Abstract
Background and Aims:
Bioactive glass S53P4 is an antibacterial bone substitute with bone-bonding and osteostimulative properties. The bone substitute has been successfully used clinically in spine; trauma; orthopedic; ear, nose, and throat; and cranio-maxillofacial surgeries. Bioactive glass S53P4 significantly reduces the amount of bacteria in vitro and possesses the capacity to kill both planktonic bacteria and bacteria in biofilm. Three patients with severe spondylodiscitis caused by Mycobacterium tuberculosis, Candida tropicalis, or Staphylococcus aureus were operatively treated due to failed conservative treatment. The vertebral defects were reconstructed using bioactive glass S53P4 and an expandable replacement device.
Material and Methods:
Decompression and a posterolateral spondylodesis, using transpedicular fixation, were performed posteriorly in combination with an anterior decompression and reconstruction using an expandable vertebral body replacement device. For patients 1 and 2, the expander was covered with bioactive glass S53P4 only, and for patient 3, the glass was mixed with autograft bone.
Results:
The patients healed well with complete neurological recovery. Fusion was observed for all patients. The total follow-up was 4 years for patient 1, 1 year and 8 months for patient 2, and 2 years and 2 months for patient 3. No relapses or complications were observed.
Conclusion:
The antibacterial properties of bioactive glass S53P4 also make it a suitable bone substitute in the treatment of severe spondylodiscitis.
Introduction
Pyogenic vertebral osteomyelitis (VO) is a rare condition. In the developed countries, a steady increase in VO related to the aging population has, however, been observed. The incidence of VO has been estimated to be 2.4 per 100,000, increasing to 6.5 per 100,000 among people >70 years of age (1). The pathogen causing VO is most often Staphylococcus aureus. VO is usually caused by hematogenous seeding, but it can also be related to spinal surgery or originate from a nearby infection (2). In cases of suspected VO, magnetic resonance imaging (MRI) is recommended. The diagnostic sensitivity is 82% and the specificity 53%–94%. Usually, the infection involves a disk space, the epicenter of the pathologic change, and two adjacent vertebral bodies (3).
Hematogenous VO can usually be treated successfully with antibiotics. Complications comprising epidural or psoas abscess formations, which are common findings, may need surgical drainage (1). In some patients, the infection causes large bony defects leading to instability and pathological fractures, resulting in the need for stabilization and reconstruction of the lost vertebral bone. Using metal implants and bone substitutes in infected fields is controversial. However, acute placement of spinal instrumentation has not been associated with recurrent infections (4–9).
In most patients, bone grafts are needed. a (BAG) S53P4, consisting of 53% SiO2, 23% Na2O, 20% CaO, and 4% P2O5, is an antibacterial, bone-bonding, and osteoconductive bone substitute, with osteogenetic and angiogenetic stimulating properties. BAG S53P4 has successfully been used in the filling of cavitary defects in the treatment of demanding osteomyelitis (10, 11), mastoiditis, and chronic frontal sinusitis for more than 10 years (12–15). Here, the operative technique and the long-term outcome of a reconstruction and stabilization method, using internal fixation and BAG S53P4 in the treatment of three demanding cases with verified severe VO caused by three different pathogens, are reported.
Materials and Methods
Patient 1 (female, 80 years of age) suffered from diffuse low back pain in 2007. Spondylodiscitis affecting lumbar vertebrae L3 and L4 and paravertebral and epidural abscess formations were observed on MRI. The patient was treated conservatively with antibiotics for 2 years without success. Progressive lytic destructions of vertebrae L3 and L4 were observed, despite the conservative treatment. The patient needed a rollator for walking because of weakness of the lower extremities (Frankel D), and in bed the neurological status was normal. The indication for operative treatment was progressive lytic deformities. In 2009, a posterior decompression of L2/3–L3/4, a posterolateral spondylodesis of L2–L5, using transpedicular fixation with an Omega 21 device (titanium alloy; Biomet Inc., Warsaw, IN, USA), in combination with lumbotomy and canalization of paravertebral abscesses, resection of vertebral bodies of L3–L4, and anterior decompression and reconstruction from L2 to L5, using a Synex 2 device (titanium alloy; Synthes Inc., Solothurn, Switzerland), were performed. The expander was covered with 0.8–1.0-mm BAG S53P4 granules (BonAlive Biomaterials, Ltd, Turku, Finland) (volume: 32 mL). Before the operation, no pathogen had been identified. Postoperatively, we confirmed by samples taken during the operation that the patient suffered from an infection caused by Mycobacterium tuberculosis.
Patient 2 (male, 53 years of age, a narcotic addict with hepatitis C) suffered from back pain, progressive kyphosis, and numbness in the lower extremities since 2006. Spondylodiscitis affecting L3 and L4 and destruction of L2–L4 were verified on MRI. Bone computed tomography (CT) biopsy confirmed that the patient suffered from the pathogen Candida tropicalis. He was treated conservatively without success, and the bodies of vertebrae L3 and L4 were completely destroyed, as well as the lower part of L2. He walked with crutches and showed some signs of cauda equina syndrome (Frankel D). In 2010, a posterior decompression of L2/3–L4/5, reduction, and posterolateral spondylodesis using a transpedicular Omega 21 device were performed. BAG S53P4 granules 2.0–3.15 mm in size (volume: 16 mL) were mixed with autologous bone and placed posterolaterally. In a second operation 5 days later, the bodies of L3 and L4 were resected and an anterior reconstruction was performed using an expandable replacement device (Obelisc, titanium alloy; Ulrich Medical GmbH & Co. KG, Ulm, Germany). The end holes of the implant were packed and the entire implant covered with 1.0–2.0 mm BAG S53P4 granules (volume: 20 mL) without any autologous bone transplant.
Patient 3 (male, 72 years of age, usually healthy) had acute back pain and fever in 2010. In 2 weeks, he developed cauda equina syndrome and paralysis of the lower left extremity. MRI revealed spondylodiscitis affecting the thoracic vertebrae T6–T8. A hemilaminectomy-type minor decompression was done and repeated twice during 9 days in another institute without any success. S. aureus was cultured in the specimens taken during the operations. Before the first operation, the patient was classified as Frankel D and after the third operation Frankel C with total cauda equina syndrome. At that stage, the patient was referred to our clinic, and a posterior decompression of vertebrae T6–T7 (laminectomia totalis) and a posterolateral spondylodesis of T4, T5, T8, andT9, using a transpedicular fixator (titanium alloy; Diapason; Stryker Corp., Kalamazoo, MI, USA), were performed. BAG S53P4 particles 2.0–3.15 mm in size (volume: 10 mL) mixed with healthy-looking pieces of the laminectomy bone were placed posterolaterally on both sides. The operation then continued with a right-sided thoracotomy, a resection of the vertebral bodies of T6–T7, an anterior decompression, and a reconstruction using an Obelisc expandable vertebral body replacement device. BAG S53P4 mixed with autologous rib bone was placed into the end holes of the implant. Part of the rib was placed as a block in the side of the implant, and the rest of the mixture of BAG/crushed rib was placed in the sides and in front of the implant.
Results
All patients recovered from the infection, and no reinfections were noted during the follow-up. The total follow-up was 4 years (patient 1), 1 year and 8 months (patient 2), and 2 years and 2 months (patient 3). For patient 1, a small psoas abscess formation was observed postoperatively; no further operative treatment was, however, needed and the abscess formation disappeared spontaneously. The antibiotic therapy lasted 6 months for patient 1. Patient 2 was prescribed continuous diflucan treatment. Patient 3 was prescribed antibiotic treatment for 2 years and 2 months. The postoperative plasma C-reactive protein (P-CRP) level was 279 mg/L (patient 1), 139 mg/L (patient 2), and 21 mg/L (patient 3). The C-reactive protein (CRP) value became and remained normal for all patients.
All three patients showed full neurologic recovery and underwent spinal fusion during follow-up. Excellent radiological results with total fusion were observed for all patients. The pre- and postoperative radiological findings for patient 3 are shown in Figs 1 to 5. In patient 1, BAG S53P4 alone was placed around the vertebral body replacement device. CT revealed that the entire region was completely fused. In patients 2 and 3, BAG S53P4 was also placed at the end holes of the device. CT in patient 3 revealed that the inside of the device was completely filled with bone. Patients 1 and 2 received only BAG in the anterior part of the spine, while patient 3 received a mixture of BAG and crushed autologous bone from the ribs. Patient 2 later died of acute leukemia.
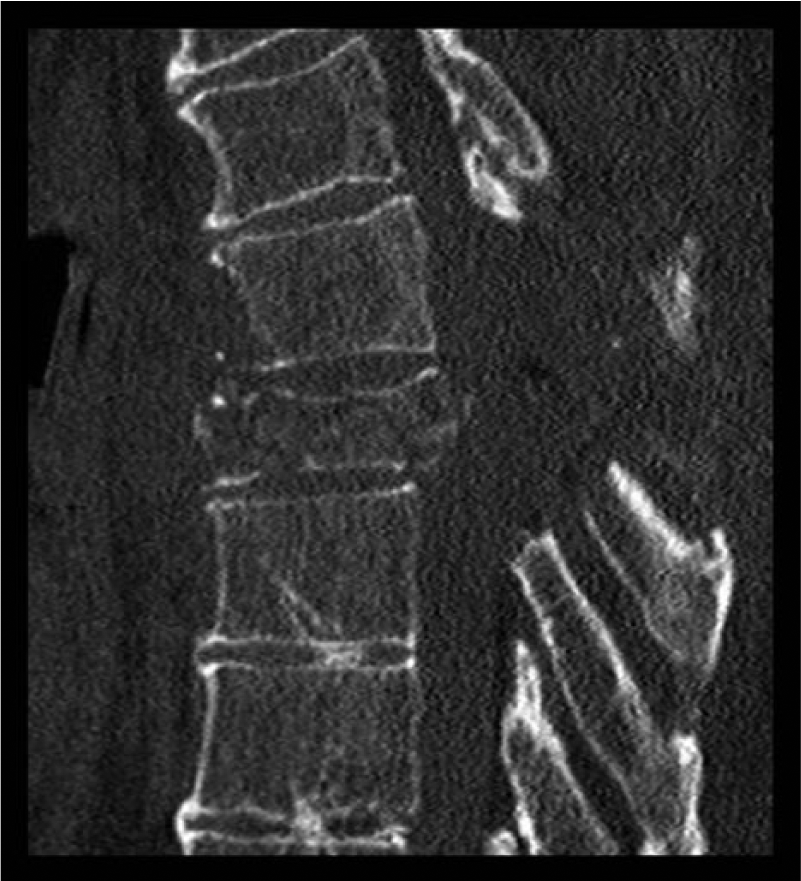
Patient 3, preoperative CT.
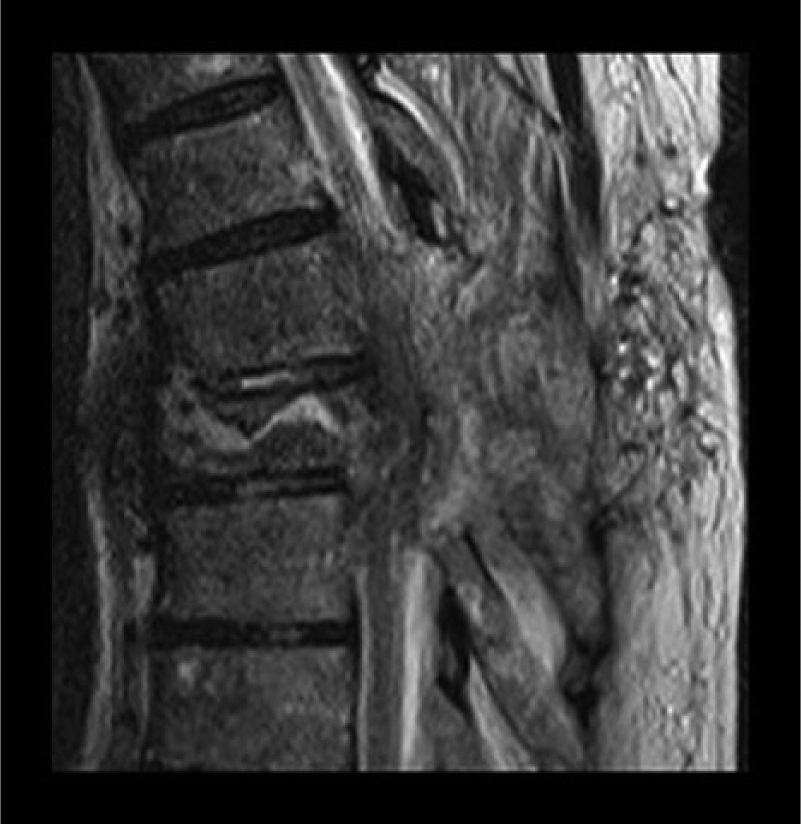
Patient 3, preoperative MRI.
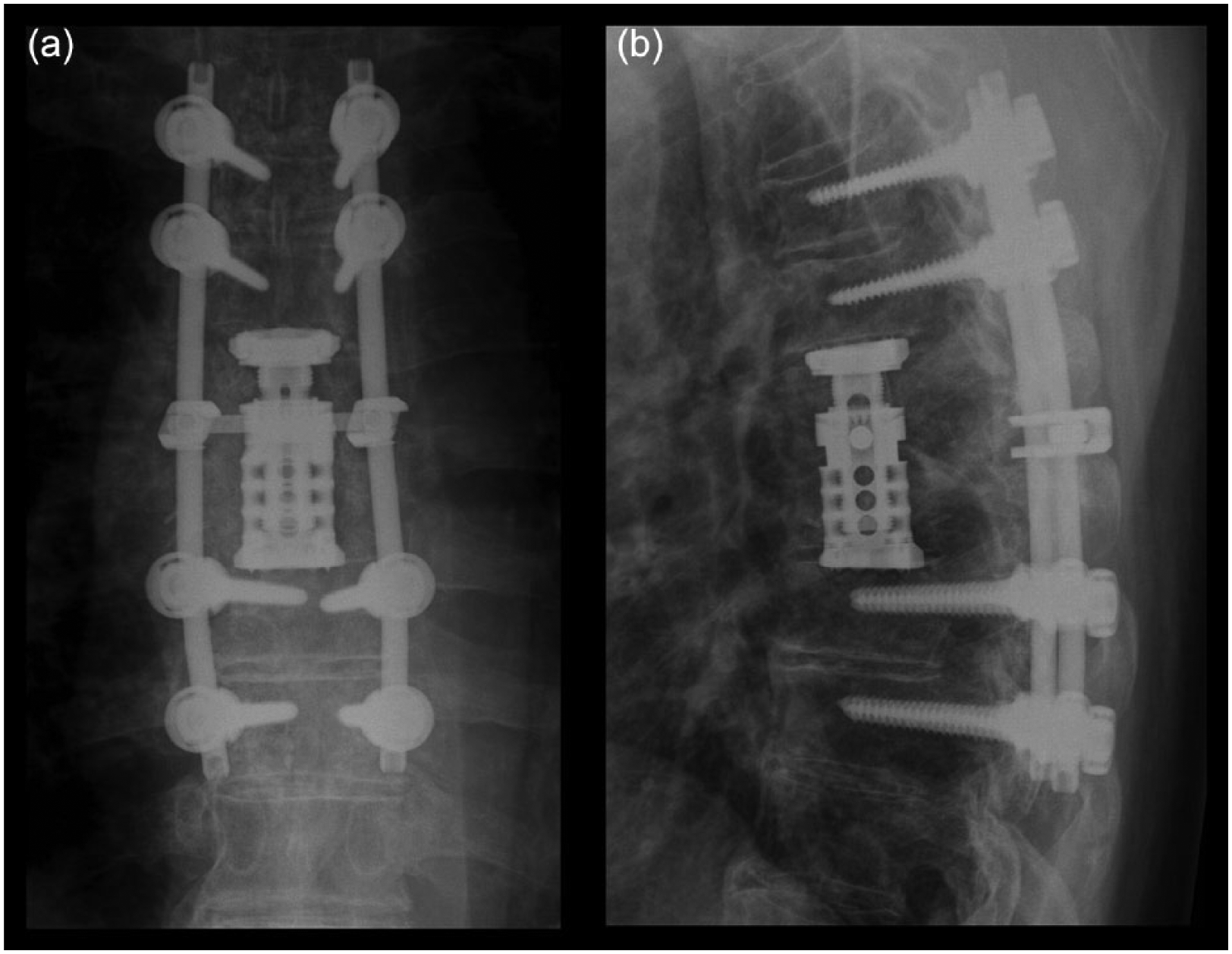
(a, b) Patient 3, postoperative X-ray at 3 months.

(a, b) Patient 3, postoperative CT at 2 years and 2 months.
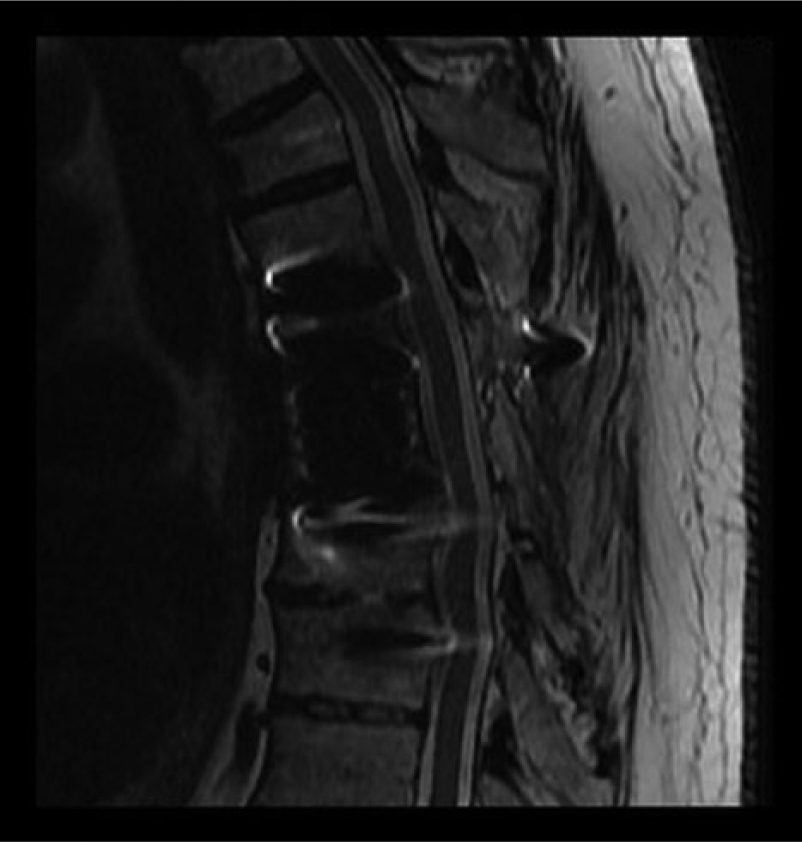
Patient 3, postoperative MRI at 2 years and 2 months.
Discussion
Surgical management of VO is demanding, especially in cases comprising large bony defects. Using implants, mainly of titanium, in the treatment of VO has been associated with low long-term recurrence risk. To use any type of implant in infected regions is, however, controversial because pathogens are able to produce biofilms on the surface of the implanted materials, thus complicating the treatment outcome. Several methods have been developed for reconstruction of destroyed vertebrae, including expandable replacement devices and cages. Fusion is usually promoted using auto- or allografts. The use of autografts is associated with morbidity at the donor site, and especially in children and adolescents, the availability of graft material can be limited. Allograft bone is associated with resorption and the possibility of spread of infections.
Use of synthetic bone grafts and implanted materials in treating osteomyelitis is generally not recommended. Bacterial colonization on implanted materials, with subsequent formation of biofilms, may hinder antibiotic treatment and recovery. However, BAG S53P4 significantly reduces S. aureus and Pseudomonas aeruginosa biofilm formation on titanium disks in vitro (16). Marked bactericidal activity of BAG S53P4 against methicillin-resistant S. aureus, Staphylococcus epidermidis, and Pseudomonas aeruginosa have been observed in vitro (11). Studies of atrophic rhinitis, a purulent disorder often caused by Klebsiella ozaenae, have shown that BAG S53P4 does not favor adhesion, colonization, or biofilm formation of K. ozaenae on its surface (17). Clinically, BAG S53P4 has been used as a bone graft substitute in cavitary defects in the treatment of osteomyelitis, with excellent clinical results (10, 11). In the majority of patients, the procedure has been performed using BAG S53P4 in a one-stage procedure.
The antibacterial effect of BAG S53P4 is well established. The properties of the glass have been associated with the initial leaching of alkali and alkaline-earth ions from the glass surface, starting immediately after implantation. The dissolution process is characterized by a rapid exchange of Na+ in the glass with H+ from the solution, resulting in a subsequent increase in pH. The high pH, which in a simulated body fluid, increases to a pHmax value of 11, and the osmotic pressure in the vicinity of the implanted glass granules may explain the antibacterial effect of BAG. Comparing the bactericidal effect of BAG S53P4 with that of several other glasses has shown that BAG S53P4 is the most effective, with the fastest killing and growth inhibitory effect. This has been observed for 29 aerobic and 17 anaerobic pathogens tested, including multiresistant pathogens (18–21).
The antibacterial effect is dependent on not only the glass composition but also the granule size. Comparing the in situ pH within the particles of BAGs in vitro has shown a relationship between pH and glass granule size. An increase in granule size corresponding to a decrease in pH, explained by a decrease in surface area, was observed (21). Various sizes of BAG S53P4 particles were used in our patients. For patient 1, a small granule size was chosen to achieve a large surface area. The same clinical results were, however, achieved despite the differences in granule size used.
In this study, fusion was observed for all patients both when used alone and as a mixture with autologous bone. A weakness in this study is that the total follow-up for patient 2 did not have a formal 2-year follow-up due to the death of the patient. In spinal surgery, long-term results using BAG S53P4 as a stand-alone bone substitute in instrumented posterolateral spondylodesis revealed a fusion rate of 88% in the treatment of spondylolisthesis and a fusion rate of 71% in the treatment of unstable lumbar fractures (22, 23). We, therefore, recommend that S53P4 be used as an expander combined with autologous bone. We also demonstrated that a favorable outcome can be achieved using antibacterial BAG S53P4 as a bone graft substitute in the treatment of severe VO.
Footnotes
Declaration of Conflicting Interests
Nina Lindfors is a clinical advisor at BonAlive® Biomaterials.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
