Abstract
Background and Aims:
Reconstruction with reconstitution of the container function of the abdominal compartment is increasingly being performed in patients with massive ventral hernia previously deemed inoperable. This situation places patients at great risk of severe intra-abdominal hypertension and abdominal compartment syndrome if organ failure ensues. Intra-abdominal hypertension and especially abdominal compartment syndrome may be devastating systemic complications with systematic and progressive organ failure and death. We thus reviewed the pathophysiology and reported clinical experiences with abnormalities of intra-abdominal pressure in the context of abdominal wall reconstruction.
Material and Methods:
Bibliographic databases (1950–2015), websites, textbooks, and the bibliographies of previously recovered articles for reports or data relating to intra-abdominal pressure, intra-abdominal hypertension, and the abdominal compartment syndrome in relation to ventral, incisional, or abdominal hernia repair or abdominal wall reconstruction.
Results:
Surgeons should thus consider and carefully measure intra-abdominal pressure and its resultant effects on respiratory parameters and function during abdominal wall reconstruction. The intra-abdominal pressure post-operatively will be a result of the new intra-peritoneal volume and the abdominal wall compliance. Strategies surgeons may utilize to ameliorate intra-abdominal pressure rise after abdominal wall reconstruction including temporizing paralysis of the musculature either temporarily or semi-permanently, pre-operative progressive pneumoperitoneum, permanently removing visceral contents, or surgically releasing the musculature to increase the abdominal container volume. In patients without complicating shock and inflammation, and in whom the abdominal wall anatomy has been so functionally adapted to maximize compliance, intra-abdominal hypertension may be transient and tolerable.
Conclusions:
Intra-abdominal hypertension/abdominal compartment syndrome in the specific setting of abdominal wall reconstruction without other complication may be considered as a quaternary situation considering the classification nomenclature of the Abdominal Compartment Society. Greater awareness of intra-abdominal pressure in abdominal wall reconstruction is required and ongoing study of these concerns is required.
Keywords
Introduction
Technical innovations coupled with a broader understanding of abdominal wall anatomy/physiology have heralded major advances in the surgical reconstruction of massive ventral hernias. Patients with so-called abdominal wall failure, who have often lost the “intra-peritoneal domain” of their viscera, now have surgical options previously unavailable or unsafe. Surgical approaches have evolved into reconstruction involving reconstructing functional dynamic abdominal walls rather than from simply patching holes (1, 2). This practically translates, however, into less robust patients being put at higher risk for increased intra-abdominal pressure (IAP) which may manifest as intra-abdominal hypertension (IAH) or the deadly abdominal compartment syndrome (ACS) (3, 4). The ACS has traditionally been understood as a complicated second hit of critical illness/trauma in pro-inflammatory, immunologically primed patients with globally enhanced vascular permeability undergoing aggressive resuscitations for catastrophic conditions. Patients undergoing abdominal wall reconstruction (AWR) should presumably be fundamentally different, being well, and not shocked, prior to the stresses of surgery. This may potentially mitigate against the development of IAH/ACS and potentially allow an increased tolerance to these conditions. There are many unknowns, however, and as AWR becomes more common, the risks and implications of IAH/ACS should be reviewed in light of the potentially serious issues regarding IAP in these patients, along with potential strategies to avoid or mitigate them.
IAH and the ACS
IAH is abnormally raised IAP within the abdominal compartment (3, 4). A working grading system for distinguishing levels of IAH has been adopted to communicate gradations of concern pending ongoing research (Table 1) (3). When IAP is repeatedly elevated to at least 20 mmHg and associated with new organ failure, ACS is diagnosed. Through a working consensus definition, 20 mmHg was adopted as the diagnostic threshold, although the critical concepts are abnormally high IAP impairing tissue and organ perfusion, affecting multiple interconnected body regions in a polycompartment mechanism, resulting in multiple organ dysfunction (5–7). Overt ACS is a highly lethal syndrome that when fully established is clinically associated with acute cardiorespiratory respiratory and renal failure, prominent among many other more subtle acute organ dysfunctions. Treatment often requires morbid therapies, such as decompressive laparotomy (8, 9). Although sporadically described for over 100 years (10), this syndrome was only widely appreciated in the 1980s in association with liberal (and often massive) crystalloid fluid administration in catastrophic clinical settings (10–12). Subsequently, it has been increasingly appreciated that almost any severe medical/surgical condition necessitating massive crystalloid resuscitation may be associated with the ACS (13–15).
Grading system for intra-abdominal hypertension.

IAH: intra-abdominal hypertension; IAP: intra-abdominal pressure.
With recent evolutions in the philosophy and practice of resuscitation, including greatly modulated crystalloid infusions and increased blood product usage, dramatically reduced ACS rates are being noted (16, 17). Thus, in institutions with appropriate awareness of the diagnosis and its ramifications, exposure to overt ACS is becoming rare. However, efforts to reconstruct failed abdominal walls in association with the reduction of extra-peritoneal viscera, without a full appreciation of the functional physiology of the abdominal compartment as a whole, raises the specter of precipitating overt ACS (18). Thus, this article will review the reported evidence concerning AWR from the viewpoint of the abdominal compartment and its pressure/volume interactions as a functional whole. The overtly stated goal is to ideally prevent the emergence of a potential new wave of ACS after AWR by reviewing the relevant physiology/pathophysiology involved.
The Abdominal Compartment Society (Wsacs.Org): A Focus on IAH
Recognizing the shifting playing field for treating the critically ill/injured and recognizing important topics not being fully addressed, the World Society of the Abdominal Compartment Syndrome recently evolved to become the Abdominal Compartment Society (www.wsacs.org) with an expanded mission to fully understand the complete form, function, and treatment of diseases and dysfunction of the abdominal compartment as a whole (19). Among many challenges, IAH remains central. IAH remains nearly ubiquitous in critical illness/injury (20, 21), yet is often ignored (22, 23). Rates of IAH have been reported as being as high as 87% in some critical care units (24). Although causality is not proven, IAH associates with increased complications and death (20, 25). Even at lower grades of IAH in animals (26, 27), there may be profound alterations in mesenteric/renal blood flow that contribute to ongoing pro-inflammatory states and propagate multi-system organ dysfunction in a multi-factorial fashion, in which any one initiating cause is hard to isolate (28).
The Abdominal Compartment and Abdominal Compliance
The abdominal compartment contains viscera within a semi-rigid container that is limited posteriorly and inferiorly by immobile bony structures, has flexible anterolateral dimensions from the abdominal wall, and potential cephalad distensibility with diaphragmatic excursion albeit with potential respiratory consequences (6, 29). The linea alba is a central tendon anchoring the rectus abdominis, oblique, and transverse abdominis muscles in order to aid respiration, posture, and movement. The transversus abdominis muscle activity is most consistently related to changes in IAP as its line of contraction is horizontal rather than vertical or diagonal (30).
As a closed container, the abdominal volume (AV) in conjunction with the distensibility of the abdominal wall defines the abdominal compliance, described by the equation Abdominal Compliance = ΔAV/ΔIAP. Blaser recently described three progressive responses of the abdominal cavity to increased pressure and/or volume. Mechanisms involved three modes of compartment behavior: (1) a reshaping phase with configuration changes and minimal change in IAP (flat slope on an abdominal pressure–volume (PV) curve), (2) a stretching phase through elastic elongation of the abdominal wall and diaphragmatic tissue (near linear slope on PV curve), and (3) rapid pressurization phase with exponential PV changes as found in a rigid space (large/exponentially increasing slope) (7). Reshaping capacity (RC) can be described as the difference between “resting/baseline intra-abdominal volume (IAV)” and maximum IAV attainable without increasing IAP (7). RC depends on age, sex, height, weight, comorbidities, and even gravity (7, 31). Novel studies of anesthetized swine in weightlessness revealed spontaneous conformational changes yielding RC without IAP change increasing the functional compliance during laparoscopic surgery in weightlessness (31–33).
The fact the abdominal cavity can accommodate modest increases in intra-abdominal volume through reshaping and stretching likely explains why marked IAP pressure changes are not seen with most modest hernia repairs. However, the model well explains (and graphically demonstrates) how once compensatory mechanisms are exceeded, IAP rises greatly and exponentially (7). Unfortunately, with massive hernia, especially those with loss of domain (LOD), this rapid pressurization phase can regularly be expected.
The Concept of LOD
LOD refers to the situation wherein intra-abdominal viscera that have herniated outside the native abdominal cavity reside in a secondary non-anatomic cavity (34). It is the result of an irreversible decrease in abdominal muscle elasticity associated with disuse atrophy and is often associated with diaphragmatic descent and dysfunction (35–37). When the linea alba is cut, the abdominal muscles quickly retract laterally with the muscles becoming thicker and shorter. Although the recti muscles are not fully mechanically unloaded and remain attached, they functionally become lateralized (37). When so mechanically studied, the internal oblique muscles in chronic hernia demonstrate increased tensile strength and mechanical toughness in conjunction with significantly reduced elasticity (37). Muscle stiffness increased nearly 60% in herniated specimens and extensibility was decreased by 27% compared to controls (37). This greatly increases the difficulty in re-approximating these muscles. In animal models of incisional hernia, the internal oblique muscles developed changes typical of mechanical unloading, namely, pathologic fibrosis, disuse atrophy, and changed muscle fiber type composition (37).
LOD is frequently used as a concept without specific definition, however (38–40). There is no official consensus on what numbers or measurements constitute LOD, with multiple non-standardized definitions proposed (Table 2) (34, 36, 41). Many authors have also endeavored to derive reproducible pre-operative markers to predict difficulties with IAH/ACS upon AWR, often utilizing novel measurement schemes (Table 3). We thus believe a standardized consensus definition of LOD is warranted, but that these grading systems require further prospective validation before general acceptance.
Suggested definitions for defining the relative peritoneal volume and/or loss of domain in massive ventral hernia.

Pre-operative factors for predicting post-operative complications for abdominal wall reconstruction.
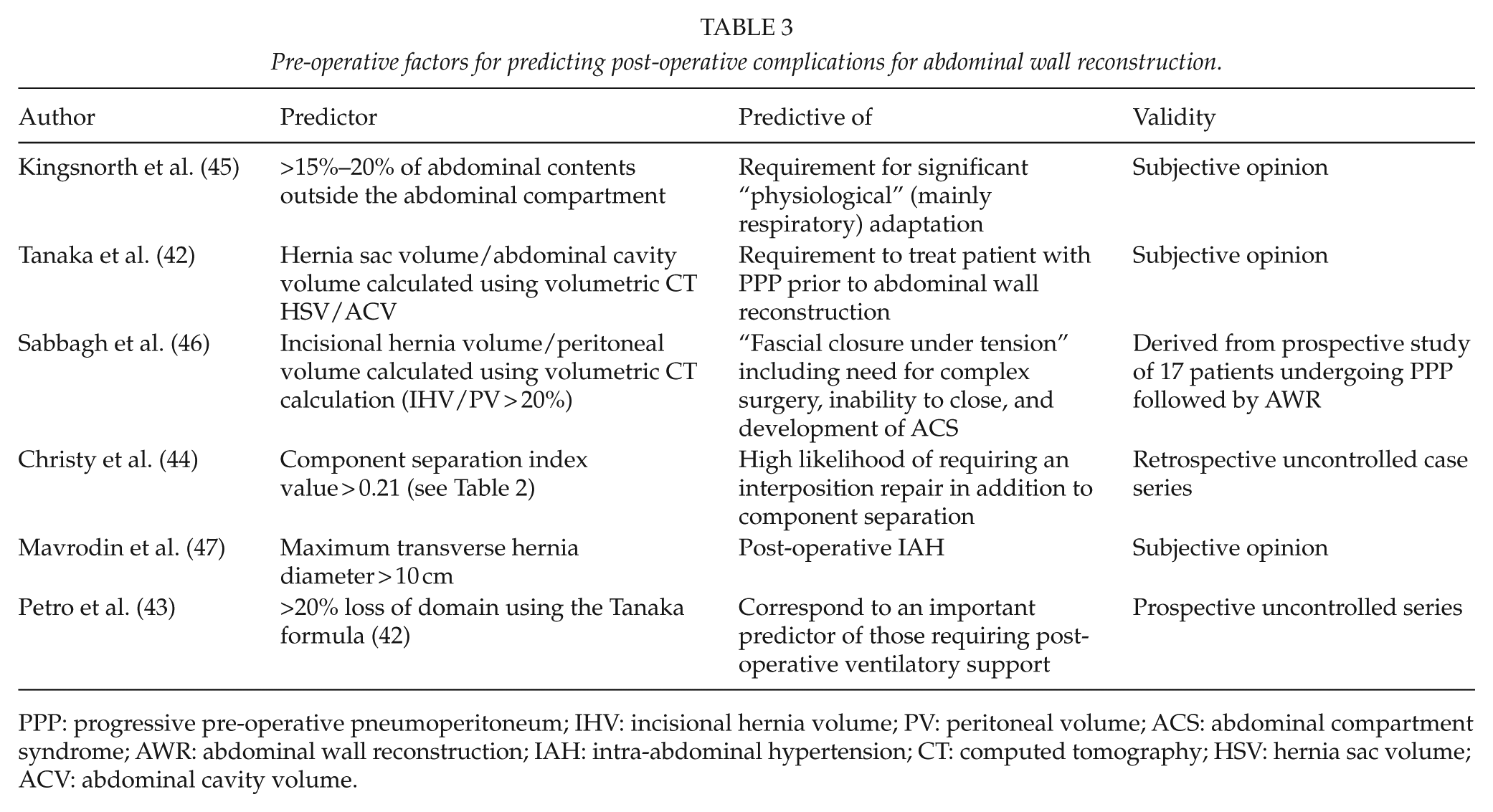
PPP: progressive pre-operative pneumoperitoneum; IHV: incisional hernia volume; PV: peritoneal volume; ACS: abdominal compartment syndrome; AWR: abdominal wall reconstruction; IAH: intra-abdominal hypertension; CT: computed tomography; HSV: hernia sac volume; ACV: abdominal cavity volume.
Epidemiology and Implications of IAH/ACS in AWR
Although it is likely intuitive to most surgeons that replacing displaced viscera within a peritoneal cavity under obvious tension during AWR can induce physiologic compromise, there is surprisingly little formal reporting. In 2015, Oliver-Allen (35) reported such an occurrence as a “rare complication” of AWR. All the authors, however, admit previously participating in Herculean closure attempts during their training without appreciating the full implications. Historically, some surgeons understood well before Kron’s (12) seminal reintroduction of the ACS concept in 1984; the general principle that reducing excessive visceral contents back within the abdominal compartment could yield catastrophic physiologic results (48). Although there are only scattered case reports (35), and brief mention of mortality/morbidity from IAH/ACS within clinical series (36, 46), we strongly suspect this is more common than generally reported. In 2015, Petro et al. (43) reported one of the first prospective studies of IAP surveillance in AWR, noting a 92% incidence of IAH (Grade I or greater) when IAP was monitored. This corroborates an epidemiological study of Losken et al. (49) who measured IAP after transverse rectus abdominis myocutaneous flap breast reconstruction. They noted that post-operative IAP was increased with more extensive procedures and also increased in relation to BMI. Those with severe IAH had significantly more general and donor-site complications (60%) (49).
Pathophysiology
IAH/ACS has traditionally been conceptualized as resulting from mechanical increases in abdominal contents when the physical capacitance of the abdominal container is exceeded by the addition of surgical packing, visceral edema, hematoma, and/or ascites. Recognition of such “classic” obvious cases was largely responsible for bringing IAH/ACS back to the attention of the trauma/critical care community at the end of the previous century. Such patients often returned to the intensive care unit (ICU) in extremis and required urgent decompression. However, it has been appreciated that the pathogenesis of the ACS may be more complex and involves all the complexities of ischemia/reperfusion and massive biomediator effects. Malbrain has theorized that IAH/ACS often develops as a result of two physiological hits. The first sequential “hit” typically occurs from sepsis or trauma-related shock causing mesenteric ischemia-reperfusion injury. Subsequently, the resultant “acute bowel injury” generates the release of pro-inflammatory mediators into the systemic circulation and especially the peritoneum. This ultimately results in neutrophil priming, increased intestinal wall permeability, extravasation of fluid into the bowel wall and mesentery, translocation of intestinal bacteria, and absorption of bacterial endotoxin. In the ensuing second hit, resultant visceral edema leads to IAH, compressing intra-abdominal lymphatics producing progressive decreases in bowel perfusion, mucosa-to-serosa intestinal necrosis, a further increase in bowel wall permeability, bacterial translocation/endotoxin absorption, and release of pro-inflammatory mediators. Even relatively mild IAH (e.g. an IAP of 15 mmHg) has been reported to decrease intestinal microcirculatory blood flow, increase bowel wall permeability, and induce irreversible gut histopathological changes, bacterial translocation, and multiorgan dysfunction syndrome. Such conceptualization may explain why AWR patients without a primary inciting cause of shock may tolerate IAH/ACS better than predicted (43).
Permissive IAH
Even in 1996, Burch et al. (9) recognized that all patients do not respond to IAP elevation uniformly. This is certainly true in AWR. The Cincinnati group recently reported that IAP was commonly elevated to the severe IAH/ACS range after AWR using a transversus abdominis release (TAR) technique, but spontaneously decreased with 24 h of intensive care management to baseline levels (43). The average IAP was 18 mmHg, and although robust details were not presented in the manuscript to understand the IAH management if any, the approach does seem congruent with the consensus guidelines of non-surgically managing mild IAH until it resolves (3, 50, 51). Others have reported that moderate IAH after AWR typically follows a spontaneous decrescendo, with spontaneous resolution of mild-moderate IAH being the norm (2). Agnew et al. (36) also reported on a series of large AWR patients in whom patients with severe Grade IV (>30 mmHg) without obvious organ dysfunction seemed to accommodate quickly and without sequelae. Finally, in Losken’s (49) experience harvesting rectus abdominis–based flaps, while they noted compatible respiratory and renal symptoms consistent with formal ACS, all resolved typically by day 2.
This seeming tolerance may relate to the fact that patients undergoing elective AWR are fundamentally different from those suffering from major trauma and inflammation (43). Thus, unless there are dramatic intra-operative complications, the elective AWR patient, even with “severe IAH,” does not enter a vicious cycle of acute bowel injury with ongoing futile fluid resuscitation. Thus, it is possible, if not probable, that their abdominal walls actually stretch further post-operatively, with increases in the IAV, and an increasing compliance overall (43). This situation would differ from other critically ill patients who continue to decrease their abdominal compliance as ongoing resuscitation generates edema and abdominal wall swelling propagating IAH.
Nomenclature: Why do we need a Quaternary Designation of IAH/ACS?
Following this construct, patients subjected to IAH/ACS after AWR are not the same population as those developing recurrent IAH/ACS after critical injury/illness. The original WSACS Consensus definitions perceived it useful to classify disorders of IAP as either primary, secondary, or recurrent according to the duration and cause of the patient’s IAH (4). The recurrent (or tertiary) syndromes are defined as being cases in which IAH/ACS redevelops following previous treatment of any cases of primary or secondary IAH/ACS (3, 4, 52). Such cases have been typically associated with a severe secondary hit in which already critically ill patients suffer markedly worse outcomes including multi-system organ dysfunction, prolonged critical care requirements, and death (52, 53). There are however, fundamental differences in the clinical settings, in which patients with recurrent or tertiary IAH/ACS are pro-inflammatory with dysregulated endothelial function and greatly altered vascular permeability. Although not well characterized, it can be assumed that most elective AWR patients will not have these risk factors unless they suffer major intra-operative complications. Thus, in an effort to better characterize disorders of IAP, and to facilitate ongoing research and communication, we suggest the distinct setting of IAH after major AWR be designated Quaternary IAH or Quaternary ACS if accompanied by the presence of new organ failure.
Strategies to Avoid Quaternary IAH/ACS after AWR
The ultimate goal of any AWR is to restore a durable, functional abdominal wall without inducing pathologic IAP increases within. A multidisciplinary approach to individualized management is desirable, involving at least general and plastic surgeons, critical care intensivists, and rehabilitation medicine specialists among others as appropriate. To reduce an increased volume into a semi-rigid container without increasing the pressure within will require modifications to the container that may involve increasing capacitance, reducing volume of the contents, or physically enlarging the container. Conceptually, possible options (other than simply avoiding surgery) can be summarized as tolerating “permissive IAH”, removing contents, relaxing the container, temporarily or semi-permanently; or actually enlarging the container.
Not all hernias can safely be repaired even in 2016 (43). Even with the many current developments, there are many cases in which it is simply not possible to reintroduce the viscera as there has been massive depletion and essential complete loss of the musculofascial tissue of the abdominal wall, often coexisting with cardiorespiratory morbidities. In such cases, a non-reductive repair with bridging mesh (either prosthetic or biologic) may be performed as a temporizing solution. We do not consider these as true AWR and will not discuss them further. Otherwise, approaches to mitigating potential quaternary IAH/ACS after AWR are summarized in Table 4.
Options for avoiding intra-abdominal hypertension/abdominal compartment syndrome after massive abdominal wall reconstruction.
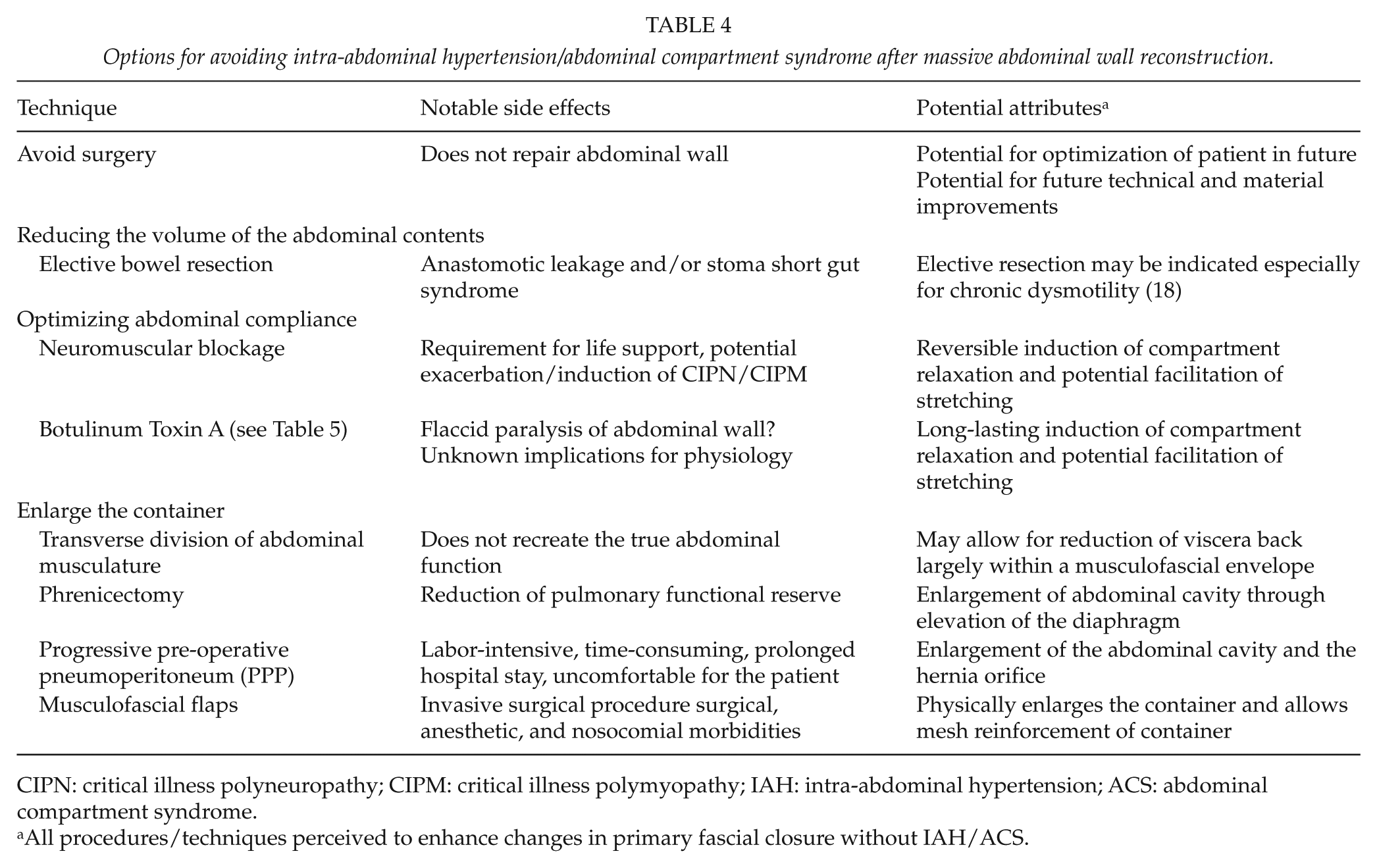
CIPN: critical illness polyneuropathy; CIPM: critical illness polymyopathy; IAH: intra-abdominal hypertension; ACS: abdominal compartment syndrome.
All procedures/techniques perceived to enhance changes in primary fascial closure without IAH/ACS.
Reducing the Volume of Abdominal Contents
Buck et al. (18) noted that patients with massive hernia and LOD often suffer from chronic bowel dysmotility. They felt that in select cases, small bowel resection should be combined with AWR, but cautioned that such should only be conducted in a University/Multidisciplinary setting preferably including a short gut specialist (18). Others have also reported the need to perform various manner of bowel resection to reduce the AV to allow closure (46, 54). The risk/benefits of this undertaking certainly require further study before it can be fully recommended however.
Improving or Optimizing the Abdominal Container Volume
Abdominal Compliance
Pharmacologic paralysis has been frequently been used to temporarily ameliorate IAH, by increasing compliance. Brief utilization of neuromuscular blockade may be considered in selected patient with mild/moderate IAH, while other interventions are used to reduce the IAP. Neuromuscular blockade (NMB) typically decreased Grade II to Grade I IAH temporarily (55). A retrospective non-randomized review found that open abdomen patients administered NMB in the first 24 h after Damage Control Laparotomy had faster and more frequent primary fascial closure by day 7 (56). Thus, the WSACS suggested (Class C Evidence) that brief trials of NMB were a potential temporizing measure in managing IAH (3). Whether NMB facilitates “stretching” and improved IAH tolerance remain pertinent research questions.
Prolonged Pharmacologic Increases in Compliance: “Chemical” Component Parts Separation
Botulinum toxin A (BTA) induces a 4- to 6-month reversible flaccid paralysis. Animal studies have demonstrated potential in AWR (Table 5). Cakmak et al. (57) compared the intra-peritoneal volumes of saline that could be contained at standardized IAP in two groups of rats randomized to intramuscular BTA versus none, finding that contained volumes significantly increased by approximately 20% with BTA. BTA effectively paralyzed the abdominal muscles and increased the abdominal wall compliance. Human experience has followed (Table 5) (58–61). Ibarra-Hurtado injected 500 U of BTA into the oblique muscles in chronic ventral hernias. One month after they measured the abdominal walls to have lengthened up to a mean of 2.59 cm and thinned a mean of 1 cm, subjectively facilitating primary closure at reoperation. Zielinski et al. (59) described BTA injection early in the course of open abdomen (OA) patients, of whom half were injected within 24 h of the initial operation. A same-stay fascial closure rate of 83% resulted, and they suggested that early BTA might avoid the extensive dissection required for formal component parts separation (CPS), but yield equivalent benefit (59).
Described botulinum A injection techniques to aid with abdominal wall reconstruction.
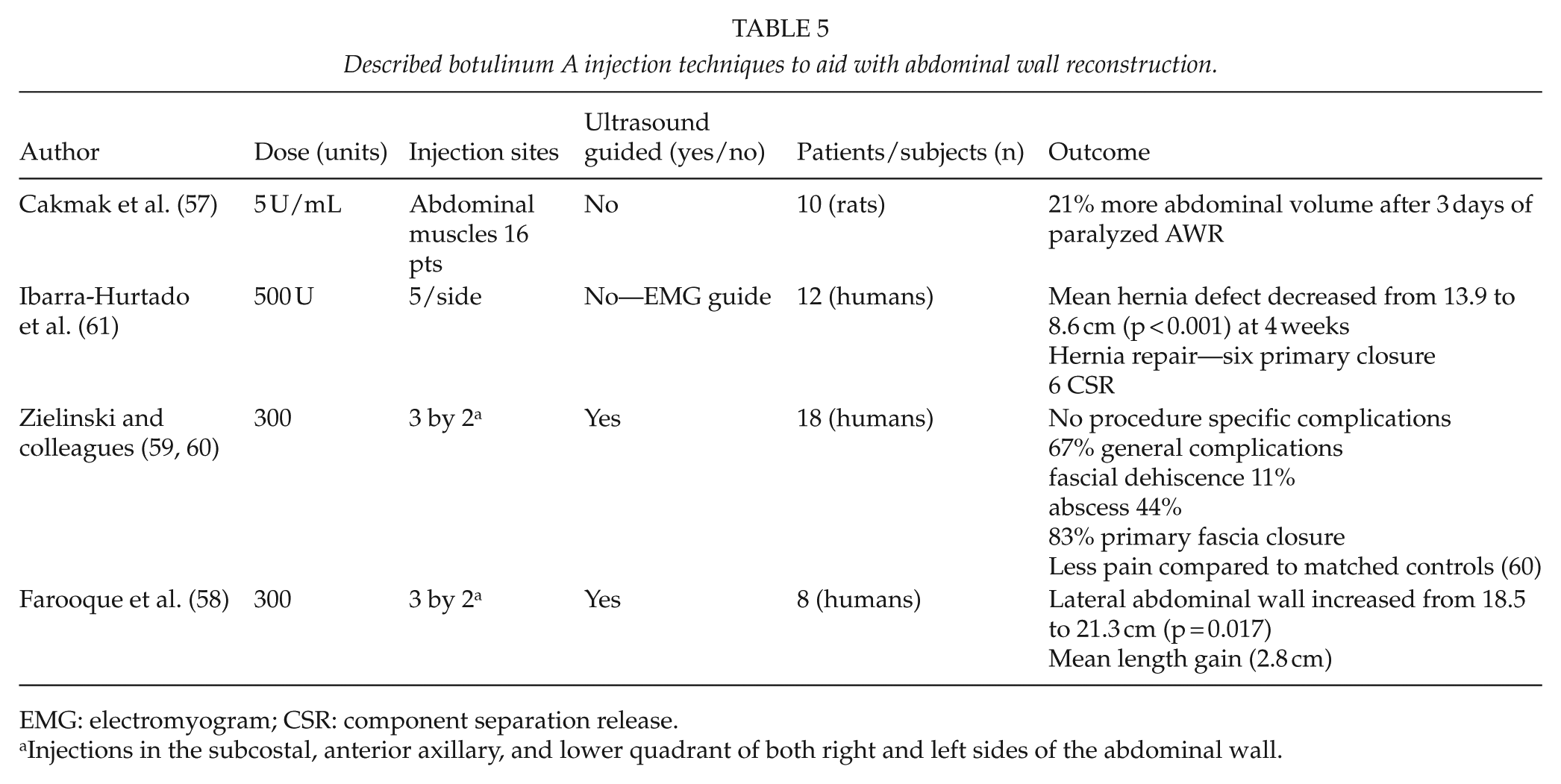
EMG: electromyogram; CSR: component separation release.
Injections in the subcostal, anterior axillary, and lower quadrant of both right and left sides of the abdominal wall.
Enlarging the Abdominal Container
In terms of simplicity, the container may be enlarged without necessarily preserving optimal function. Historical reports of neonatal gastroschisis report purposefully dividing the abdominal musculature transversely to reverse the lateralizing forces, with subsequent reconstruction (62). Strategies, such as phrenicectomy or transverse non-functional incisions, have also been proposed to enlarge the container (48, 63). Such techniques now have little role in adult AWR but are mentioned for completeness.
Pre-Operative Progressive Pneumoperitoneum
Another technique with both historical and modern usage is pre-operative progressive pneumoperitoneum (PPP) (41, 42, 48, 64, 65). Numerous reports exist, with pre-operative preparation varying from 4 to 60 days, all generally reporting favorable clinical outcomes in uncontrolled situations. Typical initial inflation volumes of air are between l000 and 4000 mL, followed by daily injections of 500–2000 mL, with the exact volume determined by patient tolerance to pain, nausea, and especially respiratory distress (41). Patients who cannot tolerate the stepwise inflation are assumed to be poor surgical candidates. Therefore, PPP may be diagnostic and therapeutic (63). While some measure IAP, others felt pressure monitoring was unnecessary and persisted with insufflations until the “flanks bulged” (48). Although long claimed that the physical gain to the “abdominal container” is due to muscular stretching (63), this was only first studied by Dumont et al. (54) in 2009. CT scanning before and after PPP revealed that although the width of the incisional defect significantly increased so did the total width of the abdominal wall musculature, resulting from anterolateral muscle stretching, rather than the recti. It is also hypothesized that diaphragmatic function may be improved through pneumoperitoneum. With chronic LOD in a ventral hernia, the diaphragm is weak and relaxed, leading to a deterioration of respiratory function. Pneumoperitoneum thus leads to an increase in IAP, thereby elevating the diaphragm with restitution of its physiological tension and function (63).
Musculofascial Flaps
The more contemporary approach compatible with contemporary health care systems is to increase the size of the peritoneal cavity through physically enlarging the abdominal container. This is accomplished through musculofascial flap techniques such as CPS and/or the TAR (36, 43, 66). Agnew et al. (36) studied CPS in 19 patients with massive AWR, with average BMI of 30, average hernia volume 915 mL, and 47% having previous repair attempts. Each was studied with both volumetric CT and pulmonary function testing before and after CS (36). The mean intra-abdominal volumes increased from a pre-operative value of 7640 to 8166 mL post-operatively (a 6% increase; p = 0.01) (36).
Occult ACS and AWR
Earlier discussion concerned the possibility that IAH/ACS in AWR might be of less clinical concern and that abnormal physiology might be more “tolerable.” While intriguing this concept needs a balanced critique. One of the most difficult conceptual issues in the field of IAP studies concerns the clinical importance of moderate levels of IAH in otherwise well or stable appearing critically ill patients (19, 28). It has been shown in animal studies that modest and even only “slightly elevated” IAH can have profound effects on multiple organ systems notably the gut with subsequent deranged perfusion, gut-barrier function, biomediator activation, and systemic inflammation (26, 27, 67, 68).
Mohan et al. (69) recently reported an elegant porcine model of quaternary ACS involving the induction of overt ACS through a simulated AWR and then rescued the animals from the resultant ACS through performing a CPS procedure. In this model, the IAPs increased a mean of 14.9 mmHg with AWR, but then decreased 9.1 mmHg with the rescue CS. In this model, the large bowel could be rescued with early CPS rescue when ACS occurred after AWR. However, the small bowel was much more fragile, in that the small bowel necrosed whenever the IAP was 20 mmHg even with CPS rescue. Thus, whether tolerating permissive IAH is really in a patient’s best interest still remains a practice with unknown risks at this time. There remains further concern that while quaternary IAH/ACS associated with muscular reconstruction/relaxation might be tolerable, tertiary IAH/ACS associated with post-operative pneumonia or an enteric leak might be initially indistinguishable. Yet, such “non-permissible” IAH/ACS might have an exponentially adverse course if not detected early.
Intra-Operative Monitoring: Continuous Intra-Abdominal and Ventilatory Pressure Monitoring
Accepting self-limiting IAH in a closely monitored situation is very different from willful ignorance. Therefore, the first step in avoiding “intolerable increases” in intra-operative IAP is to simply measure intra-operative IAP, especially during fascial closure. This may be done through a number of means, but mostly simply through the measurement of bladder pressure intra-operatively and especially during closure. The placement of a triple-lumen bladder catheter pre-operatively greatly simplifies measurement of IAP (70, 71), which should be correlated with any changes in ventilatory pressures during surgery (69). The anesthesia team should also closely follow the urine output and the dynamic and static ventilatory pressures (35), which some authors feel is the best reflection of Abdominal Compartment pressure tolerance (2, 43), although further study is required.
Conclusion
Abdominal wall failure in the form of massive ventral hernia can assume great dimensions with loss of both function and domain of the viscera. To correct this, AWR is an often dramatic endeavor that may greatly improve a patient’s quality of life, but potentially involves creating severe IAH, that may initiate multiple organ dysfunction syndrome and generate destructive storms of inflammatory biomediators. All involved, especially surgeons, need to fully appreciate the complete anatomy and physiology of the abdominal compartment to undertake such surgery such that patients are neither unnecessarily denied surgical correction nor placed at risk of an iatrogenic compartment syndrome. The specific term, Quaternary, is applied to designate these reconstructions as potentially unique situations that deserve to be fully studied to understand the fundamental physiology and clinical care requirements.
Footnotes
Declaration of Conflicting Interests
A.W.K. reports having consulted for the Canadian Space Agency, Canadian Forces Medical Services, Innovative Trauma Care, Acelity, and Cook Medical Corporations on matters not directly related to this publication. D.N. reports having advisorship and stock in Orpyx, an unrelated Biotechnology Company. M.J.R. reports having received research grants from Miromatrix, W.L. Gore, consulted for Bard, and serves on the Board of Ariste Medical on matters not directly related to this publication. D.J.R. reports no disclosures. P.B.M. reports no disclosures. C.C.P. reports having received a research grant from Miromatrix Medical Inc. F.B. reports consultancies with the Acelity, Ethicon, and Bard/Davol Corporations and having received payment for lectures from the Medtronic and Ethicon corporations. M.S. reports an Educational Consultancy and having by compensated for lecturing by the Smith and Nephew Incorporated. J.X. and C.G.B. report no disclosures.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
