fig. 4 shows the ROC curves. Based on this cutoff, 440 patients (20.5%) were classified as shocked (SBP <110) or 283/1229 (23.2%) with high-risk BD values. Interestingly, a second but smaller upward inflection of mortality risk at higher SBP (hypertensive range) was also observed. Shocked patients had a significantly higher frequency of mortality (11% versus 2%), a significantly higher median lactate level (3.0 versus 1.6), as well as larger median BD (−4 versus −1) and significantly higher frequency of ICU admission (19% versus 10%). Translated in odds ratios, those who died were 5 times more likely to have been had a SBP of <110, while those with a BD below the identified cutoff of −5 were almost 12 times more likely to be died (odds ratio (OR) = 11.5, 95% CI: 6.4–20.6). No significant difference in mean age, ICU duration, and length of hospital stay was observed by shocked status. We observed similar differences for these variables by low BD status, except that those with low BD had a significantly longer ICU and hospital stay and a lower median SBP, that is, 115 versus 124 (p < 0.001). In total, 8 out of 10 patients with a SBP <60 died, while 51% (19/37) patients with a BD ⩽−14 died. Table 1 summarizes these comparisons by shocked status and Table 2 by low BD. There was no difference in parameters between blunt and penetrating trauma sets. Table 3 summarizes these two cohorts of patients. Mortality was significantly higher in the cohort, which reached hospital within 6 h. Table 4 compares the two groups according to delay.
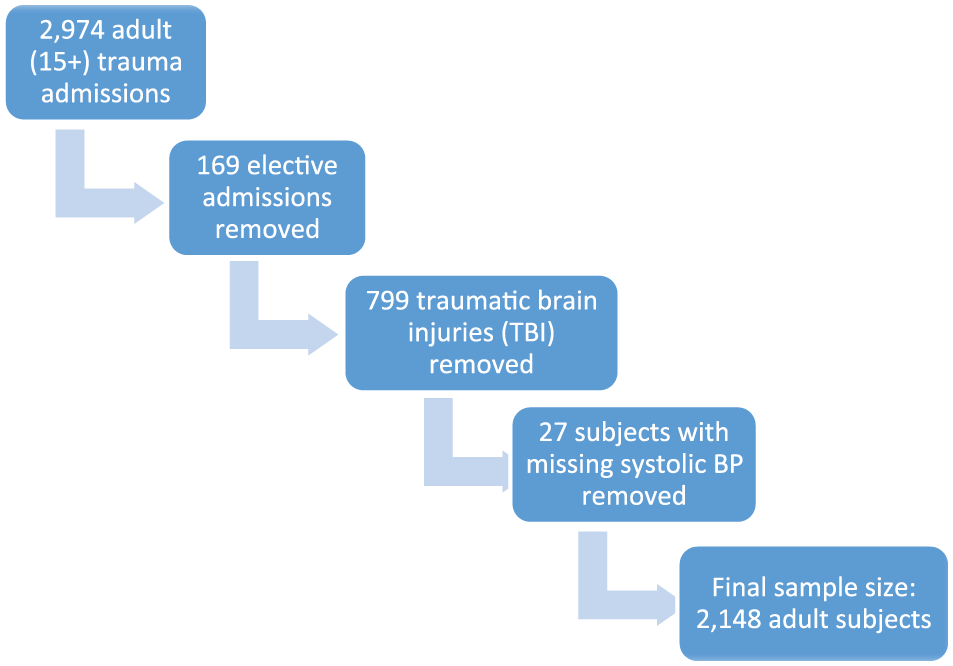
Description of study sample.
SBP and base deficit for the entire cohort.
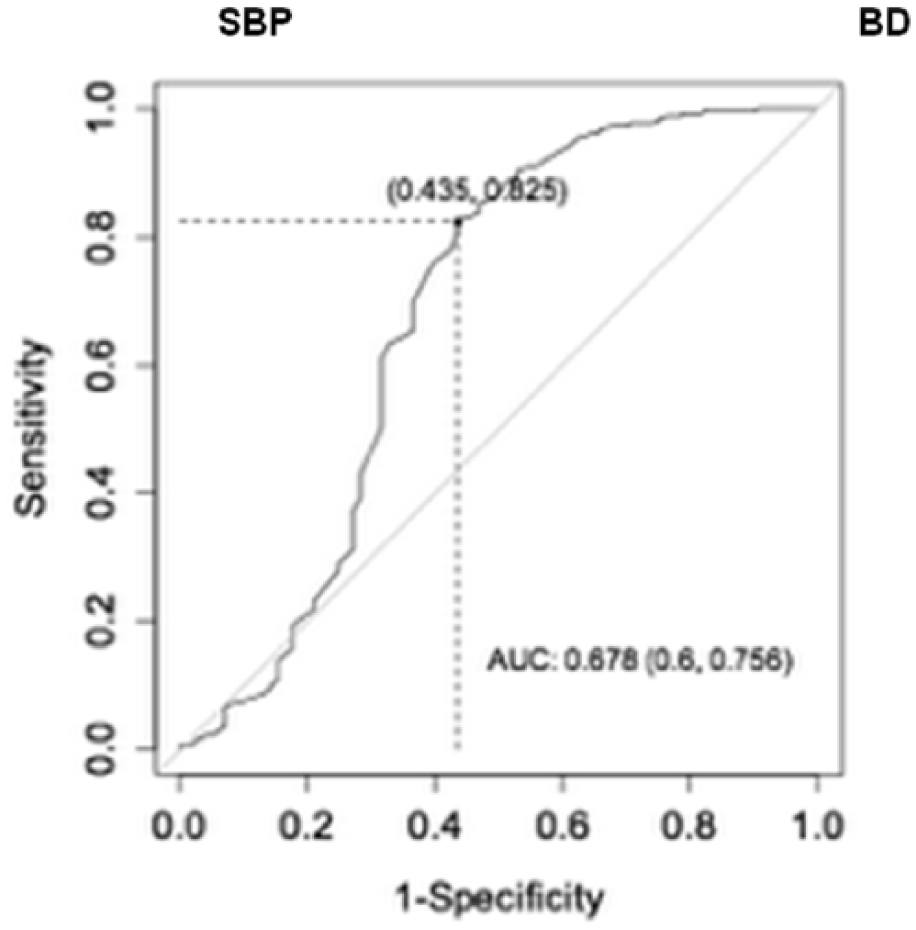
ROC curve of mortality versus SBP and base deficit.
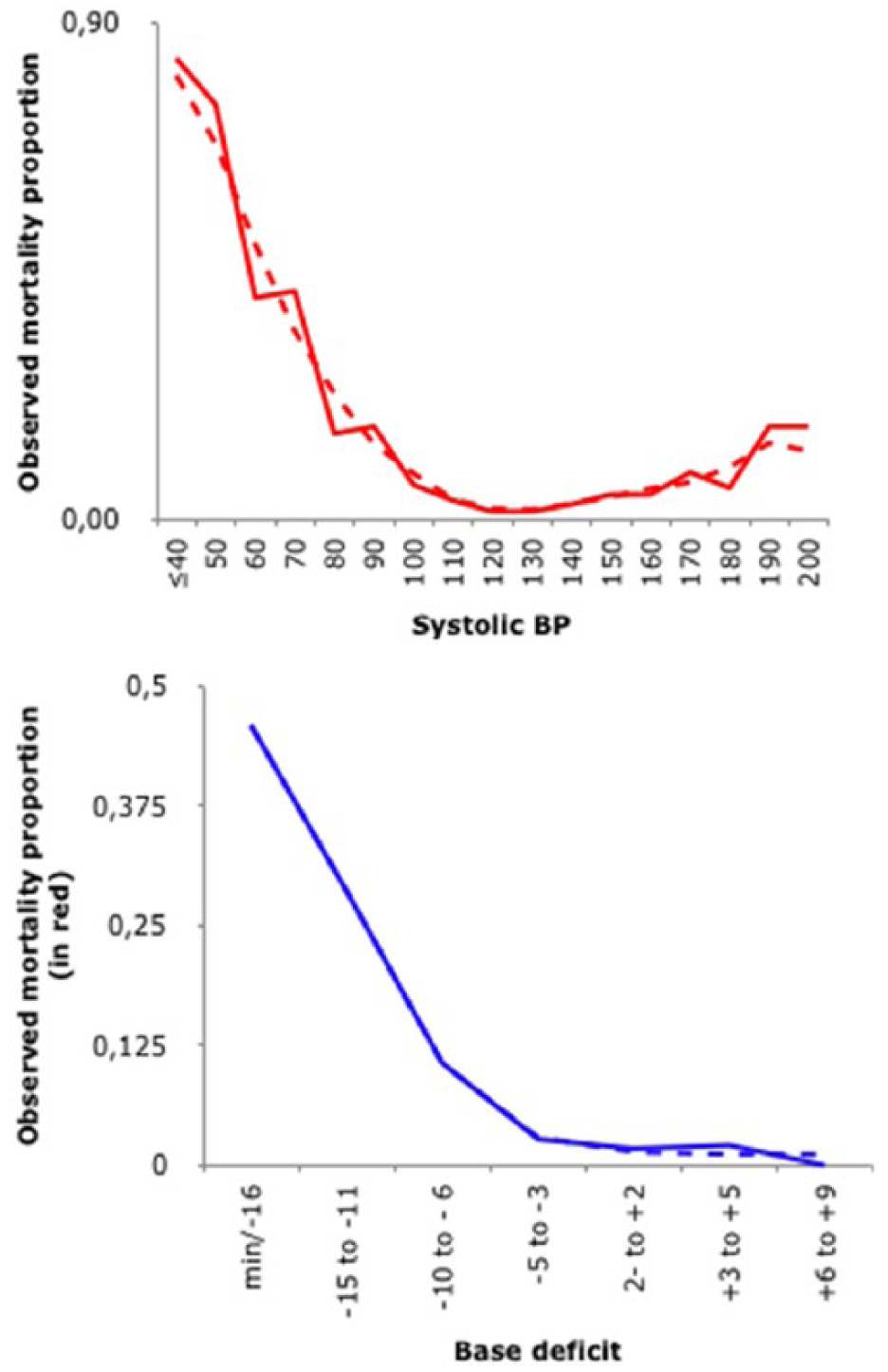
Mortality plotted against SBP and base deficit.
Summary of the entire patient cohort according to SBP.
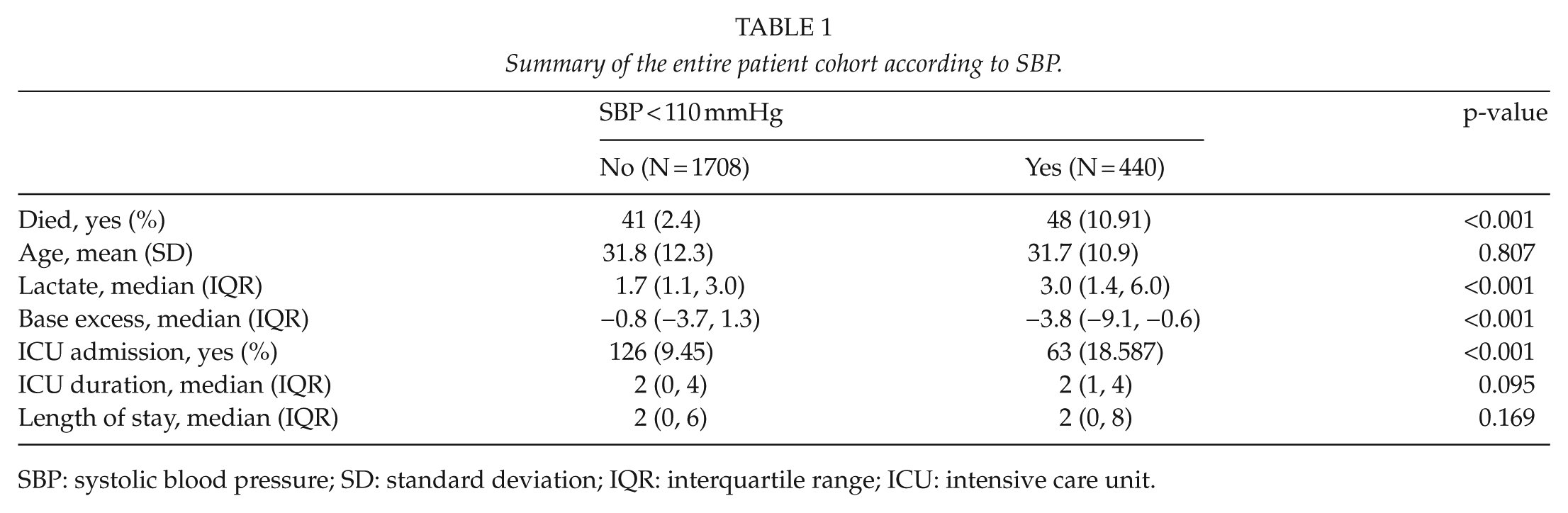
SBP: systolic blood pressure; SD: standard deviation; IQR: interquartile range; ICU: intensive care unit.
Cohort summarized according to base deficit.
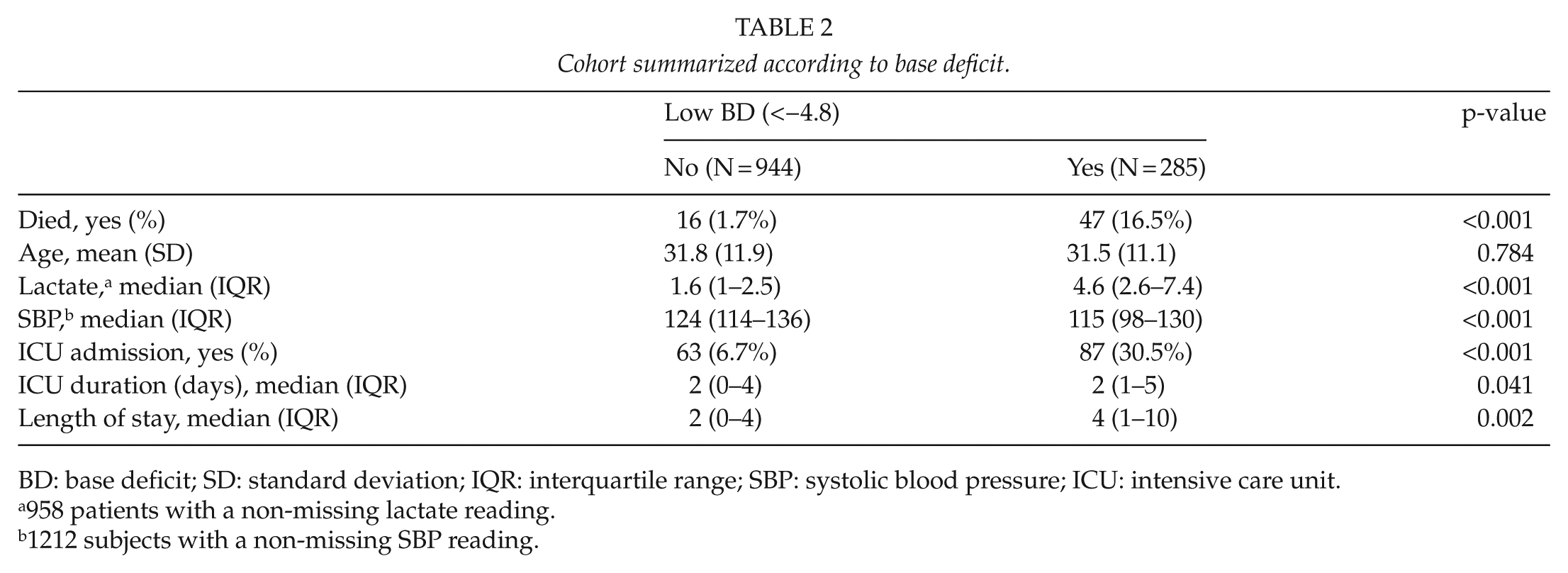
BD: base deficit; SD: standard deviation; IQR: interquartile range; SBP: systolic blood pressure; ICU: intensive care unit.
958 patients with a non-missing lactate reading.
1212 subjects with a non-missing SBP reading.
Blunt versus penetrating trauma.
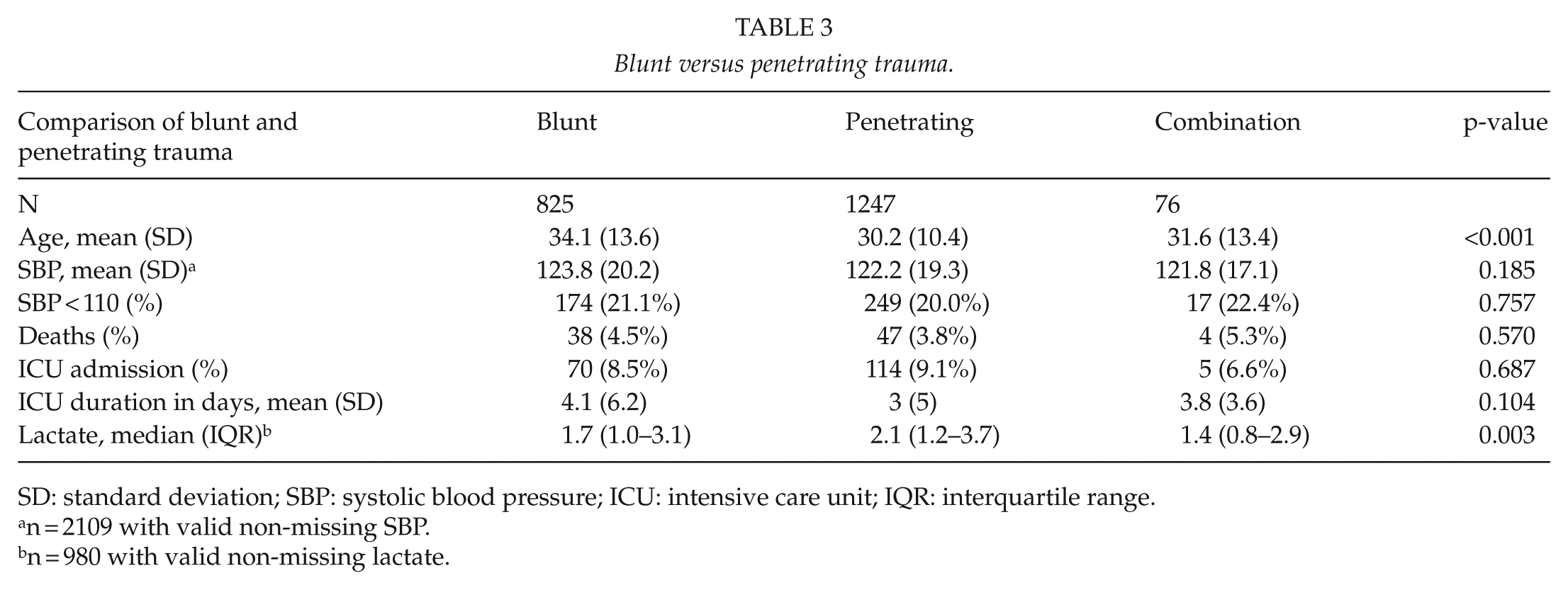
SD: standard deviation; SBP: systolic blood pressure; ICU: intensive care unit; IQR: interquartile range.
n = 2109 with valid non-missing SBP.
n = 980 with valid non-missing lactate.
Comparison of delay versus non-delay group.
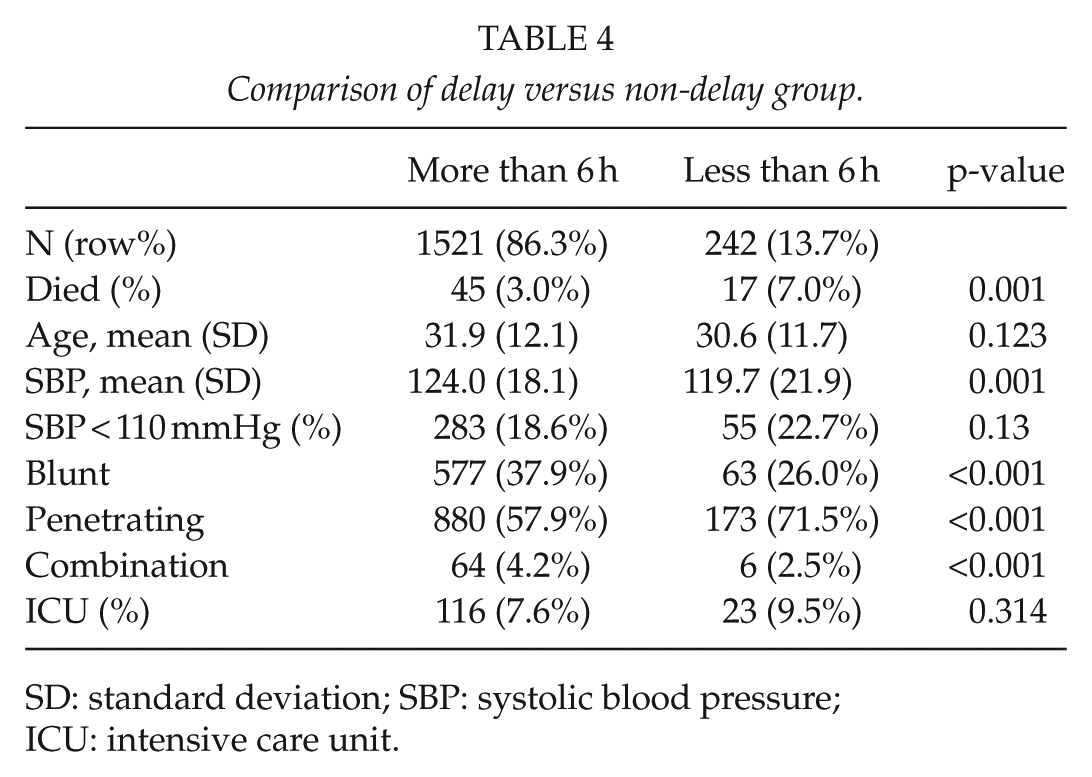
SD: standard deviation; SBP: systolic blood pressure; ICU: intensive care unit.
Modeling Outcome
Multivariate analysis was performed to identify predictors of mortality. These were then incorporated into a prediction model with a better predictive power (95.72%). We constructed a multivariable model using an accepted purposeful-selection approach. We employed a logistic regression modeling approach given the dichotomous outcome assessed. Goodness-of-fit model was also assessed. The logistic formulation allows the inclusion of continuous covariates such as uncategorized age. All covariates with a univariate p-value of <0.15 were selected for inclusion in the final multivariable model. We then checked model fit and collinearity, and we did not include ICU admission in the multivariable model. The optimal cutoff in predicted probability of death based on the model compared to the observed data revealed a model with both good sensitivity (0.8) (95% CI 0.7–0.9) and specificity (0.7) (95% CI 0.7–0.8) and an area under the ROC curve of 0.861 (0.823, 0.9). Table 5 summarizes the multivariable logistic prediction model for mortality and includes univariate results.
Univariate and multivariable logistic prediction model for mortality.
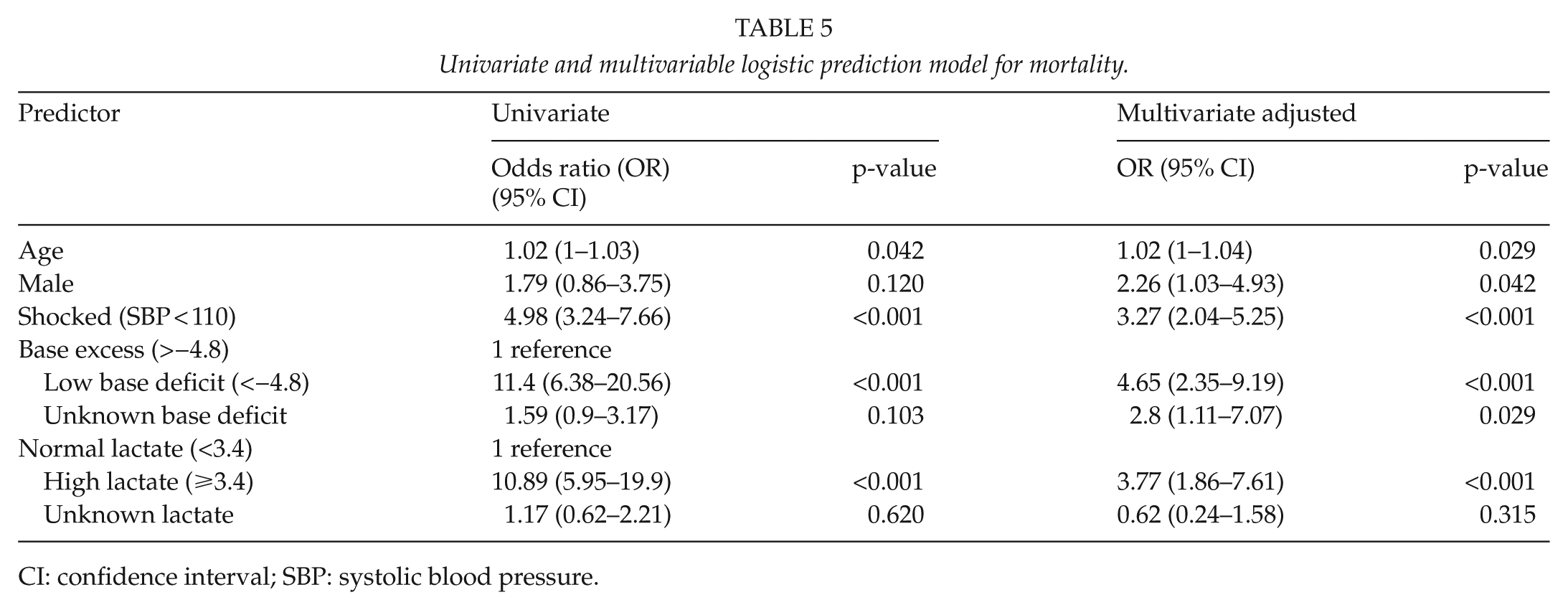
CI: confidence interval; SBP: systolic blood pressure.
Discussion
ATLS, which remains the gold standard for trauma care, traditionally taught that patients with a SBP of <90 mmHg should be triaged directly to a higher level of care. There is however increasing evidence to suggest a SBP of <90 mmHg that represents a decompensated state of shock and that triage and increased levels of concern should occur at much higher levels of SBP (4–7). ATLS has acknowledged this and the more recent 2012 course manual clearly states “no vital sign and no laboratory test can diagnose shock.” Later in the chapter, it states, “reliance solely on systolic blood pressure as an indicator of shock can result in delayed recognition of the shock state.” Over the last decade, a number of audits of large clinical databases from North America, Europe, and the military have questioned the use of a SBP of 90 mmHg as a trigger for more aggressive treatment and triage, and a number of authors have proposed new classifications of shock (15–18). Our study has applied these previously described methodologies to a single center database of trauma patients from a LMIC with a unique trauma load and has confirmed the results of these international studies. This is important corroboratory evidence as the demographics of trauma patients in South Africa are different to those in the high-income country databases and military databases. The patients are generally younger and there is a very high incidence of penetrating trauma (10, 11). Our data support the contention of the international reports that using a value of 90 mmHg to diagnose this “tipping” point is inappropriate (15–18). This is further complicated by the well-described vagaries of automated BP readings, and this is especially the case when these devices are used to measure SBP in young trauma patients (19).
Parks et al. reviewed over 115,000 blunt and penetrating trauma patients and showed that a SBP <90 mmHg is associated with markedly deranged BD readings of <20. They concluded that a SBP of <90 mmHg represents established not early shock (2). Bruns et al. (3) in a North American study from 2008 reviewed 16,000 blunt and penetrating trauma victims in the pre-hospital setting and showed that at a SBP of 110 mmHg mortality began to increase. At a SBP of ⩽70 mmHg, just over two-thirds of patients in their series died. A large multicenter series of 730,000 patients with blunt or penetrating trauma taken from the National Trauma Databank in the United States was analyzed, and the authors plotted SBP readings against mortality rates (4). This showed an inverse relationship between decreasing BP and increasing mortality and as in the previous studies identified a SBP of 110 mmHg as the inflection point at which mortality began to climb. The authors went on to show that mortality increased by 4.8% for every 10 mmHg decrease in SBP below a level of 110 mmHg. These North American findings have been supported by analysis of the TARN database from Europe and the United Kingdom (5). This group reviewed 47,927 predominantly blunt trauma victims with a median age of 51 years. In keeping with the previously quoted studies, these authors identified a SBP <110 mmHg as indicating shock. At this level, a significant increase in mortality became apparent and doubled at <100 mmHg, tripled at <90 mmHg, and increased by a factor of 5–6 at <70 mmHg, regardless of age. This group went on to focus on penetrating trauma and subsequently reported on 3444 penetrating trauma patients from their multicenter database. The authors showed that in this cohort the same finding held true as in the larger mixed cohort (6). Mortality rates doubled at a SBP of between 90 and 109 mmHg were 4-fold higher at 70–89 mmHg and 10-fold higher at <70 mmHg. Eastridge et al. (7) analyzed 7180 war casualties for the 7 years from 2002 to 2009. They looked at admission SBP, as well as BD, and mortality and went on to plot SBP against mortality. The mortality rate remained below 2% until a level of between 101 and 110 mmHg was reached. From that point onward, the slope of the curve increased almost exponentially and at a SBP of 60 mmHg, the mortality rate breached the 50% mark. There were no survivors when SBP was not recordable. All the above studies concur with the findings in this report that mortality begins to increase at an SBP of 110 mmHg and that the traditional SBP of 90–100 mmHg represents decompensated shock. However, SBP by itself is a poor predictor of mortality and a poor indicator of the presence of shock. SBP needs to be interpreted in conjunction with other data such as BD when diagnosing shock. BD appears to be a far more accurate and specific means of assessing shock as evidenced by AUC and by the correlation of BD with length of ICU stay and length of hospital stay. A more rigorous model needs to be developed to accurately quantify shock and guide therapy. Our own endeavor suggests that by incorporating vital signs like SBP and biochemical markers of hypoperfusion, such as lactate levels and BD, much better predictive models can be generated. Such a model will need to be field tested to see whether it is practical and robust enough for routine clinical use. As data continue to accrue in trauma databases around the world, researchers will be able to subject such models to rigorous empirical assessment.
There does not appear much difference in this study between the blunt and penetrating trauma cohorts in terms of demographics and outcomes. The data concerning delay are somewhat counter intuitive, and it would appear that mortality is higher in patients who arrive within 6 h. In addition, these are more likely to have sustained penetrating trauma. This suggests selection bias. Unstable patients are rushed to hospital, whereas stable ones seem to not be transported with the same degree of urgency. This does support our contention that our data set is somewhat unique as it reflects an immature pre-hospital system, unlike the data from the military and HIC civilian series, which we used as the basis of our own investigation. Of interest is the increase in mortality seen with hypertension. This needs further investigation and there are a number of potential explanations. The emerging concept that hypertension exacerbates traumatic bleeding is the focus of much research and has led to the approach of permissive hypotension in patients with torso trauma without a TBI. We do not currently have enough patients in this category to be able to provide any deeper analysis of this phenomenon and hopefully ongoing data accrual will allow for more in-depth work focusing on this in the future.
The study has a number of limitations. By the nature of all clinical registries key data sets may be missing, and this can be a major methodological limitation. However, a comparison of characteristics between those with missing and non-missing values for BD and/or lactate suggested that there were no significant differences between the patients with complete and incomplete data sets. We choose to include these as a missing category rather than using multiple imputations. We did compare the missing subjects in terms of other available socio-demographic data and it appears that there were no significant differences in this regard. Similarly, the issue of selection bias in deciding on the urgency of transfer may distort the results. It has to be acknowledged that more severely injured patients may die in the field and may not survive to reach hospital and that those who survive >6 h in the pre-hospital setting have, by definition, at least partially compensated for their injuries. However, stratifying by delay status did not affect the direction of the observed associations. Although the power is reduced for the <6-h group as there are much fewer observations in this group, the BP/shock relationship appeared to be more strongly associated with mortality in the <6-h group compared to the ⩾6-h group.
Conclusion
There is a strong body of evidence from North America, Europe, the military, and now from South Africa, which supports redefining the cutoff SBP, which indicates that impaired perfusion due to both low cardiac output and overwhelmed compensatory mechanisms is becoming critical. The data suggest that this starts to become manifest at a SBP of 110 mmHg and that a SBP reading of 90 mmHg represents an advanced state of shock. However, SBP by itself is a poor predictor of mortality and must be interpreted in conjunction with other parameters when managing acute trauma patients. BD appears to be a far better measure of shock and predictor of outcome than SBP. Future models to categorize shock will have to combine vital signs with biochemical markers of hypoperfusion.
Footnotes
Acknowledgements
D.L. Clarke contributed in concept drafting, P. Brysiewicz contributed in draft of document and references, B. Sartorius contributed in statistics, J.L. Bruce contributed in data collection and cleaning, and G.L. Laing contributed in data collection and drafting. The authors have met with all ethical requirements in the writing of this paper and the ethics number is BE 121/13.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
