Abstract
Objectives:
To evaluate the current incidence, risk factors, management, and long-term follow-up of urinary leakage following partial nephrectomy, in order to propose an algorithm for diagnosis and evaluation of postoperative urinary leakage.
Materials and Methods:
The study included 752 patients who underwent elective partial nephrectomies for renal masses between the years 1988 and 2013. Patients’ demographics, clinico-pathologic variables, and operative details were collected retrospectively. The associations between urinary leakage and patients’ variables were assessed by univariate and multivariate analyses.
Results:
Of the 752 patients, 21 (2.8%) experienced urinary leakage; 4 of the 21 patients with urinary leakage had spontaneous resolution, 1 patient underwent nephrectomy, and 16 patients were treated by retrograde ureteral stents insertion. One of them necessitated insertion of an additional percutaneous nephrostomy and another one deserved concomitant percutaneous drainage of a perirenal urinoma. The average period of time that elapsed from the operation until the insertion of stent was 8.5 ± 4.5 days. Stents were removed 68 ± 20.5 days postoperatively. None of the patients had either persistent or repeated leakage. On univariate analysis, hilar renal masses (p < 0.04) and higher preoperative creatinine levels (p < 0.01) were found to be associated with higher rates of urinary leakage. None of these variables was significant on a multivariate analysis. Review of the urinary leakage rate over time revealed it has been constantly decreasing over time, from 4% in early cases to 1.3% among the most recent ones.
Conclusion:
None of the preoperative variables that were examined in this study was significantly associated with increased risk of urinary leakage. However, cumulative surgical experience was associated with lower rates of urinary leakage, suggesting that the decrease in its incidence is related to the improved surgical skills, rather than to differences in tumors’ or patients’ characteristics.
Introduction
In recent years, nephron-sparing surgery (NSS), namely partial nephrectomy (PN), has become the standard of care in the management of clinical stage T1a and selected clinical stage T1b renal tumors (1–3). However, PN remains a technically challenging procedure, associated with increased risk of perioperative complications, including bleeding and urinary leakage (UL) (4). UL is considered as one of the most common perioperative complications of PN, with an estimated incidence ranging between 0% and 33% (3, 5–10). Conservative measures such as catheter drainage, ureteral stenting, or percutaneous nephrostomy (PCN) placement, generally resolve the UL. However, in cases of persistent ureteral obstruction and failed ureteral catheterization, reoperation may be necessary to relieve obstruction, resulting rarely in renal loss (5, 6). Several studies have identified some risk factors which might lead to UL following PN including age, depth of the lesion, and renal nephrometry score (8, 11). Nevertheless, there has been a paucity of data regarding the management of such leaks. In this study, we suggest an algorithm for the diagnosis and management of UL identified following PNs for renal masses based on our own experience and review of the literature.
Materials and Methods
Patients
This is retrospective chart review of single-center multi-surgeon series. Data included all patients who underwent elective PNs for renal masses, between the years 1987 and 2013. All procedures were performed at The Chaim Sheba Medical Center, a tertiary medical referral center.
Patients’ demographics and operative details were collected retrospectively. Clinico-pathological variables recorded included age, gender, body mass index (BMI), co-morbidities, type of operation (open or laparoscopic), year of surgery, anatomical site (right or left kidney), maximal tumor size, presence of bilateral disease or ipsilateral multifocal disease, tumor consistence (solid versus cystic), tumor location, and final pathology according to the histological report.
Urinary Leakage
UL was defined as postoperative, biochemically confirmed urine drainage persisting for at least 3 days (drainage of fluid (>50 cc/day)) with a drainage fluid to serum creatinine ratio of greater than 2) (7). In the current series, clinically significant urine leakage was defined as extra-renal urine extravasation that required prolonged maintenance of a retroperitoneal drain, re-insertion of a retroperitoneal drain, insertion of a ureteral stent, or other surgical intervention.
Follow-Up
The patients underwent outpatient follow-up conducted by the urological team, at 3- to 6-month intervals following the procedure. Follow-up included history and physical examination at each visit. Chest X-ray, abdominal computed tomography (CT), and ultrasound were performed to each patient 6 months post-surgery, and at 6–12 months intervals thereafter.
Statistical Analysis
Statistical analysis was performed using Statistical Package for Social Sciences (SPSS, Version 22.0, Chicago, IL, USA). Student’s t-test was used for analysis of continuous variables and the chi-square test was used for analysis of categorical variables. The Cox proportional hazards regression model was used for multivariate analysis models. A p value of <0.05 was considered statistically significant. Results are presented as mean ± standard deviation (SD) unless otherwise specified.
Results
Overall, 752 patients underwent PNs during the study period. Clinico-pathological demographics of these patients are provided in Table 1. Median age at surgery was 60.8 ± 12.9 years. Mean postoperative follow-up period was 38 ± 4 and 22 ± 3 months, for the open and the laparoscopic PN, respectively. The rate of UL for all PNs was 2.8% (21 of 752) including 2.7% (10 of 363) after laparoscopic partial nephrectomy (LPN) and 2.8% (11 of 389) after open partial nephrectomy (OPN). UL was identified by persistent drainage of fluid (>50 cc/day) in 19 patients and by CT in 2 patients (CT was performed for investigation of either fever of unknown origin or of prolonged ileus).
Clinico-pathologic demographics of 752 patients included in the study.
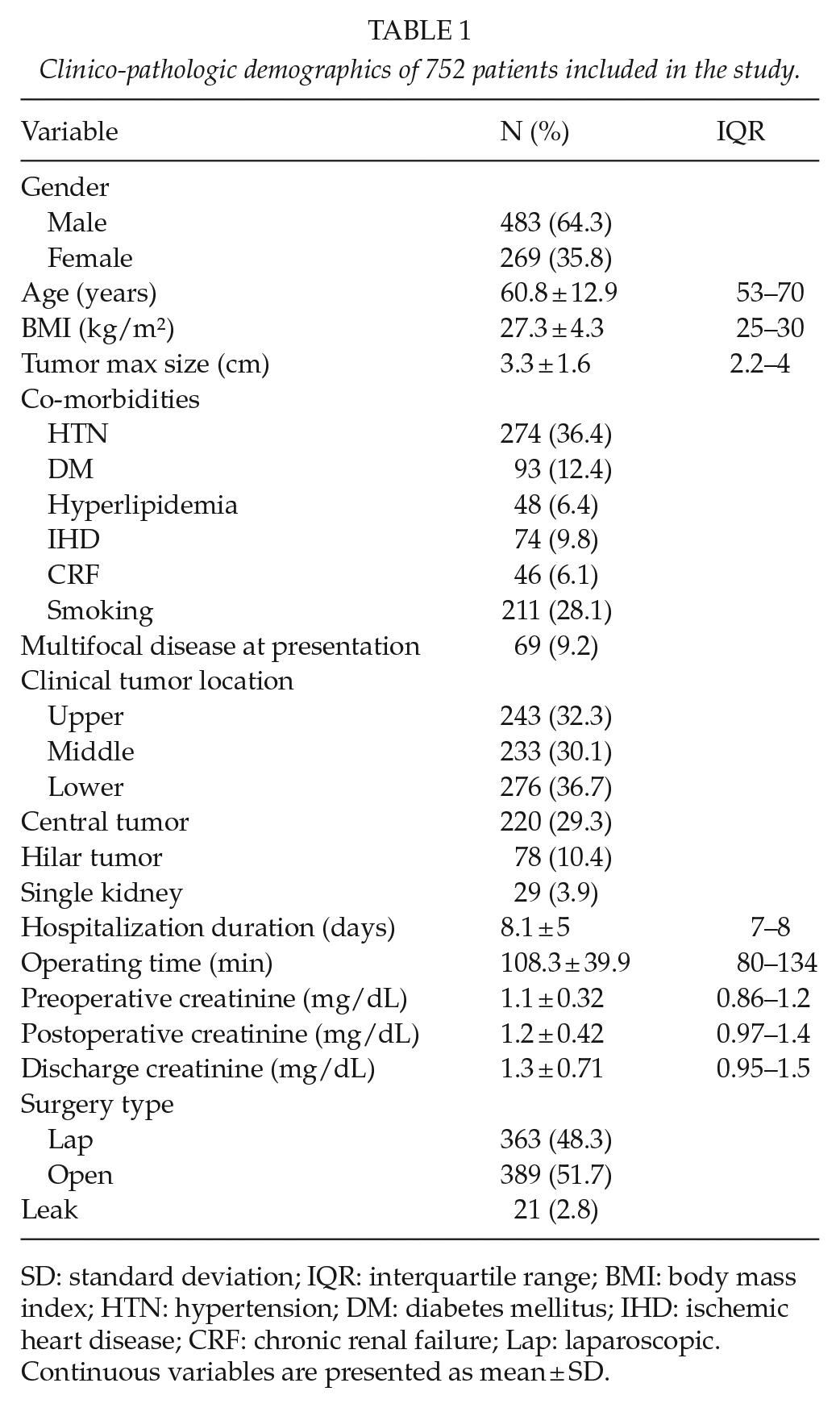
SD: standard deviation; IQR: interquartile range; BMI: body mass index; HTN: hypertension; DM: diabetes mellitus; IHD: ischemic heart disease; CRF: chronic renal failure; Lap: laparoscopic.
Continuous variables are presented as mean ± SD.
On univariate analysis (Table 2), patients with UL had significantly higher rate of hilar tumors (23.8 versus 10%, p < 0.05) and higher preoperative serum creatinine levels (1.2 versus 1.06 mg/dL, p < 0.01). Overall, no significant differences were observed in median age, BMI, co-morbidities, tumor location, or type of operation (lap versus open). Likewise, central lesions had similar rates of UL compared to peripheral lesions. None of these variables was significant on multivariate analysis. Additionally, the cumulative surgical experience was assessed as an another affecting factor; patients were stratified into five groups according to when they were operated along the study period; group 1: first 200 cases, group 2: 200–400, group 3: 400–600, group 4: 600 cases, or more. As demonstrated in Fig. 1, the rate of UL has been constantly decreasing, from 4% in the first 200 cases to 1.3% in the most recent 200.
Univariate analysis.
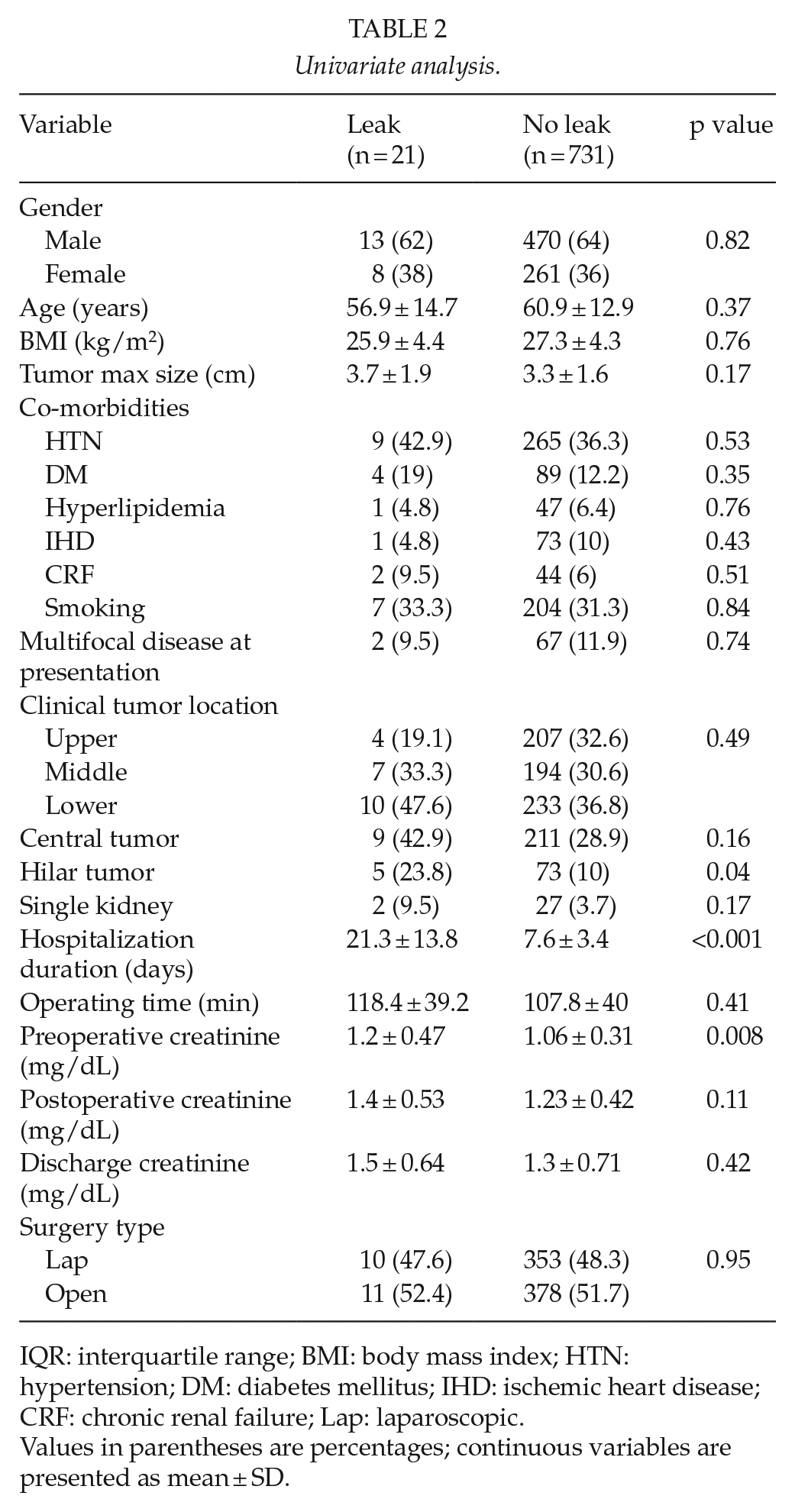
IQR: interquartile range; BMI: body mass index; HTN: hypertension; DM: diabetes mellitus; IHD: ischemic heart disease; CRF: chronic renal failure; Lap: laparoscopic.
Values in parentheses are percentages; continuous variables are presented as mean ± SD.
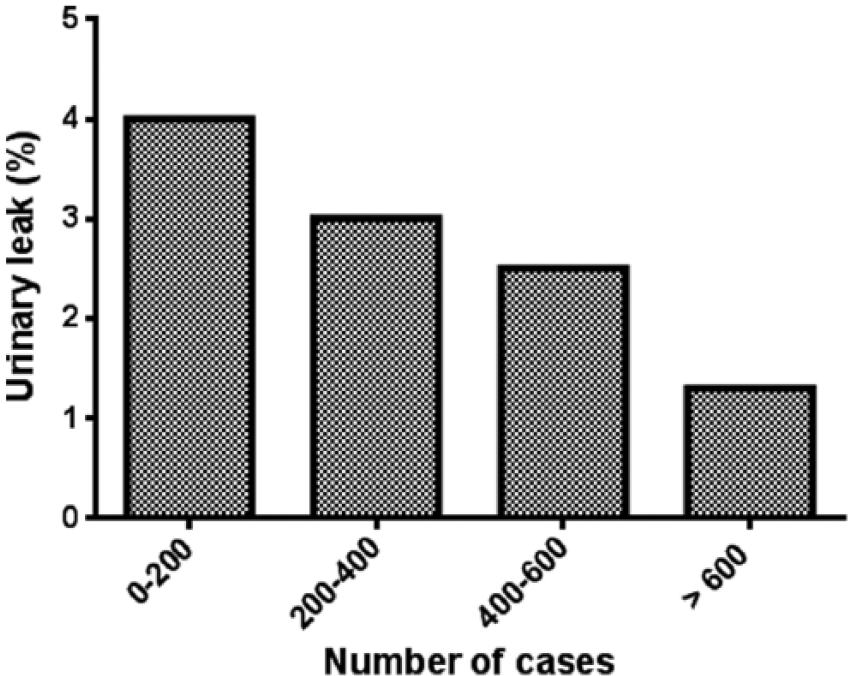
Association between surgical experience and the risk for UL.
Further analysis of the UL subgroup revealed that of the 21 patients who experienced UL, 4 patients had spontaneous resolution, 1 patient underwent nephrectomy, and 16 patients were treated by retrograde ureteral stents insertion. One of them necessitated insertion of an additional PCN and another one deserved concomitant percutaneous drainage of a perirenal urinoma. The exact location of the UL was demonstrated in nine patients by retrograde pyelography performed at the time of the insertion of the stent, originating from the renal pelvis/calyces in seven patients and from the upper ureter in one patient.
The average period of time that elapsed from the operation until the insertion of stent was 8.5 ± 4.5 days. Stents were removed 68 ± 20.5 days postoperatively. None of the patients has suffered from either persistent or repeated leakage.
Four patients with UL have eventually required renal exploration and surgical repair of the leakage. The average period of time that elapsed from the primary operation until renal exploration was 26 ± 13 days.
At surgery, leakage from open calyces was identified in two patients and an upper ureteral injury in one patient. Correction was successfully performed in these three patients. In one case, there was severe renal edema without identification of renal pelvis and the patient underwent nephrectomy.
Discussion
UL is a major postoperative complication of PN, with a reported incidence of 0%–33% (1, 4–6, 8–15) (Table 3). In this study, we evaluated the incidence; risk factors and management of UL encountered following PNs performed in a single tertiary medical center. In our experience, the overall incidence of UL was 2.8%, a rate that is comparable with the existing data (8, 12). However, as opposed to the findings of several previous studies (11, 16–19), we found that neither tumor size, nor central location of tumors are associated with a higher risk of post-surgery UL.
Incidence of post-nephron-sparing surgery urinary leakage.
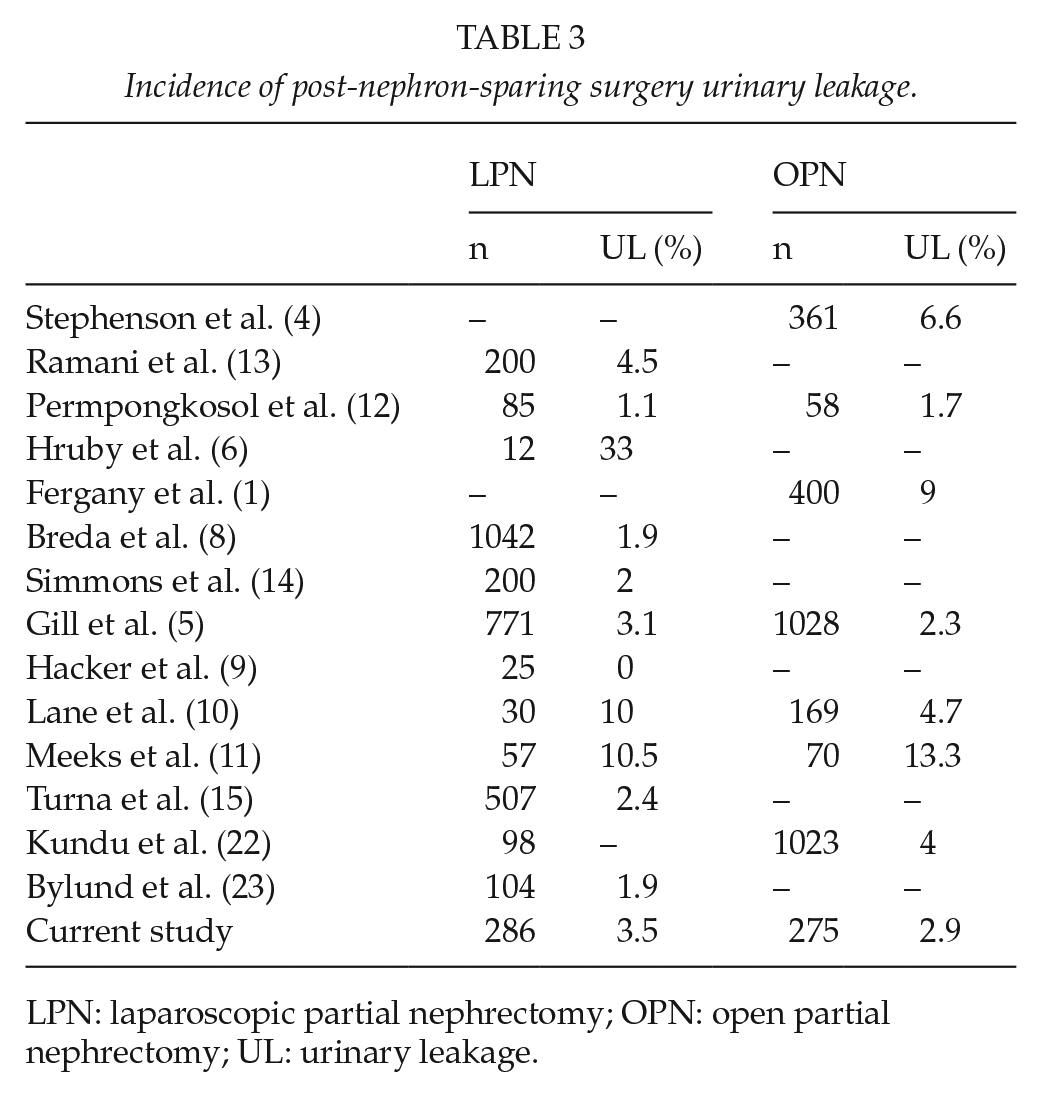
LPN: laparoscopic partial nephrectomy; OPN: open partial nephrectomy; UL: urinary leakage.
The management of post-PN UL is poorly addressed in the literature. Surprisingly, it is hardly mentioned even in the “EAU Guidelines on Iatrogenic Trauma” published on 2012 (20). Correspondingly, the chapters dealing with open and laparoscopic renal surgery briefly refer to the conservative approach as “unless there is distal obstruction to the site of leakage, most will resolve with conservative therapy after several weeks” and also recommend that “if conservative management fails, such as in cases of distal obstruction, additional intervention may be required, such as percutaneous drainage of a urinoma or ureteral stent and bladder decompression” (21). In our experience, only 4 of the 21 patients who had UL experienced spontaneous resolution and required no further intervention, 1 patient underwent nephrectomy and the remaining 16 patients were treated by retrograde insertion of ureteral stents, with 1 patient necessitating additional PCN and another patient deserving concomitant percutaneous drainage of perirenal urinoma. There is a paucity of data in the literature concerning the management of such UL and details regarding the average time interval elapsing until retrograde insertion of ureteral stents and the timing of their removal. In our series, the average relevant periods were 8.5 ± 4.5 days from surgery to stent insertion and 68 ± 20.5 days of drainage. In comparison, Meeks et al. (11) found that the average period from surgery to retrograde insertion of a ureteral stent was 25 days and the average duration until its removal was 53 days. Ramani et al. (13) and Simmons and Gill (14) reported average periods of drainage by retrograde ureteral stents of 45 and 105 days, respectively. In our series, the exact location of the UL was demonstrated in nine patients by retrograde pyelography performed at the time of the insertion of the stent, originating from the renal pelvis calyces in seven patients and from the upper ureter in two patients. In addition, four patients from the current series, with post-PN UL, have eventually required renal exploration (0.53% of the total cohort and 19% of the patients who demonstrated urinary leak), whereas one patient ended-up with nephrectomy. Review of the literature reveals that Gill et al. described 2 cases out of 24 patients with post-laparoscopic PN UL who underwent nephrectomies (5), and Hruby et al. reported similar outcome in 1 of 12 patients with such leakage. In this context, we believe that increased awareness regarding this possible complication and early intervention, namely drainage, may contribute to improved outcome.
Our study is not without limitations. First, because of its retrospective character, and the fact that operations were performed by four different surgeons, over three decades, we cannot exclude that different surgical techniques were used, and the leak may be attributable to surgical strategy rather than biology of the primary tumor. Second, the relatively small number of UL cases. We acknowledge the fact that 21 patients is a small number; however, UL post-PN is an uncommon complication, especially in recent years, probably due to increasing experience. Moreover, giving the consequence related to UL following PN, analysis of this group, despite its small number may help us learn about this complication and mainly build an evidence-based algorithm for better evaluation and management.
While there is an abundant literature addressing UL following PN, the scarcity of case series coping with the management of such complication has left the clinician with little data to develop an evidence-based algorithm for their evaluation and management.
Herein, we propose an algorithm based on our experience and review of the literature (Fig. 2). We believe that this model may help surgeons performing PN, who might be facing this uncommon situation of postoperative UL.
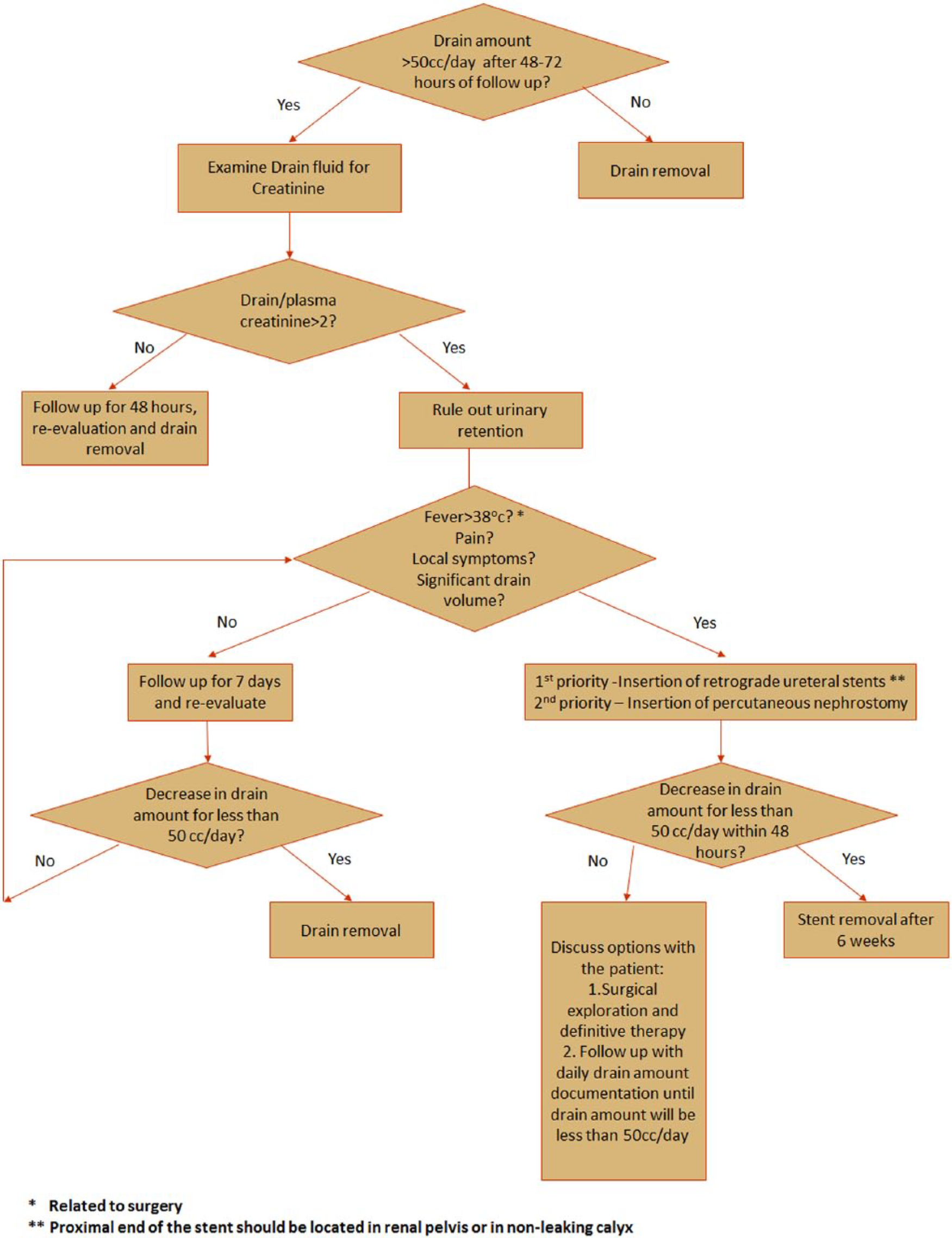
Proposed Flow chart for diagnosis and evaluation of post-operative urinary leakage.
As can be seen in Fig. 2, in the case of persistent drainage of fluid (>50 cc/day), one should start with examination of the ratio between drain creatinine and plasma creatinine levels. If the ratio is larger than 2 and urinary retention has been ruled out, UL is then identified. The next step is to evaluating whether the UL is clinically significant (i.e. fever > 38°c, pain, local symptoms, significant drain volume). If the UL is asymptomatic/not clinically significant, a non-active follow-up can be held until drain amount decreases to less than 50 cc/day. If fluid amount still exceeds 50 cc/day after 1 week, or in the case of clinically significant UL, insertion of retrograde ureteral stents or PCN is recommended. If the UL has not resolved within 48 h from stent/PCN insertion, further options should be examined, including surgical exploration and definitive repair or follow-up with daily drain amount documentation until drain amount will be less than 50 cc/day.
Conclusion
The incidence of post-PN UL in our institute is 2.8%, and is in accordance with the lower range figures previously reported in the literature. Patients treated with retrograde insertion of ureteral stent experienced shorter recovery periods, namely the time passed from the leakage diagnosis to its resolution. The single parameter which correlated with reduced UL is the cumulative experience, suggesting that the decrease in its incidence is related to improvement in the surgical skills in performing PN, rather than to differences in tumors’ and patients’ characteristics.
Footnotes
Acknowledgements
T.E. and Y.A.-G. contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
