Abstract
Background and Aims:
According to the heterogeneous results of previous studies, the prevalence of abdominal aortic aneurysm seems high among men with coronary artery disease. The associating risk factors for abdominal aortic aneurysm in this population require clarification. Our objective was to assess the prevalence of non-diagnosed abdominal aortic aneurysms in men with angiographically verified coronary artery disease and to document the associated co-morbidities and risk factors.
Material and Methods:
Altogether, 407 men with coronary artery disease were screened after invasive coronary angiography in two series at independent centers. Risk factor data were recorded and analyzed.
Results and Conclusion:
The mean age of the study cohort was 70.0 years (standard deviation: 11.0). The prevalence of previously undiagnosed abdominal aortic aneurysms in the whole screened population of 407 men was 6.1% (n = 25/407). In a multivariate analysis of the whole study population, the only significant risk factors for abdominal aortic aneurysm were age (odds ratio: 1.04, 95% confidence interval: 1.00–1.09) and history of smoking (odds ratio: 3.13, 95% confidence interval: 1.26–7.80). Non-smokers with abdominal aortic aneurysm were significantly older than smokers (mean age: 80.7 (standard deviation: 8.0) vs 68.0 (standard deviation: 11.1), p = 0.003), and age was a significant risk factor only among non-smokers (p = 0.011; p = 0.018 for interaction). Among smokers, the prevalence of abdominal aortic aneurysm was 8.8%, and 72% (n = 18/25) of all diagnosed abdominal aortic aneurysm patients were smokers. Prevalence of undiagnosed abdominal aortic aneurysms among patients with coronary artery disease is high, and history of smoking is the most significant risk factor for abdominal aortic aneurysm. Effectiveness of selective screening of abdominal aortic aneurysm in male patients with coronary artery disease warrants further studies.
Introduction
Abdominal aortic aneurysm (AAA) is a degenerative vascular disease, which manifests as a segmental pathological dilatation of abdominal aorta. Diameter of ⩾30 mm is the generally accepted threshold for diagnosis (1).
Increasing age is the predominant risk factor for AAA, and the reported prevalence of AAA has been 4%–10% among men older than 55 years (2, 3). However, according to recent evidence, the prevalence seems to be diminishing in general population even to 1.7% (4, 5). Other known risk factors for AAA are smoking, male gender, hypertension, positive family history for AAA, and other vascular diseases (6). Coronary artery disease (CAD) is a significant risk factor for AAA (7, 8), and a large proportion of the patients who develop a rupture of the abdominal aortic aneurysm (RAAA) have CAD (9).
AAA is five times more common in male than female population, and women develop RAAA on considerably advanced age. Therefore, most of the screening studies and programs for AAA focus on men (10).
AAA is usually asymptomatic and thus a poorly diagnosed condition. The diagnosis is usually incidental, and abdominal ultrasound (US) examination is the most feasible modality to diagnose AAA. The natural course of the disease is characterized by a gradual expansion accompanied by an increased risk of rupture. RAAA is a catastrophic event with overall mortality around 80% (6, 10). However, the mortality decimates to less than 5% if the aneurysm is treated electively (7, 11).
The limitation of previous studies screening AAA among CAD patients is that the results have been heterogeneous despite the fact that the prevalence of AAA seems high in most studies (8). Most studies on prevalence of AAA among CAD patients include patients with already diagnosed AAAs, thus hampering the clinical relevance of the data. The objective of our study was to determine the true prevalence and demography of non-diagnosed AAAs in men with verified CAD in coronary angiography in two independent clinical series. The secondary aim was to detect and analyze the predictors for AAA in male CAD population.
Material and Methods
This was a prospective screening study with replication of data from another unit. The study was approved by the ethical committee of the University of Eastern Finland and complies with the Declaration of Helsinki. Every participating patient provided a written informed consent. The ClinicalTrials.gov registration number for the study is NCT01897623.
The primary series of the study was conducted in North Karelia Central Hospital in Joensuu, Finland, with a catchment population of 179,000. The replication series for these data was undertaken in Tampere University Hospital, Finland, with a primary catchment population of 521,000.
Study Population
Inclusion criterion for the study was the male gender with CAD in coronary angiography with >50% narrowing in at least one major epicardial vessel or a fractional flow reserve of <0.8. Exclusion criteria were the following: previously diagnosed AAA, recently (within 1 year) excluded AAA, previously treated AAA or RAAA, patient in extremis, terminal malignant disease, or being unable to provide informed consent (demented, frailty, or delirious). Patients with stable or unstable CAD were included if they met the selection criteria. These patients were recruited and screened by the research group physicians amid their normal workflow either immediately after invasive coronary angiography or at the cardiologic ward before discharge.
In the primary series, 208 consecutive patients (who met the selection criteria) were recruited and screened between July and December in 2013. Replication series was collected between September 2013 and September 2014 by recruiting and screening 199 randomly selected patients in Tampere University Hospital. Replication cohort patients met the same inclusion criteria as the primary cohort. They were consecutive patients for the recruiting study physicians, but not consecutive for entire Tampere University Hospital unit.
Coverage for consecutiveness was 94% in the primary series. Of the 208 patients, 18 were lost to recruit due to logistic or scheduling reasons. During the recruitment period, 59 patients were excluded due to exclusion criteria and one patient refused to participate in the study (to be screened). Among those 59 excluded patients, 8 had been previously subjected to aortic repair and 24 had undergone abdominal imaging within 1 year (AAA excluded with imaging). The remaining 27 patients were excluded due to extreme fragility or end-stage malignant disease.
US Screening
The screening for AAA was done using a portable laptop US device in cardiology and intensive care units. The largest antero-posterior or transverse dimension of infra-renal aorta was registered using convex probe and vascular US program. The dimension of the vessel was measured from outer-to-outer artery wall. The threshold diameter for AAA diagnosis was 30 mm.
Demographic data of the patients were recorded. Whenever patient was diagnosed with AAA, he was informed and sent for a vascular surgeon consultation. Screening was conducted by the study group physicians after initial validation for competence by a consultant radiologist. A consultant radiologist was available and used for a second opinion in cases of poor visibility to abdominal aorta.
Statistical Methods
Demographic and descriptive data of the study population are presented using frequencies and percentages for categorical variables. For continuous variables, means and standard deviations (SDs) are reported. Statistical analyses were made using SPSS version 22.0. for MAC (IBM Corp., Armonk, NY). For categorical variables, either Pearson’s χ2-test or Fisher’s exact test was applied, and for numerical variables, independent samples t-test or Kruskal–Wallis test was applied. The risk factors for AAA were assessed with binary logistic regression analysis. Variables included in the multivariate analysis were the following: age, body mass index (BMI), cerebrovascular disease, peripheral artery disease (PAD), diabetes mellitus (DM), hypertonia (HTA), history of myocardial infarction, history of smoking (ever-smoker), chronic obstructive pulmonary disease (COPD), family history for AAA, use of statins, use of angiotensin-converting enzyme (ACE) inhibitors, and use of beta-blockers.
In all analyses, a p-value of 0.05 or less was considered statistically significant.
Results
General Characteristics
Demographic data of the combined study population are presented in Table 1. The two series were combined for further analysis because there was no significant difference in mean age (primary series vs replication (±SD): 67.5 (±11.4) vs 66.5 (±10.9), p > 0.390 for difference), indications for angiography (47% vs 46% of the patients presented with unstable angina pectoris (UAP) or acute myocardial infarct (AMI), p = 0.861), proportion of smokers between the series (50% vs 51%, p = 0.803), or prevalence of other co-morbidities such as DM, hypertension, cerebrovascular disease, or COPD (p = NS for all). In the combined study population, 37% (n = 149) of the patients had previous history of coronary angiography, 12% (n = 50) had undergone coronary artery bypass grafting (CABG), and 20% (n = 83) had had a previous percutaneous coronary intervention (PCI). PCI was performed in 51% (n = 208) of the patients during the index angiography. Previous revascularization for other atherosclerotic vascular diseases (carotid artery endarterectomy or revascularization of lower limbs) had been done in 3.2% (n = 13) of the patients.
Demographics of 407 men with coronary artery disease.
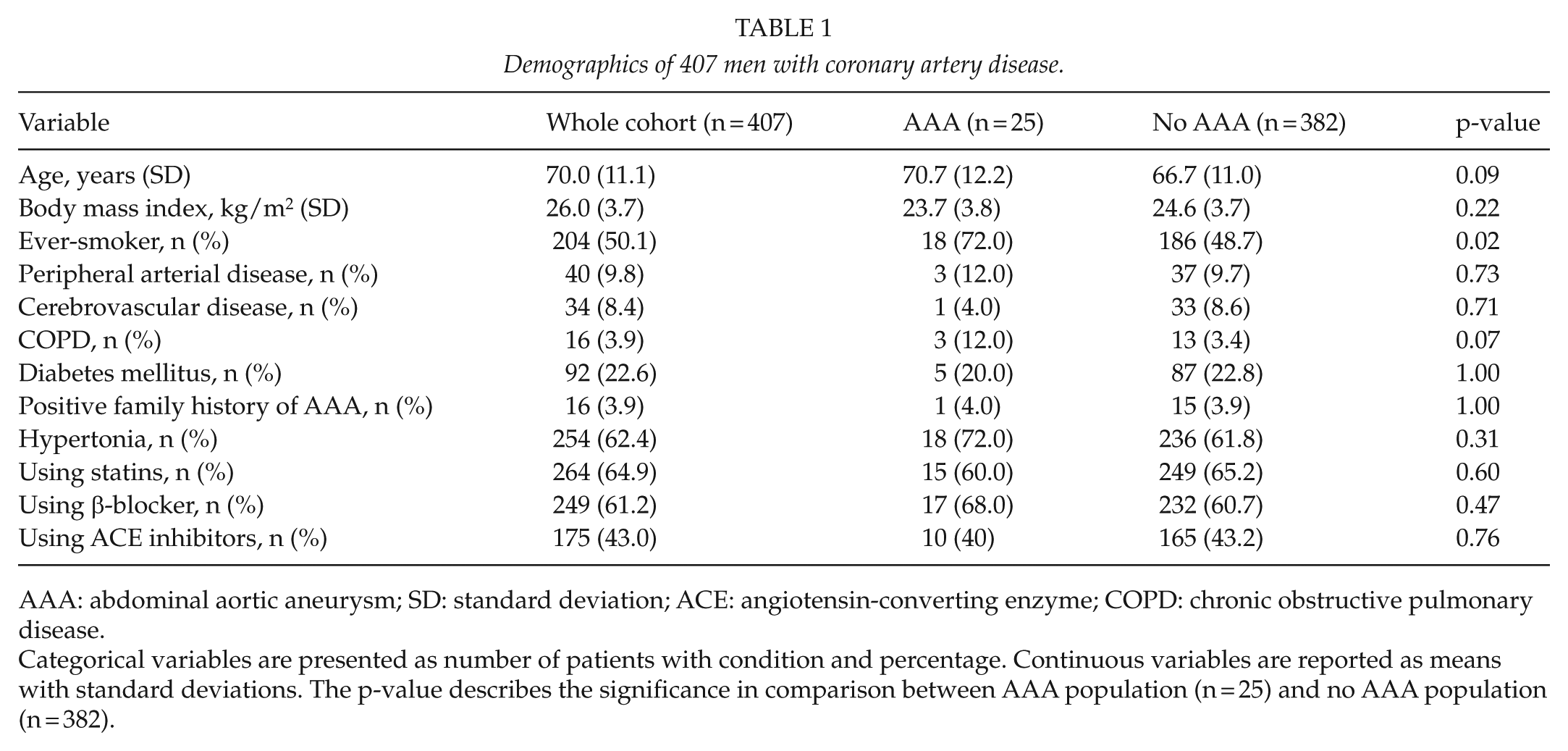
AAA: abdominal aortic aneurysm; SD: standard deviation; ACE: angiotensin-converting enzyme; COPD: chronic obstructive pulmonary disease.
Categorical variables are presented as number of patients with condition and percentage. Continuous variables are reported as means with standard deviations. The p-value describes the significance in comparison between AAA population (n = 25) and no AAA population (n = 382).
Prevalence of AAA and Risk Factors
Overall prevalence of AAA among the whole study group was 6.1% (n = 25). Overall, 15 AAAs were discovered in the primary series (prevalence of 7.2%) and 10 in the replication series (prevalence of 5.0%). The diameter of found AAAs varied between 30 and 60 mm (mean 36 mm, median 32 mm) (Table 2).
Distribution of abdominal aortic aneurysms (AAAs) by size and history of smoking.
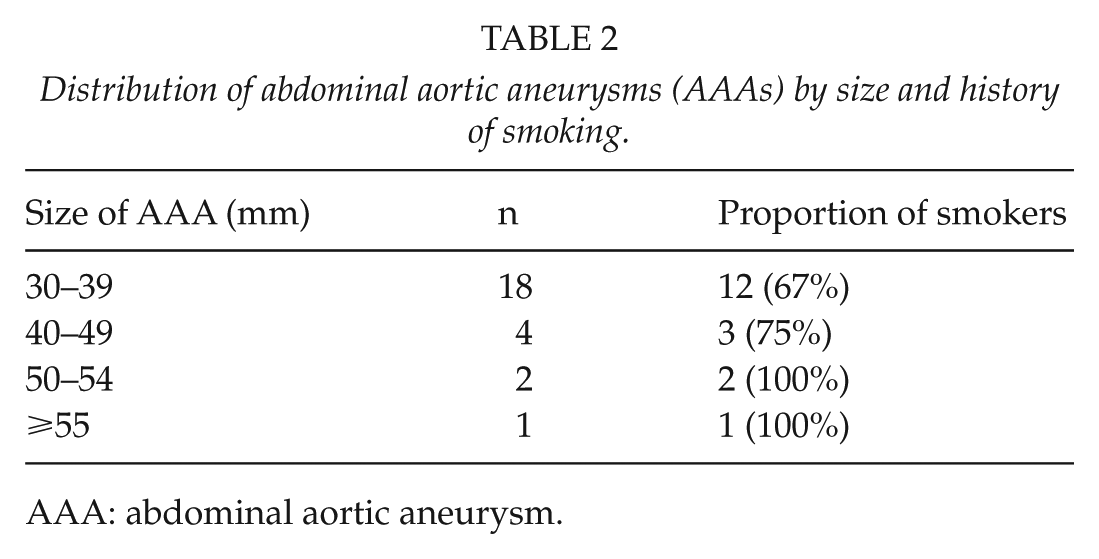
AAA: abdominal aortic aneurysm.
Only smoking and increasing age associated significantly with the risk of AAA in a multivariate analysis (odds ratio (OR) of 1.04 with 95% confidence interval (CI) of 1.00–1.09 for 1-year increase in age, p = 0.04, and OR of 3.13 with 95% CI of 1.26–7.80, p = 0.01 for smoking). Furthermore, we observed a significant interaction between age and smoking status (p = 0.02) so that increased age predisposed to AAA only in the non-smoking sub-population. For this reason, the study population was further analyzed after stratification by smoking status.
Population Stratification by Smoking Status
Overall 72% (n = 18/25) of all subjects with AAA were smokers. Proportion of smokers among AAA population also increased along with the size of the aneurysm (Table 2).
Among smokers, the subjects with AAA were not significantly older when compared to the ones without AAA (66.8 (±11.4) years vs 65.4 (±10.7) years, p = 0.61). In contrast, among non-smokers, the subjects with AAA were significantly older (80.7 (±8.0) vs 68.0 (±11.1), p = 0.003). The prevalence of AAA among smokers was 8.8% (n = 18/204), whereas the prevalence among non-smokers was 3.4% (n = 7/203).
Discussion
The main result of our study is that the prevalence of previously undiagnosed (and unsuspected) AAA is considerable among patients with CAD. The majority of AAAs was discovered in smokers or previous smokers (defined as ever-smokers). Patients with AAA but no history of smoking were significantly older when compared to ever-smokers with AAA.
In this study, the prevalence of AAA was 6.1%, exceeding clearly the prevalence of recently published findings (2.3%) based on screening of general population (4, 5). Incidence of previously undiagnosed AAAs in previous studies on CAD patients varies between 2.8% and 13% (4, 11–16) with heterogeneity in inclusion criteria and definition of CAD. Our results fall in the lower range of that spectrum, possibly due to differences in population characteristics such as age and patient selection (previously diagnosed AAAs excluded) (Fig. 1). A similar trend can be observed in the most recent results by Durieux et al. (17). Furthermore, our present results on prevalence are nearly identical to our previously published results of a screening study conducted in the same geographic area but among out-patient male population with asymptomatic coronary heart disease (wider definition also including patients without angiographic diagnosis; prevalence of AAA 6.1% vs 5.7%). However, the role of smoking as a risk factor was less significant in out-patient population (16). It is well established that the severity of the CAD correlates strongly to the prevalence of AAA (11), and the highest prevalence numbers have been reported among patients undergoing CABG (12, 18).
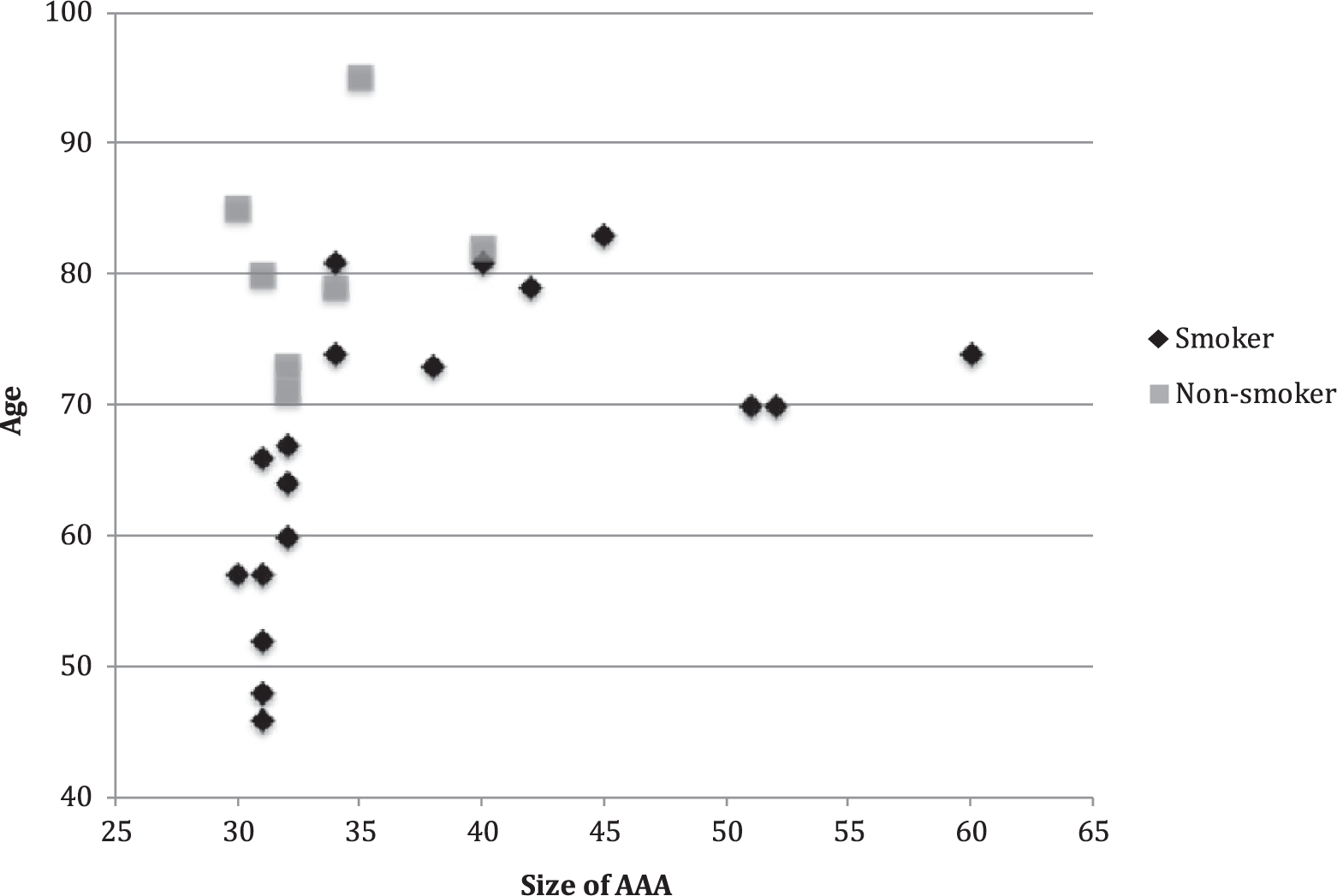
Individual characteristics of age, smoking, and aneurysm size in patients with abdominal aortic aneurysm (AAA). Age (years) is presented on Y-axis, and size of AAA (mm) is presented on X-axis.
Interestingly, according to our results, the age-adjusted prevalence of AAA is approximately threefold among patients with a history of smoking. Without age adjustment, the risk associated with smoking was only twofold. This translates to the fact that patients with AAA but no history of smoking are older (and have a higher risk factor burden) when compared to smokers with AAA. Our results support this conclusion (Fig. 1). In this study, non-smokers with AAA were relatively old with median age of 80 years, whereas ever-smokers with AAA were on average 10 years younger with one-third of the cases being 60 years or younger at the time of the screening. The same observation has been made in general population as well (6). In our study, the proportion of smokers among men with positive screening result was 72% and the prevalence of AAA was 8.8% among smokers. These numbers are in line with most previous studies reporting estimates between 65% and 80% (13, 14, 19) for the proportion of smokers among men with CAD and AAA and prevalence estimates between 6.5 and 13.4 (13, 14, 19) for AAA among smokers with CAD. Only one substantially smaller study has reported even higher prevalence of AAA among smokers (27.5%) with all discovered AAAs found among smokers with CAD (12). However, the result can be regarded as an outlier given the sample size of the study (n = 109) and substantially higher estimates that diverge from the results of other studies.
It is possible to reduce AAA-related mortality with screening programs among men in the general population, but there are controversies over the cost-effectiveness of large screening programs (5, 20–22). Several national screening programs exist: The US preventive services task force recommends one-time screening for AAA with ultrasonography in men aged 65–70 years who have ever smoked, but not among non-smokers (1). The United Kingdom and Sweden have national programs to screen all men with an age of 65 years (4, 23). Opportunistic screening for AAA is already recommended by European Society of Cardiology (24). Despite these recommendations the coverage of opportunistic screening systems remains around 40% of the eligible population and most of that screening is completed incidentally (25). A universally accepted screening system is still lacking, which is most probably due to the high associated overall costs. Therefore, a selective screening for AAA in high-risk sub-population is a compelling idea. The recent data of low prevalence in unselected population furthermore support the selective high-risk population screening strategy (4, 5). Our results concede with this.
It is noteworthy that prevalence of AAA on these types of studies can be greatly altered by applying different selection criteria (age limits and excluding conditions) on the study population. However, the underlying clinical question—how many lives could be potentially saved with screening—determines the relevance and applicability of the results. It is only of academic interest to screen those who already have had their aortas examined or are too frail or ailing for treatment. Our inclusion criteria were set with this question in mind. In the previous studies, the age limit has been set to include only patients over 50 years of age (15, 18), sometimes even ⩾65 years (12). In our population, there was no age limit and interestingly two (8%) of the AAAs were diagnosed on men <50 years of age. Both of them were smokers.
Positive family history of AAA is a significant risk factor for AAA (26). This did not manifest in our study population, which is probably explained by the study setup and inclusion criteria: all AAAs were previously undiagnosed, and, paying regard to the general awareness of the risk burden of family history in clinical practice, the first-degree relatives of our AAA patients are rather unlikely to have had their aortas screened.
US screening is a fast, easy, reliable, and convenient method for screening of AAA. There are no ethical questions about the ionizing radiation hazards or false positives. It has been documented thoroughly that aortic measurement can be done reliably even with the cardiac US probe used in trans-thoracic echocardiography (TTE) and within a timeframe of 1–4 min (27–30). Most of the patients who undergo invasive coronary angiography are also subject to TTE on the same visit, which furthermore emphasizes that AAA screening could be eloquently annexed to TTE on these patients.
Weaknesses of this Study
Owing to logistical reasons, the replication series was not performed on consecutive patients as for the primary series. However, we did not observe any significant difference between the series in demographic factors possibly affecting the risk of AAA and thus minimizing the risk of selection bias.
Smoking was recorded using categorical variables of ever-smokers (significant history of smoking or present smoker) and non-smokers. A continuous variable (like number of pack-years) could have demonstrated a dose-dependent cumulative risk of AAA and probably clarified the role of COPD as a risk factor for AAA. This dose-dependent risk has been demonstrated in the literature (6), but the use of categorical variables is easier in clinical practice, and therefore, we opted for the categorical variable.
The relatively small size of the study population probably explains the lack of association of AAA with the other well-established risk factors, such as hypertension.
Strengths of this Study
Characteristics and results are homogeneous in both series despite the difference in study center and consecutiveness. The study result of prevalence and smoking is clear and can be generalized outside the Finnish male population (Caucasian male patients). Furthermore, our study setting (focusing on previously unscreened but amendable and physically active population) emphasizes the clinical relevance and applicability of our results.
Conclusion
Prevalence of undiagnosed AAAs among patients with CAD remains high and history of smoking stands out as the strongest independent risk factor for AAA in this population. Among men with AAA, smokers are substantially younger (over a decade) than non-smokers. Effectiveness of selective screening of AAA warrants further studies.
Footnotes
Acknowledgements
Matti Turtiainen, MD, Consultant Radiologist, North Karelia Central Hospital, provided assistance and the second opinion on cases with poor visibility to aorta. Raimo Jauhiainen, MD, PhD, North Karelia Central Hospital, provided assistance in collection of clinical data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by EVO funding of Kuopio University Hospital.
