Abstract
Background and Aims:
There is a demand for a better understanding of the vascular structures around the right colonic area. Although right hemicolectomy with the recent concept of meticulous lymph node dissection is a standardized procedure for malignant diseases among most surgeons, variations in the actual anatomical vascular are not well understood. The aim of the present review was to present a detailed overview of the vascular variation pertinent to the surgery for right colon cancer.
Materials and Methods:
Medical literature was searched for the articles highlighting the vascular variation relevant to the right colon cancer surgery.
Results:
Recently, there have been many detailed studies on applied surgical vascular anatomy based on cadaveric dissections, as well as radiological and intraoperative examinations to overcome misconceptions concerning the arterial supply and venous drainage to the right colon. Ileocolic artery and middle colic artery are consistently present in all patients arising from the superior mesenteric artery. Even though the ileocolic artery passes posterior to the superior mesenteric vein in most of the cases, in some cases courses anterior to the superior mesenteric artery. The right colic artery is inconsistently present ranging from 63% to 10% across different studies. Ileocolic vein and middle colic vein is always present, while the right colic vein is absent in 50% of patients. The gastrocolic trunk of Henle is present in 46%–100% patients across many studies with variation in the tributaries ranging from bipodal to tetrapodal. Commonly, it is found that the right colonic veins, including the right colic vein, middle colic vein, and superior right colic vein, share the confluence forming the gastrocolic trunk of Henle in a highly variable frequency and different forms.
Conclusion:
Understanding the incidence and variations of the vascular anatomy of right side colon is of crucial importance. Failure to recognize the variation during surgery can result in troublesome bleeding especially during minimal invasive surgery.
Keywords
Introduction
The concept of total mesorectal excision (TME) becomes a gold standard treatment of rectal cancer, when associated with a lower recurrence rate and better oncological outcomes (1). Similarly, complete mesocolic excision (CME) has been recently introduced by Hohenberger et al. (2) in 2008, and it provides potential advantages of reduced local recurrence and improved long-term survival. However, there is still no standardized procedure for the treatment of colon cancer.
Over the last two decades, the application of minimally invasive surgery (MIS), such as laparoscopic and robotic techniques, has dramatically increased in the field of colon cancer because it has a number of advantages over open surgery (3). Notably, the use of laparoscopic techniques in treating colon cancer reduces the invasiveness of surgery without affecting the oncological outcome (4, 5). This approach, however, has several limitations, including the limited operative field of view, lack of tactile sensation, and loss of three-dimensional (3D) perception of the structure, resulting in the inability to manipulate tissues properly, longer operating times, and an increased risk of visceral and vascular injuries, especially in the patients with anatomical variations of the vascular structures or visceral obesity. Another burden in this approach is the extent of surgical resection in colon cancer to achieve better oncological outcomes by moving toward radical resection through CME and central vascular ligation (CVL), which includes preservation of the mesocolon plane by sharp dissection off the parietal plane and regional and central lymph nodes with high vascular ligation (6, 7). In addition, the main reason for open conversion for a beginner was known to be accidental bleeding from aberrant vascular injury during laparoscopic surgery (5). MIS requires thorough and comprehensive knowledge of mesenteric anatomy in terms of the 3D relation between the colonic arteries and superior mesenteric vein (SMV).
Since the last century, there have been several anatomical studies on variations in the arteries of the right colon; these have mainly focused on the incidence of ileocolic, right, and middle colic arteries (8, 9). In 1958, Sonneland et al. (8) reported the incidence of arterial branches of the right colon in 600 cadavers, where the ileocolic, right colic, and middle colic arteries showed frequencies of 100%, 29%, and 96%, respectively. Regarding the venous drainage system of the right colon, there were no descriptive details about the numbers of veins and their variations or their relation to the surgical trunk (SMV), which would be the most important landmark for laparoscopic right colectomy (10, 11).
Therefore, the aim of the present review was to present a detailed overview of the vascular variation pertinent to the surgery for right colon cancer.
Method
A review of the literature was conducted using PubMed and Google scholar to find articles related to the arterial and venous anatomy of the right colon based on cadaver, radiological, and intraoperative studies. We used the following keywords in our research “right colon; superior mesenteric artery (SMA); superior mesenteric vein (SMV); laparoscopic; robotic; right hemicolectomy; complete mesocolon excision (CME); and central vascular ligation (CVL).” We included articles written in English, recent articles from 1995 to 2015, prospective and retrospective studies, and peer-reviewed journals. We excluded non-English articles, case reports, and case series studies. After an online search we found 42 studies related to the anatomical vascular structure of the colon, and we included 17 studies “arterial (n = 11) and venous (n = 7)” to be the basic skeleton of our review related to the right colon. The decision to include the articles in this review was judged by two expert independent reviewers in the field of colonic surgery.
Applied Arterial Anatomy
Variations and their incidences
All studies regarding the incidence of arterial branches in the right colon were collected and categorized according to the method of the study (cadaveric, radiological, and intraoperative studies), as shown in Table 1. These showed that the ileocolic artery (ICA) was constantly present, while the middle colic artery (MCA) was present in 98%–100% of cases. In contrast, the rate of right colic artery (RCA) presence was highly variable, ranging from 10% to 63%. Regarding the variations of RCA branches, the studies showed that RCA may arise from the ICA and MCA, while the MCA was rarely found to arise from the ICA, hepatic, and splenic arteries (16–20). In addition, the branches of the MCA mostly arise from the main trunk (64%), while one or two arterial branches might originate directly from the SMA in 32% and 3.3% of cases, respectively (21). Similarly, multiple MCAs (i.e. two or three branches) could arise directly from the SMA (7.2%) (20).
Incidence of arterial branches of the right colon in the cadaveric, radiologic, and intraoperative studies.
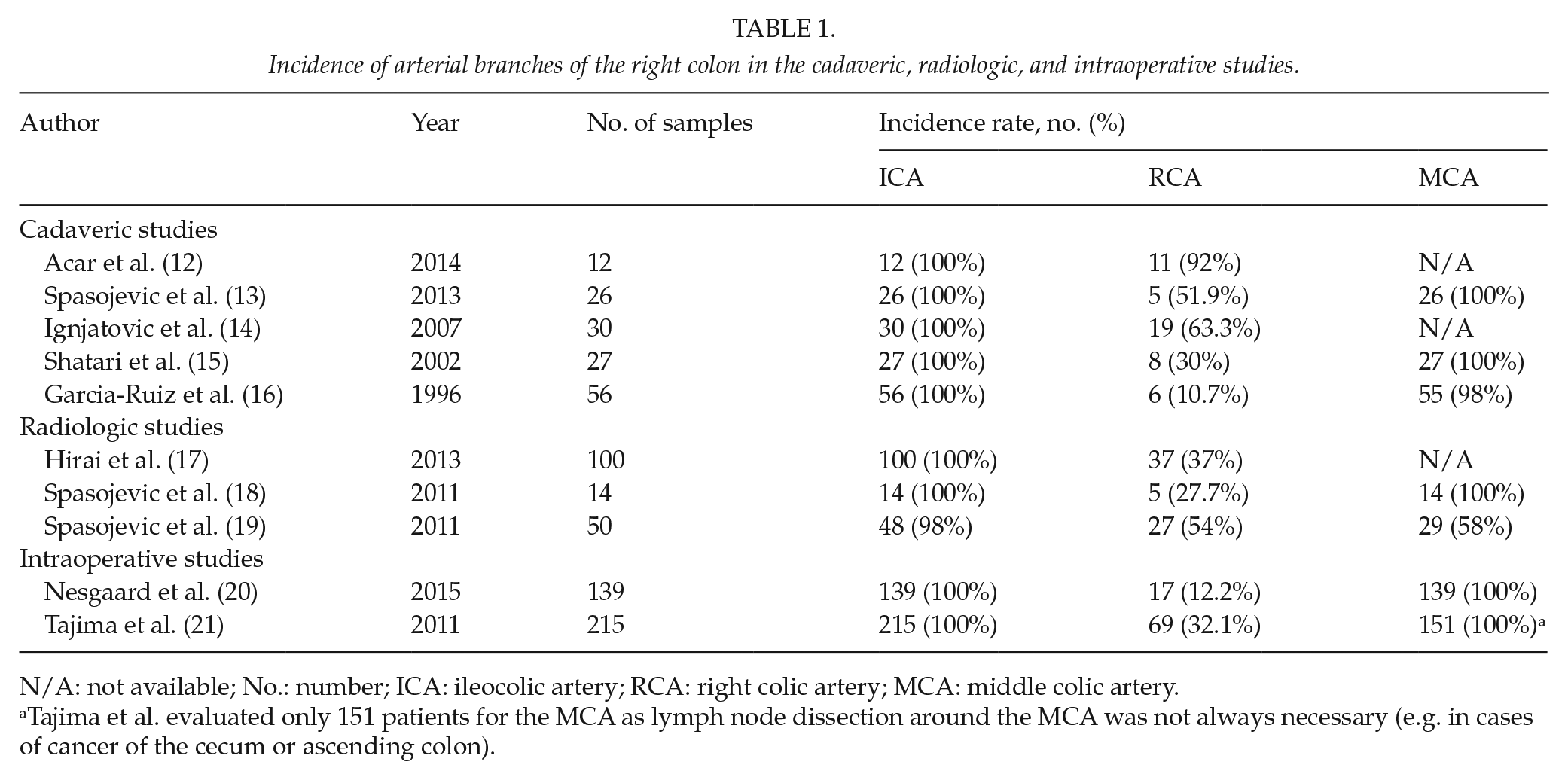
N/A: not available; No.: number; ICA: ileocolic artery; RCA: right colic artery; MCA: middle colic artery.
Tajima et al. evaluated only 151 patients for the MCA as lymph node dissection around the MCA was not always necessary (e.g. in cases of cancer of the cecum or ascending colon).
Anatomical relationship of the ICA, RCA, and SMV
One of the most important clinical relationships is the crossing pattern of the ICA and the RCA related to the SMV, as demonstrated in (Fig. 1, Table 2). The crossing pattern is defined as the ICA and the RCA crossing the SMV either posterior or anterior when they arise from the SMA. Regarding the MCA, all studies showed only anterior crossing of the SMV.
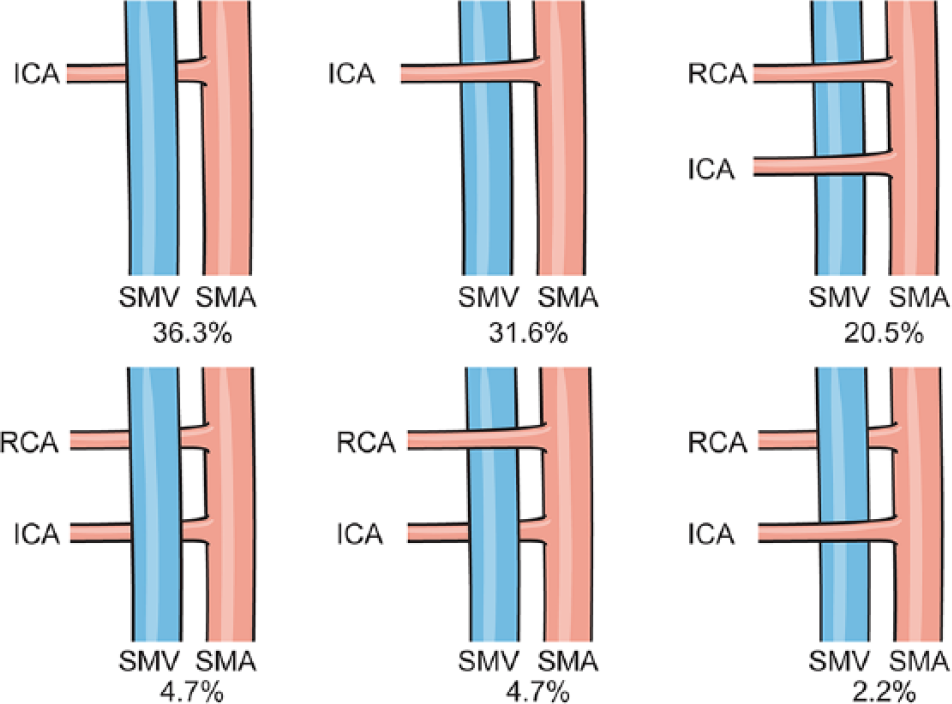
The type of the crossing pattern of the RCA and the ICA related to SMV (surgical trunk) (21).
Frequency of the crossing pattern (anterior or posterior) in cadaveric and radiologic studies of ICA and RCA related to SMV.
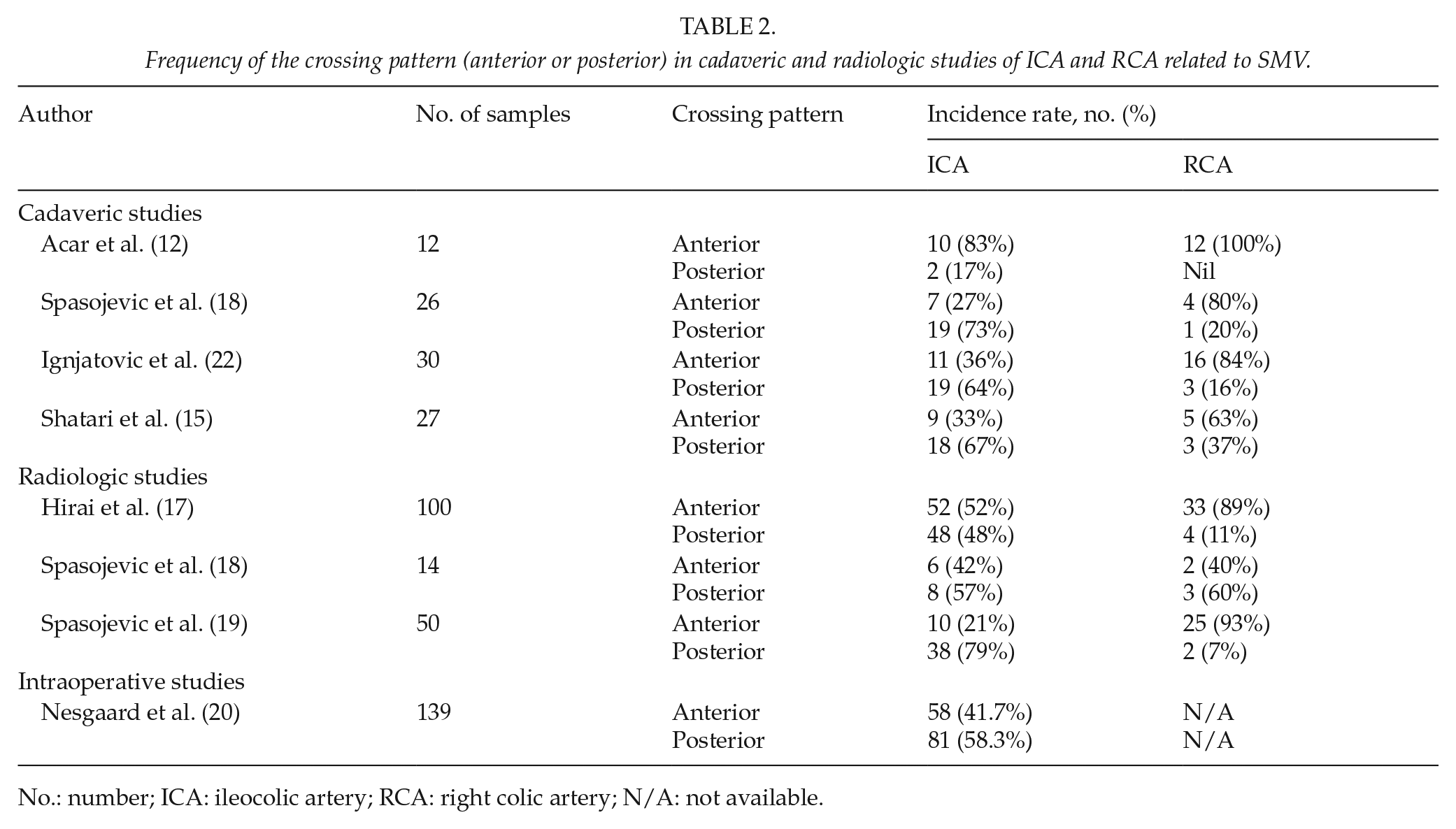
No.: number; ICA: ileocolic artery; RCA: right colic artery; N/A: not available.
The postmortem studies showed that the crossing patterns of the ICA and the RCA to the SMV were commonly anterior and posterior, respectively. Multidetector computed tomography (MDCT) angiography studies that described the crossing pattern of the right arterial colonic branches (ICA and RCA) to the SMV reported similar patterns to the postmortem and intraoperative studies.
The crossing length of the ICA and the RCA is a new concept that has clinical implications, particularly in the oncological principle. The crossing length of these arteries is defined as the distance from their origin at the SMA to the right side of the SMV. Few studies have measured the crossing length as shown in Table 3. In only one study of interest, the distance between the right colonic arteries (ICA, RCA, and MCA) was measured by MDCT. This research revealed that the mean distances between these branches were as follows: ICA to MCA, 25.2 ± 11.3 mm; ICA to RCA, 15.7 ± 4.7 mm; and RCA to MCA, 21.7 ± 11.1 mm; these findings may help the surgeons to imagine the distances between these arterial branches (20).
Estimation of the variation in crossing lengths of ICA and RCA related to SMV.
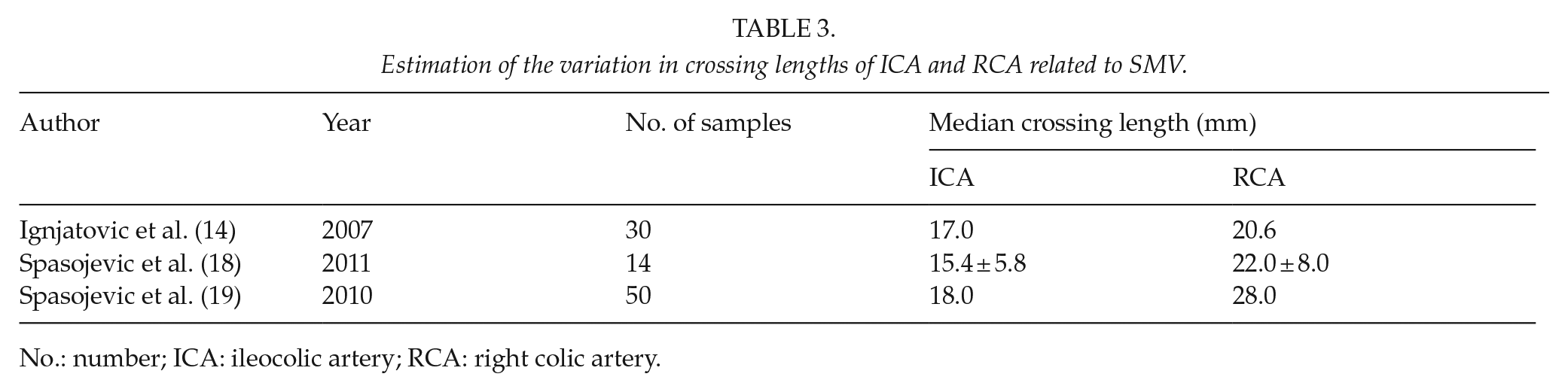
No.: number; ICA: ileocolic artery; RCA: right colic artery.
Applied Venous Anatomy
Interpretation of vein tributaries and the gastrocolic trunk of Henle
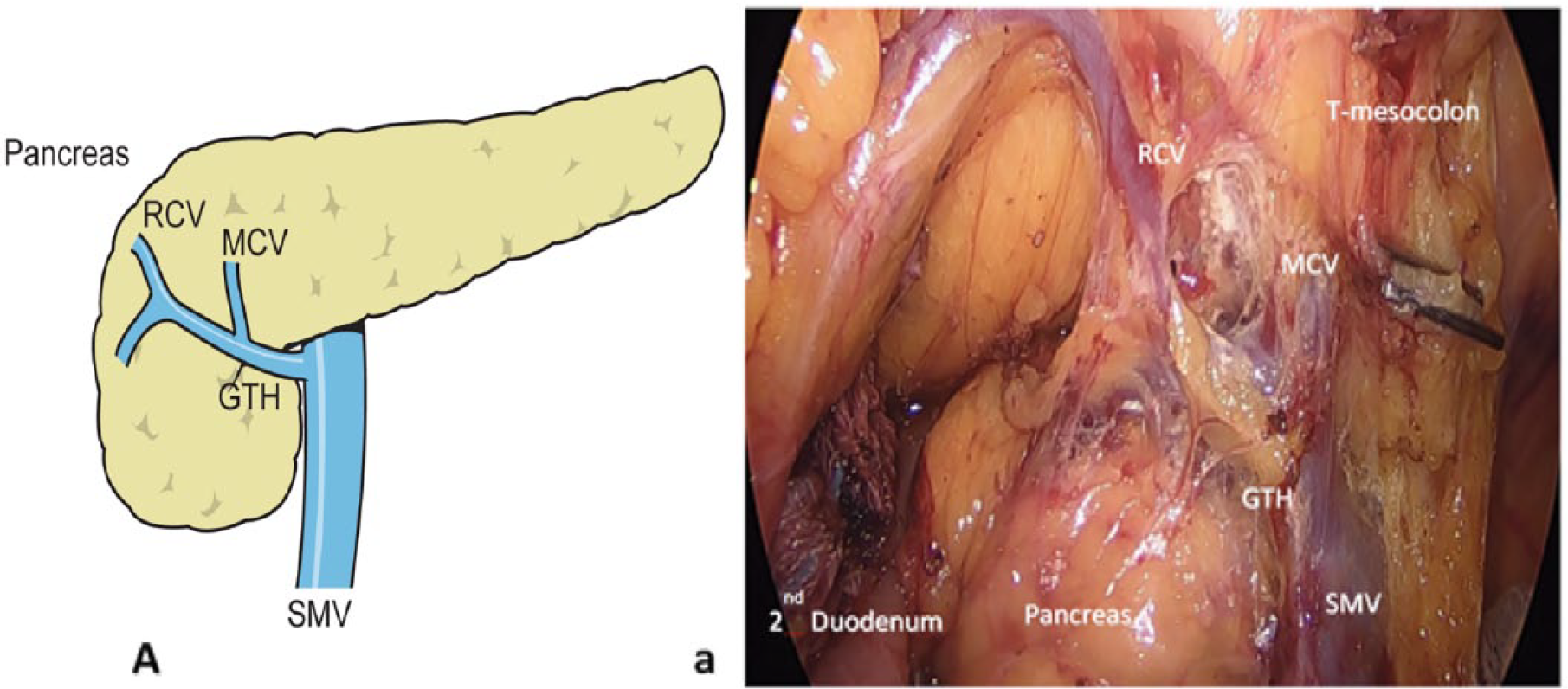
The ileocolic vein (ICV) is defined as the tributary from the ileocecal marginal veins; the right colic vein (RCV) and the middle colic vein (MCV) are defined as tributaries from the marginal veins of the ascending and transverse colon, respectively. When multiple MCVs are present, the thickest vein is defined as the main vein, while the thinner vein is called the accessory middle colic vein (aMCV) (22, 23), as shown in Fig. 2. The superior right colic vein (SRCV) is defined as the tributary from the marginal veins of the hepatic flexure. The gastrocolic trunk of Henle (GTH) was first defined by Henle in 1868 as the confluence of the superior right colic vein and the right gastroepiploic vein (RGEV), which drain into the SMV at the inferior border of the neck of the pancreas. Later, another draining vein was noted as the anterior superior pancreatic vein (11, 24) Obviously, some colic veins (MCV and RCV) sharing a confluence with the aforementioned veins to form the trunk of Henle drain into the colo-gastro-pancreatic regions, as illustrated in Fig. 3 (23, 25).
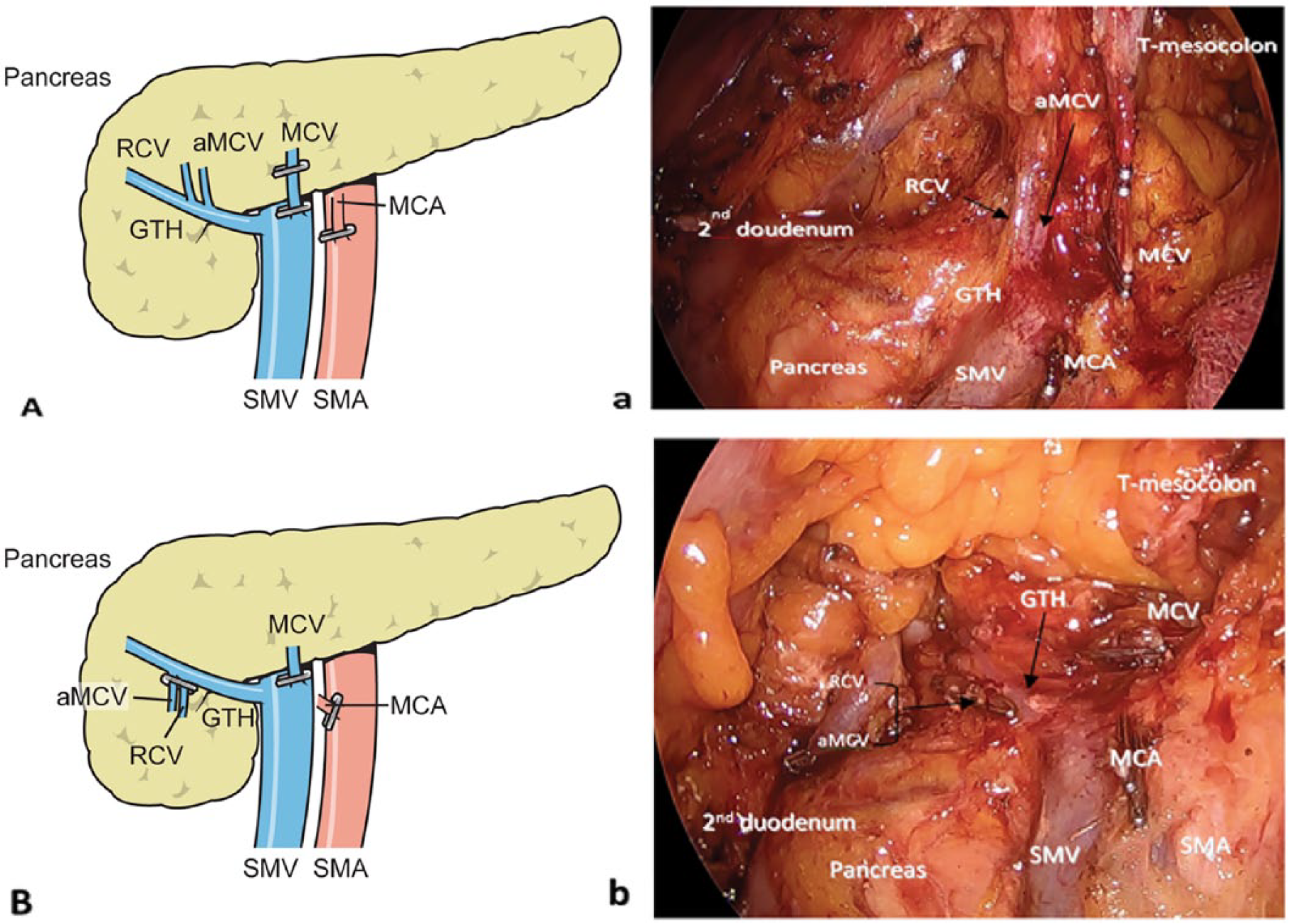
a) Operative field showing the aMCV being drained into the GTH which is thinner (as pointed by the black arrow) than the MCV that drained into the SMV. b) Operative field showing aMCV and RCV after division where draining into the GTH is clearly shown. A and B) The related operative field.
a) Operative field in laparoscopic right hemicolectomy showing a rare variation where the MCV and the RCV drained into the GTH. A) The operative field.
Incidence and variations of venous tributaries of the right colon and the GTH and their relation with SMV
ICV
All of the anatomical studies, including cadaver, intraoperative, and 3D MDCT showed that the ICV was always present and drained into the SMV, with the exception of one study showing that in 2% of cases, the ICV drained into the GTH and 98% into the SMV (25) According to this invariant finding, the ICV is considered as one of the landmark structures for laparoscopic surgery (23, 25).
RCV
The location of the RCV on the pancreatic head was observed in a cadaver study of 58 cases; it was present in 43% of cases, and 56% of the time, it drained into the SMV, and in 44% of cases, it joined the GTH (23) In another small-scale cadaver study with only nine cases, the RCV was shown to be absent in three cases, while it joined the GTH in four cases, and the SMV in three cases (26) In contrast, one anatomical 3D MDCT study reported the presence of the RCV in 93.8% of cases; 84% of those drained into the GTH and 10% ended in the SMV. Meanwhile, the RCV was absent in 6.2% of cases. In addition, it found that the RCV presented in double veins in 5% of cases (25) By using the same method, another study reported that the RCV was present in 56% of cases; here, it drained into the SMV 85% of the time and into the GTH 14% of the time (27).
MCV
The location of the MCV on the pancreatic head was found highly variable among the veins of the right colon in both cadaver and 3D MDCT studies. Particularly, in a cadaveric study, the MCV was always present; often, there were two or more MCVs (62.1%), with the main MCV draining directly into the SMV in 84.5% of cases. In 12% of cases, it drained into the GTH. In addition, the aMCV was observed in 57% of the cases, where they often drained into the SMV, while the other drained into the GTH. Interestingly, it was observed that two other cases involved drainage either into the splenic or inferior mesenteric vein (IMV) (23). In another cadaver study, the MCV was observed in all cases (nine cadavers), except for one case which drained into the GTH, all drained into the SMV (26). Regarding the anatomical 3D MDCT studies, in one study, the main MCV was found in all cases (81 patients); the MCVs were located cephalic to the MCA and drained into the SMV, the GTH, jejunal vein, and IMV at frequencies of 68%, 20%, 6%, and 5%, respectively. In addition, the MCV was observed to drain into the splenic vein (SV) in 1% of cases. Furthermore, two MCVs were reported in 45.7% of cases, which drained into the SMV and GTH in 35% of these patients, the SMV in 27%, and the GTH in 0.1% (25). In another radiological study, the MCV was described in 97 of 100 patients (97%), and 13% of the MCVs drained into the GTH, and 86% into the SMV (27).
SRCV
The SRCV is sometimes called the accessory RCV or aMCV (23, 28) Gillot et al. (11) reported the incidence of the SRCV to be 95%, and this joined the GTH in 59% of cases. Lange et al. (29) observed the SRCV in all cases; it drained into the GTH in 46% of instances whereas the rest drained directly into the SMV. One anatomical study defined the SRCV in detail; it drained mainly the hepatic flexure of the right colon. Moreover, its relation with other colic veins (MCV and RCV) were observed, where the SRCV was found in eight out of cases and drained into the GTH in all of these (26). In addition, two MDCT anatomical studies showed that the SRCV was found in 21% and 93% of cases, respectively. All of the reported SRCVs in both studies drained into the GTH (25, 27).
GTH
The GTH has been described as a confluence of the RGEV and anterior superior pancreaticoduodenal (ASPD) veins, with additional contributions of tributaries from the MCV, RCV, and SRCV. The GTH drains into the SMV at a mean distance of 2.2 cm from the inferior border of the pancreas, with a mean caliber of 5.2 mm, and the GTH follows the ventral–cranial direction toward the right side (30). The incidence of GTH has been reported in anatomical cadaver studies to be 70%–100% (23, 26, 30). In radiological studies, including MDCT and magnetic resonance imaging (MRI), the rates of the GTH presence ranged from 46%–100% (26, 27, 29, 31).
There are several variations in the formation of the GTH that depend on the number of tributaries, which are referred to as bipodal, tripodal, and tetrapodal. Based on the cadaver studies, there is one scheme that described the variations of venous structures into the confluence of the GTH according to colic veins, as shown in Fig. 4 (26). Recently, a radiological study using 3D MDCT provided a classification according to variation in the colic venous tributaries, mainly the RCV, the MCV, and the SRCV Fig. 5 (27). Interestingly, in a few studies, the distances between the GTH and other venous and arterial vessels of the right colon were measured using MDCT. When the MCA branched superiorly to the confluence of the GTH, the mean vertical distance between them was 10.0 ± 6.4 mm, while it was 10.0 ± 6.4 mm when the MCA branched inferiorly to the GTH. In addition, the distance between the ICV and the GTH was 34.2 ± 10.3 mm (20).
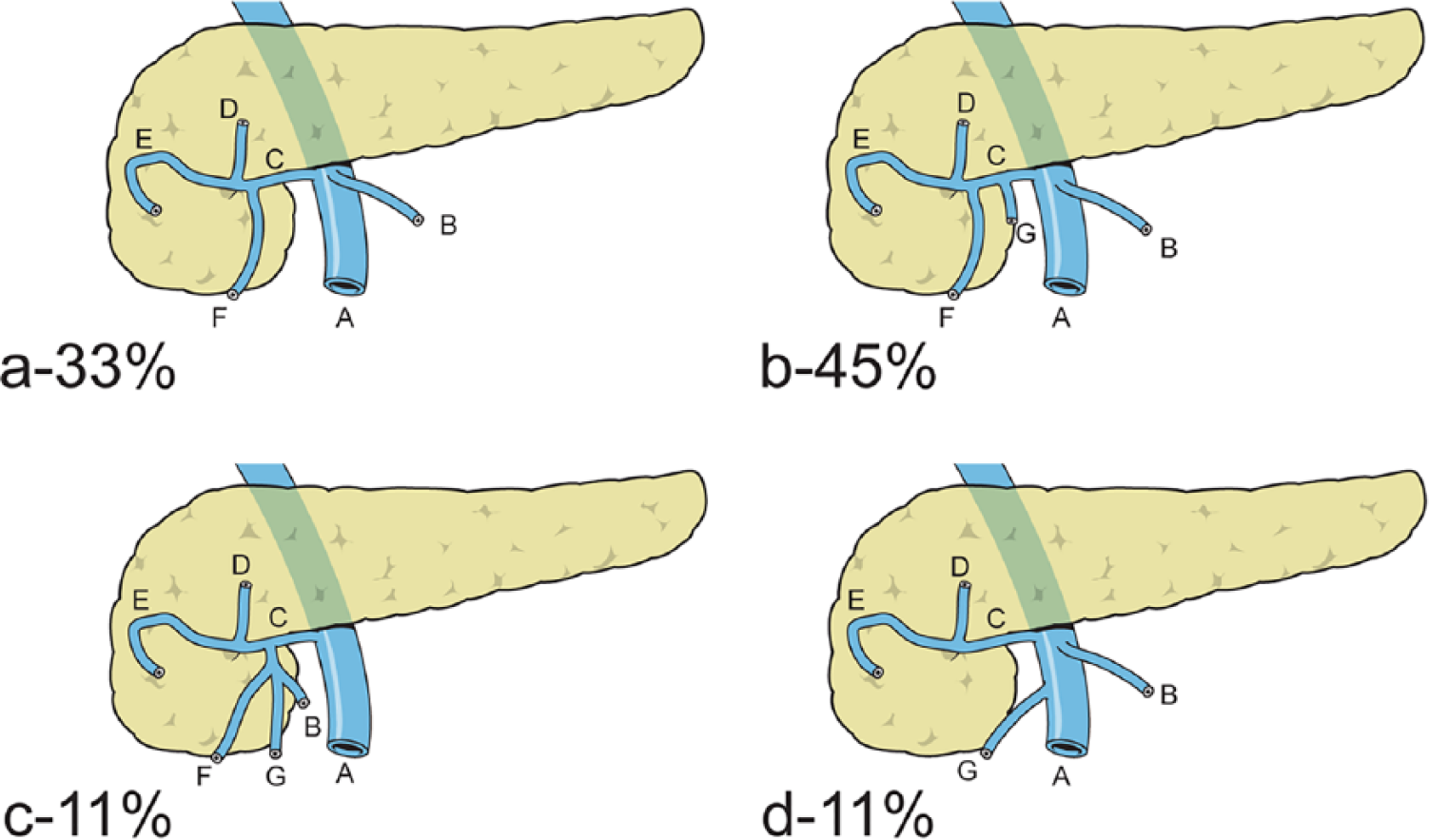
a) to d) Variations of Henle’s gastrocolic trunk according to the number of tributaries and their percentages (A: superior mesenteric vein; B: middle colic vein; C: Henle’s gastrocolic trunk; D: right gastroepiploic vein; E: anterior superior pancreaticoduodenal vein; F: superior colic vein; G: right colic vein) (26).
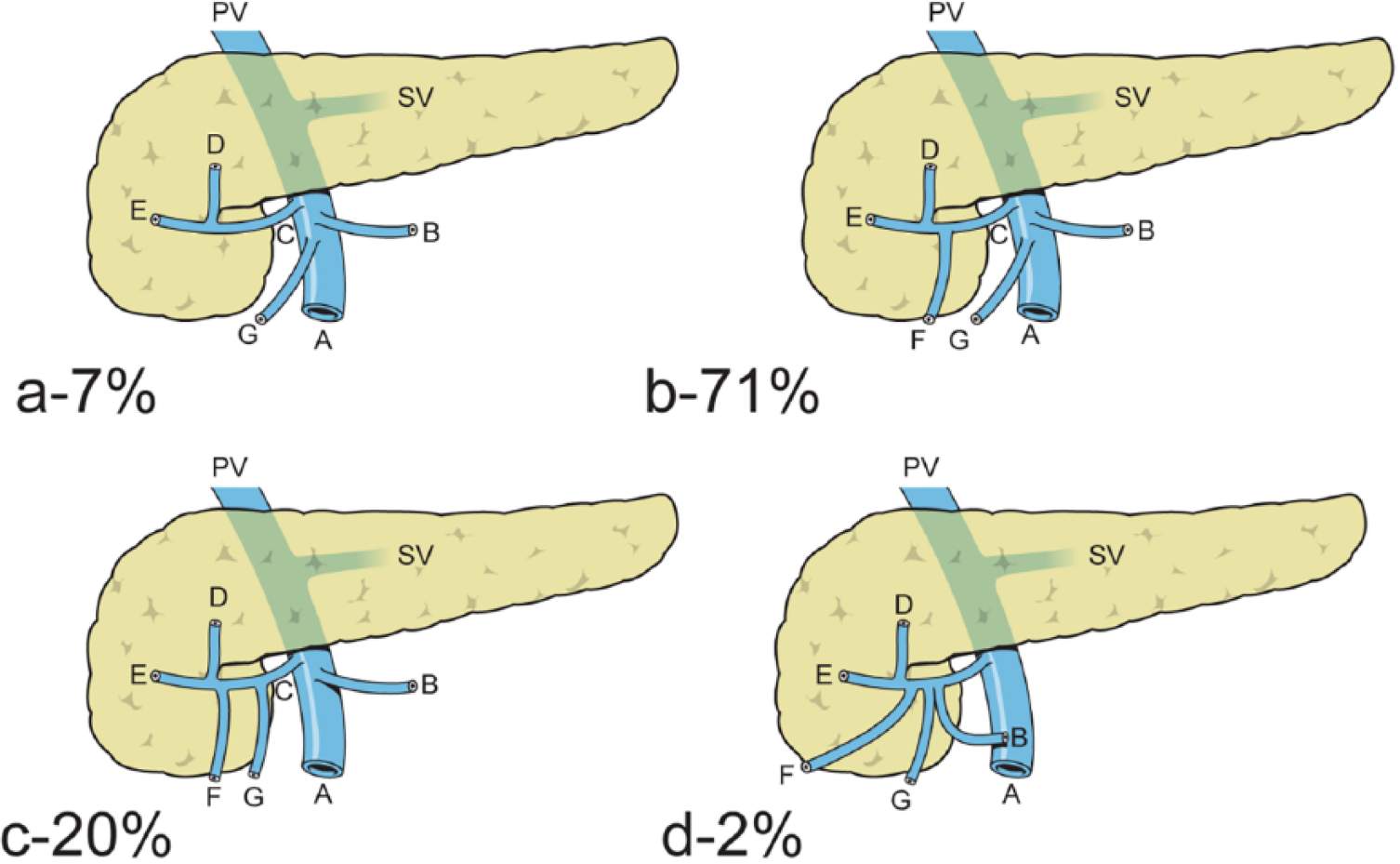
a) to d) The variations of Henle’s gastrocolic trunk according to the right colonic veins and their percentage of appearance (A: superior mesenteric vein; B: middle colic vein; C: Henle’s gastrocolic trunk; D: right gastroepiploic vein; E: anterior superior pancreaticoduodenal vein; F: superior right colic vein; G: right colic vein) (27).
Clinical Implications
Initially, all of the studies showed that the ICA and ICV are present in 100% of all samples. This finding will strongly support the ileocolic pedicle (ICA and ICV) as a landmark for starting the safe dissection of the mesocolon in the medial to lateral technique. Hohenberger et al. (2, 6) advocate radical colon resection, which entails CME and CVL in right colon cancer, including division of the ICA and the RCA on the left-hand side of the SMV and the MCA (high ligation), with additional removal of related main lymph nodes. This is almost the same as the D3 lymphadenectomy in the Japan Clinical Oncology Group (JCOG) (32, 33).
Recently, Bertelsen et al. evaluated the oncological outcomes of CME and conventional resection. They concluded that CME is associated with better disease-free survival when compared to conventional resection values of 85.8% and 75.9%, respectively (34). On the other hand, a systemic review showed that despite the high quality of the specimen, and the more excised tissue with maximal retrieved lymph node due to implementing the CME and CVL, the long-term survival benefit has not been proved yet (35). However, CME is associated with more intraoperative vascular and organ injuries and severe non-surgical complications than “conventional” resection for colonic cancer (36).
By employing this concept (CME and CVL), it is mandatory to understand the clinical anatomy of D3 lymphadenectomy in detail. This area has been described well as the cranial border of the D3 area, running 5 mm proximal to the line connecting the origins of the GTH and the MCA; the medial border of the D3 area, running along the left-hand side of the SMA; the caudal border of the D3 area, running 5 mm distal to the line connecting the origin of the ICA; and the lateral border of D3 area, running 1 cm along the right-hand side of the SMV (13).
There are several crucial issues associated with the D3 area. First, it is necessary to maintain awareness of the right colonic arterial branches in terms of the crossing pattern to the SMV, as described above, and guidance should be provided during bleeding control to prevent injuries to the main mesenteric trunks (SMA and SMV). This is especially in case of the minimally invasive approach, where the risk of bleeding reaches 3% to 9.2% (37). It is of concern that the bleeding control of the RCA or the ICA after ligation may be disastrous due to the retraction of these arterial branches, particularly when posterior crossing to the SMV is present. Recently, some vascular variations crossing the D3 area were found, such as the jejunal vein in 30% of patients and the IMV in 2.2% at the level of the ICV (20). These variations will add more awareness while approaching this area to avoid the risk of bleeding. Second, a significant length of stump containing a significant number of principle lymph nodes with a risk of metastasis reaching 11% after right colon cancer resection is required in D3 lymphadenectomy (13, 38). The remaining length of the ICA and the RCA after D2 right colonic resection in right-sided colon cancer patients may have a risk of main lymph node recurrence reaching from 3.2%–5.8% (39, 40). In addition, one of the unique cadaveric studies observed that there was a significant difference in the number of lymph nodes depending on the crossing pattern (posterior or anterior) of the ICA, whereas no significant difference was found regarding the number of retrieved lymph nodes independent of the presence or absence of the RCA and its crossing pattern. These observations demand a clinical correlation, which may add to the prognosis of the oncological outcome. Furthermore, a group of lymph nodes found at the area that does not contain arteries, such as the GTH, and posterior to the SMV, needs to be considered as well (13).
The anatomy of the region at the inferior border of the head and neck of the pancreas which is around 0.5–2.0 cm from the right side of the SMV is one of the most challenging aspects of pancreatic and right colonic resections for two reasons, namely, the fusion between the greater omentum and the transverse mesocolon at this level and the complex anatomy of the vulnerable venous tributaries of the SMV. Therefore, these veins must be identified and dissected carefully to avoid troublesome hemorrhage due to iatrogenic damage. In particular, inadvertent traction by the first assistant is a well-known cause of severance of these fragile veins, mainly the GTH. It is necessary for colorectal surgeons to comprehend the anatomical relations and variations of the RCVs to the GTH, which may guide them in controlling usual, unexpected bleeding around this region. Interestingly, one author reported that the GTH has a close relation with the RCA, with an average distance of 3.6 mm in 55.9% of cases (22). Moreover, the GTH has an important oncological impact, by representing the upper zone of D3 lymphadenectomy around the surgical trunk in right-sided colon cancer patients, in addition to the educational concept of CME and CVL (20, 41).
In fact, the usefulness of MDCT angiography in the assessment of branches arising from the SMA and SMV, particularly for the right colon, has been reported (17–19, 25, 27). Recently, there has been interest in a prospective randomized study comparing two groups of surgeons based on preoperative evaluation of MDCT angiography in laparoscopic colorectal surgery, which showed an improvement of surgical outcomes after preoperative assessment of MDCT angiography (42). Furthermore, a comparative study between MDCT and intraoperative findings was conducted to assess the vascular structures related to the right colon. This study showed that the diagnostic accuracy, sensitivity, and specificity were 97.1%, 85.7%, and 95.2%, respectively (20). MDCT angiography has some limitations. First, it is contraindicated in patients with impaired renal function or contrast allergy. Second, there is an increased risk of radiation exposure (43, 44). Finally, there are still some difficulties when it comes to defining vascular structures, especially in cases of previous surgery or vascular diseases (25).
Improving the understanding of the anatomy and embryological planes of mesentery, as well as techniques like the medial to lateral approach, will serve to guide us in road mapping to observe the incidence and variations of anatomical vascular structures as the cadaver and radiological studies have done.
By using a robotic surgical system in colectomies, which have advantages like 3D high-definition (HD) vision, visual magnification, EndoWrist technology, and the firefly system, may improve awareness and safety in right-sided colectomy. Good short-term outcomes have been shown by the preliminarily trials in this field (45).
Conclusion
In light of the above-mentioned studies, it was found that the main arterial branches supplying the right colon are the ICA and the MCA, since both branches were present in all patients; meanwhile, the RCA was found in 10%–60% of patients. In addition, the relationships of the RCA and ICA to the SMV are mostly anterior and posterior, respectively, which should be considered during CVL to avoid injuries and to do adequate lymphadenectomy. The ICV and MCV are always present, while the RCV is absent in 50% of patients. Apparently, the GTH receives venous drainage from the ascending and transverse colon in most patients, mainly from the RCV and the MCV. Therefore, it is crucial to raise awareness and comprehensively understand the variations of the 3D vascular anatomy of the right colon during surgical resection, particularly in MIS, because of the significant influence on morbidity and mortality, as well as oncological outcomes.
Footnotes
Acknowledgements
The authors would like to thank Mr Jang Dong Su for drawing illustrative figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
