Abstract
Background and Aims:
Major upper abdominal surgery is often associated with reduced health-related quality of life and reduced survival. Patients with upper abdominal malignancies often suffer from cachexia, represented by preoperative weight loss and sarcopenia (low skeletal muscle mass) and this might affect both health-related quality of life and survival. We aimed to investigate how health-related quality of life is affected by cachexia and how health-related quality of life relates to long-term survival after major upper abdominal surgery.
Materials and Methods:
From 2001 to 2006, 447 patients were included in a Norwegian multicenter randomized controlled trial in major upper abdominal surgery. In this study, six years later, these patients were analyzed as a single prospective cohort and survival data were retrieved from the National Population Registry. Cachexia was derived from patient-reported preoperative weight loss and sarcopenia as assessed from computed tomography images taken within three months preoperatively. In the original trial, self-reported health-related quality of life was assessed preoperatively at trial enrollment and eight weeks postoperatively with the health-related quality of life questionnaire Short Form 36.
Results:
A majority of the patients experienced improved mental health-related quality of life and, to a lesser extent, deteriorated physical health-related quality of life following surgery. There was a significant association between preoperative weight loss and reduced physical health-related quality of life. No association between sarcopenia and health-related quality of life was observed. Overall survival was significantly associated with physical health-related quality of life both pre- and postoperatively, and with postoperative mental health-related quality of life. The association between health-related quality of life and survival was particularly strong for postoperative physical health-related quality of life.
Conclusion:
Postoperative physical health-related quality of life strongly correlates with overall survival after major upper abdominal surgery.
Introduction
Patients undergoing major upper abdominal surgery are often weakened by weight loss (WL) (1–3) and sarcopenia (low skeletal muscle mass) (4, 5), and major complications are relatively frequent (6, 7). WL in cancer patients has been associated with reduced health-related quality of life (HRQOL) (8) but the impact of sarcopenia is uncertain (9).
Survival after upper abdominal surgery for malignant diseases is often limited, even after operations with curative intent, and long-standing functional impairment is common (10, 11). As cachexia, sarcopenia, and reduced HRQOL have all been linked to reduced survival (3, 12–14), these factors are potentially important in preoperative risk assessment and postoperative care.
Little is known about the relationship between postoperative HRQOL and survival after abdominal surgery, although some studies have indicated a positive association (15). A possible correlation between postoperative HRQOL and survival would probably reflect both advanced disease and functional impairment. Self-reported HRQOL might then represent an important tool in postoperative follow-up, both for identifying patients at risk of reduced survival and to target functional impairment.
We have investigated the relationship between cachexia, sarcopenia, and HRQOL and, furthermore, the relationship between HRQOL and long-term overall survival in a large cohort of patients undergoing major upper abdominal surgery.
Materials and Methods
From 2001 to 2006, 447 patients were included in a Norwegian multicenter randomized controlled trial (RCT), which investigated how normal food at will after all kinds of major upper abdominal surgery was tolerated (1). Further analysis has not been performed on this dataset and it has now, six years later, been treated as a single prospective cohort. We collected survival data from the Norwegian Population Registry. Survival data were collected during April 2012, six years after the last patient was included in the original trial. Patients without any preoperative or postoperative HRQOL data were excluded from further analysis.
In addition to general demographics and clinical characteristics, the prospective database included specifically: patient-reported preoperative WL, surgical procedures, and major postoperative complications. Major complications, within eight weeks after surgery, were defined a priori and listed in the original publication (1), as were the operative procedures (1). Major complications were defined as major infectious, -pulmonary, -cardiac or -surgical, for example, pneumonia, myocardial infarction, or anastomotic leakage. Detailed criteria are listed in the original publication (1).
Preoperative WL was calculated from patients’ usual pre-morbid weight as they recalled it, with no time-limit, and current weight, which was scaled preoperatively at trial enrollment. It was dichotomized into >5% or ⩽5% WL, according to international consensus (12).
Digitally stored computed tomography (CT) images for initial routine diagnostics and staging were analyzed using Slice-O-Matic software V4.2 (Tomovision, Montreal, Canada) which permitted specific tissue demarcation using Houndsfield unit threshold of −29 to +150 for skeletal muscles (16), −150 to −50 for visceral adipose tissue (17), and −190 to −30 for subcutaneous adipose tissue (16). Cross-sectional areas (cm2) were calculated for each tissue by summing tissue pixels and multiplying by the pixel surface area. A transverse CT image from the third lumbar vertebrae was assessed for each scan and tissue areas estimated (18). One single trained observer, who was blinded to all clinical and HRQOL data, analyzed all CT images. Cross-sectional area was normalized for stature (cm2/m2) and lumbar skeletal muscle index was calculated. CT images used for analysis were retrieved from routine images done within three months preoperatively. We used the cut-off for the lumbar skeletal muscle index suggested by Mourtzakis et al. (19), which corresponds to skeletal muscle mass two standard deviations from that of healthy young adults (39 cm2/m2 for women and 55 cm2/m2 for men).
Self-reported HRQOL was assessed preoperatively at trial enrollment and 8 weeks after surgery in the original trial (1). The validated Norwegian translation (20) of the generic HRQOL instrument: Short Form 36 (SF-36), Norwegian version 1.2, was utilized. While SF-36 is not a disease-specific instrument, it has been used and validated in a wide range of malignant and non-malignant diseases (21). SF-36 contains 36 questions that produce eight scales. These scales are; physical functioning, role limitation (caused by physical problems), bodily pain, general health, vitality, social functioning, role limitation (caused by emotional problems), and mental health. These scales were aggregated to produce a summary physical HRQOL score (physical component summary (PCS)) and a summary mental HRQOL score (mental component summary (MCS)) (22). These norm-based scores have a mean of 50 and a standard deviation of 10 in general adult populations (22). Reduced HRQOL were defined as summary scores lower than 50.
Patients were divided into the following disease-categories: gastroesophageal cancer, pancreatic cancer, other cancers (mainly malignant liver tumors and liver metastases), and non-cancer.
Statistics
Statistical analysis was performed with SPSS statistics software, version 22 (IBM, New York, NY, USA). A Cox proportional hazard regression analysis, stratified for disease-categories, was used for analysis of overall survival; the assumption of proportional hazards was visually inspected by log–log survival curves. Variables with a p value of <0.20 in the unadjusted analysis were included in the multivariable adjusted analysis. Pearson chi-square test and analysis of variance (ANOVA) were used for comparison of characteristics between different groups and categories. One-way t-test was used for analysis of two repeated measures. Linear regression analysis was used for analysis with HRQOL as outcome, adjusted for type of disease. The assumption of linearity and variance homogeneity was visually inspected. In all analysis, p values of <0.05 were considered statistically significant.
Results
Selection and Characteristics
The original study population comprised 447 patients. HRQOL data were missing for 53 (12.1%) patients, and survival data were not available for another 9 (2.0%) patients. Thus, survival data and complete preoperative and/or postoperative HRQOL data were available for 385 (86.1%) patients of the original study population.
Information on preoperative WL was obtained for 87.2% (336/385) of these patients, and preoperative CT images of sufficient quality were available for 44.4% (171/385). Complete preoperative or postoperative HRQOL data were available for 82.1% (316/385) and 73.8% (284/385) of the patients, respectively. In 215 patients (55.8%), complete both preoperative and postoperative HRQOL data were available.
Sixteen patients (4.2%) died within 90 days after surgery. A total of 198 patients (51.4%) died within five years. A preoperative WL in excess of 5% was observed in 116 patients (116/336 = 34.5%). Sarcopenia was encountered in 69.0% (118/171 = 69.0%) of the patients with available CT images. One or more major postoperative complications occurred in 29.6% (114/385) of the patients.
Reduced physical HRQOL (PCS < 50) was encountered preoperatively in 69.3% (219/316) and postoperatively in 78.5% (223/284) of the patients. Reduced mental HRQOL (MCS < 50) was encountered in 65.8% (208/316) preoperatively and in 51.8% (147/284) of the patients postoperatively. Pre- and postoperative PCS and MCS values in the different disease categories are illustrated in Fig. 1.
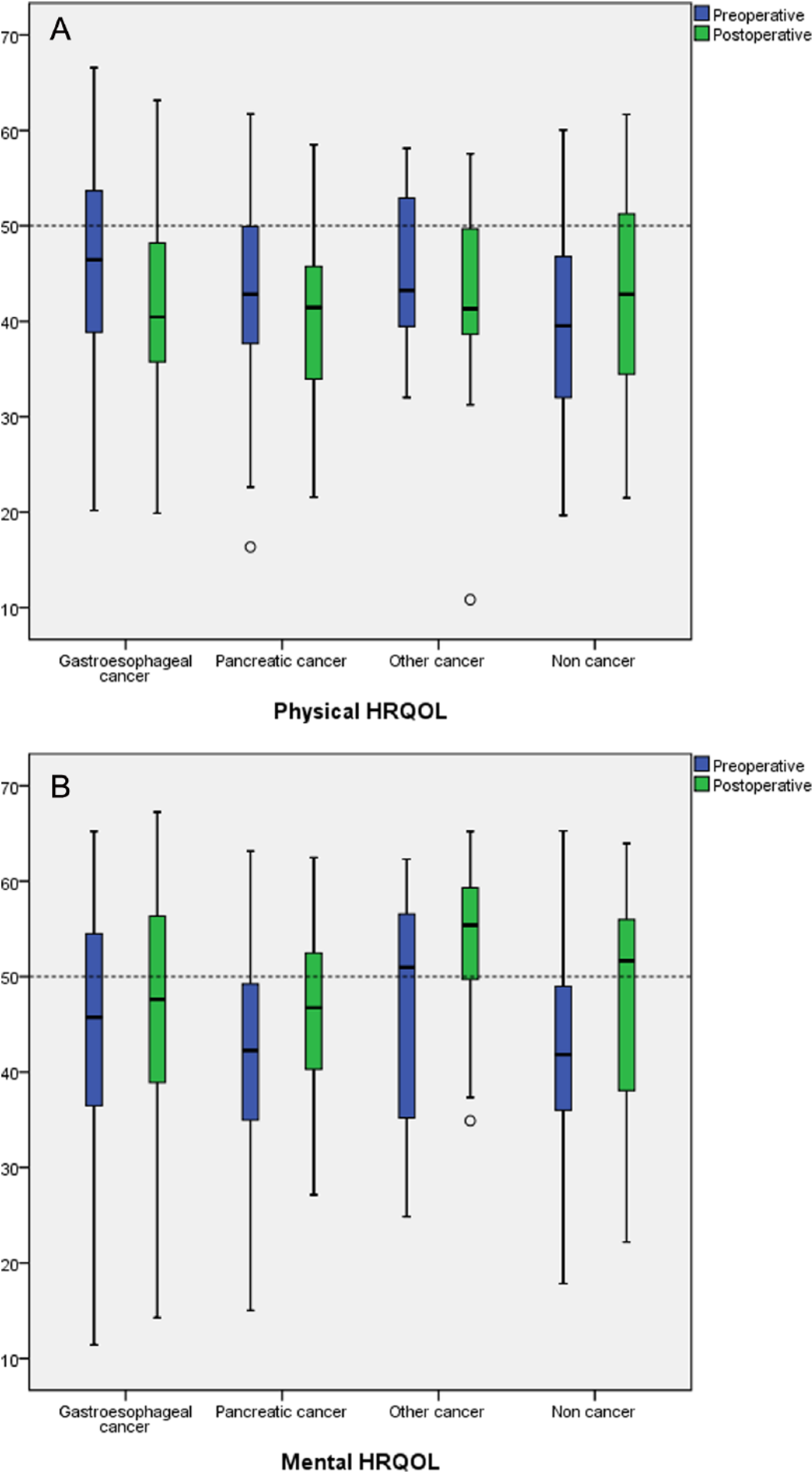
Pre- and postoperative HRQOL in the different disease categories. A) Physical HRQOL and B) mental HRQOL.
Changes in HRQOL
There was a statistically significant decrease in PCS postoperatively for patients in all disease categories except for patients without cancer (increase in PCS, p = 0.056) and patients with pancreatic cancer (decrease in PCS, p = 0.058) (Table 1). Mean decrease in PCS after surgery for the entire cohort was −2.3 (p = 0.002) (Table 1). There was a statistically significant increase of postoperative MCS in all disease categories except for patients with gastroesophageal cancer (increase in MCS, p = 0.058). Mean increase in MCS after surgery for the entire cohort was 3.9 (p < 0.001) (Table 1).
Change in HRQOL from preoperative to postoperative in all patients and in the different disease categories.
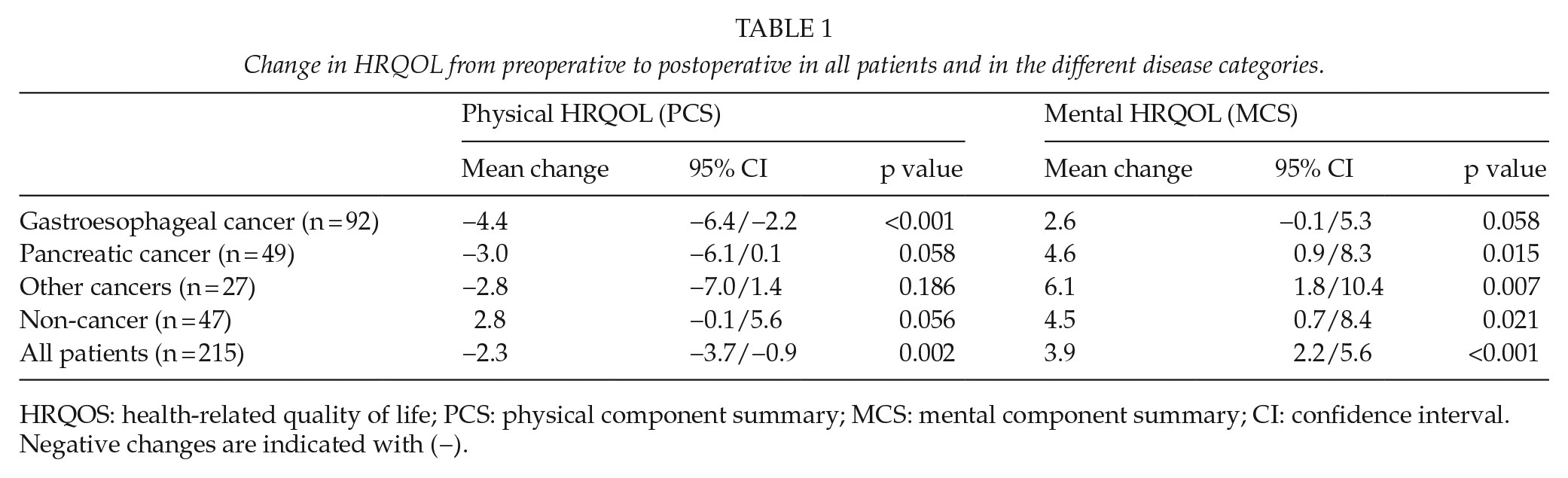
HRQOS: health-related quality of life; PCS: physical component summary; MCS: mental component summary; CI: confidence interval.
Negative changes are indicated with (−).
Risk Factors, Postoperative Complications, and HRQOL
Patients older than 65 years had higher preoperative MCS as compared to patients younger than 65 years (estimated difference (ED) = 5.2, p > 0.001). Patients with preoperative WL >5% had a significantly lower preoperative PCS than patients with preoperative WL ⩽5% (ED = −3.3, p = 0.012) (Table 2). Patients who suffered postoperative complications had significantly lower postoperative PCS (ED = −3.1, p = 0.023), with no difference in postoperative MCS (p = 0.149), compared to patients without complications.
The association between age, WL, sarcopenia, and HRQOL pre- and postoperatively.
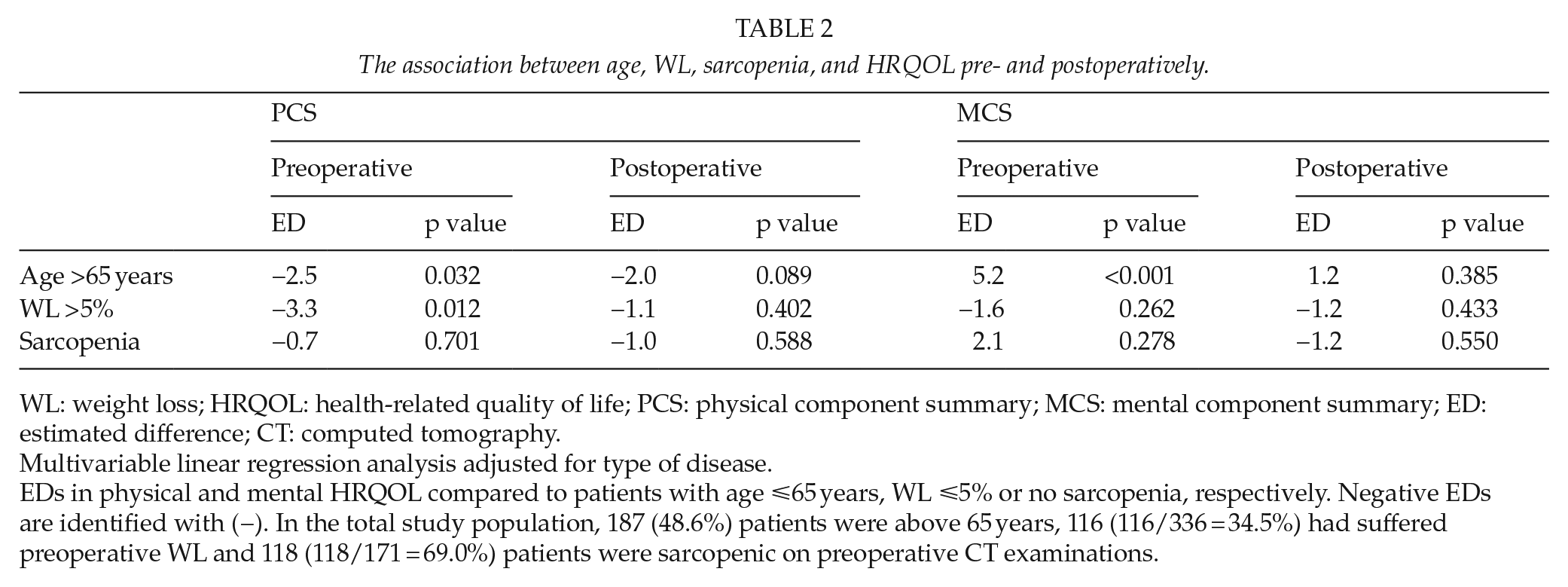
WL: weight loss; HRQOL: health-related quality of life; PCS: physical component summary; MCS: mental component summary; ED: estimated difference; CT: computed tomography.
Multivariable linear regression analysis adjusted for type of disease.
EDs in physical and mental HRQOL compared to patients with age ⩽65 years, WL ⩽5% or no sarcopenia, respectively. Negative EDs are identified with (−). In the total study population, 187 (48.6%) patients were above 65 years, 116 (116/336 = 34.5%) had suffered preoperative WL and 118 (118/171 = 69.0%) patients were sarcopenic on preoperative CT examinations.
There was no association between sarcopenia (low skeletal muscle mass) (19), and pre- or postoperative HRQOL (Table 2). There were no association between the continuous variable lumbar skeletal muscle index (adjusted for age and gender) and pre- or postoperative HRQOL.
HRQOL and Overall Survival
Pre- and postoperative PCS and postoperative MCS were all significantly associated with survival when analyzed as continuous variables.
Preoperative PCS < 50 was associated with a worse overall survival (hazard ratio (HR) = 1.38, p = 0.048) (Table 3), but no association was observed after adjustment for preoperative WL (p = 0.362).
The association between pre- and postoperative HRQOL and overall survival.
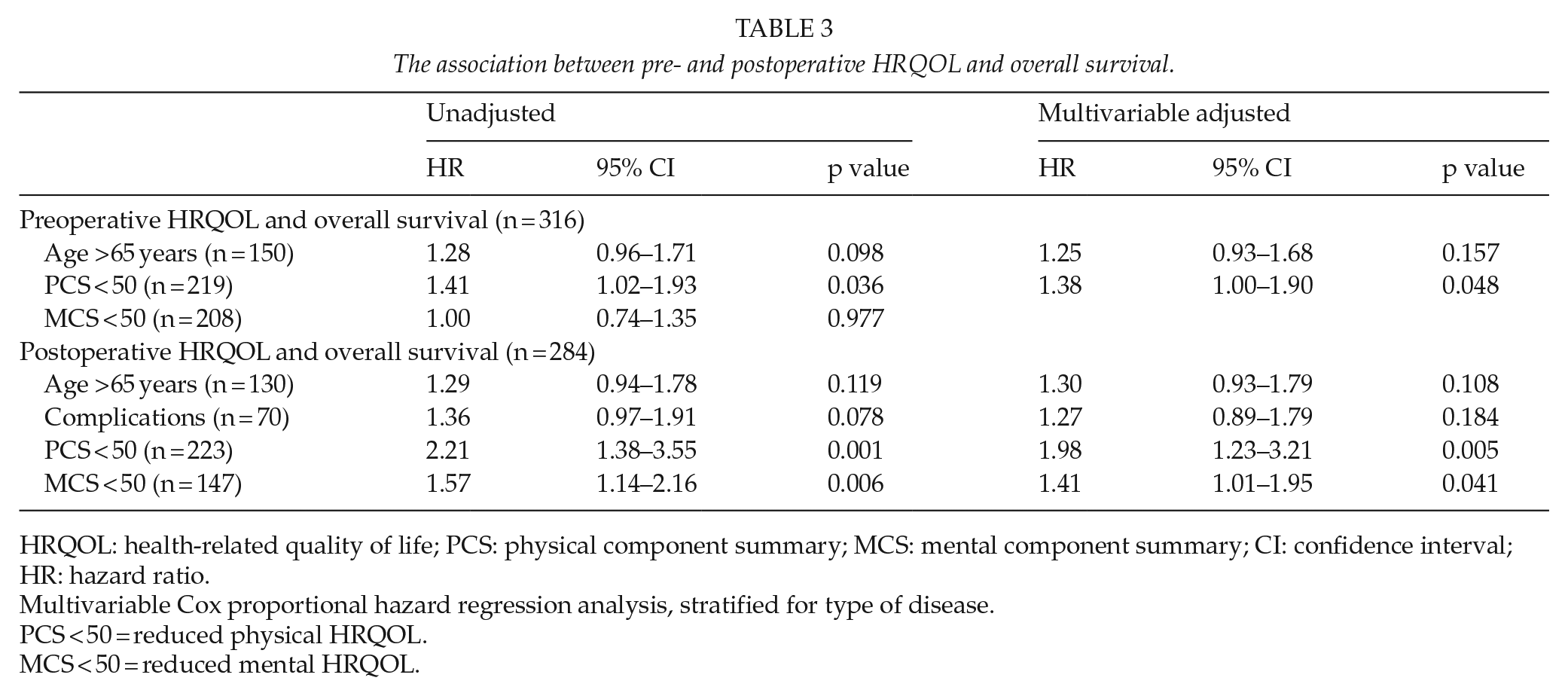
HRQOL: health-related quality of life; PCS: physical component summary; MCS: mental component summary; CI: confidence interval; HR: hazard ratio.
Multivariable Cox proportional hazard regression analysis, stratified for type of disease.
PCS < 50 = reduced physical HRQOL.
MCS < 50 = reduced mental HRQOL.
Postoperative PCS < 50 (HR = 1.98, p = 0.005) and MCS < 50 (HR = 1.41, p = 0.041) were associated with reduced overall survival, also when adjusted for age and postoperative complications (PCS < 50: HR = 1.98, p = 0.05) (Table 3). The association between PCS and overall survival was similar in all disease categories, including patients without cancer (PCS < 50: HR = 2.99, p = 0.08).
Discussion
We have demonstrated a strong association between postoperative HRQOL and overall survival after major upper abdominal surgery, particularly for physical HRQOL. A majority of patients in our cohort experienced improved mental HRQOL and, to a lesser extent, deteriorated physical HRQOL following surgery. This study is, to our knowledge, the first multicenter investigation of HRQOL in relation to cachexia and sarcopenia together with long-term survival after major upper abdominal surgery. This adds to our investigation of sarcopenia and morbidity in the same study population (23).
An association between reduced preoperative HRQOL and overall survival is well documented (14). While this association was confirmed in our study, the strong association between postoperative HRQOL and overall survival was more evident. The physical HRQOL as an outcome after major upper abdominal surgery is important as such. In addition, this quality of life measure appears to be a sensitive predictor for overall survival.
Patients with gastroesophageal cancer experienced the largest deterioration in physical HRQOL after surgery and these were the only patients in whom an improvement in mental HRQOL after surgery could not be shown. Surgery for gastroesophageal cancer (esophagectomy and gastrectomy) can cause especially severe functional impairment (24) which might explain these findings.
Upper abdominal malignancies are often associated with a poor prognosis (23). Younger cancer patients appear to be more predisposed to anxiety and depression when faced with the possibility of a huge reduction in lifespan or long-lasting functional impairment (25). This might in part explain why younger age was associated with worse preoperative mental HRQOL in our study.
Little is known about the impact of sarcopenia on HRQOL in patients with upper abdominal malignancies. Our results on WL and sarcopenia are consistent with the results in a study on patients with advanced colorectal carcinoma, where no association between sarcopenia and HRQOL was found, but where an association between WL and deteriorated HRQOL was indeed observed (9). The association between WL and HRQOL in cancer patients is well documented (8) and reduced preoperative HRQOL has been associated with advanced disease in pancreatic cancer (26). Preoperative WL might be a more global indicator of advanced disease than the amount of skeletal muscle measured at a single time-point (23), and hence be more directly linked to preoperative HRQOL.
Some limitations of our study should be addressed. For one, 14% of the patients in the original cohort were excluded because survival and/or HRQOL data were not available. Furthermore, we lacked complete pre- and postoperative HRQOL data in 18% and 26% of these patients, respectively. The percentage of patients with available CT images to assess sarcopenia and HRQOL was only 44%. Stage of disease was not included in our study, as our database lacked information on this variable.
The survival benefit from upper abdominal surgery is often limited (10, 11), and patients frequently suffer complications (6, 7) which again have been associated with reduced HRQOL (27). This is sometimes used as an argument against surgery in borderline cases. Interestingly, the patients in our study, even those who suffered postoperative complications, experienced improved mental HRQOL after surgery. Furthermore, the deterioration in physical HRQOL was less severe. These findings might be useful to consider when benefits of upper abdominal surgical procedures are evaluated.
Structured oncologic follow-up after surgery for gastric and pancreatic cancer has not improved long-term outcomes (10). Research has indicated that oncologic follow-up including routine imaging can cause increased anxiety (28) but many patients welcome regular visits to a physician (29). Postoperative physical HRQOL is probably influenced by many factors and stage of disease might be the most important. Nevertheless, postoperative HRQOL is also influenced by factors which are not necessarily directly related to stage of disease, including functional impairment, pain and nutritional depletion. Identifying patients with reduced postoperative HRQOL and paying more attention to their functional impairments may improve long-term HRQOL, and even survival, with few unwanted side-effects. Further research could investigate the benefit of intervention on functional impairment after major upper abdominal surgery.
Footnotes
Acknowledgements
EKAa and KL performed the data analysis. NJ calculated the body composition indices, blinded to all clinical data. EKAa, GT, JAS, KCF, AR, and KL participated in study conception and design. All authors participated in data acquisition and interpretation, manuscript preparation, editing, and final approval. The authors thank consultant surgeon, A. Bernstein, for data collection from Sørlandet hospital Arendal. The authors thank Professor Tom Wilsgaard, medical statistician, The Arctic University of Norway. The authors thank senior researcher Andrew Garrat, Norwegian Knowledge Centre for the Health Services, for his help in calculating norm-based component summary scores. The original trail was registered at ![]() on 23 August 2005 (NCT00134407).
on 23 August 2005 (NCT00134407).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
Inclusion into the original RCT, as well as long-term follow-up was approved by the Regional Division of the National Committee for Research Ethics, Northern Chapter (REK V). All patients provided written consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
